In the targeted therapy, nanoparticles (NPs) with specific properties, nanomedicine, are designed to specifically transport therapeutic agents to tumor sites and to release under controlled conditions. This strategy could potentially overcome the limitations of conventional methods and improve the cancer treatment outcomes by distinguishing malignant cells from non-malignant cells and selectively kill malignant cells. Bio-distribution, biocompatibility, biodegradability, and systemic clearance are the general challenges of using NPs in the targeted therapy. An effective NP-based drug delivery system should predict and control the fate of NPs in the biological environment. To develop and achieve a sound and efficient NPs-based system, we need to enhance our understanding of the nano-bio-interaction (NBI) happening between nanomaterials and a complex heterogeneous biological environment. At the cellular level, the NBI occurs at the interface of NPs surface and cell membrane. The interaction behavior of NPs is highly dependent on the physical and chemical properties of NPs.
- nano-bio-interaction
- nanoparticle
- mechanobiological properties
- cancer cells
- cell mechanics
- migratory index
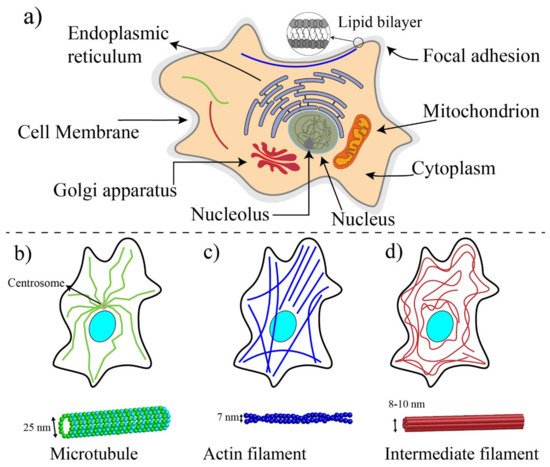
1. Basic Components of Cells and Biomechanics
2. Techniques for Mechanobiological Characterizations
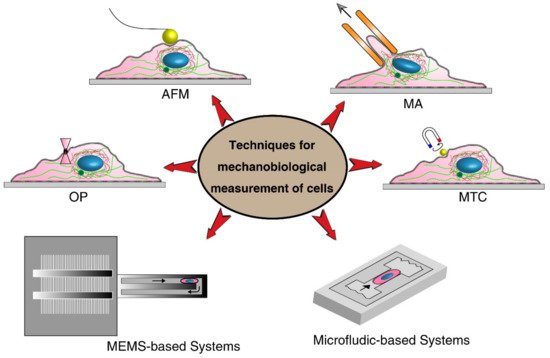
| Techniques | Cell Type | Mechanical Stimuli | Important Parameters | Advantages | Limitations | |
|---|---|---|---|---|---|---|
| Classical Techniques | Atomic Force Microscopy (AFM) | MCF7 [104][28]; Human bladder [96][20] |
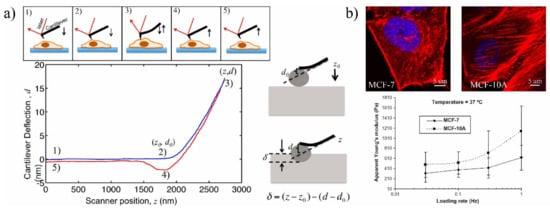
Cantilever micro indention | Tip deflection, Young’s modulus | High-resolution measurement; Provids both structural and mechanical information for local, whole, and interior measurements [23,97][29][21] | Low throughput; Mechanical hitting of AFM tip may affect cell activities and position of probe; Requires a high-resolution microscope |
| Micropipette aspiration (MA) | Human cartilage [98][22]; Colon cancer cells [105][30] |
Negative force | Young’s modulus | Low-cost and well-established method | Limited spatial resolution; Low throughput; For suspended cells only | |
| Magnetic twisting cytometry (MTC) | Melanoma [100][24]; MCF7 [106][31] |
Force is applied by magnetic beads | Stiffness and Young’s modulus | Inducing little heat and photodamages compared to optical tweezer [10][32] | Resolution limitation; Inducing non-uniform stress; Beads are localized randomly on cell; Attachment angle affects the displacement | |
| Optical tweezers (OP) | RBC [99,107][23][33] | Laser-induced surface force | Deformation index | Without physical contact | Only for suspended cells; Damaging consequence of optical heating on cells; Limited magnitude of forces |
|
| Parallel plate | Epithelial ovarian cancer [23][29]; MCF7 [106,108][31][34] |
Shear stress | Aspect ratio | Homogeneity of the applied shear stress; Simplicity; Ability to study cell population | Need bulky devices; Large amount of reagents; Difficult to visualize deformation | |
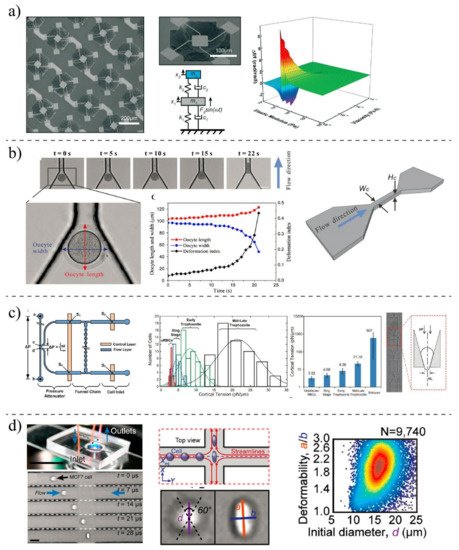
| Microfluidic Techniques | Fluid-induced deformation | PBMCs [102][26] | Fluid shear stress |
Deformation index, size | High throughput; Simultaneously, other chemical assays can be done; The measurment can be done continuously; Contactless deformation; Applicable for both suspended and adhered cells | Needing expensive high-speed camera for imaging |
