The most common route of administration of ophthalmic drugs is the topical route because it is convenient, non-invasive, and accessible to all patients. Unfortunately, drugs administered topically are not able to reach effective concentrations. Moreover, their bioavailability must be improved to decrease the frequency of administrations and their side effects, and to increase their therapeutic efficiency. For this purpose, in recent decades, particular attention has been given to the possibility of developing prolonged-release forms that are able to increase the precorneal residence time and decrease the loss of the drug due to tearing. Among these forms, gel-based materials have been studied as an ideal delivery system because they are an extremely versatile class with numerous prospective applications in ophthalmology. These materials are used in gel eye drops, in situ gelling formulations, intravitreal injections, and therapeutic contact lenses.
- gels
- hydrogels
- gel eye drops
- in situ gels
- intravitreal injection
- contact lens
1. Introduction
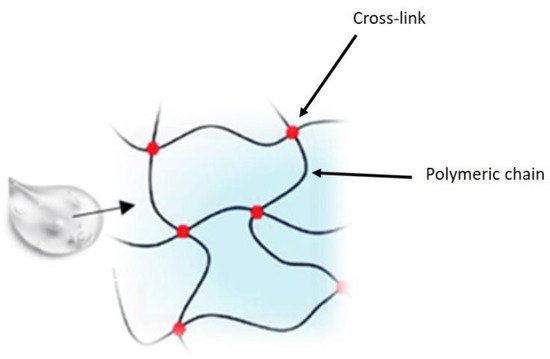
2. Ophthalmic Gels
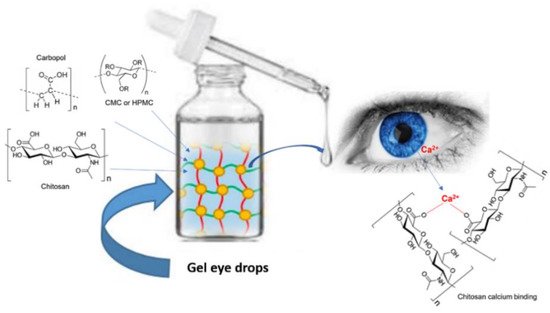
2.1. Gel Eye Drops
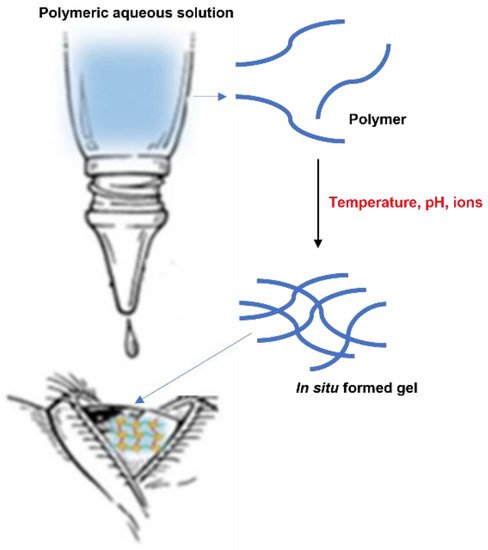
2.2. In Situ Gels
2.2.1. Temperature-Sensitive In Situ Gel Systems
2.2.2. pH-Sensitive In Situ Gel Systems
2.2.3. Ion-Sensitive In Situ Gel Systems
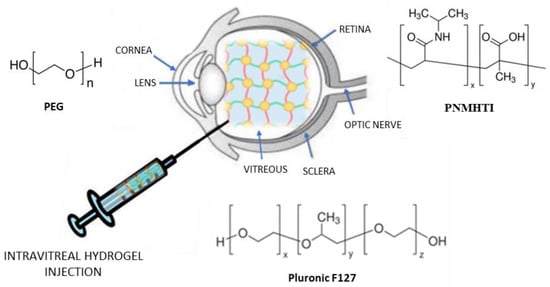
2.3. Intravitreal Injection
3. Contact Lenses
Contact lenses are ocular prosthetic devices that have several functions, such as the correction of refractive errors in the cases of myopia, hypermetropia, and astigmatism [60][58]. These devices are used to treat ocular dysfunctions, particularly corneal irregularities, and for post-surgical refractive rehabilitation. However, they can also be used as cosmetic lenses, such as colored and limbal ring lenses. Another interesting application of contact lenses concerns the prolonged administration of drugs [61][59] first described by Sedlacek in 1965 [62][60]. Subsequently, significant attention has been paid to the ability of contact lenses to improve corneal penetration and drug bioavailability [63,64][61][62]. The lens absorbs some of the drug from the tear film and then acts as a reservoir, slowly releasing the drug into the tears as the overall drug concentration in the tear film decreases. For this purpose, two methods are used. The lenses can be immersed in a solution of the drug for a period of time and then placed on the eye, resulting in a high initial release, followed by a slower and long-term release during the hours following the application, as in the administration of antibiotics or non-steroidal anti-inflammatory