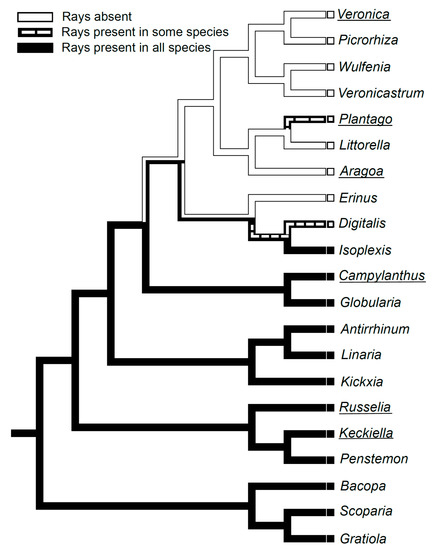
Aragoa is a shrubby genus endemic to páramo in the northern Andes representing the sister group to Plantago and Limosella. Stem and leaf structure of Aragoa corrugatifolia were studied to clarify the evolutionary pathways and ecological significance of their anatomical traits. Aragoa and Plantago share a non-fascicular primary vascular system, rayless wood and secondary phloem, and anomocytic stomata. Aragoa is distinctive from most Plantaginaceae in the presence of cortical aerenchyma and of helical thickenings in vessels. Its procambium emerges in the primary meristem ring as a continuous cylinder. The view on the ring meristem and procambial strands as developmental stages in the formation of a primary vascular system is not relevant for Aragoa, and probably for other Plantaginaceae. The raylessness is synapomorphic for the crown clade of Plantaginaceae comprising Aragoa, Littorella, Plantago, Veronica, Picrorhiza, Wulfenia, and Veronicastrum. The loss of rays is thought to be predetermined by procambium rather than by the vascular cambium. The extremely narrow vessels with helical thickenings are presumably adaptive to hydric and thermic conditions of páramo. Cortical aerenchyma is thought to be a response to the local hypoxia caused by the water retained by ericoid leaves. Trichomes on juvenile leaves are expected to be the traits of considerable taxonomic importance.
- rayless wood
- secondary phloem
- stomata
- trichomes
- procambium
- aerenchyma
- páramo
- Plantago
1. Introduction
2. Analysis on Research Results
2.1. Development of Meristematic and Conductive Tissues in Stem
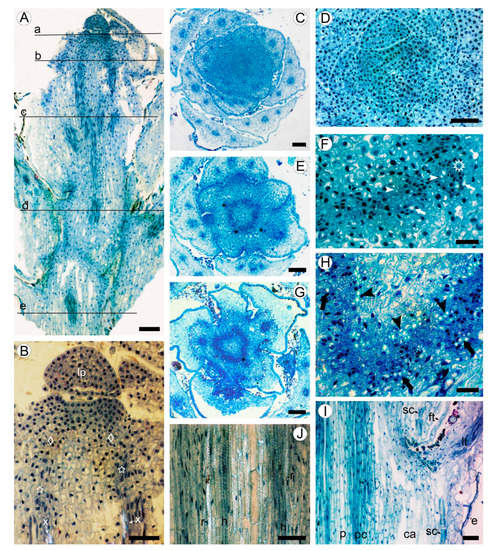
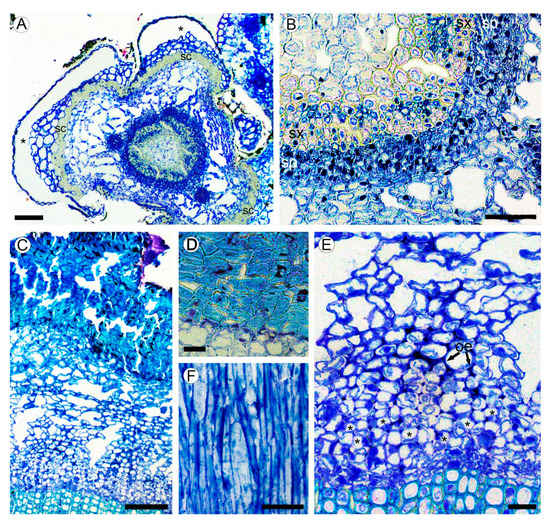
2.2. Bark Structure
2.3. Leaf Structure
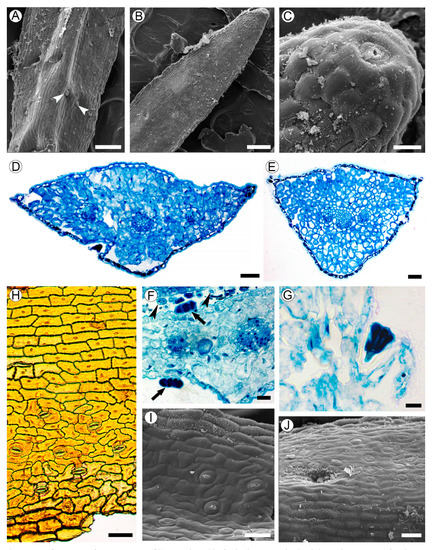
2.4. Wood Structure
