Malaria is a life-threatening global epidemic disease and has caused more than 400,000 deaths in 2019. To control and prevent malaria, the development of a vaccine is a potential method. An effective malaria vaccine should either combine antigens from all stages of the malaria parasite’s life cycle, or epitopes of multiple key antigens due to the complexity of the Plasmodium parasite. Malaria’s random constructed antigen-1 (M.RCAg-1) is one of the recombinant vaccines, which was selected from a DNA library containing thousands of diverse multi-epitope chimeric antigen genes. Moreover, besides selecting an antigen, using an adjuvant is another important procedure for most vaccine development procedures. Freund’s adjuvant is considered an effective vaccine adjuvant for malaria vaccine, but it cannot be used in clinical settings because of its serious side effects. Traditional adjuvants, such as alum adjuvant, are limited by their unsatisfactory immune effects in malaria vaccines, hence there is an urgent need to develop a novel, safe and efficient adjuvant. In recent years, Pickering emulsions have attracted increasing attention as novel adjuvant. In contrast to classical emulsions, Pickering emulsions are stabilized by solid particles instead of surfactant, having pliability and lateral mobility. In this study, we selected aluminum hydroxide gel (termed as “alum”) as a stabilizer to prepare alum-stabilized Pickering emulsions (ALPE) as a malaria vaccine adjuvant. In addition, monophosphoryl lipid A (MPLA) as an immunostimulant was incorporated into the Pickering emulsion (ALMPE) to further enhance the immune response. In vitro tests showed that, compared with alum, ALPE and ALMPE showed higher antigen load rates and could