| Constriction-induced deformation | K562 [109][35]; MDA-MB-231 [110][36] |
Mechanical squeezing | Passage time, entry times, stiffness | Wide-ranging applications in cell deformation; Applicable for different geometry structures; Adjustable dimension for different cell types |
Clogging and channel blockage; Possible effects of friction between cell and channel’s wall on measurements; Ignoring the effects of membrane rigidity and viscosity | |
| Aspiration-induced deformation | Neutrophils [24][37] | Negative pressure | Young’s modulus, cortical tension | Straightforward method; Well-established mathematical model | Leaking problem; Rectangle-like cross-section of microfluidic channels; Time-consuming process; Requiring high-vacuum pressure | |
| Optical stretcher | MCF7 [106][31]; MCF-7, MCF-10, MDA-MB-231 [111][38]; Red blood cells [99][23]; Melanoma cells [112][39] |
Optically-induced surface forces | Deformation index, cell elasticity | No physical contact; Relatively high-throughput measurements | Alignment problem; Optical heating; Thermal damage |
|
| Electrical-induced deformation | MCF-10A, MCF-7 [113][40] | Electroporation-induced swelling | Deformation index, size of cells | Fast heat dissipation; Better resolution; Automation and parallelization of test with reduced amount of samples |
High energy consumption and high voltage | |
| MEMS Techniques | Suspended microcantilever | Circulating tumor cells [114][41]; Fibroblast [101][25] | External actuator | Frequency of cantilever, passage time, transit time | All-inclusive systems; Parallel analysis; Better quality factor; Automation | Fabrication is expensive; Non-transparent channels; High stiffness of silicon; calibration process |
| MEMS resonator | MCF7 [115][42] | External actuator | Frequency of cantilever | High throughput | Expensive fabrication; Requiring external electrical system; Only for adherent cells | |
2.1. Classical Methods
2.2. MEMS- and Microfluidic-Based Techniques
3. Impacts of Nanoparticles on Structural Elements and Morphology of Cells
| Author | Cell Type | NPs Type | Methods | Cytoskeleton Changes |
|---|---|---|---|---|
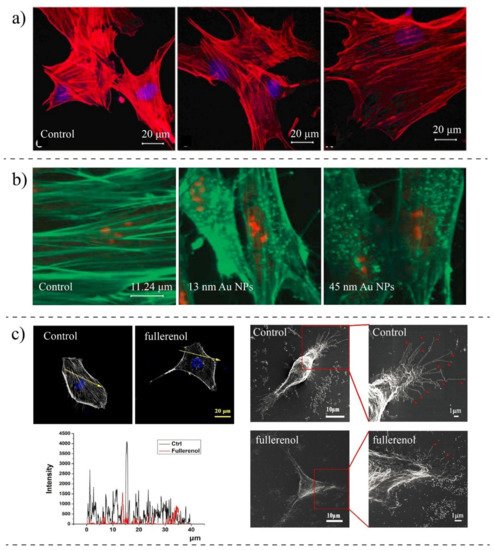
| Pernodet et al., 2007 [162][94] | CF-31 (human dermal fibroblast) | Gold NPs (13 nm) | TEM, Confocal Imaging, Migration Assay | Modification in actin networks; NPs impaired motility and adhesion |
| Pi et al., 2013 [163][95] | MCF-7 (breast cancer) | Selenium NPs | AFM, Confocal Microscopy | The organization of F-actin is changed, and they are aggregated; Actin concentration is reduced |
| Choudhury 2013 [150][82] | A549 (lung cancer) | Citrate-capped Gold NPs (20–60 nm) | Raman, FTIR, TEM, Darkfield Microscopy, UV-Visible Spectroscopy | Inhibiting the polarization of MT; MT structures are damaged, affecting the dynamic equilibrium |
| Qin et al., 2018 [151][83] | MDA-MB-231 (breast cancer) | Fullerenol NPs | SEM, Fluorescence Imaging, AFM, Scratch Assay | The concentration of actin is reduced, the migration speed is reduced, disturbing actin assembly |
| Hot et al., 2012 [145][76] | HeLa (cervical cancer) | Single-wall carbon nanotube (1 ± 0.3 nm) | Fluorescence Imaging Microscopy | NPs cause cells to have shorter F-actin; Traction force is reduced; NPs do not affect G-actin and myosin II |
| Huang et al., 2010 [141][72] | A375 (melanoma) | Silica NPs (MSNs) | TEM, Confocal Microscopy, Western Blot | The actin structure is disorganized and disrupted with NPs; Cell migration is reduced |