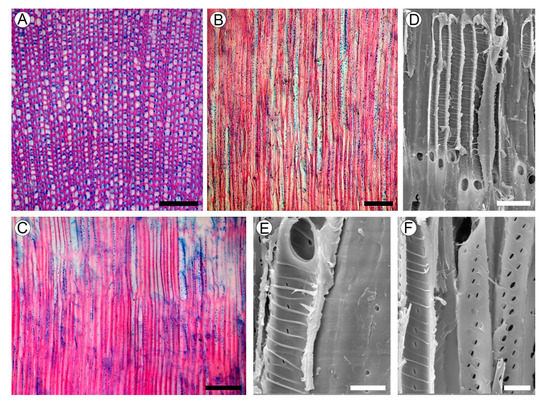
2.5. Evolution of Rayless Wood within Plantaginaceae
References
- Mennega, A.M.W. On unusual wood structures in Scrophulariaceae. Acta Bot. Neerl. 1975, 24, 359–360.
- Dill, F.E. Morphology of Veronicastrum virginicum. Trans. Kansas Acad. Sci. 1941, 44, 158–163.
- Metcalfe, C.R.; Chalk, L. Anatomy of the Dicotyledons; Clarendon Press: Oxford, UK, 1950; Volume 2, pp. 1052–1059.
- Carlquist, S. Wood anatomy of insular species of Plantago and the problem of raylessness. Bull. Torrey Bot. Club 1970, 97, 353–361.
- Carlquist, S. Living cells in wood. 2. Raylessness: Histology and evolutionary significance. Bot. J. Linn. Soc. 2015, 178, 529–555.
- Schweingruber, F.H.; Börner, A.; Schulze, E.-D. Atlas of Stem Anatomy in Herbs, Shrubs und Trees; Springer: Berlin/Heidelberg, Germany, 2011; Volume 2, pp. 1–415.
- Schweingruber, F.; Landolt, W. The Xylem Database. Swiss Federal Institute for Forest, Snow and Landscape Research. 2010. Available online: https://www.wsl.ch/dendropro/xylemdb/ (accessed on 10 April 2021).
- Kaplan, A.; Hasanoğlu, A.; Ikbal, A.I. Morphological, anatomical and palynological properties of some Turkish Veronica L. species (Scrphulariaceae). Int. J. Bot. 2007, 3, 23–32.
- Ida Christi, V.E.; Senthamarai, R. Qualitative and quantitative pharmacognostical studies on Scoparia dulcis Linn leaf. Int. J. Pharm. Pharmaceut. Res. 2015, 3, 57–74.
- Doležal, J.; Dvorský, M.; Börner, A.; Wild, J.; Schweingruber, F.H. Anatomy, Age and Ecology of High Mountain Plants in Ladakh, the Western Himalaya; Springer: Cham, Switzerland, 2018; p. 616.
- Carlquist, S. A theory of paedomorphosis in dicotyledonous woods. Phytomorphology 1962, 12, 30–45.
- Kostytschew, S. Der Bau und das Dickenwachstum der Dikotylenstämme. Ber. Dtsch. Bot. Ges. 1922, 40, 297–305.