be effectively endocytosed by J774a.1 cells. In vivo studies indicated that ALMPE could induce as high antibody titers as Freund’s adjuvant. The biocompatibility study also proved ALMPE with excellent biocompatibility. These results suggest that ALMPE is a potential adjuvant for a malaria vaccine.
- malaria vaccine
- alum stabilized Pickering emulsion
- adjuvant
- immune response
- M.RCAg-1
1. Introduction
2. Results and Discussion
2.1. Pickering Emulsions by Using Alum as Stabilizer
2.1.1. Effect Particle Concentration on Pickering Emulsions

2.1.2. Effect the Buffer Type of the Aqueous Phase on Emulsions
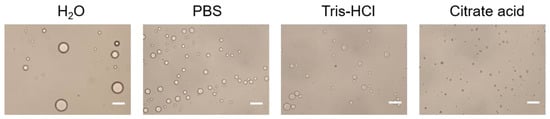
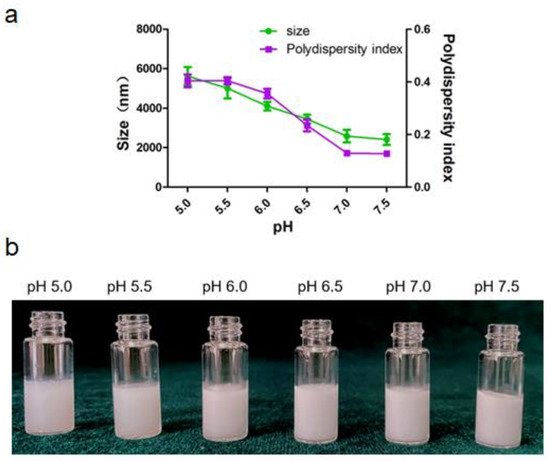
2.1.3. Effect the pH of the Aqueous Phase on Emulsion
2.1.4. Characterization of ALPE and ALMPE
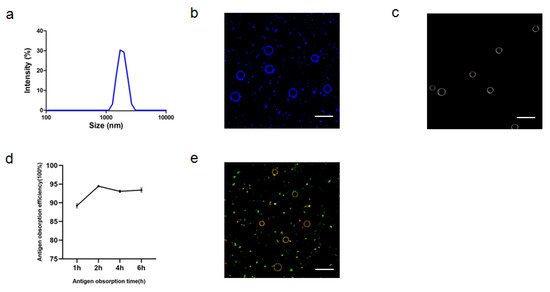
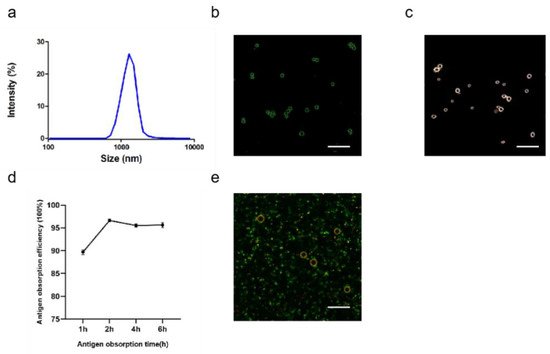
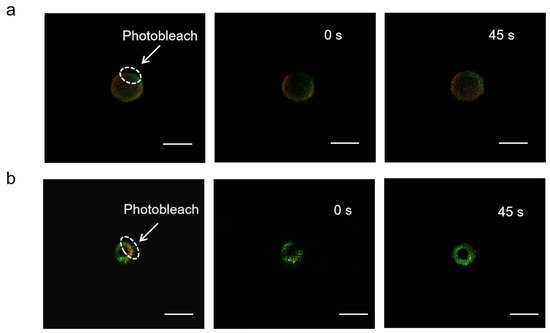
2.2. Cytosolic Localization
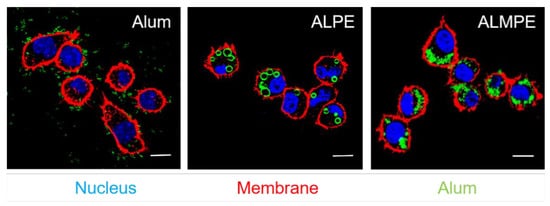
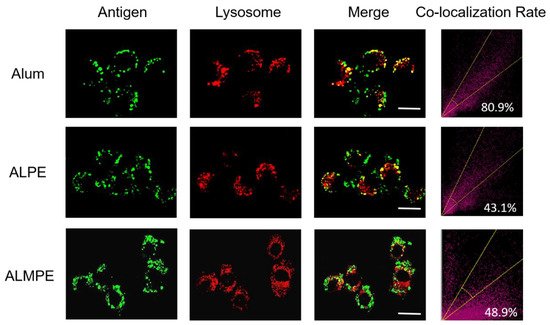
2.3. Enhanced Systemic Immunity In Vivo (OVA as Model Antigen)
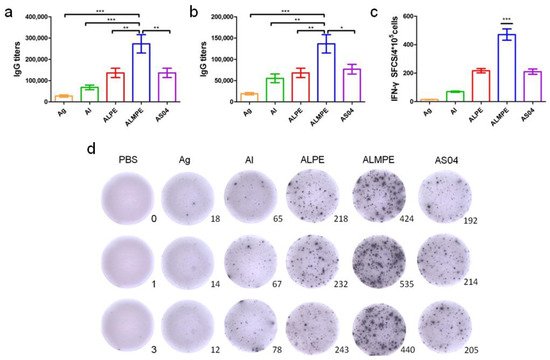
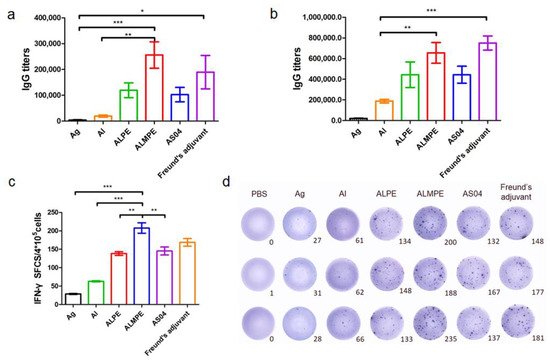
2.4. Adjuvant Effects of Pickering Emulsions for M.RCAg-1 Vaccine
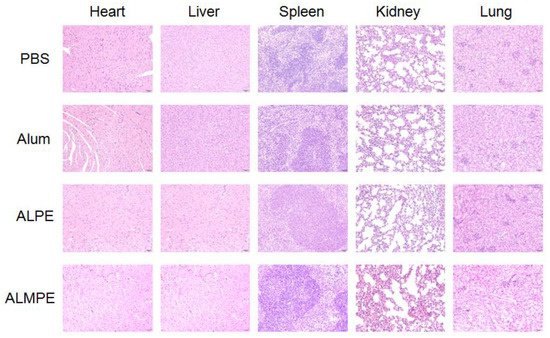
2.5. Safety and Biocompatibility
References
- Guo, F.; Liu, Y.; Zhang, C.; Wang, Q.; Gao, Y.; Deng, W.; Wang, H.; Su, Z. Stabilization of a chimeric malaria antigen in separation and purification through efficient inhibition of protease activity by imidazole. Process. Biochem. 2018, 65, 213–219.
- Schiess, N.; Villabona-Rueda, A.; Cottier, K.E.; Huether, K.; Chipeta, J.; Stins, M.F. Pathophysiology and neurologic sequelae of cerebral malaria. Malar. J. 2020, 19, 1–12.
