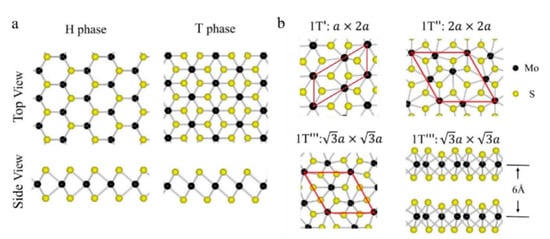
MoS2 is one of the transition metal dichalcogenides (TMDs) that has gained a high reputation in recent years due to its distinct chemical, electronic, mechanical, magnetic, and optical properties. Its unique properties enabled its use in different applications such as sensing applications, high-efficiency field effect transistors, and energy and medical (curing) applications. MoS2 exists in different crystalline structures, such as hexagonal (H), tetrahedral (T), or rhombohedral (R). It naturally exists as 2H MoS2, and its most popular structures are the semiconducting 2H and 3R phases and the 1T metallic phase, where 2H is more stable but less conductive than 1T. Metallic MoS2 has a higher conductivity (105 times) than semiconducting 2H MoS2 and high catalytic activity.
- molybdenum disulfide
- lithium-ion batteries
- sodium-ion batteries
- MoS2 energy applications
- solar cells
- hydrogen evolution reactions (HER)
- 二硫化钼
- 二硫化钼能源应用
- 太阳能电池
- 析氢反应 (HER)
- 金属二硫化钼
- 1T 二硫化钼
1. Structure and Properties
2. Energy Applications
2.1. Energy Storage Applications
2.1.1. Lithium-Ion Batteries (LIB)
| Battery Type | MoS2 Phase | Structure | Capacity | References |
|---|---|---|---|---|
| Lithium-ion | 1T (Metallic) | Nanotube-like MoS2 over graphene | Discharge capacity = 666 mA h g−1 at current density = 3500 mA g−1 |
[21] |
| Lithium-ion | 1T (Metallic) | MoS2 over carbon cloth | Reversible specific capacity = 1789 mA h g−1 at 0.1 Ag−1 Retained capacity = 853 mA h g−1 after 140 cycles at 1 Ag−1 |
[22] |
| Lithium-ion | 1T (Metallic) | 1T MoS2 + (NiMoO4) | Charged mass capacity = 940.1 mA h g−1 Discharged mass capacity = 941.6 mA h g−1 |
[23] |
| Lithium-ion | 1T (Metallic) | Pure MoS2 | Specific capacity ≈ 935 mA h g−1 for 200 cycles at 5 A g−1 can be increased to 1150 mA h g−1 |
[20] |
| Sodium-ion | 1T (Metallic) | MoS2-graphene-MoS2 | Capacity of 175 mA h g−1 at a high current density of 2 A g−1 Reverse capacity of ≈313 mA h g−1 at low current density of 50 mA g−1. Stabilizes at current density = 313 mA h g−1 after 200 cycles |
[14] |
| Sodium-ion | 2H and 1T MoS2 | Dual phase of 2H and 1T MoS2 | Capacity = 300 mA h g−1 after 200 cycles, and coulombic efficiency = 99% |
[24] |
| Sodium-ion | 2H phase transfers to 1T through chemical reactions | MoS2 and amorphous carbon (C) | Capacity = 563.5 mA h g−1 at 0.2 A g−1 Coulombic efficiency = 86.6% Cyclic stability = 484.9 mA h g−1 at 2 A g−1 |
[25] |
| Supercapacitor | 2D MoS2 | Spraying MoS2 nanosheets on Si/SiO2 | Area capacitance = 8 mF cm−2, and volumetric capacitance = 178 F cm−3 |
[26] |
| Supercapacitor | Nanoflower-like MoS2 structure | 3D-graphene/MoS2 nanohybrid | Dimensions 23.6 × 22.4 × 0.6 mm3 Specific capacitance (Csp) = 58 F g−1, energy density of 24.59 W h Kg−1, and power density of 8.8 W Kg−1 with operating window of 2.7 V (−1.5 to +1.2 V) |
[27] |
| Supercapacitor | Brush-like arrangement MoS2 | MoS2 nanowires over Ni foam | The high mass loading of MoS2 (30 mg cm−2) retains 92% of maximum capacitance after 9000 charge–discharge cycles at 5 A g−1 | [28] |
| Supercapacitor | MoS2 QSs | Exfoliated MoS2 QSs lateral size (5–10 nm) | Capacitance = 162 F g−1 Energy density = 14.4 W h kg−1 |