drugs. Alternatively, the drug can be applied to the contact lens after its application in the eye. This method is often adopted when the lens acts as a protective device (bandage lens), for example, following a corneal injury [65][63]. However, both approaches prolong the contact time of the drug by improving its penetration through the cornea [66][64]. The materials used for the manufacture of contact lenses include hydrogels (Figure 5). Because hydrogels are composed of hydrophilic monomers containing electrochemical polarities, they can allow interaction with water. In addition, they are also oxygen permeable and flexible, and capable of retaining a large percentage of water within their polymer network. Due to these characteristics, hydrogels are an attractive material for the production of contact lenses. [67,68][65][66]
References
- Gan, L.; Wang, J.; Jiang, M.; Bartlett, H.; Ouyang, D.; Eperjesi, F.; Liu, J.; Gan, Y. Recent advances in topical ophthalmic drug delivery with lipid-based nanocarriers. Drug Discov. Today 2013, 18, 290–297.
- Gholizadeh, S.; Wang, Z.; Chen, X.; Dana, R.; Annabi, N. Advanced nanodelivery platforms for topical oph-thalmic drug delivery. Drug Discov. Today 2021, 26, 1437–1449.
- Trombino, S.; Cassano, R. Special Issue on Designing Hydrogels for Controlled Drug Delivery: Guest Editors’ Introduction. Pharmaceutics 2020, 12, 57.
- Al-Kinani, A.A.; Zidan, G.; Elsaid, N.; Seyfoddin, A.; Alani, A.W.G.; Alany, R.G. Ophthalmic gels: Past, present and future. Adv. Drug Deliv. Rev. 2018, 126, 113–126.
- Dubald, M.; Bourgeois, S.; Andrieuand, V.; Fessi, H. Ophthalmic Drug Delivery Systems for Antibiothera-py-A Review. Pharmaceutics 2018, 10, 10.
- Kaur, I.P.; Kanwar, M. Ocular Preparations: The Formulation Approach. Drug Dev. Ind. Pharm. 2002, 28, 473–493.
- Rajoria, G.; Gupta, A. In Situ Gelling System: A novel approach for ocular drug delivery. Am. J. Pharm. Tech. Res. 2012, 2, 25–53.
- Cooper, R.C.; Yang, H. Hydrogel-based ocular drug delivery systems: Emerging fabrication strategies, applications, and bench-to-bedside manufacturing considerations. J. Control Release 2019, 306, 29–39.
- Kurniawansyah, I.S.; Sopyan, I.; Aditya, W.A.; Nuraini, H.; Alminda, F.D.; Nurlatifah, A. Preformed gel vs. In Situ gel: A review. Int. Res. J. Pharm. 2018, 9, 1–5.
- Ranch, K.; Patel, H.; Chavda, L.; Koli, A.; Maulvi, F.; Parikh, R.K. Development of In Situ ophthalmic gel of dexamethasone sodium phosphate and chloramphenicol: A viable alternative to conventional eye drops. J. Appl. Pharm. Sci. 2017, 7, 101–108.
- Rukari, T.G.; Jadhav, A.S.; Londhe, R.; Phalke, P.L. A Review on Ophthalmic In Situ Gels. Am. J. Pharm. Tech. Res. 2019, 9, 159–170.
