Thyroid diseases, including neoplasms, autoimmune diseases and thyroid dysfunctions, are becoming a serious social problem with rapidly increasing prevalence. The latter is increasingly linked to oxidative stress. There are many methods for determining the biomarkers of oxidative stress, making it possible to evaluate the oxidative profile in patients with thyroid diseases compared to the healthy population. This opens up a new perspective for investigating the role of elevated parameters of oxidative stress and damage in people with thyroid diseases, especially of neoplastic nature. An imbalance between oxidants and antioxidants is observed at different stages and in different types of thyroid diseases. The organ, which is part of the endocrine system, uses free radicals (reactive oxygen species, ROS) to produce hormones. Thyroid cells release enzymes that catalyse ROS generation; therefore, a key role is played by the internal defence system and non-enzymatic antioxidants that counteract excess ROS not utilised to produce thyroid hormones, acting as a buffer to neutralise free radicals and ensure whole-body homeostasis. An excess of free radicals causes structural cell damage, undermining genomic stability. Looking at the negative effects of ROS accumulation, oxidative stress appears to be implicated in both the initiation and progression of carcinogenesis.
- oxidative stress
- ROS
- thyroid diseases
- antioxidants
1. Introduction
2. Physiological Redox Signalling and the Role of ROS in Thyroid Function
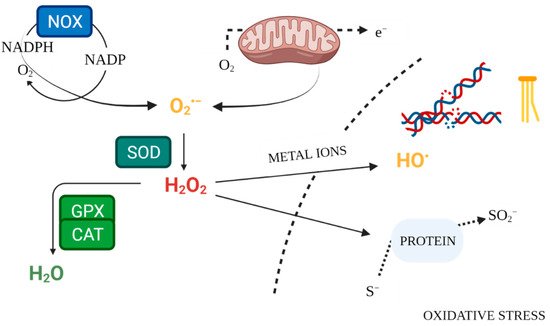
Signalling functions in immune responses are initiated when molecular oxygen is oxidised to the reactive superoxide anion radical by the NADPH oxidase (NOX) complex, itself an additional source of ROS [4]. Subsequently, the superoxide is converted by superoxide dismutase (SOD) to H2O2. Hydrogen peroxide is associated with a signalling function regulating cellular processes, due to its capacity to reversibly modify cysteine residues [20][12]. The process alters redox signalling [17][9]. Accumulation of excessive concentrations of H2O2 activates thiolate anion (Cys-S-) oxidation pathways. This is an irreversible process, resulting in permanent protein damage [40][33]. Antioxidant systems serve a protective function, preventing intracellular accumulation of ROS by reversing the modification of cysteine residues [20][12]. The role (physiological or pathological) played by ROS depends largely on their concentration and the conditions accompanying biochemical transformations. The initial concentration dictates downstream responses [7][34]. Excessive amounts of ROS at the subcellular level activates pathways leading to damage in particularly susceptible cell structures or apoptosis [40][33]. In turn, at low physiological levels, ROS play a signalling role, essential for normal cellular processes [8,41][35][36]. Reactive oxygen species also serve as intracellular mediators produced in phagocytic cells, controlling the inflammatory response and antimicrobial defence [4]. ROS play an important role in normal thyroid function. Thyroid cells release oxidases, which catalyse ROS production [42,43,44][37][38][39]. Inositols are also involved in thyroid hormone synthesis and normal