Beer is one of the most widely consumed and popular beverages in the world, and is in first place when it comes to the most-consumed alcoholic beverages. The malting and brewing industries are characterized by the generation of large amounts of residues, which account for 85% of their total by-products. In general, these by-products are used as fertilizers, drained into the sewage as waste, or get incinerated. In line with Sustainable Development Goals (SDGs), the food industry is becoming more aware of environmental changes and the negative environmental impacts of their processes and outputs. Sustainable initiatives and enthusiasm for the circular economy have led the brewing industry to reduce its environmental impact through the valorization of its by-products, either by their reduction or their re-incorporation into production processes. These materials are still nutritious, so they can be successfully utilized in various sectors (pharmaceutical, food, biotechnological, etc.).
1. Introduction
Beer is one of the most widely consumed and popular beverages in the world, and is in first place when it comes to the most-consumed alcoholic beverages. The malting and brewing industries are characterized by the generation of large amounts of residues, which account for 85% of their total by-products. In general, these by-products are used as fertilizers, drained into the sewage as waste, or get incinerated
[1]. In line with Sustainable Development Goals (SDGs), the food industry is becoming more aware of environmental changes and the negative environmental impacts of their processes and outputs. Sustainable initiatives and enthusiasm for the circular economy have led the brewing industry to reduce its environmental impact through the valorization of its by-products, either by their reduction or their re-incorporation into production processes. These materials are still nutritious, so they can be successfully utilized in various sectors (pharmaceutical, food, biotechnological, etc.).
Brewer’s spent grain (BSG) is the major solid by-product of the brewing industry, and represents around 85% of total residues
[2]. This lignocellulosic by-product is obtained after the malt saccharification and lautering processes, and consists of the husks that cover the grains, mixed with other parts of the seed (such as part of the pericarp, coat layers, etc.). BSG is generated at a ratio of ≈20 kg of BSG for every 100 L of produced beer
[3]. In general, BSG is commonly considered to be a low-value product, and it is usually mixed with exhausted hops. At present, BSG is widely used in animal feed or as a crop fertilizer, although some studies have also suggested BSG as a potential substrate for biotechnological processes, paper manufacture, or charcoal production
[4][5][4,5]. The composition of BSG is very heterogeneous, and it is greatly affected by the quality of barley, the presence of other cereals, and the production conditions (such as malting or mashing, among others)
[1][6][1,6]. Nonetheless, BSG is mainly characterized by its high contents of cellulose (12–25%), hemicellulose (20–25%) and lignin (12–28%), as well as some readily available nutrients such as sugars (~7.5–13.3%), proteins (15.9–35%) and lipids (6.4–13%)
[5][7][8][9][5,7,8,9]. Due to the high moisture content and nutritional composition, BSG has also been demonstrated to be a medium for the cultivation of microorganisms or as a carrier for immobilizing yeast during fermentation processes
[1][5][1,5]. Recently, different applications in the food industry have also been reported (bakery, confectionary, pasta production, snacks, etc.)
[10]. Apart from that, BSG is also a potentially valuable source of low-molecular-weight phenolic compounds
[5][7][11][5,7,11], among which polyphenols have attracted a large amount of interest due to their antioxidant properties and their applications in food preservation, and the cosmetic and pharmaceutical sectors
[12][13][12,13]. However, the effect of BSG supplementation in protecting food against oxidation is still underexploited.
Total phenolic content of BSG is variable (7–10 mg GAE/g d.s.), and comprises mainly bound phenolic compounds
[7][8][7,8]. Among others, some of the most abundant of these correspond to syringic, ferulic, homovanillic, sinapic and p-cumaric phenolic acids
[5][8][5,8]. For this reason, the recovery of these compounds is a niche interest in research focused on industrial applications.
