Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 1 by Wei Cui and Version 2 by Camila Xu.
Composite materials are widely applied in industrial and biomedical fields as structural materials due to their superior comprehensive properties, such as high strength, high stiffness, low weight, corrosion resistance, etc.
- toughness
- toughening mechanism
- fabrication
- application
1. Introduction
Composite materials are widely applied in industrial and biomedical fields as structural materials due to their superior comprehensive properties, such as high strength, high stiffness, low weight, corrosion resistance, etc. [1][2][3][4][1,2,3,4]. Conventional hard composites are generally composed of rigid matrices (resins, metals, ceramics, etc.) and rigid fibers or fabrics, showing isotropically high stiffness [5][6][7][5,6,7]. Soft composite, in contrast, is a sort of composite material with low flextural stiffness [8][9][10][11][12][8,9,10,11,12]. Commonly, both hard and soft composites show very high fracture stress and Young’s modulus in tension, which can reach values on the order of 100 and 101 GPa, respectively. The biggest difference between hard and soft composites is their mechanical performance upon bending. The bending stiffness of soft composites (100 MPa) is usually several orders of magnitude lower than that of hard composites (101 GPa) [5][12][5,12]. Because of a unique combination of high stiffness in tension and low hardness upon bending, soft composites are uniquely applicable to numerous applications such as soft robotics, sensors, actuators, etc. [9][11][12][9,11,12].
Although a variety of soft composites have been developed for specific applications, a timeless topic is how to toughen them, as mechanical properties always determine the reliability and lifespan of relevant products. The toughening of materials has a long history and the main concept is widely accepted for either soft or hard materials, i.e., simultaneously enhancing the size of energy dissipation zone and energy dissipation density [13][14][15][16][13,14,15,16]. In other words, a material with high toughness should, on one hand, dissipate energy at a large length scale, and on the other hand, dissipate as much as energy per unit volume [16][17][18][16,17,18]. For instance, glass is a strong, stiff, but extremely brittle material. This is because the glass plate can only dissipate energy at the atom scale, although it has a high energy dissipation density (Figure 1a) [19]. In contrast, rubber is not as strong as glass, whereas it possesses a much higher fracture toughness due to a significantly increased energy dissipation zone at the polymer chain scale (Figure 1b) [15][20][15,20]. Double network materials can show even superior fracture toughness to rubbers because their energy dissipation density is further increased due to the introduction of extra network chains (Figure 1c) [21][22][23][21,22,23]. That is, tough soft composites always possess high fracture toughness over 100 kJ m−2 and are highly resistant to crack growth even when they are notched.
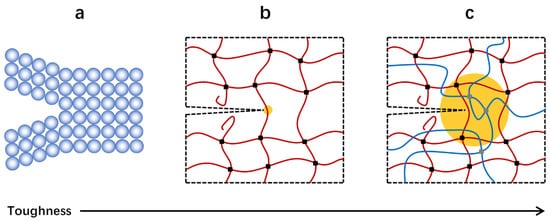
Figure 1. Different energy dissipation mechanisms give distinct toughness. (a) Breaking a layer of bonds. (b) Snapping a layer of chains. (c) Dissipating energy in the bulk.
Many biological tissues are essentially tough soft composites by exploiting the above concept [24][25][26][27][24,25,26,27]. These natural materials, generally consisting of stiff fibrous skeletons and soft extracellular matrices, are anisotropic, strong, and tough via the synergy of a large energy dissipation zone and high energy dissipation density. For example, heart valves possess fracture toughness around 1200 J m−2 while showing high resilience [28]. Tendon is a strong connective tissue that connects muscle to bone and muscle to muscle, which can sustain over 1 million cycles of loading per year and show fracture toughness as high as 20–30 kJ m−2 [28]. The efficient stress transfer between the stiff and soft phases enables these tissues to dissipate energy at a large length scale, and the energy-dissipative components both contribute to a high energy dissipation density.