- Good, M.F.; Stanisic, D.I. Biological strategies and political hurdles in developing malaria vaccines. Expert Rev. Vaccines 2021, 20, 93–95.
- Frimpong, A.; Kusi, K.A.; Ofori, M.F.; Ndifon, W. Novel strategies for malaria vaccine design. Front. Immunol. 2018, 9, 2769.
- Pirahmadi, S.; Zakeri, S.; Djadid, N.D.; Mehrizi, A.A. A review of combination adjuvants for malaria vaccines: A promising approach for vaccine development. Int. J. Parasitol. 2021, 51, 699–717.
- Dicko, A.; Greenwood, B. Malaria vaccination and rebound malaria. Lancet Infect. Dis. 2019, 19, 790–791.
- Tinto, H.; Otieno, W.; Gesase, S.; Sorgho, H.; Otieno, L.; Liheluka, E.; Valea, I.; Sing’Oei, V.; Malabeja, A.; Valia, D.; et al. Long-term incidence of severe malaria following RTS, S/AS01 vaccination in children and infants in Africa: An open-label 3-year extension study of a phase 3 randomised controlled trial. Lancet Infect. Diseases. 2019, 19, 821–832.
- Bell, G.J.; Loop, M.S.; Mvalo, T.; Juliano, J.J.; Mofolo, I.; Kamthunzi, P.; Tegha, G.; Lievens, M.; Bailey, J.; Emch, M.; et al. Environmental modifiers of RTS, S/AS01 malaria vaccine efficacy in Lilongwe, Malawi. BMC Public Health 2020, 20, 1–11.
- Draper, S.J.; Angov, E.; Horii, T.; Miller, L.H.; Srinivasan, P.; Theisen, M.; Biswas, S. Recent advances in recombinant protein-based malaria vaccines. Vaccine 2015, 33, 7433–7443.
- Pritam, M.; Singh, G.; Swaroop, S.; Singh, A.K.; Pandey, B.; Singh, S.P. A cutting-edge immunoinformatics approach for design of multi-epitope oral vaccine against dreadful human malaria. Int. J. Biol. Macromol. 2020, 158, 159–179.
- Cai, Q.-L.; Wei, F.; Lin, Y.-H.; Shao, D.-D.; Wang, H. Immunogenicity of polyepitope libraries assembled by epitope shuffling: An approach to the development of chimeric gene vaccination against malaria. Vaccine 2004, 23, 267–277.
- Binks, B.P.; Clint, J.H.; Whitby, C.P. Rheological behavior of water-in-oil emulsions stabilized by hydrophobic bentonite particles. Langmuir 2005, 21, 5307–5316.
- Aveyard, R.; Binks, B.P.; Clint, J.H. Emulsions stabilised solely by colloidal particles. Adv. Colloid Interface Sci. 2003, 100, 503–546.
- Mwangi, W.W.; Ho, K.-W.; Tey, B.-T.; Chan, E.-S. Effects of environmental factors on the physical stability of pickering-emulsions stabilized by chitosan particles. Food Hydrocoll. 2016, 60, 543–550.
- Sokolovska, A.; Hem, S.L.; HogenEsch, H. Activation of dendritic cells and induction of CD4+ T cell differentiation by aluminum-containing adjuvants. Vaccine 2007, 25, 4575–4585.
- Smith, D.M.; Simon, J.K.; Baker, J.R., Jr. Applications of nanotechnology for immunology. Nat. Rev. Immunol. 2013, 13, 592–605.
- Cimica, V.; Galarza, J.M. Adjuvant formulations for virus-like particle (VLP) based vaccines. Clin. Immunol. 2017, 183, 99–108.
- Bachmann, M.F.; Jennings, G.T. Vaccine delivery: A matter of size, geometry, kinetics and molecular patterns. Nat. Rev. Immunol. 2010, 10, 787–796.
- Zabel, F.; Kündig, T.M.; Bachmann, M.F. Virus-induced humoral immunity: On how B cell responses are initiated. Curr Opin Virol. 2013, 3, 357–362.
- Alavala, S.; Nalban, N.; Sangaraju, R.; Kuncha, M.; Jerald, M.K.; Kilari, E.K.; Sistla, R. Anti-inflammatory effect of stevioside abates Freund’s complete adjuvant (FCA)-induced adjuvant arthritis in rats. Inflammopharmacology 2020, 28, 1579–1597.
- Jin, J.W.; Tang, S.Q.; Rong, M.Z.; Zhang, M.Q. Synergistic effect of dual targeting vaccine adjuvant with aminated β-glucan and CpG-oligodeoxynucleotides for both humoral and cellular immune responses. Acta Biomater. 2018, 78, 211–223.