Another by-product of the brewing industry is brewer’s spent hops (BSH), which is generated at a ratio of ≈0.2–0.4 kg for every 100 L of produced beer. BSH is often used as a fertilizer or for animal feeding, and its composition is characterized by its contents of lipids (~1%), amino acids, and proteins (~40–52%)
[8]. Some authors have reported TPC values in BSH of 10–18 mg GAE/g d.s., and described that their main fraction consists of free polyphenols, such as vanillic, syringic, ferulic and homovanillic acids
[8]. The composition of both BSG and BSH is variable, and depends greatly on the raw materials and the brewing processes applied. Different authors have reported the importance of the extraction methods in the optimization of the recovery of these bioactive compounds from BSG and BSH. The most commonly used techniques include conventional solid–liquid extractions (employing water, organic solvents, enzymatic reactions, etc.)
[7], in which the structural and compositional features of the matrix and the extraction parameters highly influence the extraction process. Among these methods, extraction with acetone or alkaline hydrolysis has been proved to be the most efficient, and is the most commonly used. However, other emerging extraction techniques have recently been described, such as supercritical extractions and assisted high-pressure, microwave or ultrasound extractions
[7][14][15][7,14,15]. Experiments performed with these techniques have also resulted in BSG extracts with high polyphenol content, probably due to the faster heating of the solvent (e.g., in microwave-assisted extractions) or the increase of solvent penetration into the substrate (e.g., in ultrasound-assisted extractions). Nevertheless, in some cases, more research is required to improve and adapt all these extraction mechanisms in order to be able to scale them up.
3. Optimization of Extraction Conditions
3.1.1. Proximate Composition of Extracts
BSG and BSH are two by-products that still contain important amounts of nutritional components (fiber, lipids, protein, minerals, etc.); therefore, many studies have focused on their revalorization in order to integrate them into human food and animal feed
[1].
Protein composition of extracts corresponded to BSG1: 20.83 ± 0.75 mg protein/g w.s., BSG2: 24.56 ± 1.13 mg protein/g w.s., BSG3: 22.83 ± 0.28 mg protein/g w.s., BSG4: 21.98 ± 1.00 mg protein/g w.s., BSG5: 8.51 ± 0.11 mg protein/g w.s., BSH: 16.51 ± 0.71 mg protein/g w.s. Proteins in BSG are reported to be around 20% (d.m.), of which hordeins, glutelines, globulins and albumins are one of the most abundant. Approximately 30% of the protein content corresponds to essential amino acids, of which lysine is the most representative
[16][26]. Spent hops content in protein is also high, at ~23%
[1].
Ash content in samples was between 1.67 and 3.37% (w.s.). The ash content in the industrial BSG samples was very similar (2.11, 2.94, 2.21, 2.68% w.s. for BSG1, BSG2, BSG3 and BSG4, respectively, whereas the artisan BSG5 exhibited the lowest value (1.67% w.s.). Conversely, BSH presented the highest ash content, corresponding to 3.37% (w.m.), similar to the previous literature
[1]. Phosphorous, calcium, magnesium, iron, copper, potassium and manganese are some of the most important minerals of these fractions
[1].
3.1.2. Solvent Selection
In the current study, five different solvents were firstly evaluated in order to optimize the extraction of TPC in one of the samples, the BSG3 sample. The most efficient solvent corresponded to 0.75% NaOH, through which 13.24 ± 1.36 µmol GAE/g w.s. was obtained. In line with this, the potential of alkali hydrolysis to favor the release of phenolic acids from rigid lignocellulose structural components has been reported
[7]. The TPC of extracts with 50% acetone and 50% MeOH solvents presented the second best results (3.171 ± 0.094 and 1.855 ± 0.134 µmol GAE/g w.s., respectively), with no significant difference (
p > 0.05) between them. Acetone:water mixtures has also been reported to be one of the most effective solvents for extracting phenolic compounds from different natural sources
[14][17][14,27], which can probably explain the high efficiency of 50% acetone solvent. Less effective were 50% MeOH + 0.3% HCl and 50% MeOH solvents, neither of which presented differences in TPC recovery (1.45 ± 0.10 and 1.86 ± 0.13 µmol GAE/g w.s., respectively). MilliQ water was the most ineffective solvent for TPC extraction (0.38 ± 0.09 µmol GAE/g w.s.), probably due phenolic compounds often being more soluble in less polar solvents. Therefore, the mixture of water with organic solvents has been widely proposed to facilitate the extraction of these compounds from vegetable sources
[18][28]. Andres et al. (2020)
[19][29], in their study, proposed an efficient extraction methodology for recovering the phenols of BSG by means of using water, but working with longer times (~116–120 min), lower liquid/solid ratios (10–14 mg/mL
v/
w) and at lower temperatures (~30 °C). The results obtained in the current study again suggest that the efficiency of phenolic compound extraction is greatly affected by the type of solvent. Therefore, it would be advisable to carry out a cost-effectiveness study, in order to select the most suitable solvents with respect to both aspects: their recovery efficiency and their related production costs.