thyroid function, activating a cascade of processes including regulating TSH-dependent signalling (as a TSH transmitter) and generating H2O2 production used for iodination and coupling of iodotyrosine and iodothyronine [45,46,47,48][40][41][42][43]. Inositol deficiency or impairment of inositol cascades may result in insufficient synthesis of thyroid hormones, leading to hypothyroidism, which may be further compounded by an increased need for inositols in response to high TSH levels [45,48][40][43]. Myoinositol supplementation in hypothyroid patients effectively lowers TSH levels. Its effect has been demonstrated in combination with metformin and selenium compared to treatment without inositol [49,50][44][45]. The synthesis of thyroxine (T4) and triiodothyronine (T3) catalysed by thyroid peroxidase (TPO) in thyroid follicles is a very complex process involving ROS, notably, H2O2 (Figure 1) [51][46]. ROS are already essential in the initial stages of thyroid hormone production, during iodide oxidation [52][47]. Additionally, thyroid hormones perform a metabolic regulatory function by affecting mitochondrial activity [53][48]. Because of the reliance on ROS in its function, the thyroid is particularly exposed to oxidative damage [54][49]. Therefore, the antioxidant defence system of the thyroid must effectively regulate ROS production and scavenging [26,55][19][50].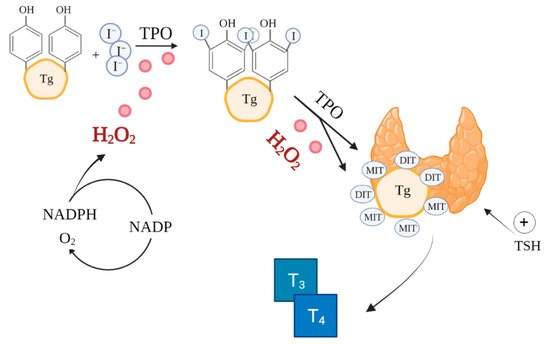
3. Biomarkers of Oxidative Stress in Thyroid Diseases
| Biomarkers | Mechanism of Development, Role | References |
|---|---|---|
| ROS | Energy metabolism in mitochondria | [68][63] |
| MDA, HNE | Lipid peroxidation products | [62][57] |
| AGE, ALE | Protein oxidation products; Advanced peroxidation end products | [64][59] |
| SOD, CAT, GPX, GR | Antioxidant enzymes | [62,68,69][57][63][64] |
| NOX, DUOX | ROS-generating enzymes | [70][65] |
| GSH/GSSG | Reduced/oxygenated glutathione | [ [64]69 ] |
| TAC, TOS | Liczba moli utleniaczy zneutralizowanych przez jeden litr płynu ustrojowego; całkowity stan oksydacyjny;Number of moles of oxidants neutralised by one litre of body fluid; total oxidative status; | [ [66]71[67] , 72 ] |
References
- Jakubczyk, K.; Dec, K.; Kałduńska, J.; Kawczuga, D.; Kochman, J.; Janda, K. Reactive Oxygen Species—Sources, Functions, Oxidative Damage. Pol. Merkur. Lek. Organ Pol. Tow. Lek. 2020, 48, 124–127.
- Tan, B.L.; Norhaizan, M.E.; Liew, W.-P.-P. Nutrients and Oxidative Stress: Friend or Foe? Oxid. Med. Cell. Longev. 2018, 2018.
- Yang, S.; Lian, G. ROS and Diseases: Role in Metabolism and Energy Supply. Mol. Cell. Biochem. 2020, 467, 1–12.
- Shekhova, E. Mitochondrial Reactive Oxygen Species as Major Effectors of Antimicrobial Immunity. PLoS Pathog. 2020, 16, e1008470.
- Yun, H.R.; Jo, Y.H.; Kim, J.; Shin, Y.; Kim, S.S.; Choi, T.G. Roles of Autophagy in Oxidative Stress. Int. J. Mol. Sci. 2020, 21, 3289.
- Sies, H. Oxidative Stress: A Concept in Redox Biology and Medicine. Redox Biol. 2015, 4, 180–183.
- Di Marzo, N.; Chisci, E.; Giovannoni, R. The Role of Hydrogen Peroxide in Redox-Dependent Signaling: Homeostatic and Pathological Responses in Mammalian Cells. Cells 2018, 7, 156.
- Sies, H.; Berndt, C.; Jones, D.P. Oxidative Stress. Annu. Rev. Biochem. 2017, 86, 715–748.
