Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 1 by Madhan Jeyaraman and Version 2 by Peter Tang.
Dental-derived MSCs possess similar phenotypes and genotypes like other sources of MSCs along with specific markers such as dentin matrix acidic phosphoprotein (DMP) -1, dentin sialophosphoprotein (DSPP), alkaline phosphatase (ALP), osteopontin (OPN), bone sialoprotein (BSP), and STRO-1. Concerning chondrogenicity, there is literature with marginal use of dental-derived MSCs.
- dental pulp
- mesenchymal stromal cell
- chondrogenicity
1. Introduction
Cartilage is an avascular and aneural structure with poorly cellularized connective tissue [1][2][1,2]. Cartilage tissue facilitates mechanical load transmission with a low frictional coefficient resulting in cartilage injury that has an inherent limited healing potential [3]. The recent idea of “Orthobiologics” led to the exploration of stem cells and regenerative medicine in treating musculoskeletal disorders [4]. Orthobiologics provide administration of osteoinductive and osteoconductive micromolecules to enhance regeneration of degenerated tissues, tendons, bones, and cartilages [4].
Tissue Engineering (TE) is defined as in-vitro or in-vivo regeneration of tissues for repairing and replacing the diseased tissue or organ to enhance and restore the tissue function and maintain tissue homeostasis and improve the biomechanical strength of the tissues [5][6][7][5,6,7]. Cartilage tissue engineering provides a new strategy by transplanting chondrogenic cells along with biocompatible 3D scaffolds and micromolecules to produce engineered cartilage tissue [6][8][6,8]. Chondrogenic cells are derived from mesenchymal stromal cells from various sources, namely, bone marrow [9], adipose tissue [10], placenta [11], amniotic fluid [12], Wharton jelly [13], umbilical cord [14], synovium [15], hair follicles [16], dental pulp [17], and gingiva [18]. The tissue engineering triad comprises mesenchymal stromal cells, scaffolds, and biomolecules such as growth factors and cytokines [8][19][8,19].
Mesenchymal stromal cells (MSCs) form an integral part of regenerative medicine for cartilage regeneration. MSCs are multipotent stem cells with clonigenicity, plasticity, self-renewal, and differentiation [20][21][20,21]. MSC differentiate into trilineage namely osteogenesis, chondrogenesis, and adipogenesis [22][23][24][22,23,24]. Chondrogenesis is mediated by various mediators such as TGF-β1 and -β3; BMP-2, -4, and -7; IGF-1; and GDF-5 [25][26][25,26]. Dental structures provide a variety of stem cells with ease of isolation, non-invasiveness, and availability [17][27][28][17,27,28]. Stem cells of dental origin have similar properties of multipotency, diverse differentiative potential, anti-inflammation, immunomodulation, immune privilege like BM-MSCs, AD-MSCs, and Sy-MSCs [29][30][29,30]. This article throws light on MSCs of dental origin in chondrogenesis and cartilage regeneration in osteoarthritic knees.
2. MSCs of Dental Origin
Stem cells of dental origin form a good therapeutic paradigm in regenerating tissues, bones, and cartilage. MSCs of dental origin include (a) dental pulp MSCs (DP-MSCs) [31][32][31,32], (b) stem cells from human exfoliated deciduous teeth (SHEDs) [33][34][33,34], (c) periodontal ligament stem cells (PDLSCs) [35][36][35,36], (d) dental follicle precursor cells (DFPCs) [37][38][37,38], (e) stem cells from apical papilla (SCAPs) [39][40][39,40], and (f) gingival derived MSCs (G-MSCs) [41][42][41,42]. Among all these sources of dental stem cells, researchers pay attention to DP-MSCs and SHEDs because of ease in their accessibility. The various sources of MSCs of dental origin are shown in Figure 1.