| Patra et al., 2007 [159][91] | A549 (lung cancer) | Gold NPs | Confocal Microscopy | The morphology is changed; Treated cells are rounded compared to non-treated |
| Pisanic et al., 2007 [149][80] | PC12M (brain) | Fe2O3 NPs | TEM, Western Blot, Fluorescent Microscopy | Reduction in the formation of actin microfilaments; They are less organized; NPs diminish the ability for differentiation |
| Wu et al., 2012 [161][93] | HAEC (aortic endothelial cells) | Diesel exhaust particles (DEPs) | AFM, Fluorescent Imaging | Cells became degraded; Cellular cytoskeletal structures were impaired |
| Wen et al., 2013 [154][86] | Acting and tubulin proteins (cell-free system) | Silver NPs | TEM, Hyperspectral Imaging, | Inducing changes in the secondary structures; Silver NPs tend to bind actin vs. tubulin |
| Cooper et al., 2015 [147][78] | B35 (neuroblastoma) | Silver NPs | Immunocytochemistry | NPs induce F-actin inclusion, disrupting the actin function |
| Rasel et al., 2015 [157][89] | Osteoblast cells | Boron nitride NPs | AFM, TEM, X-Ray | They do not affect the morphology of cells |
| Liu et al., 2017 [146][77] | HUVEC (Endothelial cells) | Gold NPs-coated with PEG (20 nm) | Fluorescent Microscopy, Traction Force Microscopy | NPs re-arranged actin filaments; Inhibition of Rock activity reduced the polymerization of actin; Reducing the focal adhesion |
| Vieira et al., 2017 [164][96] | CCD1072Sk (Normal cells-skin) | Gold NPs and silver NPs | Immunofluorescence Imaging, Cytofluorometry | NPs impair the F-actin;Cytoskeletal reorganization; Cells lose the cell polarization (without losing their viability) |
| Ali et al., 2017 [158][90] | HSC-3 (tongue cancer) | Gold nanorods coated with PEG and REG | Western Blot, DIC Microscopy, Scratch Assay | The cytoskeletal proteins are rearranged; Cytoskeletal protrusions (filopodia and lamellipoda) are reduced |
| Beaudet et al., 2017 [48][97] | HeLa (cervical cancer) | AuNPs, Swarna Bhasma | Fluorescent Imaging | Larger particles disrupted the microtubules networks |
| Ibrahim et al., 2018 [144][75] | SaOS-2 (bone cancer) | TiO2 spherical NPs | Hyperspectral Imaging, Fluorescent Imaging, Western Blot | The actin and microtubule cytoskeletal networks are disorganized |
| Kralovec et al., 2020 [143][74] | A549 (lung cancer) | Fe3O4@SiO2 | Fluorescent Imaging, Western Blot | Severe disruption of the actin filament and microtubules |
| Kota et al., 2021 [165][98] | VSMCs (vascular smooth muscle cells) | ZIF-8 NPs | AFM, Fluorescent Imaging, Polymerization Assay | Morphological changes and cytoskeletal disorganization were observed; NPs caused changes in actin filaments at basal and apical surfaces. |
References
- Tao, J.; Li, Y.; Vig, D.K.; Sun, S.X. Cell mechanics: A dialogue. Rep. Prog. Phys. 2010, 80, 036601.
- Rodriguez, M.L.; McGarry, P.J.; Sniadecki, N.J. Review on Cell Mechanics: Experimental and Modeling Approaches. Appl. Mech. Rev. 2013, 65, 060801.
- Fletcher, D.A.; Mullins, R.D. Cell mechanics and the cytoskeleton. Nature 2010, 463, 485–492.
- Hohmann, D. The Cytoskeleton—A Complex Interacting Meshwork. Cells 2019, 8, 362.
- Suresh, S. Biomechanics and biophysics of cancer cells. Acta Mater. 2007, 55, 3989–4014.
- Fechheimer, M. Cell and Molecular Biology: Concepts and Experiments. Gerald Karp. Q. Rev. Biol. 2000, 75, 454.
- Lykov, K.; Nematbakhsh, Y.; Shang, M.; Lim, C.T.; Pivkin, I.V. Probing eukaryotic cell mechanics via mesoscopic simulations. PLoS Comput. Biol. 2017, 13, 1–22.