- Bruix, A.; Adán, A.; Casaroli-Marano, R.P. Efficacy of sodium carboxymethylcellulose in the treatment of dry eye syndrome. Arch. Soc. Esp. Oftalmol. 2006, 81, 85–92.
- Aragona, P.; Benítez-Del-Castillo, J.M.; Coroneo, M.T.; Mukherji, S.; Tan, J.; Vandewalle, E.; Vingrys, A.; Liu, H.; Carlisle-Wilcox, C.; Vehige, J.; et al. Safety and Efficacy of a Preservative-Free Artificial Tear Containing Carboxymethylcellulose and Hyaluronic Acid for Dry Eye Disease: A Randomized, Controlled, Multicenter 3-Month Study. Clin. Ophthalmol. 2020, 14, 2951–2963.
- Liu, D.; Wu, Q.; Chen, W.; Lin, H.; Zhu, Y.; Liu, Y.; Liang, H.; Zhu, F. A novel FK506 loaded nanomicelles consisting of amino-terminated poly(ethylene glycol)-block-poly(D,L)-lactic acid and hydroxypropyl methylcellulose for ocular drug delivery. Int. J. Pharm. 2019, 562, 1–10.
- Yusufu, M.; Liu, X.; Zheng, T.; Fan, F.; Xu, J.; Luo, Y. Hydroxypropyl methylcellulose 2% for dry eye preven-tion during phacoemulsification in senile and diabetic patients. Int. Ophthalmol. 2018, 38, 1261–1273.
- Williams, D.; Middleton, S.; Fattahian, H.; Moridpour, R. Comparison of hyaluronic acid-containing topical eye drops with carbomer-based topical ocular gel as a tear replacement in canine keratoconjunctivitis sicca: A prospective study in twenty five dogs. Veter. Res. Forum Int. Q. J. 2012, 3, 229–232.
- López-de la Rosa, A.; Pinto-Fraga, J.; Blázquez Arauzo, F.; Urbano Rodríguez, R.; González-García, M.J. Safety and Efficacy of an Artificial Tear Containing 0.3% Hyaluronic Acid in the Management of Moder-ate-to-Severe Dry Eye Disease. Eye Contact Lens 2017, 43, 383–388.
- Williams, D.L.; Wirostko, B.M.; Gum, G.; Mann, B.K. Topical Cross-Linked HA-Based Hydrogel Accelerates Closure of Corneal Epithelial Defects and Repair of Stromal Ulceration in Companion Animals. Investig. Opthalmol. Vis. Sci. 2017, 58, 4616.
- Dubashynskaya, N.; Poshina, D.; Raik, S.; Urtti, A.; Skorik, Y.A. Polysaccharides in Ocular Drug Delivery. Pharmaceutics 2019, 12, 22.
- He, Y.; Li, J.; Zhu, J.; Jie, Y.; Wang, N.; Wang, J. Wang. The improvement of dry eye after cataract surgery by intraopera-tive using ophthalmic viscosurgical devices on the surface of cornea: The results of a consort-compliant randomized controlled trial. Medicine 2017, 96, 50–58.
- Tundisi, L.; Mostaço, G.; Carricondo, P.; Petri, D. Hydroxypropyl methylcellulose: Physicochemical properties and ocular drug delivery formulations. Eur. J. Pharm. Sci. 2021, 159, 105736.
- Aragona, P.; Papa, V.; Micali, A.; Santocono, M.; Milazzo, G. Long term treatment with sodium hyalu-ronate-containing artificial tears reduces ocular surface damage in patients with dry eye. Br. J. Ophthalmol. 2002, 86, 181–184.
- Irimia, T.; Ghica, M.V.; Popa, L.; Anuţa, V.; Arsene, A.-L.; Dinu-Pîrvu, C.-E. Strategies for Improving Ocular Drug Bioavailability and Corneal Wound Healing with Chitosan-Based Delivery Systems. Polymers 2018, 10, 1221.