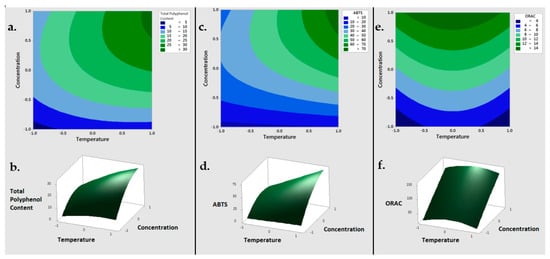
3.1.3. Optimization Results of Extraction Parameters of Concentration and Temperature through RSM
On the basis of the previous results, sodium hydroxide was selected to proceed with the optimization of TPC extraction though the Response Surface Methodology (RSM). Firstly, the extraction process was optimized to maximize the output variables of TPC, ABTS and ORAC in the BSG3 sample, while varying the input parameters of the solvent concentration (0.05, 0.75, 1.45% (
w/v)) and the temperature (4, 42, 80 °C) (Table 1). The best TPC was obtained when working with 1.45% NaOH and at 80 °C (30.05 ± 1.31 µmol GAE/g), suggesting that the extraction of TPC was improved when working at higher solvent concentration.
Table 1. Results obtained in RSM optimization using industrial BSG 3 sample.
| Temperature [°C] |
NaOH
[%] |
TPC
[µmol GAE/g w.s.] |
ABTS
[µmol TE/g w.s.] |
ORAC
[µmol GAE/g w.s.] |
| |
0.05 |
3.98 e ± 0.29 |
4.87 e ± 0.11 |
13.59 f ± 2.15 |
| 4 |
0.75 |
11.29 d ± 0.84 |
24.91 d ± 0.53 |
76.07 d ± 8.00 |
| |
1.45 |
11.66 d ± 0.35 |
31.49 d ± 2.18 |
141.98 b ± 18.85 |
| ± 10.15 |
|
0.05 |
6.32 e ± 0.46 |
| Industrial BSG 4 |
33.34 b,c ± 1.48 |
87.36 a,b | 11.96 e ± 0.84 |
± 5.5247.08 e |
165.40 a,b ± 7.18 ± 1.07 |
| 217.17 | b,c | ± 7.33 |
42 |
0.75 |
20.99 c ± 1.31 |
43.79 c |
| Artisan spent hops (BSH) | ± 3.19 |
103.45 |
24.84 d ± 1.55 | c ± 7.16 |
| 60.03 | d | ± 1.30 |
152.58 b,c ± 8.24 |
- |
|
1.45 |
23.36 b ± 1.09 |
33.83 d ± 1.35 |
115.94 c |
| Artisan BSG5 |
36.52 a,b ± 0.81 | ± 18.38 |
| 90.51 | a | ± 6.41 |
188.61 a ± 12.00 |
188.74 c ± 7.66 |
|
0.05 |
6.24 e ± 1.10 |
7.44 e ± 0.70 |
25.16 f ± 1.08 |
| 80 |
0.75 |
27.17 a ± 0.41 |
51.80 b ± 3.312 |
59.21 d,e ± 0.93 |
| |
1.45 |
30.05 a ± 1.31 |
85.49 a ± 2.96 |
163.73 a ± 3.76 |