Until now, immense efforts using synthetic approaches have been taken in an endeavor to fabricate tough soft composites. Generally, soft composites can be classified by their energy dissipation mechanisms at different length scales (Figure 2). At the molecular level, double network (DN) material is a typical soft composite, which dissipates energy by breaking polymer chains [29][30][29,30]. At the nanometer scale, phase-separated material is an example, dissipating energy via the rupture of nanophases [31][32][33][31,32,33]. At the micron scale, common soft composites are micro-fiber-reinforced polymers. The energy dissipation relies on the stress transfer between the stiff fibrils and soft matrices [34][35][36][37][34,35,36,37]. At the millimeter scale, macroscopic fiber fabrics are combined with soft polymer matrices, generating soft composites that dissipate energy by breaking both fibers and matrices [18][38][39][40][18,38,39,40].
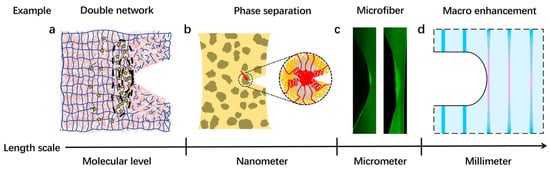
Figure 2. Energy dissipation mechanisms at different length scales of soft composites. (a) Double network materials with sacrificial bonds at the molecular scale. Reproduced with permission from reference [29] Copyright 2014 The American Association for the Advancement of Science. (b) Phase-separated materials with hard phases at the nanoscale. Reproduced with permission from reference [31] Copyright 2021 American Chemical Society. (c) Micro-fiber-reinforced soft composites dissipate energy at the micrometer scale. Reproduced with permission from reference [37] Copyright 2019 National Academy of Sciences. (d) Soft composites with macroscopic reinforcing phases dissipate energy at the millimeter scale. Reproduced with permission from reference [38] Copyright 2019 National Academy of Sciences.
2. Fabrication
2.1. Double Network
The double network (DN) method is one of the most pioneering strategies to toughen soft materials, especially for hydrogels and elastomers. The general concept of the DN strategy is to combine two interpenetrating polymer networks with contrasting structures (Figure 3a) [41][44]. The first network is usually densely cross-linked, which is rigid and brittle. The second network is a sparsely cross-linked neutral polymer with a much higher concentration, which is soft and stretchable [42][45]. A DN gel or elastomer can be deemed as a molecular-level soft composite with high heterogeneity, although it behaves like a homogenous material.
The key factors to construct an efficient DN network instead of a simple interpenetrating network are, on one hand, to highly pre-stretch the first network, resulting in taut molecular chains with high stiffness, on the other hand, to swell the first network as much as possible in the second monomer solution with a low concentration of the cross-linking agent, enabling the high concentration ratio of the second network to the first network. In classical hydrogel systems, such a contrasting DN structure is realized by utilizing polyelectrolytes as the first network, which dramatically swell in the second neutral monomer solution due to high osmotic pressure. This effect leads to highly extended first network chains and a high concentration ratio between two networks (Figure 3b) [43][46]. Because both networks are chemically cross-linked, the classical DN hydrogels cannot self-recover after damage. Physical DN hydrogels are then developed to enable recoverable energy dissipation mechanisms. A typical example is the Ca2+-alginate/polyacrylamide system, which exploits the unzipping of ionic crosslinks between Ca2+ and alginate to dissipate energy and the re-zipping of the ionic bonds to heal the damage [23]. Another example is the physical DN gels based on an amphiphilic triblock copolymer that contains strong hydrophobic domains and sacrificial hydrogen bonds [44][47]. The reversible physical interactions of the first network enable the resulting DN gels to partially restore the mechanical properties after damage. The fabrication of physical DN gels is different from that of chemical ones. Only a one-pot method is required when preparing physical DN gels because the first network is usually a pre-polymer, which can dissolve in the second monomer solution by either stirring or heating (Figure 3c) [43][46]. As long as the concentration ratio between the first and second networks is carefully controlled, physical hydrogels with DN features can also be successfully fabricated. The DN method was firstly employed in the hydrogel systems and later proven to be applicable in elastomer systems [29][45][46][47][29,48,49,50]. The preparation of typical DN elastomers is slightly different from that of DN hydrogels since neutral monomers are commonly utilized to construct the first network. A multi-step swelling method was introduced to sufficiently pre-stretch the first network and form a high concentration contrast between the soft and the rigid networks. A drawback of this multi-step method is that it consumes significant time and energy. To overcome this dilemma, Matsuda et al. employed polyelectrolytes as the first network and dissolved them in organic solvents that have a similar dielectric constant to the monomer, giving rise to the formation of highly extended first network chains and enabling the dramatic swelling of the first network in second neutral monomer solutions (Figure 3d) [48][51]. In this way, a DN elastomer can be manufactured using the traditional method for DN hydrogels.
