Lateral flow technology (also known as lateral flow assay) plays a critical role in POC testing, as the technique is rapid, cost-effective, and can be operated by untrained personnel. Lateral flow technologies can be classified as follows: lateral flow immunoassay (LFIA), nucleic acid lateral flow assay (NLFA), and nucleic acid lateral flow immunoassay (NALFIA). LFIA is able to detect antibodies/antigens, while NLFA uses a DNA or RNA probe to detect nucleic acid. Moreover, NALFIA uses both antibodies/antigens and nucleic acid as biomarkers for the detection of antigens/antibodies or amplicons.
- COVID-19
- SARS-CoV-2
- lateral flow assay
- point-of-care testing
1. Introduction
Accurate and effective diagnosis at COVID-19′s early stages is critical for reducing the risk of transmission, as it allows for quick isolation, contact tracing, and earlier treatment. An ideal diagnostic technique would be cost-effective, portable, rapid, and robust with high sensitivity and specificity [7,8][1][2]. This would allow for point-of-care (POC) testing and patient self-administration, resulting in rapid and adequate results and better epidemiological surveillance.
Currently available diagnostic techniques for COVID-19 are based on the detection of the viral gene, antigen, or human antibodies (serological test) and human metabolites [9,10,11,12,13,14,15,16][3][4][5][6][7][8][9][10]. Among these techniques, the detection of viral RNA sequences by reverse transcription polymerase chain reaction (RT-PCR), reverse transcription loop-mediated isothermal amplification (RT-LAMP), and reverse transcription quantitative polymerase chain reaction (RT-qPCR) have been the most reliable methods. RT-qPCR uses signal amplification to achieve a high degree of accuracy [17,18,19][11][12][13]. RT-LAMP is a newly established technique in which amplification occurs at a single temperature [20,21,22][14][15][16]. RT-qPCR is able to directly detect SARS-CoV-2 by monitoring the amplification of a targeted DNA molecule during the PCR [13][7]. Moreover, some novel technologies for detecting viral gene, such as next-generation sequencing (NGS) and Clustered Regularly Interspaced Short Palindromic Repeats (CRISPR), draw great attention due to their better accuracy and higher throughput [23,24][17][18]. However, these methods are expensive, time-consuming, and limited to well-trained professional operators. Therefore, they are often not amenable to extensive population-based or POC testing [25,26][19][20].
Virus antigens or host antibodies can also be detected serologically. The enzyme-linked immunosorbent assay (ELISA) is a rapid and inexpensive technique for detecting specific antibodies in blood samples. In a recent study, an ELISA test was used to detect human SARS-CoV-2 seroconverters [27][21]. This test enabled the detection of distinct antibody types as early as three days after the onset of symptoms. However, similar to RT-PCR techniques, the ELISA method also needs to be performed by well-trained personnel. It also relies on specialized equipment, making it difficult to use at POC testing.
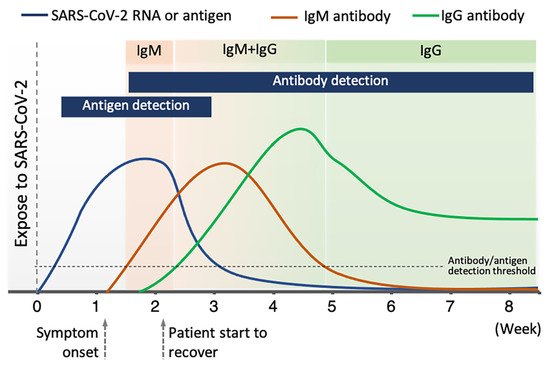
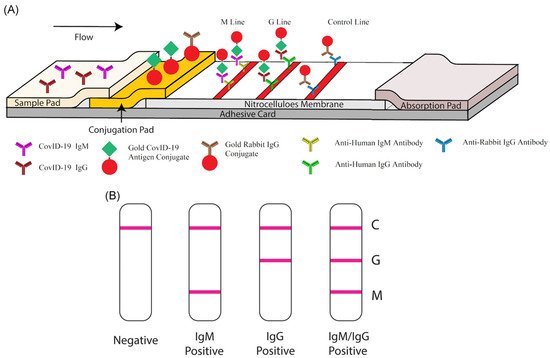
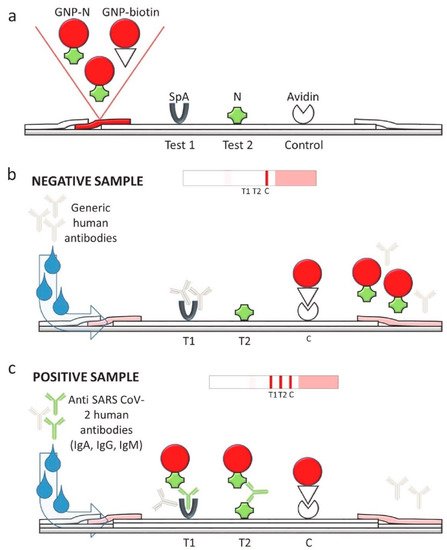
Among available POC testing techniques, the lateral flow immunoassay (LFIA) has been extensively researched and used for COVID-19 diagnosis, owing to its low cost, speed, and accessibility [13,14,25][7][8][19]. To diagnose COVID-19, lateral flow tests combine SARS-CoV-2 pathogen assays with antibodies in patients. LFIA tests usually take around 10–30 min, while the conventional ELISA takes approximately 2–5 h. The sensitivity of COVID-19 detection by LFIA ranges from 61% to 88% (10 days after the first onset of symptoms) to 100% (after 3 weeks) [28,29][22][23]. However, early detection of the disease is a real challenge for LFIA, due to its low accuracy in detection. The accuracy of an LFIA device is evaluated in terms of its sensitivity and specificity. Thus, many efforts have been made to achieve higher sensitivity and specificity for SARS-CoV-2 detection in order to reduce false negative/positive predictive results. In a recent report, Xiang et al. showed that redesigned LFIA can obtain comparable sensitivity to ELISA [30][24]. Similarly, Smith et al. evaluated the sensitivity of the Quidel SARS Sofia rapid antigen flow immunoassay (USA) against RT-qPCR [31][25]. All tests achieved higher than 98% sensitivity to detect infected patients if tests were administered every three days. These evaluations confirmed the possibility of developing an ultrasensitive, highly specific LFIA for POC testing.