- Silva, M.; Calado, R.; Marto, J.; Bettencourt, A.; Almeida, A.; Gonçalve, L. Chitosan nanoparticles as a mu-coadhesive drug delivery system for ocular administration. Mar. Drugs. 2017, 15, 370.
- Chhonker, Y.S.; Prasad, Y.D.; Chandasana, H.; Vishvkarma, A.; Mitra, K.; Shukla, P.K.; Bhatta, R.S. Amphotericin-B entrapped lecithin/chitosan nanoparticles for prolonged ocular application. Int. J. Biol. Macromol. 2015, 72, 1451–1458.
- Tiwari, S.; Goyal, A.K.; Mishra, N.; Khatri, K.; Vaidya, B.; Mehta, A.; Wu, Y.; Vyas, S.P. Development and characterization of novel carrier gel core liposomes based transmission blocking malaria vaccine. J. Control Release 2009, 140, 157–165.
- Mona, A.; Moustafa, M.; El-Refaie, W.M.; Yosra, S.R.; Ossama, R.; Abdallah, Y. Gel in core carbosomes as novel ophthalmic vehicles with enhanced corneal permeation and residence. Int. J. Pharm. 2018, 546, 166–175.
- Duan, Y.; Cai, X.; Du, H.; Zhai, G. Novel In Situ gel systems based on P123/TPGS mixed micelles and gellan gum for ophthalmic delivery of curcumin. Colloids Surf. B Biointerfaces 2015, 128, 322–330.
- Devasani, S.R.; Dev, A.; Rathod, S.; Deshmukh, G. An overview of In Situ gelling systems. Pharm. Biol. Eval. 2016, 3, 60–69.
- Cao, Y.; Zhang, C.; Shen, W.; Cheng, Z.; Yu, L.; Ping, Q. Poly(N-isopropylacrylamide)–chitosan as thermosensitive In Situ gel-forming system for ocular drug delivery. J. Control Release 2007, 120, 186–194.
- Sheshala, R.; Kok, Y.; Ng, J.; Thakur, R.; Dua, K. In Situ Gelling Ophthalmic Drug Delivery System: An Overview and Its Applications. Recent Patents Drug Deliv. Formul. 2015, 9, 242–253.
- Wu, Y.; Liu, Y.; Li, X.; Kebebe, D.; Zhang, B.; Ren, J.; Lu, J.; Li, J.; Du, S.; Liu, Z. Research progress of In Situ gel-ling ophthalmic drug delivery system. Asian J. Pharm. Sci. 2019, 14, 1–15.
- Patel, N.; Nakrani, H.; Raval, M.; Navin, S. Development of loteprednol etabonate-loaded cationic nanoemulsified In Situ ophthalmic gel for sustained delivery and enhanced ocular bioavailability. Drug Deliv. 2016, 23, 3712–3723.
- Zhu, M.; Wang, J.; Li, N. A novel thermo-sensitive hydrogel-based on poly(N-isopropylacrylamide)/hyalu-ronic acid of ketoconazole for ophthalmic delivery. Artificial Cells Nanomed. Biotechnol. 2018, 46, 1282–1287.
- Okur, N.U.; Yozgatlı, V.; Okur, M.E.; Yoltaş, A.; Siafaka, P.I. Improving therapeutic efficacy of voriconazole against fungal keratitis: Thermo-sensitive In Situ gels as ophthalmic drug carriers. J. Drug Deliv. Sci. Technol. 2019, 49, 323–333.
- Gugleva, V.; Titeva, S.; Ermenlieva, N.; Tsibranska, S.; Tcholakova, S.; Rangelov, S.; Momekova, D. Development and evaluation of doxycycline niosomal thermoresponsive In Situ gel for ophthalmic delivery. Int. J. Pharm. 2020, 15, 120010–120021.
- Wei, Y.; Li, C.; Zhu, Q.; Zhang, X.; Guan, J.; Mao, S. Comparison of thermosensitive In Situ gels and drug-resin complex for ocular drug delivery: In Vitro drug release and In Vivo tissue distribution. Int. J. Pharm. 2020, 578, 119184.