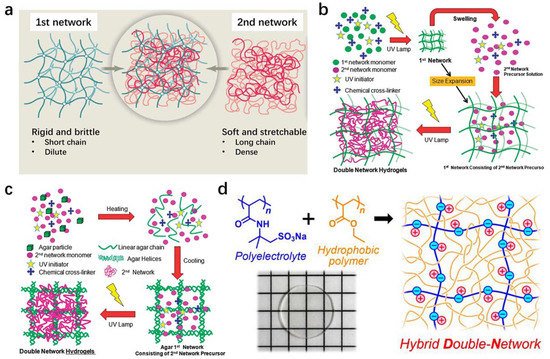
Figure 3. Fabrication of double network hydrogels and elastomers. (a) The composition of conventional chemical double network hydrogels. Reproduced with permission from reference [41][44] Copyright 2014 The American Association for the Advancement of Science. (b) Fabrication of conventional chemical double network hydrogels. Reproduced with permission from reference [43][46] Copyright 2015 Royal Society of Chemistry. (c) Fabrication of physical double network hydrogels. Reproduced with permission from reference [43][46] Copyright 2015 Royal Society of Chemistry. (d) Fabrication of double network elastomers. Reproduced with permission from reference [48][51] Copyright 2019 American Chemical Society.
2.2. Phase Separation
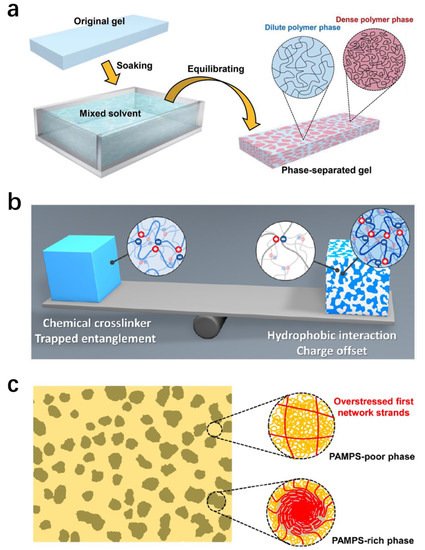
Phase separation results in hydrogels or elastomers that possess dilute and dense polymer phases [49][50][51][52,53,54]. The dilute phase is soft, consisting of polymers with low volume fractions. On the contrary, the dense phase is relatively hard, which is composed of polymers with high volume fractions. The energy dissipation of phase-separated materials depends on the rupture of massive dense polymer phases at the nanometer scale. Therefore, phase-separated materials can be simply considered nano-scale soft composites.
There are a variety of methods to fabricate hydrogels or elastomers with phase-separated structures. One simple way is to utilize mixed-solvent-induced phase separation. Equilibrating a neutral hydrogel in a mixture of both good and poor solvents induces phase separation, resulting in an inhomogeneous network structure with bicontinuous domains (Figure 4a) [52][55]. Because of the low polymer-solvent affinity between the network chains and the poor solvent, a portion of polymer chains aggregate into local dense phases, which possess a considerably high polymer volume fraction to induce inter-/intra- polymer interactions. In contrast, other polymer chains in low volume fractions form the dilute polymer phases, which have relatively low modulus. Another example is the polyampholyte (PA) hydrogel system (Figure 4b) [49][53][52,56]. Unlike the solvent-initiated phase separation, the PA gels show a bicontinuous network structure due to the distribution of ionic bonds. The different density of ionic bonds leads to the local aggregation of polymer backbones via hydrophobic association, giving rise to a network structure with soft and hard phases. The phase separation method is also applicable in elastomer systems by using an ionic polymer as the first network and a nonpolar polymer as the second network (Figure 4c) [31]. Firstly, the polyelectrolyte network is polymerized and then soaked into the second monomer solution using a cosolvent of a high dielectric constant. Due to the high osmotic pressure, the first network is highly pre-stretched and brittle. Afterward, the second network is polymerized and formed within the first network, producing DN materials with a highly contrasting architecture. A DN elastomer is formed by finally removing the cosolvent.