- Filomeni, G.; De Zio, D.; Cecconi, F. Oxidative Stress and Autophagy: The Clash between Damage and Metabolic Needs. Cell Death Differ. 2015, 22, 377–388.
- Gu, Y.; Han, J.; Jiang, C.; Zhang, Y. Biomarkers, Oxidative Stress and Autophagy in Skin Aging. Ageing Res. Rev. 2020, 59, 101036.
- Vostrikova, S.M.; Grinev, A.B.; Gogvadze, V.G. Reactive Oxygen Species and Antioxidants in Carcinogenesis and Tumor Therapy. Biochem. Mosc. 2020, 85, 1254–1266.
- Mahdavi, A.; Naeini, A.A.; Najafi, M.; Maracy, M.; Ghazvini, M.A. Effect of Levetiracetam Drug on Antioxidant and Liver Enzymes in Epileptic Patients: Case-Control Study. Afr. Health Sci. 2020, 20, 984–990.
- Jakubczyk, K.; Kałduńska, J.; Dec, K.; Kawczuga, D.; Janda, K. Antioxidant Properties of Small-Molecule Non-Enzymatic Compounds. Pol. Merkur. Lek. Organ Pol. Tow. Lek. 2020, 48, 128–132.
- Kowalska, K.; Brodowski, J.; Pokorska-Niewiada, K.; Szczuko, M. The Change in the Content of Nutrients in Diets Eliminating Products of Animal Origin in Comparison to a Regular Diet from the Area of Middle-Eastern Europe. Nutrients 2020, 12, 2986.
- Marrocco, I.; Altieri, F.; Peluso, I. Measurement and Clinical Significance of Biomarkers of Oxidative Stress in Humans. Oxid. Med. Cell. Longev. 2017, 2017, 6501046.
- Sies, H.; Jones, D.P. Reactive Oxygen Species (ROS) as Pleiotropic Physiological Signalling Agents. Nat. Rev. Mol. Cell Biol. 2020, 21, 363–383.
- Fukai, T.; Ushio-Fukai, M. Cross-Talk between NADPH Oxidase and Mitochondria: Role in ROS Signaling and Angiogenesis. Cells 2020, 9, 1849.
- Kim, Y.-M.; Kim, S.-J.; Tatsunami, R.; Yamamura, H.; Fukai, T.; Ushio-Fukai, M. ROS-Induced ROS Release Orchestrated by Nox4, Nox2, and Mitochondria in VEGF Signaling and Angiogenesis. Am. J. Physiol. Cell Physiol. 2017, 312, C749–C764.
- Aldosari, S.; Awad, M.; Harrington, E.O.; Sellke, F.W.; Abid, M.R. Subcellular Reactive Oxygen Species (ROS) in Cardiovascular Pathophysiology. Antioxid. Basel Switz. 2018, 7, 14.
- Irazabal, M.V.; Torres, V.E. Reactive Oxygen Species and Redox Signaling in Chronic Kidney Disease. Cells 2020, 9, 1342.
- Garmendia Madariaga, A.; Santos Palacios, S.; Guillén-Grima, F.; Galofré, J.C. The Incidence and Prevalence of Thyroid Dysfunction in Europe: A Meta-Analysis. J. Clin. Endocrinol. Metab. 2014, 99, 923–931.
- Canaris, G.J.; Manowitz, N.R.; Mayor, G.; Ridgway, E.C. The Colorado Thyroid Disease Prevalence Study. Arch. Intern. Med. 2000, 160, 526–534.
- Kasagi, K.; Takahashi, N.; Inoue, G.; Honda, T.; Kawachi, Y.; Izumi, Y. Thyroid Function in Japanese Adults as Assessed by a General Health Checkup System in Relation with Thyroid-Related Antibodies and Other Clinical Parameters. Thyroid 2009, 19, 937–944.