[29] |
| Hybrid Supercapacitor |
N-3DG and 3D-IEMoS2@G |
Prepared using solvothermal process | Energy density = 140 W h kg−1 at 630 W kg−1, and 43 W h kg−1 at power density of 103 kW kg−1 Lifecycle over 10,000 |
[30] |
2.1.2. Sodium-Ion Batteries (NIB)
2.1.3. Supercapacitors
2.2. Energy Generation Applications
2.2.1. Hydrogen Evolution Reactions (HER)
| Type of Reaction | Catalyst Used | Specification | References |
|---|---|---|---|
| HER | (MoS2/CoSe2) | Tafel slope = 36 mV dec−1 Onset potential = −11 mV Exchange current density = 7.3 × 10−2 mA cm−2 |
[44] |
| HER | 1T MoS2 | Overpotential = 156 mV, at 10 mA cm−2 Tafel slope = 42.7 mV dec−1 |
[45] |
| HER/OER | Amorphous Ni–Co complexes hybridized with 1T MoS2 | Overpotentials = 70 mV HER and 235 mV for OER at 10 mA cm−2 Tafel slope = 38.1 to 45.7 mV dec−1 |
[46] |
| OER | Rhombohedral MoS2 microspheres over conductive Ni | Overpotential ≈ 310 mV Tafel slope ≈ 105 mV dec−1 |
[47] |
| OER | MoS2 quantum dots (MSQDs) | Overpotential = 280 mV Tafel slope = 39 mV dec−1 |
[48] |
| CO2 reduction | Vertically aligned MoS2 nanoflakes (2H and 1T phases coexist) |
Overpotential = 54 mV Reduction current density = 130 mA cm−2 at −0.764 V |
[49] |
| CO2 reduction | p–n junction Bi2S3/MoS2 composite |
Photocatalytic CO2 reduction 20 times higher than single catalysts under visible light irradiation |
[50] |
| CO2 reduction | 3R MoS2 nanoflower powder | Synthesized using CVD CO production < 0.01 μmol-gcat−1 hr−1 at 25 °C which is negligible |
[51] |
2.2.2. Oxygen Evolution Reactions (OER)
References
- Dai, Z.; Jin, W.; Grady, M.; Sadowski, J.T.; Dadap, J.I.; Osgood, R.M.; Pohl, K. Surface Structure of Bulk 2H-MoS2(0001) and Exfoliated Suspended Monolayer MoS2: A Selected Area Low Energy Electron Diffraction Study. Surf. Sci. 2017, 660, 16–21.
- Li, X.; Zhu, H. Two-Dimensional MoS2: Properties, Preparation, and Applications. J. Mater. 2015, 1, 33–44.
- Toh, R.J.; Sofer, Z.; Luxa, J.; Sedmidubský, D.; Pumera, M. 3R Phase of MoS2 and WS2 Outperforms the Corresponding 2H Phase for Hydrogen Evolution. Chem. Commun. 2017, 53, 3054–3057.
- Terrones, H.; López-Urías, F.; Terrones, M. Novel Hetero-Layered Materials with Tunable Direct Band Gaps by Sandwiching Different Metal Disulfides and Diselenides. Sci. Rep. 2013, 3, 1549.
- Kadantsev, E.S.; Hawrylak, P. Electronic Structure of a Single MoS2 Monolayer. Solid State Commun. 2012, 152, 909–913.
- Lin, Y.-C.; Dumcenco, D.O.; Huang, Y.-S.; Suenaga, K. Atomic Mechanism of the Semiconducting-to-Metallic Phase Transition in Single-Layered MoS2. Nat. Nanotechnol. 2014, 9, 391–396.
- Kappera, R.; Voiry, D.; Yalcin, S.E.; Branch, B.; Gupta, G.; Mohite, A.D.; Chhowalla, M. Phase-Engineered Low-Resistance Contacts for Ultrathin MoS2 Transistors. Nat. Mater. 2014, 13, 1128–1134.
- Hu, L.; Shan, X.; Wu, Y.; Zhao, J.; Lu, X. Laser Thinning and Patterning of MoS2 with Layer-by-Layer Precision. Sci. Rep. 2017, 7, 15538.
- Cho, S.; Kim, S.; Kim, J.H.; Zhao, J.; Seok, J.; Keum, D.H.; Baik, J.; Choe, D.-H.; Chang, K.J.; Suenaga, K.; et al. Phase Patterning for Ohmic Homojunction Contact in MoTe2. Science 2015, 349, 625–628.