- Dahl, K.N.; Ribeiro, A.J.S.; Lammerding, J. Nuclear shape, mechanics, and mechanotransduction. Circ. Res. 2008, 102, 1307–1318.
- Pullarkat, P.A.; Fernández, P.A.; Ott, A. Rheological properties of the Eukaryotic cell cytoskeleton. Phys. Rep. 2007, 449, 29–53.
- Cotton, F.A.; Wilkinson, G.; Murillo, C.A.; Bochmann, M. Advanced Inorganic Chemistry, 6th ed.; Wiley: Hoboken, NJ, USA, 1999.
- Huang, H.; Dai, C.; Shen, H.; Gu, M.; Wang, Y.; Liu, J.; Chen, L.; Sun, L. Recent Advances on the Model, Measurement Technique, and Application of Single Cell Mechanics. Int. J. Mol. Sci. 2020, 21, 6248.
- Sears, D. Protein structure prediction: Concepts and applications. Biochem. Mol. Biol. Educ. 2007, 35, 79–80.
- Pellegrin, S.; Mellor, H. Actin stress fibers. J. Cell Sci. 2007, 120, 3491–3499.
- Nguyen, A.V.; Trompetto, B.; Tan, X.H.M.; Scott, M.B.; Hu, K.H.-H.; Deeds, E.; Butte, M.J.; Chiou, P.Y.; Rowat, A.C. Differential Contributions of Actin and Myosin to the Physical Phenotypes and Invasion of Pancreatic Cancer Cells. Cell. Mol. Bioeng. 2019, 13, 27–44.
- Ford, L.E. Mechanics of Motor Proteins and the Cytoskeleton (review). Perspect. Biol. Med. 2002, 45, 305–307.
- Surcel, A.; Robinson, D.N. Meddling with myosin’s mechanobiology in cancer. Proc. Natl. Acad. of Sci. USA 2006, 116, 15322–15323.
- Mofrad, M.R.K.; Kamm, R.D. Cytoskeletal mechanics: Models and Measurements; Cambridge University Press: Cambridge, UK, 2006.
- Calzado-Martín, A.; Encinar, M.; Tamayo, J.; Calleja, M.; Paulo, A.S. Effect of Actin Organization on the Stiffness of Living Breast Cancer Cells Revealed by Peak-Force Modulation Atomic Force Microscopy. ACS Nano 2016, 10, 3365–3374.
- Kashani, A.S.; Packirisamy, M. Cancer cells optimize elasticity for efficient migration: Migratory index. R. Soc. Open Sci. 2020, 7, 10.
- Lekka, M.; Laidler, P.; Gil, D.; Lekki, J.; Stachura, Z.; Hrynkiewicz, A.Z. Elasticity of normal and cancerous human bladder cells studied by scanning force microscopy. Eur. Biophys. J. 1999, 28, 312–316.
- Li, M.; Dang, D.; Liu, L.; Xi, N.; Wang, Y. Atomic force microscopy in characterizing cell mechanics for biomedical applications: A review. IEEE Trans. Nanobiosci. 2017, 16, 523–540.
- Trickey, W.R.; Lee, G.M.; Guilak, F. Viscoelastic properties of chondrocytes from normal and osteoarthritic human cartilage. J. Orthop. Res. 2000, 18, 891–898.
- Bellini, N.; Vishnubhatla, K.C.; Bragheri, F.; Ferrara, L.; Minzioni, P.; Ramponi, R.; Cristiani, I.; Osellame, R. Femtosecond laser fabricated monolithic chip for optical trapping and stretching of single cells. Opt. Express 2010, 18, 4679–4688.
- Coughlin, M.F.; Puig-De-Morales, M.; Bursac, P.; Mellema, M.; Millet, E.; Fredberg, J.J. Filamin-A and Rheological Properties of Cultured Melanoma Cells. Biophys. J. 2006, 90, 2199–2205.
- Byun, S.; Son, S.; Amodei, D.; Cermak, N.; Shaw, J.; Kang, J.H.; Hecht, V.C.; Winslow, M.M.; Jacks, T.; Mallick, P.; et al. Characterizing deformability and surface friction of cancer cells. Proc. Natl. Acad. Sci. USA 2013, 110, 7580–7585.
- Gossett, D.R.; Tse, H.T.K.; Lee, S.A.; Ying, Y.; Lindgren, A.G.; Yang, O.O.; Rao, J.; Clark, A.T.; di Carlo, D. Hydrodynamic stretching of single cells for large population mechanical phenotyping. Proc. Natl. Acad. Sci. USA 2012, 109, 7630–7635.
- Rajagopalan, J.; Saif, M.T.A. MEMS sensors and microsystems for cell mechanobiology. J. Micromechanics Microengineering 2011, 21, 054002–054012.
- Li, Q.; Lee, G.; Ong, C.N.; Lim, C. AFM indentation study of breast cancer cells. Biochem. Biophys. Res. Commun. 2008, 374, 609–613.
