Asthma is a highly heterogeneous disease, but the pathogenesis of asthma is still unclear. It is well known that the airway inflammatory immune response is the pathological basis of asthma. Metabolomics is a systems biology method to analyze the difference of low molecular weight metabolites (<1.5 kDa) and explore the relationship between metabolic small molecules and pathophysiological changes of the organisms.
- metabolomics
- asthma
- inflammation
- pathogenesis
1. Introduction
2. Integrative Analysis of Asthma-Related Metabolites and Metabolic Pathways in Different Samples
High-throughput sequencing technology has been widely applied to the phenotypic identification, diagnosis, and intervention of highly heterogeneous and complex diseases such as asthma. The metabolomic analysis of various samples. i.e., blood serum plasma samples, urine samples, local tissue samples, exhaled breath condensate (EBC) samples, bronchoalveolar lavage fluid (BALF) samples, induced sputum, and stool samples showed that the different metabolites associated with asthma in different samples and the integration of disordered metabolic pathways may provide evidence for the pathogenesis of asthma. This review explores the potential association of different asthma metabolites in various samples with airway inflammation, airway obstruction, and mucus secretion during the development of asthma, and explains the possible pathogenesis of asthma from a metabolic perspective as shown in Figure 1.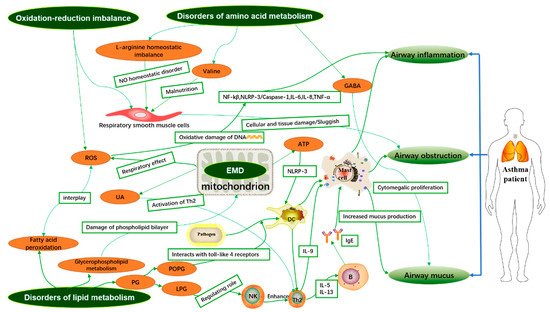
| Author and Year | Subjects | Sample/Methods | Significant Metabolites | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Up | Down | |||||||||||||||||
| Pang, Z. et al. (2018) [6] | eosinophilic asthmatics (EA, | n | = 13), noneosinophilic asthmatics (NEA, | n | = 16), and healthy controls (HC, | n | = 15) | Serum/UPLC-MS/MS | Monosaccharides, LysoPC(18:1), Retinyl ester, PC(18:1/2:0), LysoPC(o-18:0), Arachidonic acid, PE(18:3/14:0), PC(16:0/18:1) | Glycerophosphocholine, PS(18:0/22:5), Cholesterol glucuronide, Phytosphingosine, Sphinganine, LysoPC(p-18:1), Retinols, PC(20:4/16:1) | ||||||||
| Guo, C. et al. (2021) [7] | 51 asthma patients and 9 healthy individuals | Serum/LC-MS | No report | SM 34:2, SM 38:1, SM 40:1 | ||||||||||||||
| Chiu, C.-Y. et al. (2020) [8] | Asthma ( | n | = 28) and healthy controls ( | n | = 26) | Plasma and urine/NMR | Histidine | 1-methylnicotinamide, trimethylamine N-oxide (TMAO) | ||||||||||
| Turi, K.N. et al. (2021) [9] | 600 infants from 3 independent cohorts | Plasma/LC-MS | Succinate, N-(2-furoyl)glycine | Iminodiacetate (IDA) | ||||||||||||||
| Jiang, T. et al. (2021) [10] | 28 healthy controls and 33 outpatients with asthma | Plasma/LC-MS/MS | Phosphatidylethanolamine (PE) (18:1p/22:6), PE (20:0/18:1), PE (38:1), sphingomyelin (SM) (d18:1/18:1), triglyceride (TG) (16:0/16:0/18:1) | Phosphatidylinositol (PI) (16:0/20:4), TG (17:0/18:1/18:1), phosphatidylglycerol (PG) (44:0), ceramide (Cer) (d16:0/27:2), lysophosphatidylcholine (LPC) (22:4) | ||||||||||||||