2. Lateral Flow Technologies for COVID-19 Detection
2.1. Gene Detection
2.2. Antigen Detection
2.3. Antibody Detection
| Type | Test Kit | Sample Type | Biomarker | Detection Method | Sensitivity | Test Time | Characteristics | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Antigen detection | BinaxNOW COVID-19 Ag Card, Abbott Diagnostics Scarborough, Inc. [74] | BinaxNOW COVID-19 Ag Card, Abbott Diagnostics Scarborough, Inc. [40] | Nasal swab | N protein | Visual | 22.5 TCID | 50 | /swab | 15 min | POC testing; performance depends on following careful testing instructions | |
| CareStart COVID-19 Antigen test, Access Bio, Inc. [75] | CareStart COVID-19 Antigen test, Access Bio, Inc. [41] | Nasopharyngeal Swab | N protein | Visual | 8 × 10 | 2 | TCID | 50 | /mL | 10 min | Requires sample preparation step; operated by trained personnel |
| Lumira Dx SARS-CoV-2 Ag Test, Lumira Dx UK Ltd. [76] | Lumira Dx SARS-CoV-2 Ag Test, Lumira Dx UK Ltd. [42] | Nasal swab | N protein | Fluorescence | 32 TCID | 50 | /mL | 12 min | Requires Lumira Dx Platform; operated by trained personnel | ||
| Sofia 2 Flu + SARS Antigen Flow Immunoassay, Quidel Corporation [77] | Sofia 2 Flu + SARS Antigen Flow Immunoassay, Quidel Corporation [43] |
Nasal, Nasopharyngeal swabs |
N protein | Fluorescence | 4.17 × 10 | 5 | TCID | 50 | /mL | 15 min | Detection of SARS-CoV-2, Influenza A Virus, and Influenza B Virus; limited to Sofia 2 Instrument; operated by trained personnel |
| Antibodies detection | Biohit SARS-CoV-2 IgM/IgG Antibody Test Kit, Biohit Healthcare (Hefei) Co., Ltd. [78] | Biohit SARS-CoV-2 IgM/IgG Antibody Test Kit, Biohit Healthcare (Hefei) Co., Ltd. [44] | Serum, plasma, venous whole blood (heparin, EDTA, and sodium citrate) | IgM and IgG | Visual | 96.7% | 10–20 min | Operated by trained personnel | |||
| COVID-19 IgG/IgM Rapid Test Cassette, Healgen Scientific LLC [79] | COVID-19 IgG/IgM Rapid Test Cassette, Healgen Scientific LLC [45] | Serum, plasma, whole blood | IgM and IgG | Visual | 100% | 10 min | Operated by trained personnel | ||||
| Diagnostic Kit for IgM/IgG Antibody to Coronavirus (SARS-CoV-2), Zhuhai Livzon Diagnostics Inc. [80] | Diagnostic Kit for IgM/IgG Antibody to Coronavirus (SARS-CoV-2), Zhuhai Livzon Diagnostics Inc. [46] | Serum, plasma, venous whole blood | IgM and IgG | Visual | 90.6% | 15 min | - | ||||
| qSARS-CoV-2 IgG/IgM Rapid Test, Cellex Inc. [81] | qSARS-CoV-2 IgG/IgM Rapid Test, Cellex Inc. [47] | Serum, plasma (EDTA or citrate), venous whole blood | IgM and IgG | Visual | - | 15 min | Operated by trained personnel | ||||
| Sienna-Clarity COVIBLOCK COVID-19 IgG/IgM Rapid Test Cassette, Salofa Oy [82] | Sienna-Clarity COVIBLOCK COVID-19 IgG/IgM Rapid Test Cassette, Salofa Oy [48] | Serum, plasma, fingerstick whole blood | IgM and IgG | Visual | 93.3% | 15–20 min | Operated by trained personnel | ||||
| SARS-CoV-2 IgG IgM Antibody Rapid Test Kit, Lumigenex Co., Ltd. [83] | SARS-CoV-2 IgG IgM Antibody Rapid Test Kit, Lumigenex Co., Ltd. [49] | Serum, plasma, fingerstick whole blood | IgM and IgG | Visual | 100% | 15 min | Operated by trained personnel | ||||
| SARS-CoV-2 Antibody Test, Guangzhou Wondfo Biotech Co., Ltd. [84] | SARS-CoV-2 Antibody Test, Guangzhou Wondfo Biotech Co., Ltd. [50] | Serum, plasma, whole blood | IgM and IgG | Visual | 86.4% | 15 min | - | ||||
| RapCov Rapid COVID-19 Test, ADVAITE, Inc. [85] | RapCov Rapid COVID-19 Test, ADVAITE, Inc. [51] | Fingerstick whole blood | IgG | Visual | 90% | 15 min | Operated by trained personnel | ||||
| Rapid COVID-19 IgM/IgG Combo Test Kit, Megna Health, Inc. [86] | Rapid COVID-19 IgM/IgG Combo Test Kit, Megna Health, Inc. [52] | Serum, acid citrate dextrose plasma, fingerstick whole blood | IgM and IgG | Visual | 100% | 10–20 min | Operated by trained personnel |
In a comparison study, the average sensitivity of commercial kits (≈65%) was lower than the sensitivity of laboratory-based kits (≈88%) and of other serological methods (>80%) [87][53]. Moreover, although the claimed sensitivity and specificity of some commercial kits are high, the clinical accuracy of COVID-19 diagnosis is much lower, with the positive predictive value ranging from 11% to 50% [88][54]. In addition, LFIA detection devices are also associated with other challenges relating to difficulties controlling the fluid velocity and capillary force; the interferent porous membrane; the analysis time; and the sample nature [89,90][55][56]. Therefore, further efforts are needed to enhance the sensitivity and specificity of LFIA and ensure LFIA’s practical application in disease control and surveillance.