- Krieg, M.; Flaeschner, G.; Alsteens, D.; Gaub, B.M.; Roos, W.H.; Wuite, G.J.L.; Gaub, H.E.; Gerber, C.; Dufrêne, Y.F.; Müller, D.J. Atomic force microscopy-based mechanobiology. Nat. Rev. Phys. 2018, 1, 41–57.
- Pachenari, M.; Seyedpour, S.; Janmaleki, M.; Shayan, S.B.; Taranejoo, S.; Hosseinkhani, H. Mechanical properties of cancer cytoskeleton depend on actin filaments to microtubules content: Investigating different grades of colon cancer cell lines. J. Biomech. 2014, 47, 373–379.
- Wu, P.-H.; Aroush, D.R.-B.; Asnacios, A.; Chen, W.-C.; Dokukin, M.E.; Doss, B.L.; Durand-Smet, P.; Ekpenyong, A.; Guck, J.; Guz, N.V.; et al. A comparison of methods to assess cell mechanical properties. Nat. Methods 2018, 15, 491–498.
- Price, L.S.L.; Stern, S.T.; Deal, A.M.; Kabanov, A.V.; Zamboni, W.C. A reanalysis of nanoparticle tumor delivery using classical pharmacokinetic metrics. Sci. Adv. 2020, 6, 9249.
- Dao, M.; Lim, C.T.; Suresh, S. Mechanics of the human red blood cell deformed by optical tweezers. J. Mech. Phys. Solids 2003, 51, 2259–2280.
- Kashani, A.S.; Packirisamy, M. Cellular deformation characterization of human breast cancer cells under hydrodynamic forces. AIMS Biophys. 2017, 4, 400–414.
- Lange, J.R.; Steinwachs, J.; Kolb, T.; Lautscham, L.A.; Harder, I.; Whyte, G.; Fabry, B. Microconstriction Arrays for High-Throughput Quantitative Measurements of Cell Mechanical Properties. Biophys. J. 2015, 109, 26–34.
- Mak, M.; Erickson, D. A serial micropipette microfluidic device with applications to cancer cell repeated deformation studies. Integr. Biol. 2013, 5, 1374–1384.
- Guo, Q.; Park, S.; Ma, H. Microfluidic micropipette aspiration for measuring the deformability of single cells. Lab a Chip 2012, 12, 2687–2695.
- Guck, J.; Schinkinger, S.; Lincoln, B.; Wottawah, F.; Ebert, S.; Romeyke, M.; Lenz, D.; Erickson, H.M.; Ananthakrishnan, R.; Mitchell, D.; et al. Optical deformability as an inherent cell marker for testing malignant transformation and metastatic compe-tence. Biophys. J. 2005, 88, 3689–3698.
- Yang, T.; Paie, P.; Nava, G.; Bragheri, F.; Vázquez, R.M.; Minzioni, P.; Veglione, M.; DI Tano, M.; Mondello, C.; Osellame, R.; et al. An integrated optofluidic device for single-cell sorting driven by mechanical properties. Lab. Chip. 2015, 15, 1262–1266.
- Bao, N.; Zhan, Y.; Lu, C. Microfluidic Electroporative Flow Cytometry for Studying Single-Cell Biomechanics. Anal. Chem. 2008, 80, 7714–7719.
- Bagnall, J.S.; Byun, S.; Begum, S.; Miyamoto, D.T.; Hecht, V.C.; Maheswaran, S.; Stott, S.L.; Toner, M.; Hynes, R.O.; Manalis, S.R. Deformability of Tumor Cells versus Blood Cells. Sci. Rep. 2015, 5, 18542.
- Corbin, E.A.; Kong, F.; Lim, C.T.; King, W.P.; Bashir, R. Biophysical properties of human breast cancer cells measured using silicon MEMS resonators and atomic force microscopy. Lab. Chip. 2015, 15, 839–847.
- Puig-De-Morales, M.; Grabulosa, M.; Alcaraz, J.; Mullol, J.; Maksym, G.N.; Fredberg, J.J.; Navajas, D. Measurement of cell microrheology by magnetic twisting cytometry with frequency domain de-modulation. J. Appl. Physiol. 2001, 91, 1152–1159.
- Unal, M.; Alapan, Y.; Jia, H.; Varga, A.G.; Angelino, K.; Aslan, M.; Sayin, I.; Han, C.; Jiang, Y.; Zhang, Z.; et al. Micro and Nano-Scale Technologies for Cell Mechanics. Nanobiomedicine 2014, 1, 5.
- Kim, D.-H.; Wong, P.K.; Park, J.; Levchenko, A.; Sun, Y. Microengineered Platforms for Cell Mechanobiology. Annu. Rev. Biomed. Eng. 2009, 11, 203–233.
- Vigmostad, S.C.; Krog, B.L.; Nauseef, J.T.; Henry, M.; Keshav, V. Alterations in cancer cell mechanical properties after fluid shear stress exposure: A micropipette aspiration study. Cell Heal. Cytoskelet. 2015, 7, 25–35.