- Sun, K.; Liu, Y.; Pan, Y.; Zhu, H.; Zhao, J.; Zeng, L.; Liu, Z.; Liu, C. Targeted Bottom-up Synthesis of 1T-Phase MoS2 Arrays with High Electrocatalytic Hydrogen Evolution Activity by Simultaneous Structure and Morphology Engineering. Nano Res. 2018, 11, 4368–4379.
- Yang, S.; Zhang, K.; Wang, C.; Zhang, Y.; Chen, S.; Wu, C.; Vasileff, A.; Qiao, S.-Z.; Song, L. Hierarchical 1T-MoS2 Nanotubular Structures for Enhanced Supercapacitive Performance. J. Mater. Chem. A 2017, 5, 23704–23711.
- Geng, X.; Zhang, Y.; Han, Y.; Li, J.; Yang, L.; Benamara, M.; Chen, L.; Zhu, H. Two-Dimensional Water-Coupled Metallic MoS2 with Nanochannels for Ultrafast Supercapacitors. Nano Lett. 2017, 17, 1825–1832.
- Li, P.; Yang, Y.; Gong, S.; Lv, F.; Wang, W.; Li, Y.; Luo, M.; Xing, Y.; Wang, Q.; Guo, S. Co-Doped 1T-MoS2 Nanosheets Embedded in N, S-Doped Carbon Nanobowls for High-Rate and Ultra-Stable Sodium-Ion Batteries. Nano Res. 2019, 12, 2218–2223.
- Geng, X.; Jiao, Y.; Han, Y.; Mukhopadhyay, A.; Yang, L.; Zhu, H. Freestanding Metallic 1T MoS2 with Dual Ion Diffusion Paths as High Rate Anode for Sodium-Ion Batteries. Adv. Funct. Mater. 2017, 27, 1702998.
- Tang, W.; Wang, X.; Xie, D.; Xia, X.; Gu, C.; Tu, J. Hollow Metallic 1T MoS2 Arrays Grown on Carbon Cloth: A Freestanding Electrode for Sodium Ion Batteries. J. Mater. Chem. A 2018, 6, 18318–18324.
- Shang, C.; Fang, Y.Q.; Zhang, Q.; Wang, N.Z.; Wang, Y.F.; Liu, Z.; Lei, B.; Meng, F.B.; Ma, L.K.; Wu, T.; et al. Superconductivity in the Metastable 1 T’ and 1 T’’’ Phases of MoS2 Crystals. Phys. Rev. B 2018, 98, 184513.
- Qian, X.; Liu, J.; Fu, L.; Li, J. Quantum Spin Hall Effect in Two-Dimensional Transition Metal Dichalcogenides. Science 2014, 346, 1344–1347.
- Kappera, R.; Voiry, D.; Yalcin, S.E.; Jen, W.; Acerce, M.; Torrel, S.; Branch, B.; Lei, S.; Chen, W.; Najmaei, S.; et al. Metallic 1T Phase Source/Drain Electrodes for Field Effect Transistors from Chemical Vapor Deposited MoS2. APL Mater. 2014, 2, 092516.
- Saha, D.; Kruse, P. Editors’ Choice—Review—Conductive Forms of MoS2 and Their Applications in Energy Storage and Conversion. J. Electrochem. Soc. 2020, 167, 126517.
- Jiao, Y.; Mukhopadhyay, A.; Ma, Y.; Yang, L.; Hafez, A.M.; Zhu, H. Ion Transport Nanotube Assembled with Vertically Aligned Metallic MoS2 for High Rate Lithium-Ion Batteries. Adv. Energy Mater. 2018, 8, 1702779.
- Xiang, T.; Fang, Q.; Xie, H.; Wu, C.; Wang, C.; Zhou, Y.; Liu, D.; Chen, S.; Khalil, A.; Tao, S.; et al. Vertical 1T-MoS2 Nanosheets with Expanded Interlayer Spacing Edged on a Graphene Frame for High Rate Lithium-Ion Batteries. Nanoscale 2017, 9, 6975–6983.
- Wu, M.; Zhan, J.; Wu, K.; Li, Z.; Wang, L.; Geng, B.; Wang, L.; Pan, D. Metallic 1T MoS2 Nanosheet Arrays Vertically Grown on Activated Carbon Fiber Cloth for Enhanced Li-Ion Storage Performance. J. Mater. Chem. A 2017, 5, 14061–14069.