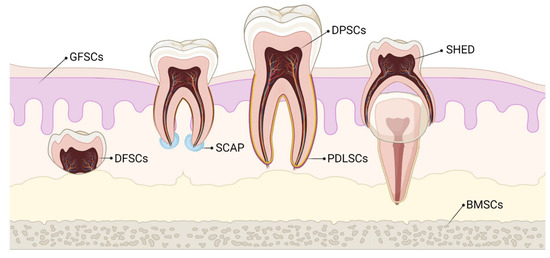
Figure 1. Various sources of stem cells of dental origin. BMSCs–Bone Marrow Stem Cells; DFSCs–Dental Follicle Stem Cells; DPSCs–Dental Pulp Stem Cells; GFSCs–Gingival Fibroblastic Stem Cells; PDLSCs–Periodontal Ligament Stem Cells; SCAP–Stem Cells of Apical Papilla; SHED–Stem cells of Human Exfoliated Deciduous teeth.
3. Characterization of Dental-Derived MSCs (D-MSCs)
DP-MSCs and SHEDs possess similar immunophenotype like BM-MSCs, AD-MSCs, and Sy-MSCs [43][44][45][108,109,110]. DP-MSCs and SHEDs possess cell markers of MSCs (CD-13 [alanyl aminopeptidase], -44, -73 [ecto-5′-nucleotidase], -90, -105 [endoglin], -146, and -166, STRO-1) [46][111], osteogenic markers (BMP-2, OCN, OPN, osteonectin, and COL-1) [47][112], adipogenic markers (LPL and PPAR-γ) [48][113], chondrogenic markers (SOX-9 and COL-2) [45][47][110,112], myogenic markers (myosin, myogenin, and SMA-α) [49][114], neurogenic markers (nestin, GFAP, MAP-2, and β3 tubulin) [50][51][52][115,116,117], and pluripotency markers (OCT-4, SOX-2, Nanog, and IGF-1R) [53][54][118,119]. They demonstrate negative staining for hematopoietic markers (CD-14, -19, -34, -45, and HLA-DR) [44][55][109,120].
The specific markers for D-MSCs are markers of odontoblast differentiation [dental matrix protein-1 (DMP-1) and dentine sialophosphoprotein (DSPP)] [56][121], markers of extracellular matrix [alkaline phosphatase (ALP)] [57][122], makers of osteogenic differentiation [osteopontin (OPN)] [58][123], markers of mineralized tissue differentiation [bone sialoprotein (BSP)] [59][124], and markers of differentiating potential of D-MSCs [STRO-1] [60][125].
Biodentine, a bioactive dentine substitute, is capable of inducing DP-MSCs differentiation of odontoblasts. Luo et al. demonstrated odontoblast differentiation of DP-MSCs by increased expression of ALP, OCN, DSPP, DMP1, and BSP [61][126]. Optimal mechanical compression increased the expression of DSPP, BMP-7, and Wnt10a genes for odontoblast differentiation by DP-MSCs [62][127]. BBX gene expression induces the differentiation of odontoblasts by DP-MSCs [63][128]. DNA methylation and PTEN expression were increased in DP-MSCs, which are responsible for lineage differentiation and reduced oncogenesis when compared with BM-MSCs [64][129].
4. Harvesting and Delivery Methods of D-MSCs
Various regenerative medicine experts followed different methods to extract and harvest stromal cells from dental pulp.
Raoof et al. used three different methods to isolate DP-MSCs, namely, (a) digestion of dental pulp tissue with collagenase and placement of isolated trypsinized cells in petri dishes, (b) explantation of undigested dental pulp pieces to culture plates, and (c) explantation of trypsinized dental pulp tissues to petri dishes for outgrowth [65][130]. These tissues are plated to MEM medium supplemented with 20% fetal bovine serum at 37 °C with a 5% CO2 incubator. A total of 60% cellular confluency was achieved within days of culture and checked for pluripotency markers by RT/PCR analysis [65][130].
Naz et al. expanded DP-MSCs and SHEDs via the explant culture method after extirpation of dental pulp tissues from deciduous teeth [66][131]. As a result of culture expansion, MSCs exhibit fibroblast-like cells with long cytoplasmic processes. DP-MSCs and SHEDs characterization was done and cryopreserved for future use as shown in Figure 2.