- Empson, M.; Flood, V.; Ma, G.; Eastman, C.J.; Mitchell, P. Prevalence of Thyroid Disease in an Older Australian Population. Intern. Med. J. 2007, 37, 448–455.
- Rostami, R.; Aghasi, M.R.; Mohammadi, A.; Nourooz-Zadeh, J. Enhanced Oxidative Stress in Hashimoto’s Thyroiditis: Inter-Relationships to Biomarkers of Thyroid Function. Clin. Biochem. 2013, 46, 308–312.
- Ameziane El Hassani, R.; Buffet, C.; Leboulleux, S.; Dupuy, C. Oxidative Stress in Thyroid Carcinomas: Biological and Clinical Significance. Endocr. Relat. Cancer 2019, 26, R131–R143.
- Fahim, Y.A.; Sharaf, N.E.; Hasani, I.W.; Ragab, E.A.; Abdelhakim, H.K. Assessment of Thyroid Function and Oxidative Stress State in Foundry Workers Exposed to Lead. J. Health Pollut. 2020, 10, 200903.
- Lassoued, S.; Mseddi, M.; Mnif, F.; Abid, M.; Guermazi, F.; Masmoudi, H.; El Feki, A.; Attia, H. A Comparative Study of the Oxidative Profile in Graves’ Disease, Hashimoto’s Thyroiditis, and Papillary Thyroid Cancer. Biol. Trace Elem. Res. 2010, 138, 107–115.
- Mehran, L.; Amouzegar, A.; Rahimabad, P.K.; Tohidi, M.; Tahmasebinejad, Z.; Azizi, F. Thyroid Function and Metabolic Syndrome: A Population-Based Thyroid Study. Horm. Metab. Res. 2017, 49, 192–200.
- Du, F.-M.; Kuang, H.-Y.; Duan, B.-H.; Liu, D.-N.; Yu, X.-Y. Effects of Thyroid Hormone and Depression on Common Components of Central Obesity. J. Int. Med. Res. 2019, 47, 3040–3049.
- Song, R.-H.; Wang, B.; Yao, Q.-M.; Li, Q.; Jia, X.; Zhang, J.-A. The Impact of Obesity on Thyroid Autoimmunity and Dysfunction: A Systematic Review and Meta-Analysis. Front. Immunol. 2019, 10, 2349.
- Heinonen, S.; Buzkova, J.; Muniandy, M.; Kaksonen, R.; Ollikainen, M.; Ismail, K.; Hakkarainen, A.; Lundbom, J.; Lundbom, N.; Vuolteenaho, K.; et al. Impaired Mitochondrial Biogenesis in Adipose Tissue in Acquired Obesity. Diabetes 2015, 64, 3135–3145.
- Parra, M.D.; Martínez de Morentin, B.E.; Martínez, J.A. Postprandial Insulin Response and Mitochondrial Oxidation in Obese Men Nutritionally Treated to Lose Weight. Eur. J. Clin. Nutr. 2005, 59, 334–340.
- Anderson, E.J.; Lustig, M.E.; Boyle, K.E.; Woodlief, T.L.; Kane, D.A.; Lin, C.-T.; Price, J.W.; Kang, L.; Rabinovitch, P.S.; Szeto, H.H.; et al. Mitochondrial H2O2 Emission and Cellular Redox State Link Excess Fat Intake to Insulin Resistance in Both Rodents and Humans. J. Clin. Investig. 2009, 119, 573–581.
- Saraf-Bank, S.; Ahmadi, A.; Paknahad, Z.; Maracy, M.; Nourian, M. Effects of Curcumin Supplementation on Markers of Inflammation and Oxidative Stress among Healthy Overweight and Obese Girl Adolescents: A Randomized Placebo-Controlled Clinical Trial. Phytother. Res. 2019, 33, 2015–2022.