- Kostytschew, S. Der Bau und das Dickenwachstum der Dikotylenstämme. Beih. Bot. Zentralbl. 1924, 40, 295–350.
- Helm, J. Untersuchungen über die Differenzierung der Sprossscheitelmeristeme von Dikotylen, unter besonderer Berücksichtig- ung des Prokambiums. Planta 1931, 15, 105–191.
- Krumbiegel, A.; Kästner, A. Sekundäres Dickenwachstum von Sproß und Wurzel bei annuellen Dicotylen. Biosyst. Ecol. 1993, 4, 1–49.
- Hussain, K. Bacopa monnieri (L.) Lennell—A good biomarker of water pollution/contamination. J. Stress Physiol. Biochem. 2010, 6, 91–101.
- Hamed, K.A.; Hassan, S.A.; Mohamed, A.-S.H.; Hosney, N.K. Morphological and anatomical study on Plantaginaceae Juss. and some related taxa of Scrophulariaceae Juss. Egypt. J. Exp. Biol. Bot. 2014, 10, 135–146.
- Ahmed, E.M.; Desoukey, S.Y.; Fouad, M.A.; Kamel, M.S. A pharmacognostical study of Russelia equisetiformis Sch. & Cham. Int. J. Pharmacog. Phytochem. Res. 2016, 8, 174–192.
- Beck, C.B. An Introduction to Plant Structure and Development; Plant Anatomy for the Twenty-First Century; Cambridge University Press: Cambridge, CA, USA, 2010; pp. 1–464.
- Claßen-Bockhoff, R.; Franke, D.; Krämer, H. Early ontogeny defines the diversification of primary vascular bundle systems in angiosperms. Bot. J. Linn. Soc. 2021, 195, 281–307.
- Cumbie, B.G. Development and structure of the xylem in Canavalia (Leguminosae). Bull. Torrey Bot. Club 1967, 94, 162–175.
- Cumbie, B.G. Developmental changes in the xylem and vascular cambium of Apocynum sibiricum. Bull. Torrey Bot. Club 1969, 96, 629–640.
- Soh, W.Y. Early ontogeny of vascular cambium II. Aucuba japonica and Weigela coraeensis. Bot. Mag. 1974, 87, 17–32.
- Soh, W.Y. Early ontogeny of vascular cambium III. Robinia pseudo-acacia and Syringa oblata. Bot. Mag. 1974, 87, 99–112.
- Myśkow, E. Procambium–cambium transition during vascular meristem development in Diospyros lotus. Botany 2010, 88, 985–993.
- InsideWood. 2004-Onwards. Available online: http://insidewood.lib.ncsu.edu/ (accessed on 10 April 2021).
- Michener, D.S. Wood and leaf anatomy of Keckiella (Scrophulariaceae): Ecological considerations. Aliso 1981, 10, 39–57.
- Kalashnikova, O.A.; Bildanova, L.I.; Ryzhov, V.M.; Tarasenko, L.V. Morphological and anatomical analysis of Gratiola officinalis L. In Pharmaceutical Botany: Its Present State and Perspectives; Kurkin, V.A., Ed.; SamGMU: Samara, Russia, 2017; pp. 92–103. (In Russian)
- Albach, D.C.; Meudt, H.M.; Oxelman, B. Piecing together the “new” Plantaginaceae. Am. J. Bot. 2005, 92, 297–315.
- Mower, J.P.; Guo, W.; Partha, R.; Fan, W.; Levsen, N.; Wolff, K.; Nugent, J.M.; Pabón-Mora, N.; González, F. Plastomes from tribe Plantagineae (Plantaginaceae) reveal infrageneric structural synapormorphies and localized hypermutation for Plantago and functional loss of ndh genes from Littorella. Mol. Phylogenet. Evol. 2021, 162, 107217.
- Hoggard, R.K.; Kores, P.J.; Molvray, M.; Hoggard, G.D.; Broughton, D.A. Molecular systematics and biogeography of the amphibious genus Littorella (Plantaginaceae). Am. J. Bot. 2003, 90, 429–435.