3. Enhancement of Sensitivity and Specificity
During the COVID-19 pandemic and beyond, the need for low-cost, simple, rapid, and highly accurate methods of disease detection is urgent. However, false negative and false positive test results due to low sensitivity and specificity make it difficult for lateral flow devices to detect disease at its early stages. Therefore, results of lateral flow devices are required to be confirmed with RT-PCR, in order to inform decision-making surrounding isolation and treatment. Up to now, many efforts have been made to enhance sensitivity and specificity of lateral flow technologies. Several methods have been developed, such as sample pre-concentration and amplification, signal enhancement using nanoparticles or an external signal reader, optimizing assay time, and the use of high affinity agents. A summary of methods for enhancing the sensitivity and specificity of lateral flow assays can be found in Table 2.
Table 3.During the COVID-19 pandemic and beyond, the need for low-cost, simple, rapid, and highly accurate Mmethods for enhancing sensof disease detection is urgent. However, false negative and false positive test results due to low sensitivity and specificity make it difficult for lateral flow devices to detect disease at its early stages. Therefore, results of lateral flow devices are required to be confirmed with RT-PCR, in order to inform decision-making surrounding isolation and treatment. Up to now, many efforts have been made to enhance sensitivity and specificity of lateral flow technologies. Several methods have been developed, such as sample pre-concentration and amplification, signal enhancement using nanoparticles or an external signal reader, optimizing assay time, and the use of high affinity agents. A summary of methods for enhancing the sensitivity and specificity of lateral flow assays can be found in Table 2.
|
|
Strategy |
Method |
|---|
| Type | Test Kit | Sample Type | Biomarker | Detection Method | Sensitivity | Test Time | Characteristics |
|---|
|
Enhance sensitivity |
Sample enrichment |
Pre-concentration: · Filtration · Centrifugation · Magnetic pre-concentration · Applying electric field PCR pre-amplification of nucleic acid analytes |
|
Signal enhancement
|
Nanomaterials as reporter: · Modified-AuNPs, quantum dots, fluorescent nanodiamond, selenium NPs, up-converting phosphor particles, lanthanide-doped NPs, aggregation-induced emission (AIE) NPs. External reader: · Optical reader · Fluorescence · Chemiluminescence · Raman |
|
|
Enhance specificity |
Maximizing specific binding |
High-affinity molecules Phage display technology for antibody selection CRISPR/Cas-mediated lateral flow assays |
|
Minimizing non-specific binding |
Pre-filtration or centrifugation Optimizing size and concentration Surface modification with proteins, sugars, PEG. Optimizing running buffer Membrane blocking |
| Antigen detection | BinaxNOW COVID-19 Ag Card, Abbott Diagnostics Scarborough, Inc. [40] | Nasal swab | N protein | Visual | 22.5 TCID50/swab | 15 min | POC testing; performance depends on following careful testing instructions |
| CareStart COVID-19 Antigen test, Access Bio, Inc. [41] | Nasopharyngeal Swab | N protein | Visual | 8 × 102 TCID50/mL | 10 min | Requires sample preparation step; operated by trained personnel | |
| Lumira Dx SARS-CoV-2 Ag Test, Lumira Dx UK Ltd. [42] | Nasal swab | N protein | Fluorescence | 32 TCID50/mL | 12 min | Requires Lumira Dx Platform; operated by trained personnel | |
| Sofia 2 Flu + SARS Antigen Flow Immunoassay, Quidel Corporation [43] |
Nasal, Nasopharyngeal swabs |
N protein | Fluorescence | 4.17 × 105 TCID50/mL | 15 min | Detection of SARS-CoV-2, Influenza A Virus, and Influenza B Virus; limited to Sofia 2 Instrument; operated by trained personnel | |
| Antibodies detection | Biohit SARS-CoV-2 IgM/IgG Antibody Test Kit, Biohit Healthcare (Hefei) Co., Ltd. [44] | Serum, plasma, venous whole blood (heparin, EDTA, and sodium citrate) | IgM and IgG | Visual | 96.7% | 10–20 min | Operated by trained personnel |
| COVID-19 IgG/IgM Rapid Test Cassette, Healgen Scientific LLC [45] | Serum, plasma, whole blood | IgM and IgG | Visual | 100% | 10 min | Operated by trained personnel | |
| Diagnostic Kit for IgM/IgG Antibody to Coronavirus (SARS-CoV-2), Zhuhai Livzon Diagnostics Inc. [46] | Serum, plasma, venous whole blood | IgM and IgG | Visual | 90.6% | 15 min | - | |
| qSARS-CoV-2 IgG/IgM Rapid Test, Cellex Inc. [47] | Serum, plasma (EDTA or citrate), venous whole blood | IgM and IgG | Visual | - | 15 min | Operated by trained personnel | |