| Bian, X. et al. (2017) [11] | 15 healthy human and 15 asthma patients | Serum/UHPLC-Q-TOF-MS | Ursodeoxycholic acid, Deoxycholic acid, Isodeoxycholic acid, EPA | Palmitic acid, Lauric acid | ||||||||||||||
| Matysiak, J. et al. (2020) [12] | asthmatic children ( | n | = 13) and the control group ( | n | = 17) | Blood/LC-MS/MS | l | -Arginine, Β-Alanine, Ƴ-Amino-N-Butyric Acid, | l | -Histidine, Hydroxy- | l | -Proline | d,l | -Β-Aminoisobutyric Acid, Taurine, | l | -Tryptophan, | l | -Valine |
| Ghosh, N. et al. (2020) [13] | (i) controls = 33 (ii) asthma = 34 (iii) COPD = 30 and (iv) ACO = 35 | Serum/GC-MS | 2-palmitoylglycerol, cholesterol, serine, threonine, Ethanolamine, Glucose, Stearic acid, Linoleic acid, | d | -Mannose, Succinic acid | Lactic acid, 2-palmitoylglycerol | ||||||||||||
| Liang, Y. et al. (2019) [14] | A total of 17 patients with mildly persistent asthma, 17 patients with stable COPD, and 15 healthy subjects | Serum/LC-MS | Hypoxanthine, P-chlorophenylalanine, Inosine, Theophylline, Bilirubin, Palmitic acid | l | -Glutamine, Glycerophosphocholine, Succinate, Xanthine, Arachidonic Acid, | l | -Pyroglutamic acid, Indoxyl sulfate, | l | -Valine, | l | -Norleucine, | l | -Leucine, | l | -Phenylalanine | |||
| Chiu, C.-Y. et al. (2018) [15] | Asthma ( | n | = 30) and healthy controls ( | n | = 30) | Urine/NMR | Guanidoacetic acid | 1-methylnicotinamide, allantoin | ||||||||||
| Li, S. et al. (2020) [16] | Asthmatic children ( | n | = 30) and healthy controls ( | n | = 30) | Urine/GC-MS | l | -allothreonine 1, stearic acid, succinic acid, 2-hydroxybutanoic acid, azelaic acid, gentiobiose 2, tyramine, leucine, | d | -altrose 1, | d | -erythrosphingosine 1, citraconic acid 4 | Valine, uric acid, methionine 1, 3,4-dihydroxycinnamic acid, purine riboside, malonic acid 1, cysteine, erythrose 1, lactamide 1 | |||||
| Chawes, B.L. et al.(2018) [17] | 171 and 161 healthy neonates born from mothers with asthma | Urine/UPLC-MS | bile acid taurochenodeoxycholate-3-sulfate, fatty acid 3-hydroxytetradecanedioic acid | glucoronidated steroid compound | ||||||||||||||
| Carraro, S. et al. (2018) [18] | Children for transient wheezing ( | n | = 16) and early-onset asthma ( | n | = 16) | Urine/UPLC-MS | 4-(4-deoxy-α-d-gluc-4-enuronosyl)-d-galacturonate, Glutaric acid, 4-hydroxynonenal, Phosphatidyl glycerol, 3-methyluridine, Steroid O-sulfate, 5-hydroxy-l-tryptophan, 3-indoleacetic acid, Tiglylglycine, Indole, Cytosine, N-acetylputrescine, Indole-3-acetamide, 6-methyladenine, 5-methylcytosine, N-acryloylglycine, Hydroxyphenyllactic acid | Oxoadipic acid, (-)-epinephrine, l-tyrosine, 3-hydroxyhippuric acid, Benzoic acid,3-hydroxy-sebacic acid, Dihydroferulic acid 4-sulfate, p-cresol, Indolelactic acid, N-acetyl-l-phenylalanine, N2-acetyl-ornithine | ||||||||||
| Tao, J.-L. et al. (2019) [19] | Children for healthy control ( | n | = 29), uncontrolled asthma ( | n | = 37) or controlled asthma ( | n | = 43) | Urine/GC-MS | Aspartic acid, Xanthosine, Hypoxanthine, N-acetylgalactosamine |
Stearic acid, Heptadecanoic acid, Uric acid, | d | -threitol | ||||||