- Li, Z.; Zhan, X.; Zhu, W.; Qi, S.; Braun, P.V. Carbon-Free, High-Capacity and Long Cycle Life 1D–2D NiMoO4 Nanowires/Metallic 1T MoS2 Composite Lithium-Ion Battery Anodes. ACS Appl. Mater. Interfaces 2019, 11, 44593–44600.
- Wu, J.; Liu, J.; Cui, J.; Yao, S.; Ihsan-Ul-Haq, M.; Mubarak, N.; Quattrocchi, E.; Ciucci, F.; Kim, J.-K. Dual-Phase MoS2 as a High-Performance Sodium-Ion Battery Anode. J. Mater. Chem. A 2020, 8, 2114–2122.
- Pan, Q.; Zhang, Q.; Zheng, F.; Liu, Y.; Li, Y.; Ou, X.; Xiong, X.; Yang, C.; Liu, M. Construction of MoS2/C Hierarchical Tubular Heterostructures for High-Performance Sodium Ion Batteries. ACS Nano 2018, 12, 12578–12586.
- Cao, L.; Yang, S.; Gao, W.; Liu, Z.; Gong, Y.; Ma, L.; Shi, G.; Lei, S.; Zhang, Y.; Zhang, S.; et al. Direct Laser-Patterned Micro-Supercapacitors from Paintable MoS2 Films. Small 2013, 9, 2905–2910.
- Singh, K.; Kumar, S.; Agarwal, K.; Soni, K.; Ramana Gedela, V.; Ghosh, K. Three-Dimensional Graphene with MoS2 Nanohybrid as Potential Energy Storage/Transfer Device. Sci. Rep. 2017, 7, 9458.
- Manuraj, M.; Kavya Nair, K.V.; Unni, K.N.N.; Rakhi, R.B. High Performance Supercapacitors Based on MoS2 Nanostructures with near Commercial Mass Loading. J. Alloy. Compd. 2020, 819, 152963.
- Nardekar, S.S.; Krishnamoorthy, K.; Pazhamalai, P.; Sahoo, S.; Mariappan, V.K.; Kim, S.-J. Exceptional Interfacial Electrochemistry of Few-Layered 2D MoS2 Quantum Sheets for High Performance Flexible Solid-State Supercapacitors. J. Mater. Chem. A 2020, 8, 13121–13131.
- Zhan, C.; Liu, W.; Hu, M.; Liang, Q.; Yu, X.; Shen, Y.; Lv, R.; Kang, F.; Huang, Z.-H. High-Performance Sodium-Ion Hybrid Capacitors Based on an Interlayer-Expanded MoS2/RGO Composite: Surpassing the Performance of Lithium-Ion Capacitors in a Uniform System. NPG Asia Mater. 2018, 10, 775–787.
- Su, J.; Pei, Y.; Yang, Z.; Wang, X. Ab Initio Study of Graphene-like Monolayer Molybdenum Disulfide as a Promising Anode Material for Rechargeable Sodium Ion Batteries. RSC Adv. 2014, 4, 43183–43188.
- Kühne, M.; Börrnert, F.; Fecher, S.; Ghorbani-Asl, M.; Biskupek, J.; Samuelis, D.; Krasheninnikov, A.V.; Kaiser, U.; Smet, J.H. Reversible Superdense Ordering of Lithium between Two Graphene Sheets. Nature 2018, 564, 234–239.
- Chepkasov, I.V.; Ghorbani-Asl, M.; Popov, Z.I.; Smet, J.H.; Krasheninnikov, A.V. Alkali Metals inside Bi-Layer Graphene and MoS2: Insights from First-Principles Calculations. Nano Energy 2020, 75, 104927.
- Acerce, M.; Voiry, D.; Chhowalla, M. Metallic 1T Phase MoS2 Nanosheets as Supercapacitor Electrode Materials. Nat. Nanotechnol. 2015, 10, 313–318.
- Miao, L.; Song, Z.; Zhu, D.; Li, L.; Gan, L.; Liu, M. Recent Advances in Carbon-Based Supercapacitors. Mater. Adv. 2020, 1, 945–966.
- Liang, Z.; Zhao, C.; Zhao, W.; Zhang, Y.; Srimuk, P.; Presser, V.; Feng, G. Molecular Understanding of Charge Storage in MoS2 Supercapacitors with Ionic Liquids. Energy Environ. Mater. 2021, eem2.12147.