- Shojaei-Baghini, E.; Zheng, Y.; Sun, Y. Automated Micropipette Aspiration of Single Cells. Ann. Biomed. Eng. 2013, 41, 1208–1216.
- Rebelo, L.M.; de Sousa, J.S.; Filho, J.M.; Radmacher, M. Comparison of the viscoelastic properties of cells from different kidney cancer phenotypes measured with atomic force microscopy. Nanotechnology 2013, 24, 055102.
- Rodriguez-Ramos, J.; Pabijan, J.; García, R.G.; Lekka, M. The softening of human bladder cancer cells happens at an early stage of the malignancy process. Beilstein J. Nanotechnol. 2014, 5, 447–457.
- Ketene, A.N.; Schmelz, E.M.; Roberts, P.C.; Agah, M. The effects of cancer progression on the viscoelasticity of ovarian cell cytoskeleton structures. Nanomed. Nanotechnol. Biol. Med. 2012, 8, 93–102.
- Omidvar, R.; Tafazzoli-Shadpour, M.; Shokrgozar, M.A.; Rostami, M. Atomic force microscope-based single cell force spectroscopy of breast cancer cell lines: An approach for evaluating cellular invasion. J. Biomech. 2014, 47, 3373–3379.
- Binnig, G.; Quate, C.F.; Gerber, C. Atomic Force Microscope. Phys. Rev. Lett. 1986, 56, 930–933.
- Senapati, S.; Lindsay, S. Recent Progress in Molecular Recognition Imaging Using Atomic Force Microscopy. Accounts Chem. Res. 2016, 49, 503–510.
- Vinckier, A.; Semenza, G. Measuring elasticity of biological materials by atomic force microscopy. FEBS Lett. 1998, 430, 12–16.
- Nguyen, T.D.; Gu, Y. Investigation of Cell-Substrate Adhesion Properties of Living Chondrocyte by Measuring Adhesive Shear Force and Detachment Using AFM and Inverse FEA. Sci. Rep. 2016, 6, 1–13.
- Thomas, G.; Burnham, N.A.; Camesano, T.A.; Wen, Q. Measuring the Mechanical Properties of Living Cells Using Atomic Force Microscopy. J. Vis. Exp. 2013, 76, e50497.
- Rosen, Y.; Gurman, P. MEMS and Microfluidics for Diagnostics Devices. Curr. Pharm. Biotechnol. 2011, 11, 366–375.
- Zheng, Y.; Sun, Y. Microfluidic devices for mechanical characterisation of single cells in suspension. Micro Nano Lett. 2011, 6, 327–331.
- Godin, M.; Bryan, A.K.; Burg, T.P.; Babcock, K.; Manalis, S.R. Measuring the mass, density, and size of particles and cells using a suspended microchannel resonator. Appl. Phys. Lett. 2007, 91, 123121.
- Luo, Z.; Guven, S.; Gözen, I.; Chen, P.; Tasoglu, S.; Anchan, R.M.; Bai, B.; Demirci, U. Deformation of a single mouse oocyte in a constricted microfluidic channel. Microfluid. Nanofluidics 2015, 19, 883–890.
- Guo, Q.; Reiling, S.; Rohrbach, P.; Ma, H. Microfluidic biomechanical assay for red blood cells parasitized by Plasmodium falciparum. Lab. Chip. 2012, 12, 1143–1150.
- Weibel, D.B.; DiLuzio, W.R.; Whitesides, G.M. Microfabrication meets microbiology. Nat. Rev. Genet. 2007, 5, 209–218.
- Shelby, J.P.; White, J.; Ganesan, K.; Rathod, P.K.; Chiu, D.T. A microfluidic model for single-cell capillary obstruction by Plasmodium falciparum-infected erythrocytes. Proc. Natl. Acad. Sci. USA 2003, 100, 14618–14622.
- Valizadeh, A.; Khosroushahi, A.Y. Single-cell analysis based on lab on a chip fluidic system. Anal. Methods 2015, 7, 8524–8533.
- Eluru, G.; Srinivasan, R.; Gorthi, S.S. Deformability Measurement of Single-Cells at High-Throughput with Imaging Flow Cytometry. J. Light. Technol. 2015, 33, 3475–3480.
- Behzadi, S.; Serpooshan, V.; Tao, W.; Hamaly, M.A.; Alkawareek, M.Y.; Dreaden, E.C.; Brown, D.; Alkilany, A.M.; Farokhzad, O.C.; Mahmoudi, M. Cellular uptake of nanoparticles: Journey inside the cell. Chem. Soc. Rev. 2017, 46, 4218–4244.
- Lundqvist, M.; Stigler, J.; Cedervall, T.; Berggård, T.; Flanagan, M.B.; Lynch, I.; Elia, G.; Dawson, K.A. The Evolution of the Protein Corona around Nanoparticles: A Test Study. ACS Nano 2011, 5, 7503–7509.