Figure 4. Fabrication of phase-separated hydrogels and elastomers. (a) Fabrication via mixed-solvent-induced phase separation. Reproduced with permission from reference [52][55] Copyright 2021 Royal Society of Chemistry. (b) The bicontinuous phase-separated structure of polyampholyte hydrogels. Reproduced with permission from reference [49][52] Copyright 2020 American Chemical Society. (c) Elastomers fabricated via nanophase separation. Reproduced with permission from reference [31] Copyright 2021 American Chemical Society.
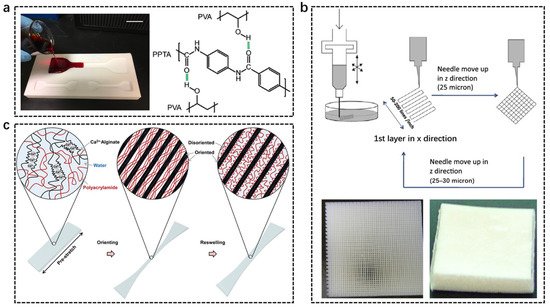
2.3. Microscopic Reinforcement
Introducing microfibers into a soft matrix is a well-established way to develop soft composites [54][55][56][57][57,58,59,60]. Usually, the added microfibers have a relatively low volume fraction, which, however, can lead to obvious mechanical enhancement of the soft composites. Interfacial interactions between the fibers and the matrix are crucial for efficient stress transmission and resulting energy dissipation. In this part, we give several common ways to fabricate tough soft composites by introducing micro-fibers as the enhancement phases.
The most common way to fabricate micro-fiber-reinforced soft composites is direct blending. A great number of examples can be listed because this method is simple and efficient. For the hydrogel system, it is worth noting that the micro-fiber should interact strongly with the hydrophilic polymer matrix to prevent interfacial delamination upon load. For instance, aramid micro-fibers can form massive hydrogen bonding with polyvinyl alcohol chains, the mixture of which gives rise to a strong and tough composite hydrogel (Figure 5a) [58][61]. The efficient stress transfer between the stiff fibers and the soft matrices enables the gel to show self-organization behaviors, resembling biological tissues. Apart from common fillers, 3D printed fibers can also be used to fabricate strong soft composites. By employing a 3D rapid prototyping technique, crossed log-piles of elastic fibers are fabricated (Figure 5b) [59][62]. Epoxy-based hydrogels are combined with the fibers to form an interpreting structure. Adjusting the construct geometry, corresponding mechanical properties of the soft composite such as strength, modulus, and toughness can be facilely regulated. The above methods mainly involve the introduction of external fiber fillers, which must carefully consider the interfacial interactions between the fillers and the matrices. An emerging method that can ignore the interface problem is to form microfibers in situ in soft composites [60][63]. This strategy is originally inspired by biological fibrous tissues. In bio-tissues, microfibers impregnate thoroughly with soft extracellular matrices, without the concern of interfacial problems. The key to the formation of microfibers is the utilization of rigid polymers with a large persistence length. By orienting and drying such polymers, strong microfibers can be formed, which are capable of maintaining the fibrous structure after reswelling. On the contrary, an oriented soft polymer with a small persistence length tends to restore the isotropic network structure after reswelling. Based on the contrasting reswelling behaviors, soft hydrogel composites can be fabricated by orienting and reswelling two interpreting networks with different persistence lengths (Figure 5c) [61][64].