| Adamko, D.J. et al. (2015) [20] | Adults with asthma ( | n | = 58) and COPD ( | n | = 24) | Urine/NMR | Glutamine, succinate, uracil, pantothenate | Arginine, dimethylamine, 3-Hydroxyisovalerate, betaine, choline, glucose, 1-methylnicotinamide | ||||||||||
| Ravi, A. et al. (2021) [21] | Healthy controls ( | n | = 7) and patients with severe asthma ( | n | = 9) | BECs/UPLC-MS | Phosphatidylcholines, lysophosphatidylcholines, lysophosphatidylethanolamines, bis(monoacylglycero)phosphates | No report | ||||||||||
| Chang-Chien, J. et al.(2020) [22] | stable asthma ( | n | = 92) and non-asthmatic controls ( | n | = 73) | EBC/NMR | lactate, formate, butyric acid, isobutyrate | No report | ||||||||||
| Ferraro, V.A. et al. (2020) [23] | asthmatic children ( | n | = 26) and healthy children ( | n | = 16) | EBC/UPLC-MS | 9-amino-nonanoic acid, 12-amino-dodecanoic acid, lactone of PGF-MUM, N-linoleoyl taurine, 17-phenoxy trinor PGF2α ethyl amide, lysoPC (18:2(9Z,12Z)) | No report | ||||||||||
| Kang, Y.P. et al. (2014) [24] | 38 asthma patients and 13 healthy subjects | BALF/HPLC-QTOF-MS | lysophosphatidylcholine (LPC), phosphatidylcholine (PC), phosphatidylglycerol (PG), phosphatidylserine (PS), sphingomyelin (SM), triglyceride (TG) | No report | ||||||||||||||
| Tian, M. et al. (2017) [25] | 15 healthy controls and 20 asthma patients | Sputum/UHPLC-QTOF-MS | Glycerol 1-stearate_1, 1-Hexadecanoyl-sn-glycerol_1, Cytidine 2′,3′-cyclic phosphate, 1-Hexadecanoyl-2-(9Z-octadecenoyl)-sn-glycero-3-phospho-(1′-rac-glycerol), 1-Octadecanoyl-2-(9Z-octadecenoyl)-sn-glycero-3-phosphoserine | His-Pro, Thr-Phe_1, Arg-Phe_1, Adenine_1, Phe-Tyr_1, Phe-Gln_1, Tyr-Ala_2, Phe-Ser_1, Urocanic acid | ||||||||||||||
Up: metabolites are higher in asthmatics than in normal people; Down: metabolites are lower in asthmatics than in normal people. UPLC-MS/MS: ultra-performance liquid chromatography-tandem mass spectrometry; LC-MS: liquid chromatography-mass spectrometry; NMR: nuclear magnetic resonance; LC-MS/MS: liquid chromatography-tandem mass spectrometry; GC-MS: Gas chromatography-mass spectrometry; UPLC-MS: ultra-performance liquid chromatography-mass spectrometry; HPLC-QTOF-MS: high performance liquid chromatography tandem quadrupole time-of-flight mass spectrometry; UHPLC-QTOF-MS: ultra-performance liquid chromatography tandem quadrupole time-of-flight mass spectrometry.
2.1. Analysis of Biomarkers Associated with Asthma in Blood/Serum/Plasma Samples
2.1.1. Phenotypic Identification and Treatment of Asthma
2.1.2. Diagnosis of Asthma
References
- Kaur, R.; Chupp, G. Phenotypes and endotypes of adult asthma: Moving toward precision medicine. J. Allergy Clin. Immunol. 2019, 144, 1–12.
- Szefler, S.J.; Wenzel, S.; Brown, R.; Erzurum, S.C.; Fahy, J.V.; Hamilton, R.G.; Hunt, J.F.; Kita, H.; Liu, A.H.; Panettieri, R.A.; et al. Asthma outcomes: Biomarkers. J. Allergy Clin. Immunol. 2012, 129, S9–S23.
- Aaron, S.D.; Boulet, L.P.; Reddel, H.; Gershon, A.S. Underdiagnosis and Overdiagnosis of Asthma. Am. J. Respir. Crit. Care Med. 2018, 198, 1012–1020.