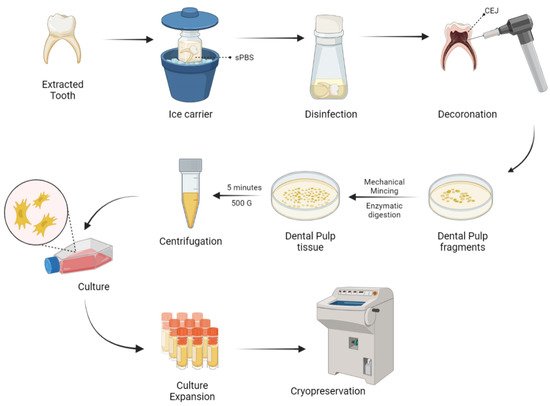

Figure 2. Harvesting and delivery method of DP-MSCs. sPBS—sterile phosphate buffer saline; CEJ—cementum enamel junction.
No significant change was observed in the differentiating capabilities and immunophenotypic properties of cryopreserved and non-cryopreserved DP-MSCs isolated from dental pulp, but there were significant differences in the morphology and proliferative potential of cryopreserved DP-MSCs than non-cryopreserved DP-MSCs [67][132].
The survival rates of DP-MSCs in DMSO free medium by static magnetic cryopreservation increased by 2 to 2.5 fold when the cells were exposed to 0.4 or 0.8-T static magnetic fields [68][133]. Gioventù et al. demonstrated that cryopreserved teeth by laser piercing have maintained cellular viability [69][134].
To isolate a pure population of DP-MSCs, the identification of cell surface markers like LNGFR and THY-1 are significant [70][71][87,88]. The assessment of the number of colonies showed that LNGFRLow+THY-1High+ cells in the dental pulp have a significantly higher colony-forming potential than LNGFR+THY-1+ cells in the bone marrow [72][73][74][135,136,137].
5. Chondrogenicity of Dental-Derived MSCs
Though dental-derived stem cells possess higher osteogenic potential, they are being explored very marginally for chondrogenicity. DP-MSCs act as a promising source for cartilage tissue engineering and regeneration. DP-MSCs possess a strong potential to differentiate into hyaline and fibrocartilage [75][138]. Sophia et al. demonstrated that hyaline cartilage contains few chondrocytes in their extracellular matrix rich in GAGs and type 2 collagen [2], whereas Allen et al. stated that fibrocartilage contains fibroblastic cells with small amounts of GAGs and type 1 collagen [76][139].
Longoni et al. expanded DP-MSCs from seven molar teeth and induced chondrogenesis in a 3D pellet culture system [77][63]. These culture-expanded DP-MSCs display GAGs, aggrecan, and type 2 collagen after three weeks. The assessment of culture-expanded cells revealed fibroblastic cells with long cytoplasmic processes with a predominance of type 1 collagen to state the formation of fibrocartilage. They concluded that DP-MSCs regenerate fibrocartilage in joints, rather than hyaline cartilage.
DP-MSCs provide a rapid ex-vivo expansion and chondrogenic differentiation potential and hence provide a favorable cell type for treating cartilage disorders. Khajeh et al. demonstrated a significant role of hypoxia mimicking agent and cobalt chloride on chondrogenesis with DP-MSCs [78][140]. Cobalt chloride exposure to DP-MSCs increases the cellular pellet mass in culture, cellular morphology and integrity, ECM deposition, and cellular organizations. There were elevated levels of GAGs and type 2 collagen expression [79][141]. Cobalt chloride enhances the stemness of DP-MSCs where flow cytometry reveals the increased expression of STRO-1+ cells [80][142].