- Mahboobian, M.M.; Mohammadi, M.; Mansouri, Z. Development of thermosensitive In Situ gel nanoemul-sions for ocular delivery of acyclovir. J. Drug Del. Sci. Technol. 2020, 55, 101400–101409.
- Kim, Y.C.; Shin, M.D.; Hackett, S.F.; Hsueh, H.T.; E Silva, R.L.; Date, A.; Han, H.; Kim, B.-J.; Xiao, A.; Kim, Y.; et al. Gelling hypotonic polymer solution for extended topical drug delivery to the eye. Nat. Biomed. Eng. 2020, 4, 1053–1062.
- Lajri, G.; Ravindranath, S. Ophthalmic pH Sensitive In situ Gel: A Revie. J. Drug Del. Therap. 2019, 9, 682–689.
- Dawood, B.Y.; Kassab, H.J. Preparation and In Vitro evaluation of naproxen as a pH sensitive ocular In Situ gel. Int. J App. Pharm. 2019, 11, 37–44.
- Wadetwar, R.N.; Agrawal, A.R.; Kanojiya, P.S. In Situ gel containing bimatoprost solid lipid nanoparticles for ocular delivery: In Vitro and Ex-Vivo evaluation. J. Drug Deliv. Sci. Technol. 2020, 56, 101575.
- Kouchak, M.; Mahmoodzadeh, M.; Farrahi, F. Designing of a pH-Triggered Carbopol®®/HPMC In Situ gel for Ocular Delivery of Dorzolamide HCl: In Vitro, In Vivo, and Ex Vivo Evaluation. AAPS PharmSciTech 2019, 20, 210–218.
- Sheshala, R.; Ming, N.J.; Kok, Y.Y.; Singh, T.R.R.; Dua, K. Formulation and characterization of pH induced In Situ gels containing sulfacetamide sodium for ocular drug delivery: A combination of Carbo-pol®®/HPMC polymer. Indian J. Pharm. Ed. Res. 2019, 53, 654–662.
- Jain, P.; Jaiswal, P.C.; Mirza, M.A.; Anwer, K.M.; Iqbal, Z. Preparation of levofloxacin loaded insitu gel for sustained ocular delivery: In Vitro and Ex Vivo evaluations. Drug Devel. Ind. Pharm. 2019, 1, 1–8.
- Majeed, A.; Khan, N.A. Ocular In Situ gel: An overview. J. Drug Deliv. Ther. 2019, 9, 337–347.
- Rozier, A.; Mazuel, C.; Grove, J.; Plazonnet, B. Gelrite®: A novel, ion-activated, In Situ gelling polymer for ophthalmic vehicles. Effect on bioavailability of timolol. Int. J. Pharm. 1989, 57, 163–168.
- Rupenthal, I.D.; Green, C.R.; Alany, R.G. Comparison of ion-activated In Situ gelling systems for ocular drug delivery. Part 1: Physicochemical characterisation and In Vitro release. Int. J. Pharm. 2011, 411, 69–77.
- Zhu, L.; Ao, J.; Li, P. A novel In Situ gel base of deacetylase gellan gum for sustained ophthalmic drug deliv-ery of ketotifen: In Vitro and In Vivo evaluation. Drug Des. Dev. Ther. 2015, 9, 3943–3949.
- Chanrasekaran, R.; Thailambal, V.G. The influence of calcium ions, acetate and l-glycerate groups on the gellan double helix. Carbohydr. Polym. 1990, 12, 431–432.
- Morsi, N.; Ibrahim, M.; Refai, H.; El Sorogy, H. Nanoemulsion-based electrolyte triggered In Situ gel for ocu-lar delivery of acetazolamide. Eur. J. Pharm. Sci. 2017, 104, 302–314.
- Kotreka, U.K.; Davis, V.L.; Adeyeye, M.C. Development of topical ophthalmic In Situ gel-forming estradiol delivery system intended for the prevention of age-related cataracts. PLoS ONE 2017, 12, e0172306.