- Kelly, R.S.; Dahlin, A.; McGeachie, M.J.; Qiu, W.; Sordillo, J.; Wan, E.S.; Wu, A.C.; Lasky-Su, J. Asthma Metabolomics and the Potential for Integrative Omics in Research and the Clinic. Chest 2017, 151, 262–277.
- Kuruvilla, M.E.; Lee, F.E.-H.; Lee, G.B. Understanding Asthma Phenotypes, Endotypes, and Mechanisms of Disease. Clin. Rev. Allergy Immunol. 2019, 56, 219–233.
- Pang, Z.; Wang, G.; Wang, C.; Zhang, W.; Liu, J.; Wang, F. Serum Metabolomics Analysis of Asthma in Different Inflammatory Phenotypes: A Cross-Sectional Study in Northeast China. BioMed Res. Int. 2018, 2018, 1–14.
- Guo, C.; Sun, L.; Zhang, L.; Dong, F.; Zhang, X.; Yao, L.; Chang, C. Serum sphingolipid profile in asthma. J. Leukoc. Biol. 2021, 110, 53–59.
- Chiu, C.-Y.; Cheng, M.-L.; Chiang, M.-H.; Wang, C.-J.; Tsai, M.-H.; Lin, G. Metabolomic Analysis Reveals Distinct Profiles in the Plasma and Urine Associated with IgE Reactions in Childhood Asthma. J. Clin. Med. 2020, 9, 887.
- Turi, K.N.; McKennan, C.; Gebretsadik, T.; Snyder, B.; Seroogy, C.M.; Lemanske, R.F.; Zoratti, E.; Havstad, S.; Ober, C.; Lynch, S.; et al. Unconjugated bilirubin is associated with protection from early-life wheeze and childhood asthma. J. Allergy Clin. Immunol. 2021.
- Jiang, T.; Dai, L.; Li, P.; Zhao, J.; Wang, X.; An, L.; Liu, M.; Wu, S.; Wang, Y.; Peng, Y.; et al. Lipid metabolism and identification of biomarkers in asthma by lipidomic analysis. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2021, 1866, 158853.
- Bian, X.; Sun, B.; Zheng, P.; Li, N.; Wu, J.-L. Derivatization enhanced separation and sensitivity of long chain-free fatty acids: Application to asthma using targeted and non-targeted liquid chromatography-mass spectrometry approach. Anal. Chim. Acta 2017, 989, 59–70.
- Matysiak, J.; Klupczynska, A.; Packi, K.; Mackowiak-Jakubowska, A.; Bręborowicz, A.; Pawlicka, O.; Olejniczak, K.; Kokot, Z.J.; Matysiak, J. Alterations in Serum-Free Amino Acid Profiles in Childhood Asthma. Int. J. Environ. Res. Public Health 2020, 17, 4758.
- Ghosh, N.; Choudhury, P.; Kaushik, S.R.; Arya, R.; Nanda, R.; Bhattacharyya, P.; Roychowdhury, S.; Banerjee, R.; Chaudhury, K. Metabolomic fingerprinting and systemic inflammatory profiling of asthma COPD overlap (ACO). Respir. Res. 2020, 21, 1–16.
- Liang, Y.; Gai, X.Y.; Chang, C.; Zhang, X.; Wang, J.; Li, T.T. Metabolomic Profiling Differences among Asthma, COPD, and Healthy Subjects: A LC-MS-based Metabolomic Analysis. Biomed. Environ. Sci. 2019, 32, 659–672.
- Chiu, C.-Y.; Lin, G.; Cheng, M.-L.; Chiang, M.-H.; Tsai, M.-H.; Su, K.-W.; Hua, M.-C.; Liao, S.-L.; Lai, S.-H.; Yao, T.-C.; et al. Longitudinal urinary metabolomic profiling reveals metabolites for asthma development in early childhood. Pediatr. Allergy Immunol. 2018, 29, 496–503.
- Li, S.; Liu, J.; Zhou, J.; Wang, Y.; Jin, F.; Chen, X.; Yang, J.; Chen, Z. Urinary Metabolomic Profiling Reveals Biological Pathways and Predictive Signatures Associated with Childhood Asthma. J. Asthma Allergy 2020, ume 13, 713–724.
