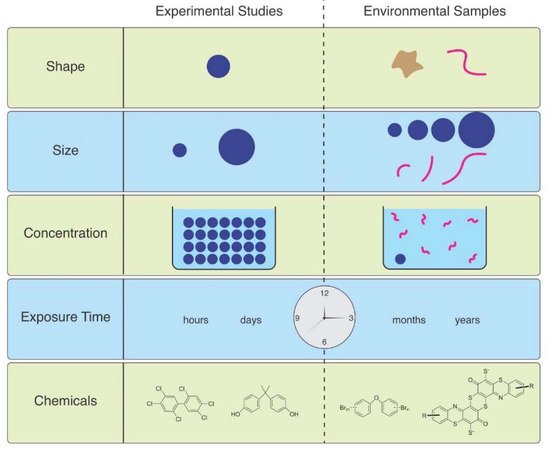
2.1. Aquatic Effects
As Ward et al. (2019) point out, microspheres are available to be purchased with uniform size and shape, with added fluorescence or dyes allowing them to be visualized in tissues [6]. Therefore, they are useful for studies of ingestion and egestion in many species. However useful they are, they are not representative of MPs in the environment, and are therefore not realistic ( Figure 1
). Despite not being so easily obtained, other researchers have found ways to study effects of microfibers, which are the predominant shape of MPs in the water, air, and soil. They can be acquired from ropes, lint in dryers, textile samples, microfilaments used to make synthetic textiles, and other methods.
Figure 1.
The mismatch between MPs used in laboratory studies and those in the environment.
Microfiber (MF) uptake by Asian clams (
Corbicula fluminea
) varied by polymer type and size. The uptake of fibers was greater in those exposed to greater concentrations than lower concentrations, and clams were more likely to take up polyester fibers of smaller size [7]. Horn et al. (2019) used polypropylene rope as a source of microfibers, the concentrations of which were based on concentrations found in the beach from where the experimental subjects, the mole crab, Emerita analoga
, were collected [8]. Adult crabs exposed to these microfibers had increased mortality and impaired embryo development. Larval midges, Chironomus riparius
, were exposed to PET MFs, and effects including survival, time till emergence, growth, head capsule length, and general stress response, were examined in 28-day sediment chronic toxicity tests [9]. They used artificial sediments spiked with MFs at concentrations of 500, 5000, and 50,000 particles/kg sediment dry weight. The lowest concentration was comparable to 500 particles/kg sediment dry weight in Lake Ontario, Canada and 4900 particles/kg sediment dry weight as in the Rhine River. Larvae ingested the microfibers which were later found in the adults. However, no significant effects were seen on time until emergence, head capsule lengths, weight, or HSP 70 (stress response) compared to control organisms. Alnajar et al. (2021) conducted a seven-day exposure of the mussel Mytilus galloprovinciallis
to MFs at 56–180 mg L
−1
(far higher than environmental levels) and observed a reduction in mean clearance rate, abnormality in gills and digestive gland, and an increase in DNA damage [10]. They felt that these effects were due to a combination of the fibrous material itself and chemicals mobilized from the polymers into seawater or the digestive tract, the latter being consistent with an increase in trace elements (e.g., zinc) in the exposure medium with increasing lint concentration. Lobster ( Homarus americanus
) larvae were exposed to 0, 1, 10 and 25 MF mL
−1
[11]. Only the highest concentration decreased early larval survival, and the timing or rate of molting was not altered. All larval and post-larval stages accumulated MFs under the carapace, and ingestion increased with larval stage and with MF concentration; oxygen consumption rates were reduced in later larval stages exposed to high concentrations.
Some studies have compared responses to and effects of fibers vs. spheres. Ward et al. (2019) indicated that mussels reject most of the fibers and spheres they ingest, but a much smaller percentage of spheres were rejected than polyester fibers (ca. 65–260 μm long × 16 μm wide) [6]. The amphipod Hyalella azteca
had slower egestion of fibers than microspheres, but eventually both showed complete egestion [12]. MFs had greater toxicity than microbeads however, possibly because of slower gut passage. In a 28-day feeding experiment, Blarer and Burkhart-Holm (2016) studied effects of fibers and spheres on the feeding rate, assimilation efficiency and change in wet weight of the amphipod Gammarus fossarum
[13]. While both types were ingested and egested, only the fibers impaired the health of the animals. Lower concentrations of MF had more severe effects on the amphipod G. fossarum
than higher concentrations of MP particles. Frydkjær et al. (2017) reported that elevated concentrations of PE particles decreased the mobility of
Daphnia magna
, while irregular shaped fragments (10–75 µm) had greater effects than beads (10–106 µm) [14]. D. magna
egested regular-shaped PE faster than irregular ones, indicating that “spiky” particles are retained longer and have more severe effects. Polyethylene terephthalate granular particles (
p
-PET, approximately 150 μm diam) and fibers (
f
-PET, approximately 3–5 mm L and 20 μm diam) were compared for effects on development of zebrafish (
Danio rerio
) embryos and their joint effects with cadmium [15]. Both types of MP accelerated blood flow and heart rate and inhibited hatching. Both forms decreased the toxicity of Cd. The detoxification effect of f
-PET was greater than that of
p
-PET. Mendrik et al. (2021) found that MP fibers, but not spheres, reduced photosynthesis of algal symbionts of
Acropora
sp. corals, with a 41% decrease in photochemical efficiency after 12 days [16]. Grass shrimp ( Palaemonetes pugio
) were exposed to either sediment, polyethylene spheres, polypropylene fragments, tire fragments, polyester fibers, or clean-water for 96 h at a nominal concentration of 50,000 particles/L before a bacterial challenge with
V. campbellii
[17]. Mortality was not observed in any of the exposures, and survival following the bacterial challenge was similar among shrimp exposed to particle-free water, sediment, polypropylene fragments, polyethylene spheres, tire fragments, and polyester fibers. The grass shrimp cleared most of the ingested particles and all of the ventilated particles within 48 h.