Figure 5. Fabrication of micro-fiber-reinforced soft composites. (a) Micro aramid fiber-reinforced hydrogels. Reproduced with permission from reference [58][61] Copyright 2017 Wiley. (b) Hydrogel composites reinforced by 3D printed skeletons. Reproduced with permission from reference [59][62] Copyright 2012 Elsevier. (c) Hydrogel composites fabricated via reswelling disparity of two oriented polymers with contrasting persistence lengths. Reproduced with permission from reference [61][64] Copyright 2021 Royal Society of Chemistry.
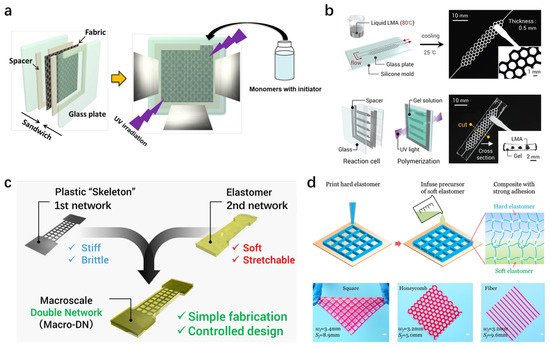
2.4. Macroscopic Reinforcement
Soft composites with satisfactory mechanical properties can also be produced by using macroscopic reinforcement, which includes, but is not limited to, fiber fabrics, metal meshes, plastic grids, etc. The preparation method of soft composites with macroscopic enhancement is similar to that of micro-filler-reinforced soft materials and the interface is still a key point that should be taken into account. Here, we give some examples for developing soft composites with macroscopic reinforcing phases.
A very familiar system with macroscopic phases is the fabric-reinforced soft composite. Unlike soft composites enhanced by microfibers, fabric-reinforced soft composites are usually fabricated by combining soft matrices with woven fiber fabrics (Figure 6a) [18]. In this case, the fiber volume fraction is relatively high, and the mechanical properties are mainly dominated by the fabric phases. Moreover, the mechanical and structural anisotropy of the soft composites can be regulated by exploiting a variety of weave patterns of the fabrics, which are different from the random distribution of fillers in micro-fiber-reinforced soft composites. Besides common fabrics, some novel rigid phases are also applicable to develop soft composites. Hydrogel composites with a series of desired properties, such as excellent mechanical performance, shape memory, and thermal healing, are created by integrating a low-melting-point alloy into a hydrogel system (Figure 6b) [62][65]. The alloy is able to transform from a load-bearing solid state to a free-deformable liquid state upon temperature increase, enabling the release of stress concentration between the soft and rigid phases. The resulting hydrogel composites can be uniquely applied in controlled electrochemical reactions and channel-structure templating by virtue of the special metal-hydrogel combination. The 3D printed rigid plastic grids are also proven efficient to construct tough soft composites. By simply combining silicon rubber and such rigid grids, macroscopic double network composites are fabricated (Figure 6c) [63][66]. The topological interlocking enables significant force transmission between the soft and rigid phases, preventing delamination. The optimal mechanical properties appear when the grid/matrix strength ratio is approaching one. Interestingly, soft composites with macroscopic reinforcement can also be fabricated from the same type of elastomers with different rigidity. By tuning the cross-linking density, a hard elastomer and a soft elastomer can be prepared, which shows contrasting Young’s modulus. The hard elastomer acts as the macroscopic reinforcing phase while the soft elastomer is the matrix (Figure 6d) [64][65][67,68]. Such a soft/hard combination allows the composite elastomers to show excellent fatigue resistance by virtue of the efficient stress de-concentration.

Figure 6. Fabrication of soft composites with macroscopic reinforcing phases. (a) Fabrication of traditional woven fabric-reinforced soft composites. Reproduced with permission from reference [18] Copyright 2020 Wiley. (b) Soft composites made up of low-melting-point alloy reinforced hydrogels. Reproduced with permission from reference [62][65] Copyright 2018 Wiley. (c) Elastomer composites with 3D printed plastic skeletons as the reinforcing phases. Reproduced with permission from reference [63][66] Copyright 2019 American Chemical Society. (d) Combining two elastomers with different stiffness into a tough soft composite. Reproduced with permission from reference [64][67] Copyright 2020 Elsevier.