- Yin, X.; Lanza, I.R.; Swain, J.M.; Sarr, M.G.; Nair, K.S.; Jensen, M.D. Adipocyte Mitochondrial Function Is Reduced in Human Obesity Independent of Fat Cell Size. J. Clin. Endocrinol. Metab. 2014, 99, E209–E216.
- Fischer, B.; Schöttl, T.; Schempp, C.; Fromme, T.; Hauner, H.; Klingenspor, M.; Skurk, T. Inverse Relationship between Body Mass Index and Mitochondrial Oxidative Phosphorylation Capacity in Human Subcutaneous Adipocytes. Am. J. Physiol. Endocrinol. Metab. 2015, 309, E380–E387.
- Christe, M.; Hirzel, E.; Lindinger, A.; Kern, B.; von Flüe, M.; Peterli, R.; Peters, T.; Eberle, A.N.; Lindinger, P.W. Obesity Affects Mitochondrial Citrate Synthase in Human Omental Adipose Tissue. ISRN Obes. 2013, 2013, 826027.
- Schmid, D.; Ricci, C.; Behrens, G.; Leitzmann, M.F. Adiposity and Risk of Thyroid Cancer: A Systematic Review and Meta-Analysis. Obes. Rev. Off. J. Int. Assoc. Study Obes. 2015, 16, 1042–1054.
- Schieber, M.; Chandel, N.S. ROS Function in Redox Signaling and Oxidative Stress. Curr. Biol. CB 2014, 24, R453–R462.
- Sies, H. Hydrogen Peroxide as a Central Redox Signaling Molecule in Physiological Oxidative Stress: Oxidative Eustress. Redox Biol. 2017, 11, 613–619.
- Ameziane-El-Hassani, R.; Schlumberger, M.; Dupuy, C. NADPH Oxidases: New Actors in Thyroid Cancer? Nat. Rev. Endocrinol. 2016, 12, 485–494.
- Cardoso, L.C.; Martins, D.C.; Figueiredo, M.D.; Rosenthal, D.; Vaisman, M.; Violante, A.H.; Carvalho, D.P. Ca2+/Nicotinamide Adenine Dinucleotide Phosphate-Dependent H2O2 Generation Is Inhibited by Iodide in Human Thyroids. J. Clin. Endocrinol. Metab. 2001, 86, 4339–4343.
- Dupuy, C.; Virion, A.; Ohayon, R.; Kaniewski, J.; Dème, D.; Pommier, J. Mechanism of Hydrogen Peroxide Formation Catalyzed by NADPH Oxidase in Thyroid Plasma Membrane. J. Biol. Chem. 1991, 266, 3739–3743.
- Piras, C.; Pibiri, M.; Leoni, V.P.; Balsamo, A.; Tronci, L.; Arisci, N.; Mariotti, S.; Atzori, L. Analysis of Metabolomics Profile in Hypothyroid Patients before and after Thyroid Hormone Replacement. J. Endocrinol. Investig. 2021, 44, 1309–1319.
- Ohye, H.; Sugawara, M. Dual Oxidase, Hydrogen Peroxide and Thyroid Diseases. Exp. Biol. Med. Maywood NJ 2010, 235, 424–433.
- Benvenga, S.; Nordio, M.; Laganà, A.S.; Unfer, V. The Role of Inositol in Thyroid Physiology and in Subclinical Hypothyroidism Management. Front. Endocrinol. 2021, 12, 662582.
- Grasberger, H.; Van Sande, J.; Hag-Dahood Mahameed, A.; Tenenbaum-Rakover, Y.; Refetoff, S. A Familial Thyrotropin (TSH) Receptor Mutation Provides in Vivo Evidence That the Inositol Phosphates/Ca2+ Cascade Mediates TSH Action on Thyroid Hormone Synthesis. J. Clin. Endocrinol. Metab. 2007, 92, 2816–2820.