- Geiser, M.; Rothen-Rutishauser, B.; Kapp, N.; Schürch, S.; Kreyling, W.; Schulz, H.; Semmler, M.; Hof, V.I.; Heyder, J.; Gehr, P. Ultrafine Particles Cross Cellular Membranes by Nonphagocytic Mechanisms in Lungs and in Cultured Cells. Environ. Heal. Perspect. 2005, 113, 1555–1560.
- Wu, Y.-L.; Putcha, N.; Ng, K.W.; Leong, D.T.; Lim, C.T.; Loo, S.C.J.; Chen, X. Biophysical Responses upon the Interaction of Nanomaterials with Cellular Interfaces. Accounts Chem. Res. 2012, 46, 782–791.
- Panariti, A.; Miserocchi, G.; Rivolta, I. The effect of nanoparticle uptake on cellular behavior: Disrupting or enabling functions? Nanotechnol. Sci. Appl. 2012, 5, 87–100.
- Tian, F.; Cui, D.; Schwarz, H.; Estrada, G.G.; Kobayashi, H. Cytotoxicity of single-wall carbon nanotubes on human fibroblasts. Toxicol. Vitr. 2006, 20, 1202–1212.
- Huang, X.; Teng, X.; Chen, D.; Tang, F.; He, J. The effect of the shape of mesoporous silica nanoparticles on cellular uptake and cell function. Biomaterials 2010, 31, 438–448.
- Meng, H.; Yang, S.; Li, Z.; Xia, T.; Chen, J.; Ji, Z.; Zhang, H.; Wang, X.; Lin, S.; Huang, C.; et al. Aspect Ratio Determines the Quantity of Mesoporous Silica Nanoparticle Uptake by a Small GTPase-Dependent Macropinocytosis Mechanism. ACS Nano 2011, 5, 4434–4447.
- Královec, K.; Melounková, L.; Slováková, M.; Mannová, N.; Sedlák, M.; Bartáček, J.; Havelek, R. Disruption of cell adhesion and cytoskeletal networks by thiol-functionalized silica-coated iron oxide nanoparticles. Int. J. Mol. Sci. 2020, 21, 9350.
- Ibrahim, M.; Schoelermann, J.; Mustafa, K.; Cimpan, M.R. TiO2 nanoparticles disrupt cell adhesion and the architecture of cytoskeletal networks of human osteoblast-like cells in a size dependent manner. J. Biomed. Mater. Res. Part A 2018, 106, 2582–2593.
- Holt, B.D.; Shams, H.; Horst, T.A.; Basu, S.; Rape, A.D.; Wang, Y.L.; Rohde, G.K.; Mofrad, M.R.; Islam, M.F.; Dahl, K.N. Altered Cell Mechanics from the Inside: Dispersed Single Wall Carbon Nanotubes Integrate with and Re-structure Actin. J. Funct. Biomater. 2012, 3, 398–417.
- Liu, Y.; Rogel, N.; Harada, K.; Jarett, L.; Maiorana, C.H.; German, G.K.; Mahler, G.J.; Doiron, A.L. Nanoparticle size-specific actin rearrangement and barrier dysfunction of endothelial cells. Nanotoxicology 2017, 11, 846–856.
- Cooper, R.J.; Spitzer, N. Silver nanoparticles at sublethal concentrations disrupt cytoskeleton and neurite dynamics in cultured adult neural stem cells. NeuroToxicology 2015, 48, 231–238.
- Ogneva, I.V.; Buravkov, S.V.; Shubenkov, A.N.; Buravkova, L.B. Mechanical characteristics of mesenchymal stem cells under impact of silica-based nanoparticles. Nanoscale Res. Lett. 2014, 9, 284.
- Pisanic, T.R.; Blackwell, J.D.; Shubayev, V.I.; Fiñones, R.R.; Jin, S. Nanotoxicity of iron oxide nanoparticle internalization in growing neurons. Biomaterials 2007, 28, 2572–2581.
- Mironava, T.; Hadjiargyrou, M.; Simon, M.; Jurukovski, V.; Rafailovich, M.H. Gold nanoparticles cellular toxicity and re-covery: Effect of size, concentration and exposure time. Nanotoxicology 2010, 4, 120–137.
- Choudhury, D.; Xavier, P.L.; Chaudhari, K.; John, R.; Dasgupta, A.K.; Pradeep, T.; Chakrabarti, G. Unprecedented inhibition of tubulin polymerization directed by gold nanoparticles inducing cell cycle arrest and apoptosis. Nanoscale 2013, 5, 4476–4489.
- Qin, Y.; Chen, K.; Gu, W.; Dong, X.; Lei, R.; Chang, Y.; Bai, X.; Xia, S.; Zeng, L.; Zhang, J.; et al. Small size fullerenol nanoparticles suppress lung metastasis of breast cancer cell by disrupting actin dynamics. J. Nanobiotechnol. 2018, 16, 54.