- Anroop, B.N.; Shah, J.; Shery, J.; Bandar, E.A.; Nagaraja, S.; Mohamed, A.M.; Sumeet, G.; Mahesh, A.; Pot-tathil, S.; Katharigatta, N.V. Experimental design, formulation and In Vivo evaluation of a novel topical In Situ gel system to treat ocular infections. PLoS ONE 2021, 19, e0248857.
- Bochot, A.; Fattal, E. Liposomes for intravitreal drug delivery: A state of the art. J. Control. Release 2012, 161, 628–634.
- Ilochonwu, B.C.; Urtti, A.; Hennink, W.E.; Vermonden, T. Intravitreal hydrogels for sustained release of therapeutic proteins. J. Control Release 2020, 326, 419–441.
- Pachis, K.; Blazaki, S.; Tzatzarakis, M.; Klepetsanis, P.; Naoumidi, E.; Tsilimbaris, M.; Antimisiaris, S.G. Sus-tained release of intravitreal flurbiprofen from a novel drug-in-liposome-in-hydrogel formulation. Eur. J. Pharm. Sci. 2017, 109, 324–333.
- Sapino, S.; Peira, E.; Chirio, D.; Chindamo, G.; Guglielmo, S.; Oliaro-Bosso, S.; Barbero, R.; Vercelli, C.; Re, G.; Brunella, V.; et al. Thermosensitive Nanocomposite Hydrogels for Intravitreal Delivery of Cefuroxime. Nanomaterials 2019, 9, 1461.
- Moreddu, R.; Vigolo, D.; Yetisen, A.K. Contact Lens Technology: From Fundamentals to Applications. Adv. Healthc. Mater. 2019, 8, e1900368.
- Swanson, M.W. A Cross-Sectional Analysis of U.S. Contact Lens User Demographics. Optom. Vis. Sci. 2012, 89, 839–848.
- Sedlacek, J. Possibilities of application of eye drugs with the aid of gel-contact lenses. Cesk Oftalmol. 1965, 21, 509–512.
- Silbert, J.A. A review of therapeutic agents and contact lens wear. J. Am. Optom. Assoc. 1996, 67, 165–171.
- Karlgard, C.C.S.; Wong, N.S.; Jones, L.W.; Moresoli, C. In Vitro uptake and release studies of ocular pharma-ceutical agents by silicon-containing and p-HEMA hydrogel contact lens materials. Int. J. Pharm. 2003, 257, 141–151.
- De Paolis, M.D. The role of contact lenses in the management of the radial keratotomy patient. Optom. Clin. Off. Publ. Prentice Soc. 1994, 4, 25–34.
- Hehl, E.M.; Beck, R.; Luthard, K.; Guthoff, R.; Drewelow, B. Improved penetration of aminoglycosides and fluorozuinolones into the aqueous humour of patients by means of Acuvue contact lenses. Eur. J. Clin. Pharmacol. 1999, 55, 317–323.
- Vidal-Rohr, M.; Wolffsohn, J.S.; Davies, L.N.; Cerviño, A. Effect of contact lens surface properties on com-fort, tear stability and ocular physiology. Cont. Lens Anterior Eye 2018, 41, 117–121.
- Mahinroosta, M.; Farsangi, Z.J.; Allahverdi, A.; Shakoori, Z. Hydrogels as intelligent materials: A brief review of synthesis, properties and applications. Mater. Today Chem. 2018, 8, 42–55.
- Hu, X.; Tan, H.; Hao, L. Functional hydrogel contact lens for drug delivery in the application of oculopathy therapy. J. Mech. Behav. Biomed. Mater. 2016, 64, 43–52.
- Horne, R.R.; Judd, K.E.; Pitt, W.G. Rapid loading and prolonged release of latanoprost from a silicone hy-drogel contact lens. J. Drug Del. Sci. Technol. 2017, 41, 410–418.
- Silva, D.; de Sousa, H.C.; Gil, M.H.; Santos, L.F.; Oom, M.S.; Alvarez-Lorenzo, C.; Saramago, B.; Serro, A.P. Moxifloxacin-imprinted silicone-based hydrogels as contact lens materials for extended drug release. Eur. J. Pharm. Sci. 2020, 156, 105591.