- Chawes, B.L.; Giordano, G.; Pirillo, P.; Rago, D.; Rasmussen, M.A.; Stokholm, J.; Bønnelykke, K.; Bisgaard, H.; Baraldi, E. Neonatal Urine Metabolic Profiling and Development of Childhood Asthma. Metabolites 2019, 9, 185.
- Carraro, S.; Bozzetto, S.; Giordano, G.; El Mazloum, D.; Stocchero, M.; Pirillo, P.; Zanconato, S.; Baraldi, E. Wheezing preschool children with early-onset asthma reveal a specific metabolomic profile. Pediatr. Allergy Immunol. 2018, 29, 375–382.
- Tao, J.-L.; Chen, Y.-Z.; Dai, Q.-G.; Tian, M.; Wang, S.-C.; Shan, J.-J.; Ji, J.-J.; Lin, L.-L.; Li, W.-W.; Yuan, B. Urine metabolic profiles in paediatric asthma. Respirology 2019, 24, 572–581.
- Adamko, D.J.; Nair, P.; Mayers, I.; Tsuyuki, R.T.; Regush, S.; Rowe, B.H. Metabolomic profiling of asthma and chronic obstructive pulmonary disease: A pilot study differentiating diseases. J. Allergy Clin. Immunol. 2015, 136, 571–580.
- Ravi, A.; Goorsenberg, A.W.; Dijkhuis, A.; Dierdorp, B.S.; Dekker, T.; van Weeghel, M.; Piñeros, Y.S.S.; Shah, P.L.; Hacken, N.H.T.; Annema, J.T.; et al. Metabolic differences between bronchial epithelium from healthy individuals and patients with asthma and the effect of bronchial thermoplasty. J. Allergy Clin. Immunol. 2021, S0091–S6749, 00170-6.
- Chang-Chien, J.; Huang, H.; Tsai, H.; Lo, C.; Lin, W.; Tseng, Y.; Wang, S.; Ho, H.; Cheng, M.; Yao, T. Metabolomic differences of exhaled breath condensate among children with and without asthma. Pediatr. Allergy Immunol. 2021, 32, 264–272.
- Ferraro, V.A.; Carraro, S.; Pirillo, P.; Gucciardi, A.; Poloniato, G.; Stocchero, M.; Giordano, G.; Zanconato, S.; Baraldi, E. Breathomics in Asthmatic Children Treated with Inhaled Corticosteroids. Metabolites 2020, 10, 390.
- Kang, Y.P.; Lee, W.J.; Hong, J.Y.; Lee, S.B.; Park, J.H.; Kim, D.; Park, S.; Park, C.-S.; Park, S.-W.; Kwon, S.W. Novel Approach for Analysis of Bronchoalveolar Lavage Fluid (BALF) Using HPLC-QTOF-MS-Based Lipidomics: Lipid Levels in Asthmatics and Corticosteroid-Treated Asthmatic Patients. J. Proteome Res. 2014, 13, 3919–3929.
- Tian, M.; Chen, M.; Bao, Y.-L.; Xu, C.-D.; Qin, Q.-Z.; Zhang, W.-X.; He, Y.-T.; Shao, Q. Sputum metabolomic profiling of bronchial asthma based on quadruple time-of-flight mass spectrometry. Int. J. Clin. Exp. Pathol. 2017, 10, 10363–10373.
- Chung, K.F. Asthma phenotyping: A necessity for improved therapeutic precision and new targeted therapies. J. Intern. Med. 2016, 279, 192–204.
- Simpson, J.L.; Scott, R.; Boyle, M.J.; Gibson, P.G. Inflammatory subtypes in asthma: Assessment and identification using induced sputum. Respirology 2006, 11, 54–61.
- Mims, J.W. Asthma: Definitions and pathophysiology. Int. Forum Allergy Rhinol. 2015, 5, S2–S6.
- Fahy, J.V. Type 2 inflammation in asthma—Present in most, absent in many. Nat. Rev. Immunol. 2015, 15, 57–65.
- Bara, I.; Ozier, A.; De Lara, J.-M.T.; Marthan, R.; Berger, P. Pathophysiology of bronchial smooth muscle remodelling in asthma. Eur. Respir. J. 2010, 36, 1174–1184.