- Morgante, G.; Musacchio, M.C.; Orvieto, R.; Massaro, M.G.; De Leo, V. Alterations in Thyroid Function among the Different Polycystic Ovary Syndrome Phenotypes. Gynecol. Endocrinol. 2013, 29, 967–969.
- Pace, C.; Tumino, D.; Russo, M.; Le Moli, R.; Naselli, A.; Borzì, G.; Malandrino, P.; Frasca, F. Role of Selenium and Myo-Inositol Supplementation on Autoimmune Thyroiditis Progression. Endocr. J. 2020, 67, 1093–1098.
- Thanas, C.; Ziros, P.G.; Chartoumpekis, D.V.; Renaud, C.O.; Sykiotis, G.P. The Keap1/Nrf2 Signaling Pathway in the Thyroid—2020 Update. Antioxidants 2020, 9, 1082.
- Massart, C.; Hoste, C.; Virion, A.; Ruf, J.; Dumont, J.E.; Van Sande, J. Cell Biology of H2O2 Generation in the Thyroid: Investigation of the Control of Dual Oxidases (DUOX) Activity in Intact Ex Vivo Thyroid Tissue and Cell Lines. Mol. Cell. Endocrinol. 2011, 343, 32–44.
- Venditti, P.; Puca, A.; Di Meo, S. Effects of Thyroid State on H2O2 Production by Rat Heart Mitochondria: Sites of Production with Complex I- and Complex II-Linked Substrates. Horm. Metab. Res. 2003, 35, 55–61.
- Paunkov, A.; Chartoumpekis, D.V.; Ziros, P.G.; Chondrogianni, N.; Kensler, T.W.; Sykiotis, G.P. Impact of Antioxidant Natural Compounds on the Thyroid Gland and Implication of the Keap1/Nrf2 Signaling Pathway. Curr. Pharm. Des. 2019, 25, 1828–1846.
- Poncin, S.; Gérard, A.-C.; Boucquey, M.; Senou, M.; Calderon, P.B.; Knoops, B.; Lengelé, B.; Many, M.-C.; Colin, I.M. Oxidative Stress in the Thyroid Gland: From Harmlessness to Hazard Depending on the Iodine Content. Endocrinology 2008, 149, 424–433.
- Szanto, I.; Pusztaszeri, M.; Mavromati, M. H2O2 Metabolism in Normal Thyroid Cells and in Thyroid Tumorigenesis: Focus on NADPH Oxidases. Antioxidants 2019, 8, 126.
- Eleutherio, E.C.A.; Magalhães, R.S.S.; de Araújo Brasil, A.; Neto, J.R.M.; de Holanda Paranhos, L. SOD1, More than Just an Antioxidant. Arch. Biochem. Biophys. 2021, 697.
- Sepasi Tehrani, H.; Moosavi-Movahedi, A.A. Catalase and Its Mysteries. Prog. Biophys. Mol. Biol. 2018, 140, 5–12.
- Couto, N.; Wood, J.; Barber, J. The Role of Glutathione Reductase and Related Enzymes on Cellular Redox Homoeostasis Network. Free Radic. Biol. Med. 2016, 95, 27–42.
- Metere, A.; Frezzotti, F.; Graves, C.E.; Vergine, M.; De Luca, A.; Pietraforte, D.; Giacomelli, L. A Possible Role for Selenoprotein Glutathione Peroxidase (GPx1) and Thioredoxin Reductases (TrxR1) in Thyroid Cancer: Our Experience in Thyroid Surgery. Cancer Cell Int. 2018, 18, 7.
- Torun, A.N.; Kulaksizoglu, S.; Kulaksizoglu, M.; Pamuk, B.O.; Isbilen, E.; Tutuncu, N.B. Serum Total Antioxidant Status and Lipid Peroxidation Marker Malondialdehyde Levels in Overt and Subclinical Hypothyroidism. Clin. Endocrinol. 2009, 70, 469–474.