| Sienna-Clarity COVIBLOCK COVID-19 IgG/IgM Rapid Test Cassette, Salofa Oy [48] | Serum, plasma, fingerstick whole blood | IgM and IgG | Visual | 93.3% | 15–20 min | Operated by trained personnel | |
| SARS-CoV-2 IgG IgM Antibody Rapid Test Kit, Lumigenex Co., Ltd. [49] | Serum, plasma, fingerstick whole blood | IgM and IgG | Visual | 100% | 15 min | Operated by trained personnel | |
| SARS-CoV-2 Antibody Test, Guangzhou Wondfo Biotech Co., Ltd. [50] | Serum, plasma, whole blood | IgM and IgG | Visual | 86.4% | 15 min | - | |
| RapCov Rapid COVID-19 Test, ADVAITE, Inc. [51] | Fingerstick whole blood | IgG | Visual | 90% | 15 min | Operated by trained personnel | |
| Rapid COVID-19 IgM/IgG Combo Test Kit, Megna Health, Inc. [52] | Serum, acid citrate dextrose plasma, fingerstick whole blood | IgM and IgG | Visual | 100% | 10–20 min | Operated by trained personnel |
3.1. Sample Pre-Concentration and Pre-Amplification
3.1. Sample Pre-Concentration and Pre-Amplification
If the same quantity of sample is used, conventional lateral flow technologies only achieve an average sensitivity of 66%, which is much lower than other serological assays. However, pre-concentration of the sample, before it is processed with lateral flow test, can significantly improve the assay’s sensitivity. Sharma et al. have used a magnetic field to pre-concentrate the analytes from the sample matrices to achieve 10 times higher sensitivity [91][57]. In addition, the antigen–reporter complex can also be concentrated during the lateral flow assay running process by applying an electric field. The so-called isotachophoresis method allows for improved equilibrium binding and thus lowers the detection limit up to 400 times [93][58]. Pre-amplification is another excellent technique for increasing sensitivity. Amplifying DNA/RNA targeted samples by PCR prior to the lateral flow assay process can significantly boost lateral flow assay sensitivity up to RT-PCR’s sensitivity level [94][59]. However, the PCR amplification technique requires expensive instruments and well-trained personnel. Although sample enrichment methods can help increase sensitivity by ten to hundreds of times (even reaching ultrasensitivity), these methods still require additional equipment, extra preparation steps, or prolong the testing period, making it difficult for them to use for POC testing.
3.2. Signal Enhancement
Signal enhancement for lateral flow assays involves either the development of a new optical reporter system or utilizing an external signal reader to amplify the signal intensity and contrast. AuNPs with a nominal size around 20–40 nm have been widely used for conventional lateral flow assays. So far, most LFIAs for COVID-19 detection were developed using traditional AuNPs, but their sensitivities were not very high. In recent years, fluorescent nanoparticles have been increasingly applied in disease diagnosis and are promising alternative reporters, as their unique chemical and optical properties mean that they have the potential to enhance the sensitivity of lateral flow assays. Many fluorescence nanoparticles were utilized for SARS-CoV-2 detection, including QDs [95][60], FNDs [96][61], selenium nanoparticles [65,97][62][63], up-converting phosphor particles [98][64], lanthanide-doped nanoparticles [64][65], and aggregation-induced emission (AIE) nanoparticles [66].3.3. Method for Improving Specificity
3.3.1. Phage Display Technique for SARS-CoV-2 Antibody Selection
Specificity is another important factor that directly affects the accuracy of the lateral flow assay. Lateral flow assays for COVID-19 detection may be inaccurate due to the cross-reactivity of the SARS-CoV-2 virus with other coronaviruses. The cross-reactivity can reduce the specificity of the test, thus generating false positive results. To overcome this issue, phage display can be used to select SARS-CoV-2 antibodies with the strongest affinity. The phage display technique is a powerful method within the field of molecular biology that was awarded the 2018 Nobel Prize in Chemistry and has been widely used for the selection of antibodies, peptides, and disease-specific antigens [122][67]. In phage display, an exogenous DNA fragment encoding a protein of interest is inserted into a phage coat protein gene on the exposed surface, which is then capable of interacting with various external target molecules. This phenotype–genotype interaction enables researchers to isolate target-specific ligands [123][68].