- Pite, H.; Morais-Almeida, M.; Rocha, S. Metabolomics in asthma. Curr. Opin. Pulm. Med. 2018, 24, 94–103.
- Gai, X.Y.; Zhang, L.J.; Chang, C.; Guo, C.L.; Abulikemu, M.; Li, W.X.; Wang, J.; Yao, W.Z.; Zhang, X. Metabolomic Anal-ysis of Serum Glycerophospholipid Levels in Eosinophilic and Neutrophilic Asthma. Biomed. Environ. Sci. 2019, 32, 96–106.
- Reinke, S.N.; Gallart-Ayala, H.; Gómez, C.; Checa, A.; Fauland, A.; Naz, S.; Kamleh, M.A.; Djukanović, R.; Hinks, T.S.; Wheelock, C.E. Metabolomics analysis identifies different metabotypes of asthma severity. Eur. Respir. J. 2017, 49, 1601740.
- Crestani, E.; Harb, H.; Charbonnier, L.-M.; Leirer, J.; Motsinger-Reif, A.; Rachid, R.; Phipatanakul, W.; Kaddurah-Daouk, R.; Chatila, T.A. Untargeted metabolomic profiling identifies disease-specific signatures in food allergy and asthma. J. Allergy Clin. Immunol. 2020, 145, 897–906.
- Chiu, C.; Chou, H.; Chang, L.; Fan, W.; Dinh, M.C.V.; Kuo, Y.; Chung, W.; Lai, H.; Hsieh, W.; Su, S. Integration of metagenomics-metabolomics reveals specific signatures and functions of airway microbiota in mite-sensitized childhood asthma. Allergy 2020, 75, 2846–2857.
- di Palmo, E.; Cantarelli, E.; Catelli, A.; Ricci, G.; Gallucci, M.; Miniaci, A.; Pession, A. The Predictive Role of Biomarkers and Genetics in Childhood Asthma Exacerbations. Int. J. Mol. Sci. 2021, 22, 4651.
- Rastogi, D.; Fraser, S.; Oh, J.; Huber, A.M.; Schulman, Y.; Bhagtani, R.H.; Khan, Z.S.; Tesfa, L.; Hall, C.; Macian, F. Inflammation, Metabolic Dysregulation, and Pulmonary Function among Obese Urban Adolescents with Asthma. Am. J. Respir. Crit. Care Med. 2015, 191, 149–160.
- Periyalil, H.A.; Gibson, P.G.; Wood, L.G. Immunometabolism in Obese Asthmatics: Are We There Yet? Nutrients 2013, 5, 3506–3530.
- Shore, S.A.; Cho, Y. Obesity and Asthma: Microbiome–Metabolome Interactions. Am. J. Respir. Cell Mol. Biol. 2016, 54, 609–617.
- Miethe, S.; Guarino, M.; Alhamdan, F.; Simon, H.-U.; Renz, H.; Dufour, J.-F.; Potaczek, D.P.; Garn, H. The effects of obesity on asthma: Immunometabolic links. Pol. Arch. Intern. Med. 2018, 128, 469–477.
- Liu, Y.; Zheng, J.; Zhang, H.P.; Zhang, X.; Wang, L.; Wood, L.; Wang, G. Obesity-Associated Metabolic Signatures Correlate to Clinical and Inflammatory Profiles of Asthma: A Pilot Study. Allergy Asthma Immunol. Res. 2018, 10, 628–647.
- Gomez-Llorente, M.A.; Martínez-Cañavate, A.; Chueca, N.; Rico, M.D.L.C.; Romero, R.; Anguita-Ruiz, A.; Aguilera, C.M.; Gil-Campos, M.; Mesa, M.D.; Khakimov, B.; et al. A Multi-Omics Approach Reveals New Signatures in Obese Allergic Asthmatic Children. Biomedicines 2020, 8, 359.
- Winnica, D.; Corey, C.; Mullett, S.; Reynolds, M.; Hill, G.; Wendell, S.; Que, L.; Holguin, F.; Shiva, S. Bioenergetic Differences in the Airway Epithelium of Lean Versus Obese Asthmatics Are Driven by Nitric Oxide and Reflected in Circulating Platelets. Antioxid. Redox Signal. 2019, 31, 673–686.