- Issaad, D.; Moustaoui, H.; Medjahed, A.; Lalaoui, L.; Spadavecchia, J.; Bouafia, M.; de la Chapelle, M.L.; Djaker, N. Scattering Correlation Spectroscopy and Raman Spectroscopy of Thiophenol on Gold Nanoparticles: Comparative Study between Nanospheres and Nanourchins. J. Phys. Chem. C 2017, 121, 18254–18262.
- Kashani, A.S.; Badilescu, S.; Piekny, A.; Packirisamy, M. Differing Affinities of Gold Nanostars and Nanospheres toward HeLa and HepG2 Cells: Implications for Cancer Therapy. ACS Appl. Nano Mater. 2020, 3, 4114–4126.
- Wen, Y.; Geitner, N.; Chen, R.; Ding, F.; Chen, P.; Andorfer, R.E.; Govindan, P.N.; Ke, P.C. Binding of cytoskeletal proteins with silver nanoparticles. RSC Adv. 2013, 3, 22002–22007.
- Sinclair, W.E.; Chang, H.H.; Dan, A.; Kenis, P.J.A.; Murphy, C.J.; Leckband, D.E. Gold nanoparticles disrupt actin organization and pulmonary endothelial barriers. Sci. Rep. 2020, 10, 1–12.
- Septiadi, D.; Crippa, F.; Moore, T.L.; Rothen-Rutishauser, B.; Petri-Fink, A. Nanoparticle–Cell Interaction: A Cell Mechanics Perspective. Adv. Mater. 2018, 30, 1–30.
- Rasel, M.A.; Li, T.; Nguyen, T.D.; Singh, S.; Zhou, Y.; Xiao, Y.; Gu, Y. Biophysical response of living cells to boron nitride nanoparticles: Uptake mechanism and biomechanical characterization. J. Nanopart. Res. 2015, 17, 1–13.
- Ali, M.R.K.; Wu, Y.; Tang, Y.; Xiao, H.; Chen, K.; Han, T.; Fang, N.; Wu, R.; El-Sayed, M.A. Targeting cancer cell integrins using gold nanorods in photothermal therapy inhibits migration through affecting cytoskeletal proteins. Proc. Natl. Acad. Sci. USA 2017, 114, E5655–E5663.
- Patra, H.; Banerjee, S.; Chaudhuri, U.; Lahiri, P.; Dasgupta, A.K. Cell selective response to gold nanoparticles. Nanomed. Nanotechnol. Biol. Med. 2007, 3, 111–119.
- Subbiah, R.; Ramasundaram, S.; Du, P.; Hyojin, K.; Sung, D.; Park, K.; Lee, N.E.; Yun, K.; Choi, K.J. Evaluation of cytotoxicity, biophysics and biomechanics of cells treated with functionalized hybrid nano-materials. J. R. Soc. Interface 2013, 10, 20130694.
- Wu, Y.; Yu, T.; Gilbertson, T.A.; Zhou, A.; Xu, H.; Nguyen, K.T. Biophysical Assessment of Single Cell Cytotoxicity: Diesel Exhaust Particle-Treated Human Aortic Endothelial Cells. PLoS ONE 2012, 7, e36885.
- Pernodet, N.; Fang, X.; Sun, Y.; Bakhtina, A.; Ramakrishnan, A.; Sokolov, J.; Ulman, A.; Rafailovich, M. Adverse Effects of Citrate/Gold Nanoparticles on Human Dermal Fibroblasts. Small 2006, 2, 766–773.
- Pi, J.; Yang, F.; Jin, H.; Huang, X.; Liu, R.; Yang, P.; Cai, J. Selenium nanoparticles induced membrane bio-mechanical property changes in MCF-7 cells by disturbing mem-brane molecules and F-actin. Bioorganic Med. Chem. Lett. 2013, 23, 6296–6303.
- Vieira, L.F.d.; Lins, M.P.; Viana, I.M.M.N.; Santos, J.E.d.; Smaniotto, S.; Reis, M.D.d.S. Metallic nanoparticles re-duce the migration of human fibroblasts in vitro. Nanoscale Res. Lett. 2017, 12, 200.
- Beaudet, D.; Badilescu, S.; Kuruvinashetti, K.; Kashani, A.S.; Jaunky, D.; Ouellette, S.; Piekny, A.; Packirisamy, M. Comparative study on cellular entry of incinerated ancient gold particles (Swarna Bhasma) and chemically synthesized gold particles. Sci. Rep. 2017, 7, 10678.
- Kota, D.; Kang, L.; Rickel, A.; Liu, J.; Smith, S.; Hong, Z.; Wang, C. Low doses of zeolitic imidazolate framework-8 nanoparticles alter the actin organization and contractility of vascular smooth muscle cells. J. Hazard. Mater. 2021, 414, 125514.