3.3.2. CRISPR/Cas-Mediated Lateral Flow Nucleic Acid Assay
3.3.3. Minimizing Non-Specific Binding
Specificity can also be enhanced by minimizing non-specific binding and non-specific interactions of the reporter to the targeted analytes and the membrane [88][54]. To reduce non-specific binding, a pre-filtration or centrifugation step can be applied to remove undesirable substances in the whole blood [132][71]. Optimizing reporter size and concentration and blocking the conjugated reporter by surface modification can also help minimize non-specific binding. Several proteins, sugars, and PEG polymer can be a surface coating or chemically conjugated to the reporter to enhance stability [90][56]. In addition, the running buffer also strongly affects the specificity of tests. Surfactants can help reduce non-specific binding; however, in high concentrations, it also reduces specific binding [88][54]. The pH and ionic strength of the buffer solution also need to be considered when optimizing the running buffer.4. Conclusions
Lateral flow technologies developed during the COVID-19 pandemic are portable, fast-acting, inexpensive, and easy to use, and therefore, they are becoming one of the most suitable techniques to practice POC testing. A comparison of recent COVID-19 detection methods has been described in Figure 4. Although false negative and false positive issues limit their clinical use, researchers around the world have worked together to improve the efficiency and accuracy of lateral flow tests in the hopes of creating a universal test for COVID-19.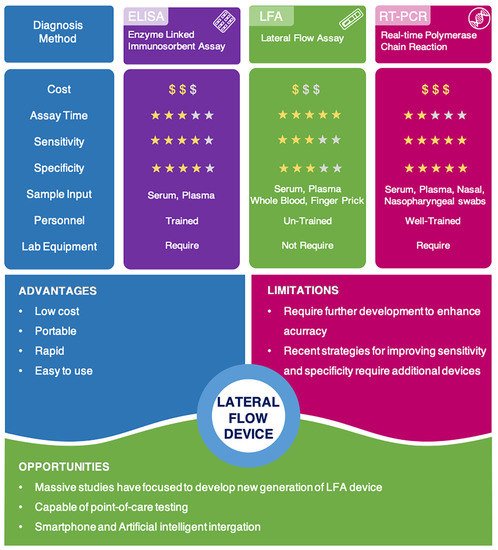
References
- Drain, P.K.; Hyle, E.P.; Noubary, F.; Freedberg, K.A.; Wilson, D.; Bishai, W.R.; Rodriguez, W.; Bassett, I.V. Diagnostic point-of-care tests in resource-limited settings. Lancet Infect. Dis. 2014, 14, 239–249.
- Gubala, V.; Harris, L.F.; Ricco, A.J.; Tan, M.X.; Williams, D.E. Point of care diagnostics: Status and future. Anal. Chem. 2012, 84, 487–515.
- Udugama, B.; Kadhiresan, P.; Kozlowski, H.N.; Malekjahani, A.; Osborne, M.; Li, V.Y.; Chen, H.; Mubareka, S.; Gubbay, J.B.; Chan, W.C. Diagnosing COVID-19: The disease and tools for detection. ACS Nano 2020, 14, 3822–3835.
- Cui, F.; Zhou, H.S. Diagnostic methods and potential portable biosensors for coronavirus disease 2019. Biosens. Bioelectron. 2020, 165, 112349.
- Pokhrel, P.; Hu, C.; Mao, H. Detecting the coronavirus (COVID-19). ACS Sens. 2020, 5, 2283–2296.
- Weiss, C.; Carriere, M.; Fusco, L.; Capua, I.; Regla-Nava, J.A.; Pasquali, M.; Scott, J.A.; Vitale, F.; Unal, M.A.; Mattevi, C. Toward nanotechnology-enabled approaches against the COVID-19 pandemic. ACS Nano 2020, 14, 6383–6406.
- Eftekhari, A.; Alipour, M.; Chodari, L.; Maleki Dizaj, S.; Ardalan, M.R.; Samiei, M.; Sharifi, S.; Zununi Vahed, S.; Huseynova, I.; Khalilov, R. A comprehensive review of detection methods for SARS-CoV-2. Microorganisms 2021, 9, 232.
- Nguyen, N.N.; McCarthy, C.; Lantigua, D.; Camci-Unal, G. Development of diagnostic tests for detection of SARS-CoV-2. Diagnostics 2020, 10, 905.
- Carter, L.J.; Garner, L.V.; Smoot, J.W.; Li, Y.; Zhou, Q.; Saveson, C.J.; Sasso, J.M.; Gregg, A.C.; Soares, D.J.; Beskid, T.R.; et al. Assay techniques and test development for COVID-19 diagnosis. ACS Cent. Sci. 2020, 6, 591–605.
- De Vries, R.; Vigeveno, R.M.; Mulder, S.; Farzan, N.; Vintges, D.R.; Goeman, J.J.; Bruisten, S.; van den Corput, B.; Geelhoed, J.J.M.; Visser, L.G.; et al. Ruling out SARS-CoV-2 infection using exhaled breath analysis by electronic nose in a public health setting. medRxiv 2021.
- Antiochia, R. Developments in biosensors for CoV detection and future trends. Biosens. Bioelectron. 2021, 173, 112777.
- Shen, M.; Zhou, Y.; Ye, J.; Al-Maskri, A.A.A.; Kang, Y.; Zeng, S.; Cai, S. Recent advances and perspectives of nucleic acid detection for coronavirus. J. Pharm. Anal. 2020, 10, 97–101.
- Waggoner, J.J.; Stittleburg, V.; Pond, R.; Saklawi, Y.; Sahoo, M.K.; Babiker, A.; Hussaini, L.; Kraft, C.S.; Pinsky, B.A.; Anderson, E.J. Triplex real-time RT-PCR for severe acute respiratory syndrome coronavirus 2. Emerg. Infect. Dis. 2020, 26, 1633.
- Kashir, J.; Yaqinuddin, A. Loop mediated isothermal amplification (LAMP) assays as a rapid diagnostic for COVID-19. Med. Hypotheses 2020, 141, 109786.
- Huang, W.E.; Lim, B.; Hsu, C.C.; Xiong, D.; Wu, W.; Yu, Y.; Jia, H.; Wang, Y.; Zeng, Y.; Ji, M. RT-LAMP for rapid diagnosis of coronavirus SARS-CoV-2. Microb. Biotechnol. 2020, 13, 950–961.