- Patel, M.; Pilcher, J.; Reddel, H.K.; Pritchard, A.; Corin, A.; Helm, C.; Tofield, C.; Shaw, D.; Black, P.; Weatherall, M.; et al. Metrics of salbutamol use as predictors of future adverse outcomes in asthma. Clin. Exp. Allergy 2013, 43, 1144–1151.
- McGeachie, M.J.; Dahlin, A.; Qiu, W.; Croteau-Chonka, D.C.; Savage, J.; Wu, A.C.; Wan, E.S.; Sordillo, J.E.; Al-Garawi, A.; Martinez, F.D.; et al. The metabolomics of asthma control: A promising link between genetics and disease. Immun. Inflamm. Dis. 2015, 3, 224–238.
- Yang, Y.; Uhlig, S. The role of sphingolipids in respiratory disease. Ther. Adv. Respir. Dis. 2011, 5, 325–344.
- Nixon, G.F. Sphingolipids in inflammation: Pathological implications and potential therapeutic targets. Br. J. Pharmacol. 2009, 158, 982–993.
- Yu, M.; Jia, H.-M.; Cui, F.-X.; Yang, Y.; Zhao, Y.; Yang, M.-H.; Zou, Z.-M. The Effect of Chinese Herbal Medicine Formula mKG on Allergic Asthma by Regulating Lung and Plasma Metabolic Alternations. Int. J. Mol. Sci. 2017, 18, 602.
- Ran, S.; Sun, F.; Song, Y.; Wang, X.; Hong, Y.; Han, Y. The Study of Dried Ginger and Linggan Wuwei Jiangxin Decoction Treatment of Cold Asthma Rats Using GC–MS Based Metabolomics. Front. Pharmacol. 2019, 10, 284.
- You, Y.-N.; Xing, Q.-Q.; Zhao, X.; Ji, J.-J.; Yan, H.; Zhou, T.; Dong, Y.-M.; Ren, L.-S.; Hou, S.-T.; Ding, Y.-Y. Gu-Ben-Fang-Xiao decoction modulates lipid metabolism by activating the AMPK pathway in asthma remission. Biomed. Pharmacother. 2021, 138, 111403.
- Bhavsar, P.; Hew, M.; Khorasani, N.; Torrego, A.; Barnes, P.J.; Adcock, I.; Chung, K.F. Relative corticosteroid insensitivity of alveolar macrophages in severe asthma compared with non-severe asthma. Thorax 2008, 63, 784–790.
- Pang, Z.; Ran, N.; Yuan, Y.; Wang, C.; Wang, G.; Lin, H.; Hsu, A.C.-Y.; Liu, J.; Wang, F. Phenotype-Specific Therapeutic Effect of Rhodiola wallichiana var. cholaensis Combined with Dexamethasone on Experimental Murine Asthma and Its Comprehensive Pharmacological Mechanism. Int. J. Mol. Sci. 2019, 20, 4216.
- Su, L.; Shi, L.; Liu, J.; Huang, L.; Huang, Y.; Nie, X. Metabolic profiling of asthma in mice and the interventional effects of SPA using liquid chromatography and Q-TOF mass spectrometry. Mol. BioSyst. 2017, 13, 1172–1181.
- Mochimaru, T.; Fukunaga, K.; Miyata, J.; Matsusaka, M.; Masaki, K.; Kabata, H.; Ueda, S.; Suzuki, Y.; Goto, T.; Urabe, D.; et al. 12-OH-17,18-Epoxyeicosatetraenoic acid alleviates eosinophilic airway inflammation in murine lungs. Allergy 2018, 73, 369–378.
- Comhair, S.A.A.; McDunn, J.; Bennett, C.; Fettig, J.; Erzurum, S.C.; Kalhan, S.C.; Fetig, J. Metabolomic Endotype of Asthma. J. Immunol. 2015, 195, 643–650.