- Balat, M. Potential Importance of Hydrogen as a Future Solution to Environmental and Transportation Problems. Int. J. Hydrog. Energy 2008, 33, 4013–4029.
- Gao, M.-R.; Chan, M.K.Y.; Sun, Y. Edge-Terminated Molybdenum Disulfide with a 9.4-Å Interlayer Spacing for Electrochemical Hydrogen Production. Nat. Commun. 2015, 6, 7493.
- Ye, K.; Li, M.; Luo, J.; Wu, B.; Lai, L. The H2O Dissociation and Hydrogen Evolution Performance of Monolayer MoS2 Containing Single Mo Vacancy: A Theoretical Study. IEEE Trans. Nanotechnol. 2020, 19, 163–167.
- Tang, Q.; Jiang, D. Mechanism of Hydrogen Evolution Reaction on 1T-MoS2 from First Principles. ACS Catal. 2016, 6, 4953–4961.
- Ye, K.; Li, M.; Luo, J.; Wu, B.; Lai, L. Activating Inert Basal Plane of MoS2 for H2O Dissociation and HER via Formation of Vacancy Defects: A DFT Study. In Proceedings of the 2019 IEEE 19th International Conference on Nanotechnology (IEEE-NANO), Macao, China, 22–26 July 2019; IEEE: Macao, China, 2019; pp. 48–53.
- Li, J.; Joseph, T.; Ghorbani-Asl, M.; Kolekar, S.; Krasheninnikov, A.V.; Batzill, M. Mirror Twin Boundaries in MoSe2 Monolayers as One Dimensional Nanotemplates for Selective Water Adsorption. Nanoscale 2021, 13, 1038–1047.
- Ye, K.; Lai, L.; Li, M.; Luo, J.; Wu, B.; Ren, Z. Strain Effect on the Hydrogen Evolution Reaction of V Mo -SLMoS2. IEEE Trans. Nanotechnol. 2020, 19, 192–196.
- Gao, M.-R.; Liang, J.-X.; Zheng, Y.-R.; Xu, Y.-F.; Jiang, J.; Gao, Q.; Li, J.; Yu, S.-H. An Efficient Molybdenum Disulfide/Cobalt Diselenide Hybrid Catalyst for Electrochemical Hydrogen Generation. Nat. Commun. 2015, 6, 5982.
- Li, Y.; Wang, L.; Zhang, S.; Dong, X.; Song, Y.; Cai, T.; Liu, Y. Cracked Monolayer 1T MoS2 with Abundant Active Sites for Enhanced Electrocatalytic Hydrogen Evolution. Catal. Sci. Technol. 2017, 7, 718–724.
- Li, H.; Chen, S.; Jia, X.; Xu, B.; Lin, H.; Yang, H.; Song, L.; Wang, X. Amorphous Nickel-Cobalt Complexes Hybridized with 1T-Phase Molybdenum Disulfide via Hydrazine-Induced Phase Transformation for Water Splitting. Nat. Commun. 2017, 8, 15377.
- Yan, K.; Lu, Y. Direct Growth of MoS2 Microspheres on Ni Foam as a Hybrid Nanocomposite Efficient for Oxygen Evolution Reaction. Small 2016, 12, 2975–2981.
- Mohanty, B.; Ghorbani-Asl, M.; Kretschmer, S.; Ghosh, A.; Guha, P.; Panda, S.K.; Jena, B.; Krasheninnikov, A.V.; Jena, B.K. MoS2 Quantum Dots as Efficient Catalyst Materials for the Oxygen Evolution Reaction. ACS Catal. 2018, 8, 1683–1689.
- Asadi, M.; Kumar, B.; Behranginia, A.; Rosen, B.A.; Baskin, A.; Repnin, N.; Pisasale, D.; Phillips, P.; Zhu, W.; Haasch, R.; et al. Robust Carbon Dioxide Reduction on Molybdenum Disulphide Edges. Nat. Commun. 2014, 5, 4470.
- Kim, R.; Kim, J.; Do, J.Y.; Seo, M.W.; Kang, M. Carbon Dioxide Photoreduction on the Bi2S3/MoS2 Catalyst. Catalysts 2019, 9, 998.
- Meier, A.J.; Garg, A.; Sutter, B.; Kuhn, J.N.; Bhethanabotla, V.R. MoS2 Nanoflowers as a Gateway for Solar-Driven CO2 Photoreduction. ACS Sustain. Chem. Eng. 2019, 7, 265–275.