- Yu, L.; Wu, S.; Hao, X.; Dong, X.; Mao, L.; Pelechano, V.; Chen, W.-H.; Yin, X. Rapid detection of COVID-19 coronavirus using a reverse transcriptional loop-mediated isothermal amplification (RT-LAMP) diagnostic platform. Clin. Chem. 2020, 66, 975–977.
- Ackerman, C.M.; Myhrvold, C.; Thakku, S.G.; Freije, C.A.; Metsky, H.C.; Yang, D.K.; Ye, S.H.; Boehm, C.K.; Kosoko-Thoroddsen, T.-S.F.; Kehe, J.; et al. Massively multiplexed nucleic acid detection with Cas13. Nature 2020, 582, 277–282.
- Yelagandula, R.; Bykov, A.; Vogt, A.; Heinen, R.; Özkan, E.; Strobl, M.M.; Baar, J.C.; Uzunova, K.; Hajdusits, B.; Kordic, D.; et al. Multiplexed detection of SARS-CoV-2 and other respiratory infections in high throughput by SARSeq. Nat. Commun. 2021, 12, 3132.
- Antiochia, R. Paper-Based Biosensors: Frontiers in Point-of-Care Detection of COVID-19 Disease. Biosensors 2021, 11, 110.
- Nayak, S.; Blumenfeld, N.R.; Laksanasopin, T.; Sia, S.K. Point-of-care diagnostics: Recent developments in a connected age. Anal. Chem. 2017, 89, 102–123.
- Amanat, F.; Stadlbauer, D.; Strohmeier, S.; Nguyen, T.H.; Chromikova, V.; McMahon, M.; Jiang, K.; Arunkumar, G.A.; Jurczyszak, D.; Polanco, J. A serological assay to detect SARS-CoV-2 seroconversion in humans. Nat. Med. 2020, 26, 1033–1036.
- Adams, E.R.; Ainsworth, M.; Anand, R.; Andersson, M.I.; Auckland, K.; Baillie, J.K.; Barnes, E.; Beer, S.; Bell, J.I.; Berry, T. Antibody testing for COVID-19: A report from the National COVID Scientific Advisory Panel. Wellcome Open Res. 2020, 5.
- Wu, J.-L.; Tseng, W.-P.; Lin, C.-H.; Lee, T.-F.; Chung, M.-Y.; Huang, C.-H.; Chen, S.-Y.; Hsueh, P.-R.; Chen, S.-C. Four point-of-care lateral flow immunoassays for diagnosis of COVID-19 and for assessing dynamics of antibody responses to SARS-CoV-2. J. Infect. 2020, 81, 435–442.
- Xiang, J.; Yan, M.; Li, H.; Liu, T.; Lin, C.; Huang, S.; Shen, C. Evaluation of enzyme-linked immunoassay and colloidal gold-immunochromatographic assay kit for detection of novel coronavirus (SARS-CoV-2) causing an outbreak of pneumonia (COVID-19). medRxiv 2020.
- Smith, R.L.; Gibson, L.L.; Martinez, P.P.; Ke, R.; Mirza, A.; Conte, M.; Gallagher, N.; Conte, A.; Wang, L.; Fredrickson, R. Longitudinal assessment of diagnostic test performance over the course of acute SARS-CoV-2 infection. medRxiv 2021.
- Yu, S.; Nimse, S.B.; Kim, J.; Song, K.-S.; Kim, T. Development of a lateral flow strip membrane assay for rapid and sensitive detection of the SARS-CoV-2. Anal. Chem. 2020, 92, 14139–14144.
- Zhu, X.; Wang, X.; Han, L.; Chen, T.; Wang, L.; Li, H.; Li, S.; He, L.; Fu, X.; Chen, S. Multiplex reverse transcription loop-mediated isothermal amplification combined with nanoparticle-based lateral flow biosensor for the diagnosis of COVID-19. Biosens. Bioelectron. 2020, 166, 112437.
- Wang, D.; He, S.; Wang, X.; Yan, Y.; Liu, J.; Wu, S.; Liu, S.; Lei, Y.; Chen, M.; Li, L. Rapid lateral flow immunoassay for the fluorescence detection of SARS-CoV-2 RNA. Nat. Biomed. Eng. 2020, 4, 1150–1158.
- Baker, A.N.; Richards, S.-J.; Guy, C.S.; Congdon, T.R.; Hasan, M.; Zwetsloot, A.J.; Gallo, A.; Lewandowski, J.R.; Stansfeld, P.J.; Straube, A. The SARS-CoV-2 spike protein binds sialic acids and enables rapid detection in a lateral flow point of care diagnostic device. ACS Cent. Sci. 2020, 6, 2046–2052.
- Diao, B.; Wen, K.; Chen, J.; Liu, Y.; Yuan, Z.; Han, C.; Chen, J.; Pan, Y.; Chen, L.; Dan, Y. Diagnosis of acute respiratory syndrome coronavirus 2 infection by detection of nucleocapsid protein. medRxiv 2020.
- Grant, B.D.; Anderson, C.E.; Williford, J.R.; Alonzo, L.F.; Glukhova, V.A.; Boyle, D.S.; Weigl, B.H.; Nichols, K.P. SARS-CoV-2 coronavirus nucleocapsid antigen-detecting half-strip lateral flow assay toward the development of point of care tests using commercially available reagents. Anal. Chem. 2020, 92, 11305–11309.
- Zhao, J.; Yuan, Q.; Wang, H.; Liu, W.; Liao, X.; Su, Y.; Wang, X.; Yuan, J.; Li, T.; Li, J.; et al. Antibody Responses to SARS-CoV-2 in Patients with Novel Coronavirus Disease 2019. Clin. Infect. Dis. 2020, 71, 2027–2034.