SHEDs lose stemness and compromise the therapeutic effects when cultured and expanded in vitro for the long term. Hypoxia is a major factor in the maintenance of stemness of MSCs [81][82][143,144]. SHEDs, when co-cultured with cobalt chloride, increased the hypoxia-inducible factor-1α in a dose-dependent manner, resulted in increased expression of STRO-1+ cells and stem/stromal cell markers such as OCT4, NANOG, SOX2, and c-Myc and decreased osteogenic differentiation by reducing ALP levels [79][141]. Hypoxia suppresses chondrogenic hypertrophy in agarose or alginate-chondroitin sulfate-platelet lysate hydrogel and 3D pellet culture system in cartilage tissue engineering [83][84][145,146]. At the protein level, the inhibitors of chondrocyte hypertrophy are PTHrP, TGF-β3, BMP-4, -7, and -13, GG86/2, Dorsomorphin, and FK506, whereas, at the gene level, Nkx3.2, SOX-9, Smad6, HDAC4, ChM1, sFlt-1, and C-1-1 are responsible for inhibiting chondrocyte hypertrophy during culture [85][86][87][88][89][147,148,149,150,151]. The chondrogenesis due to hypoxia is mediated through SOX-9 gene transcription or p38 MAPK gene activation [90][152]. Hypoxia promotes chondrogenic differentiation and cartilage extracellular matrix synthesis and suppresses terminal chondrocyte differentiation and hence the hypoxia phenomenon preserves chondrocyte phenotype and function during chondrogenesis [91][153].
Hsu et al. cultured human gingival fibroblast (HGF) cells on chitosan membrane to observe in-vitro chondrogenesis. On culture, increased spheroid formation resulted, which indicates the stemness of HGF. Spheroid formation by HGF was supported by Rho/Rho-associated kinases and the connexion 43 pathway. Hence, they concluded that culturing HGF on chitosan membrane induces spheroid formation, which further induces chondrogenesis by the ROCK pathway [92][154].
Ferre et al. demonstrated osteogenesis and chondrogenesis by human gingival stem cells in vitro in 3D floating micromass pellet cultures in a specified medium. Osteogenic cells exhibited the increased expression of Runx-2, ALP, presence of osteoid-like mass, and osterix expression whereas chondrogenic cells exhibited increased expression of type 2 collagen, GAGs, and SOX-9 transcription gene [93][155].
SOX-9, the master gene for chondrogenesis, helps in the proliferation of chondrocytes, but not in chondrocyte hypertrophy [94][156]. SOX-9 gene was expressed in human gingival stem cells at the basal level without chondrogenic stimulation [95][157]. Such basal expression of SOX-9 explains efficient chondrogenesis. SOX-9 gene knock-out mice were unable to regenerate normal cartilage despite MSC condensation [96][97][98][99][158,159,160,161]. The chondrogenesis by SOX-9 is due to
- (1) chondrocyte expression of SOX-9 until growth plate hypertrophy and in articular cartilage throughout life in adults,
- (2) to secure lineage specificity towards chondrogenesis in fetal and postnatal growth plates,
- (3) to maintain adult cartilage homeostasis,
- (4) and to repress non-chondrogenic lineages in gene level [50][51][52][115,116,117].
Ferre et al. demonstrated the differentiation of type 10 collagen secreting hypertrophic chondrocytes and fibroblast-like synoviocytes by human gingival stem cells. Under hypoxia and hypoxic mimicking environment, G-MSCs express high levels of VEGF-α, which promote vasculogenesis for regenerative therapies [93][155].
NOTCH ligand signaling plays a major role in the chondrogenic differentiation of cells [100][162]. NOTCH-2 modulates the activities of NOTCH-3 and -1, hence influence the growth and development, and homeostasis of chondrocytes and articular cartilage [101][102][103][163,164,165]. NOTCH-3 represses the proliferation of terminally differentiated chondrocytes within the cartilaginous tissues [104][166]. In a 3D-cultured chondrogenesis, there is a downregulation of NOTCH ligands and receptors [105][106][167,168]. While MSC undergoes terminal chondrocyte differentiation, NOTCH-3 receptors were upregulated and were highly expressed [100][107][162,169].