- Izawa, K.; Isobe, M.; Matsukawa, T.; Ito, S.; Maehara, A.; Takahashi, M.; Yamanishi, Y.; Kaitani, A.; Oki, T.; Okumura, K.; et al. Sphingomyelin and ceramide are physiological ligands for human LMIR3/CD300f, inhibiting FcεRI-mediated mast cell activation. J. Allergy Clin. Immunol. 2014, 133, 270–273.e7.
- Schjødt, M.S.; Gürdeniz, G.; Chawes, B. The Metabolomics of Childhood Atopic Diseases: A Comprehensive Pathway-Specific Review. Metabolites 2020, 10, 511.
- Kelly, R.S.; Chawes, B.; Blighe, K.; Virkud, Y.V.; Croteau-Chonka, D.C.; McGeachie, M.J.; Clish, C.; Bullock, K.; Celedón, J.C.; Weiss, S.T.; et al. An Integrative Transcriptomic and Metabolomic Study of Lung Function in Children with Asthma. Chest 2018, 154, 335–348.
- Kelly, R.S.; Virkud, Y.; Giorgio, R.; Celedón, J.C.; Weiss, S.T.; Lasky-Su, J. Metabolomic profiling of lung function in Costa-Rican children with asthma. Biochim. Biophys. Acta Mol. Basis Dis. 2017, 1863, 1590–1595.
- Wang, S.; Tang, K.; Lu, Y.; Tian, Z.; Huang, Z.; Wang, M.; Zhao, J.; Xie, J. Revealing the role of glycerophospholipid metabolism in asthma through plasma lipidomics. Clin. Chim. Acta 2021, 513, 34–42.
- Funk, C.D. Prostaglandins and Leukotrienes: Advances in Eicosanoid Biology. Science 2001, 294, 1871–1875.
- Anthonisen, N.R.; Lindgren, P.G.; Tashkin, D.P.; Kanner, R.E.; Scanlon, P.D.; Connett, J.E. Bronchodilator response in the lung health study over 11 yrs. Eur. Respir. J. 2005, 26, 45–51.
- Liu, C.-L.; Wu, C.-L.; Lu, Y.-T. Effects of Age on 1-Second Forced Expiratory Volume Response to Bronchodilation. Int. J. Gerontol. 2009, 3, 149–155.
- Kelly, R.S.; Sordillo, J.E.; Lutz, S.M.; Avila, L.; Soto-Quiros, M.; Celedón, J.C.; McGeachie, M.J.; Dahlin, A.; Tantisira, K.; Huang, M.; et al. Pharmacometabolomics of Bronchodilator Response in Asthma and the Role of Age-Metabolite Interactions. Metaboites 2019, 9, 179.
- Sordillo, J.E.; Lutz, S.M.; Kelly, R.S.; McGeachie, M.J.; Dahlin, A.; Tantisira, K.; Clish, C.; Lasky-Su, J.; Wu, A.C. Plasmalogens Mediate the Effect of Age on Bronchodilator Response in Individuals With Asthma. Front. Med. 2020, 7, 38.
- Kelly, R.S.; Sordillo, J.; Lasky-Su, J.; Dahlin, A.; Perng, W.; Rifas-Shiman, S.L.; Weiss, S.T.; Gold, D.R.; Litonjua, A.; Hivert, M.-F.; et al. Plasma metabolite profiles in children with current asthma. Clin. Exp. Allergy 2018, 48, 1297–1304.
- Van Der Sluijs, K.F.; Van De Pol, M.; Kulik, W.; Dijkhuis, A.; Smids, B.S.; Van Eijk, H.W.; Karlas, J.; Molenkamp, R.; Wolthers, K.C.; Johnston, S.; et al. Systemic tryptophan and kynurenine catabolite levels relate to severity of rhinovirus-induced asthma exacerbation: A prospective study with a parallel-group design. Thorax 2013, 68, 1122–1130.
- Collipp, P.J.; Chen, S.Y.; Sharma, R.K.; Balachandar, V.; Maddaiah, V.T. Tryptophane metabolism in bronchial asthma. Ann. Allergy 1975, 35, 153–158.
- Fogarty, A.; Broadfield, E.; Lewis, S.; Lawson, N.; Britton, J. Amino acids and asthma: A case-control study. Eur. Respir. J. 2004, 23, 565–568.