- Okba, N.M.A.; Müller, M.A.; Li, W.; Wang, C.; GeurtsvanKessel, C.H.; Corman, V.M.; Lamers, M.M.; Sikkema, R.S.; de Bruin, E.; Chandler, F.D.; et al. Severe Acute Respiratory Syndrome Coronavirus 2-Specific Antibody Responses in Coronavirus Disease Patients. Emerg. Infect. Dis. 2020, 26, 1478–1488.
- Stadlbauer, D.; Amanat, F.; Chromikova, V.; Jiang, K.; Strohmeier, S.; Arunkumar, G.A.; Tan, J.; Bhavsar, D.; Capuano, C.; Kirkpatrick, E.; et al. SARS-CoV-2 Seroconversion in Humans: A Detailed Protocol for a Serological Assay, Antigen Production, and Test Setup. Curr. Protoc. Microbiol. 2020, 57, e100.
- Bates, T.A.; Weinstein, J.B.; Farley, S.; Leier, H.C.; Messer, W.B.; Tafesse, F.G. Cross-reactivity of SARS-CoV structural protein antibodies against SARS-CoV-2. Cell Rep. 2021, 34, 108737.
- Wen, T.; Huang, C.; Shi, F.-J.; Zeng, X.-Y.; Lu, T.; Ding, S.-N.; Jiao, Y.-J. Development of a lateral flow immunoassay strip for rapid detection of IgG antibody against SARS-CoV-2 virus. Analyst 2020, 145, 5345–5352.
- Li, Z.; Yi, Y.; Luo, X.; Xiong, N.; Liu, Y.; Li, S.; Sun, R.; Wang, Y.; Hu, B.; Chen, W. Development and clinical application of a rapid IgM-IgG combined antibody test for SARS-CoV-2 infection diagnosis. J. Med. Virol. 2020, 92, 1518–1524.
- Cavalera, S.; Colitti, B.; Rosati, S.; Ferrara, G.; Bertolotti, L.; Nogarol, C.; Guiotto, C.; Cagnazzo, C.; Denina, M.; Fagioli, F. A multi-target lateral flow immunoassay enabling the specific and sensitive detection of total antibodies to SARS-COV-2. Talanta 2021, 223, 121737.
- Sterlin, D.; Mathian, A.; Miyara, M.; Mohr, A.; Anna, F.; Claër, L.; Quentric, P.; Fadlallah, J.; Devilliers, H.; Ghillani, P. IgA dominates the early neutralizing antibody response to SARS-CoV-2. Sci. Transl. Med. 2021, 13, eabd2223.
- BinaxNOW COVID-19 Ag Card. Abbott Diagnostics Scarborough, Inc. Available online: https://www.fda.gov/media/141570/download (accessed on 20 July 2021).
- CareStart COVID-19 Anti-gen Test. Access Bio, Inc. Available online: https://www.fda.gov/media/142919/download (accessed on 20 July 2021).
- Lumira Dx SARS-CoV-2 Ag Test. Lumira Dx UK Ltd. Available online: https://www.fda.gov/media/141302/download (accessed on 20 July 2021).
- Sofia 2 Flu + SARS Antigen FIA, Quidel Corporation. Available online: https://www.fda.gov/media/142704/download (accessed on 20 July 2021).
- Biohit SARS-CoV-2 IgM/IgG Antibody Test Kit. Biohit Healthcare (Hefei) Co., Ltd. Available online: https://www.fda.gov/media/139283/download (accessed on 20 July 2021).
- COVID-19 IgG/IgM Rapid Test Cassette. Healgen Scientific LLC. Available online: https://www.fda.gov/media/138438/download (accessed on 20 July 2021).
- Diagnostic Kit for IgM/IgG Antibody to Coronavirus (SARS-CoV-2). Zhuhai Livzon Diagnostics Inc. Available online: http://www.livzondiagnostics.com/en-us/info/17.html (accessed on 20 July 2021).
- qSARS-CoV-2 IgG/IgM Rapid Test. Cellex Inc. Available online: https://www.fda.gov/media/136622/download (accessed on 20 July 2021).
- Sienna-Clarity COVIBLOCK COVID-19 IgG/IgM Rapid Test Cassette. Salofa Oy. Available online: https://www.fda.gov/media/140082/download (accessed on 20 July 2021).
- SARS-CoV-2 Ig-GIgM Antibody Rapid Test Kit. Lumigenex Co., Ltd. Available online: http://www.lumigenex.com/dynamic/363.html (accessed on 20 July 2021).
- SARS-CoV-2 Antibody Test. Guangzhou Wondfo Biotech Co., Ltd. Available online: https://www.bilcare.com/SARS-CoV-2%20Antibody%20Test%20(Lateral%20Flow%20Method).pdf (accessed on 20 July 2021).
- RapCov Rapid COVID-19 Test. ADVAITE, Inc. Available online: https://www.fda.gov/media/145080/download (accessed on 20 July 2021).
- Rapid COVID-19 IgM/IgG Combo Test Kit. Megna Health, Inc. Available online: https://www.fda.gov/media/140297/download (accessed on 20 July 2021).
- Bastos, M.L.; Tavaziva, G.; Abidi, S.K.; Campbell, J.R.; Haraoui, L.-P.; Johnston, J.C.; Lan, Z.; Law, S.; MacLean, E.; Trajman, A. Diagnostic accuracy of serological tests for COVID-19: Systematic review and meta-analysis. BMJ 2020, 370.