- Erdamar, H.; Cimen, B.; Gülcemal, H.; Saraymen, R.; Yerer, B.; Demirci, H. Increased Lipid Peroxidation and Impaired Enzymatic Antioxidant Defense Mechanism in Thyroid Tissue with Multinodular Goiter and Papillary Carcinoma. Clin. Biochem. 2010, 43, 650–654.
- Loomis, S.J.; Chen, Y.; Sacks, D.B.; Christenson, E.S.; Christenson, R.H.; Rebholz, C.M.; Selvin, E. Cross-Sectional Analysis of AGE-CML, SRAGE, and EsRAGE with Diabetes and Cardiometabolic Risk Factors in a Community-Based Cohort. Clin. Chem. 2017, 63, 980–989.
- Ruggeri, R.M.; Giovinazzo, S.; Barbalace, M.C.; Cristani, M.; Alibrandi, A.; Vicchio, T.M.; Giuffrida, G.; Aguennouz, M.H.; Malaguti, M.; Angeloni, C.; et al. Influence of Dietary Habits on Oxidative Stress Markers in Hashimoto’s Thyroiditis. Thyroid Off. J. Am. Thyroid Assoc. 2021, 31, 96–105.
- Kasai, H. Analysis of a Form of Oxidative DNA Damage, 8-Hydroxy-2′-Deoxyguanosine, as a Marker of Cellular Oxidative Stress during Carcinogenesis. Mutat. Res. Mutat. Res. 1997, 387, 147–163.
- Rovcanin, B.R.; Gopcevic, K.R.; Kekic, D.L.; Zivaljevic, V.R.; Diklic, A.D.; Paunovic, I.R. Papillary Thyroid Carcinoma: A Malignant Tumor with Increased Antioxidant Defense Capacity. Tohoku J. Exp. Med. 2016, 240, 101–111.
- Ates, I.; Arikan, M.F.; Altay, M.; Yilmaz, F.M.; Yilmaz, N.; Berker, D.; Guler, S. The Effect of Oxidative Stress on the Progression of Hashimoto’s Thyroiditis. Arch. Physiol. Biochem. 2018, 124, 351–356.
- Bednarek, J.; Wysocki, H.; Sowinski, J. Oxidation Products and Antioxidant Markers in Plasma of Patients with Graves’ Disease and Toxic Multinodular Goiter: Effect of Methimazole Treatment. Free Radic. Res. 2004, 38, 659–664.
- Rostami, R.; Nourooz-Zadeh, S.; Mohammadi, A.; Khalkhali, H.R.; Ferns, G.; Nourooz-Zadeh, J. Serum Selenium Status and Its Interrelationship with Serum Biomarkers of Thyroid Function and Antioxidant Defense in Hashimoto’s Thyroiditis. Antioxidants 2020, 9, 1070.
- Fortunato, R.S.; Braga, W.M.O.; Ortenzi, V.H.; Rodrigues, D.C.; Andrade, B.M.; Miranda-Alves, L.; Rondinelli, E.; Dupuy, C.; Ferreira, A.C.F.; Carvalho, D.P. Sexual Dimorphism of Thyroid Reactive Oxygen Species Production Due to Higher NADPH Oxidase 4 Expression in Female Thyroid Glands. Thyroid Off. J. Am. Thyroid Assoc. 2013, 23, 111–119.
- Faam, B.; Ghadiri, A.A.; Ghaffari, M.A.; Totonchi, M.; Khorsandi, L. Comparing Oxidative Stress Status Among Iranian Males and Females with Malignant and Non-Malignant Thyroid Nodules. Int. J. Endocrinol. Metab. 2021, 19, e105669.
- Ates, I.; Yilmaz, F.M.; Altay, M.; Yilmaz, N.; Berker, D.; Güler, S. The Relationship between Oxidative Stress and Autoimmunity in Hashimoto’s Thyroiditis. Eur. J. Endocrinol. 2015, 173, 791–799.