- Liu, Y.; Zhan, L.; Qin, Z.; Sackrison, J.; Bischof, J.C. Ultrasensitive and highly specific lateral flow assays for point-of-care diagnosis. ACS Nano 2021, 15, 3593–3611.
- Chen, P.; Gates-Hollingsworth, M.; Pandit, S.; Park, A.; Montgomery, D.; AuCoin, D.; Gu, J.; Zenhausern, F. Paper-based Vertical Flow Immunoassay (VFI) for detection of bio-threat pathogens. Talanta 2019, 191, 81–88.
- Sajid, M.; Kawde, A.-N.; Daud, M. Designs, formats and applications of lateral flow assay: A literature review. J. Saudi Chem. Soc. 2015, 19, 689–705.
- Sharma, A.; Tok, A.I.Y.; Lee, C.; Ganapathy, R.; Alagappan, P.; Liedberg, B. Magnetic field assisted preconcentration of biomolecules for lateral flow assaying. Sens. Actuators B Chem. 2019, 285, 431–437.
- Moghadam, B.Y.; Connelly, K.T.; Posner, J.D. Two orders of magnitude improvement in detection limit of lateral flow assays using isotachophoresis. Anal. Chem. 2015, 87, 1009–1017.
- Kim, J.; Mohamed, M.A.A.; Zagorovsky, K.; Chan, W.C. State of diagnosing infectious pathogens using colloidal nanomaterials. Biomaterials 2017, 146, 97–114.
- Wang, C.; Shi, D.; Wan, N.; Yang, X.; Liu, H.; Gao, H.; Zhang, M.; Bai, Z.; Li, D.; Dai, E. Development of spike protein-based fluorescence lateral flow assay for the simultaneous detection of SARS-CoV-2 specific IgM and IgG. Analyst 2021, 146, 3908–3917.
- Hui, Y.Y.; Chen, O.J.; Lin, H.-H.; Su, Y.-K.; Chen, K.Y.; Wang, C.-Y.; Hsiao, W.W.-W.; Chang, H.-C. Magnetically Modulated Fluorescence of Nitrogen-Vacancy Centers in Nanodiamonds for Ultrasensitive Biomedical Analysis. Anal. Chem. 2021, 93, 7140–7147.
- Wang, Z.-Z.; Zheng, Z.; Wang, X.-C.; Zheng, P.-M.; Cui, F.-C.; Zhou, Q.-W.; Hu, H.-Z.; Li, X.-Q.; Zhang, H.-L.; Wei, Y.-X.; et al. Rapid detection of anti-SARS-CoV-2 IgM and IgG using a selenium nanoparticle-based lateral flow immunoassay. Res. Sq. 2021.
- Wang, Z.; Zheng, Z.; Hu, H.; Zhou, Q.; Liu, W.; Li, X.; Liu, Z.; Wang, Y.; Ma, Y. A point-of-care selenium nanoparticle-based test for the combined detection of anti-SARS-CoV-2 IgM and IgG in human serum and blood. Lab Chip 2020, 20, 4255–4261.
- Tjon Kon Fat, E.M.; Abrams, W.R.; Niedbala, R.S.; Corstjens, P.L. Lateral flow sandwich assay utilizing upconverting phosphor (UCP) reporters. Methods Cell Biol. 2012, 112, 203–234.
- Chen, Z.; Zhang, Z.; Zhai, X.; Li, Y.; Lin, L.; Zhao, H.; Bian, L.; Li, P.; Yu, L.; Wu, Y. Rapid and sensitive detection of anti-SARS-CoV-2 IgG, using lanthanide-doped nanoparticles-based lateral flow immunoassay. Anal. Chem. 2020, 92, 7226–7231.
- Chen, R.; Ren, C.; Liu, M.; Ge, X.; Qu, M.; Zhou, X.; Liang, M.; Liu, Y.; Li, F. Early Detection of SARS-CoV-2 Seroconversion in Humans with Aggregation-Induced Near-Infrared Emission Nanoparticle-Labeled Lateral Flow Immunoassay. ACS Nano 2021, 15, 8996–9004.
- Anand, T.; Virmani, N.; Bera, B.C.; Vaid, R.K.; Vashisth, M.; Bardajatya, P.; Kumar, A.; Tripathi, B.N. Phage Display Technique as a Tool for Diagnosis and Antibody Selection for Coronaviruses. Curr. Microbiol. 2021, 78, 1124–1134.
- Parmley, S.F.; Smith, G.P. Antibody-selectable filamentous fd phage vectors: Affinity purification of target genes. Gene 1988, 73, 305–318.
- Broughton, J.P.; Deng, X.; Yu, G.; Fasching, C.L.; Servellita, V.; Singh, J.; Miao, X.; Streithorst, J.A.; Granados, A.; Sotomayor-Gonzalez, A. CRISPR–Cas12-based detection of SARS-CoV-2. Nat. Biotechnol. 2020, 38, 870–874.
- Hou, T.; Zeng, W.; Yang, M.; Chen, W.; Ren, L.; Ai, J.; Wu, J.; Liao, Y.; Gou, X.; Li, Y. Development and evaluation of a rapid CRISPR-based diagnostic for COVID-19. PLoS Pathog. 2020, 16, e1008705.
- Schramm, E.C.; Staten, N.R.; Zhang, Z.; Bruce, S.S.; Kellner, C.; Atkinson, J.P.; Kyttaris, V.C.; Tsokos, G.C.; Petri, M.; Connolly, E.S. A quantitative lateral flow assay to detect complement activation in blood. Anal. Biochem. 2015, 477, 78–85.
