Cooled preservation of semen is usually associated with artificial insemination and genetic improvement programs in livestock species. Several studies have reported an increase in reactive oxidative species and a decrease in antioxidant substances and sperm quality parameters during long-term semen storage at refrigerated temperatures. The supplementation of antioxidants in extenders before refrigeration could reduce this detrimental effect. Various antioxidants have been tested, both enzymatic, such as superoxide dismutase and catalase, and non-enzymatic, such as reduced glutathione, vitamins E and C and melatonin. However, the problem of oxidative stress in semen storage has not been fully resolved. The effects of antioxidants for semen-cooled storage have not been reviewed in depth. Therefore, the objective of the present study was to review the efficiency of the supplementation of antioxidants in the extender during cooled storage of semen in livestock species.
- antioxidant
- oxidative stress
- refrigerated storage
- semen
- livestock species
1. Introduction
2. Treatments with Antioxidants in the Preservation Process
2.1. Enzymatic Antioxidants
| Antioxidant | Concentration | Opt | A/C | Temp | Time | Species | In Vitro Effects | Ref. | |
|---|---|---|---|---|---|---|---|---|---|
| CAT | 50–150 U/mL | 100 | A | 4 °C | 30 h | bovine | Increased sperm motility and decreased dead or abnormal spermatozoa, and acrosomal abnormalities compared with control group. | [79] | [26] |
| CAT | 100 U/mL | A | 4 °C | 72 h | canine | Reduced total ROS, increased sperm motility. | [105] | [27] | |
| CAT | |||||||||
| BHT90–3600 U/mL | A | 5 °C | 72 h | equine | No effect or detrimental effect at high concentrations. | [95] | [ | ||
| SOD + GPx | |||||||||
| 100 and 5 U/mL respectively | |||||||||
| C | |||||||||
| 4 °C | |||||||||
| 96 h | canine | No effect in sperm motility, DNA or acrosome status with compared with control group. Increased viability. | [ | 101 | ] | [ | 22 | ] |
2.2. Non-Enzymatic Antioxidants
2.2.1. Amino Acids and Small Peptides
| Antioxidant | Conc | Opt | Temp | Time | Species | In Vitro Effects | Ref. | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| cysteine | 5 mM | 5 °C | 72 h | caprine | No effect on sperm motility and HOST. | [48] | |||||||||||||||||
| cysteine | 2–4 mM | 5 °C | 96 h | ovine | Slightly increased motility and viability. | [ | 49 | 0.5–2 mM] | A | 5 °C | 72 h | equine | Detrimental effect on motility. | 28] | |||||||||
| [ | 29 | ] | cysteine | 0.25–5 mM | 5 mM | 10 °C | 7–14 d | porcine | Increased sperm viability. | [43] | |||||||||||||
| Idebenone | Bq | 1–8 µM | A | 5 °C | 72 h | ovine | Increased motility, progressivity, viability and T-AOC. | [71 | CAT | 100–200 U/mL | A | ||||||||||||
| ] | hypotaurine | 5 mM | 5 °C | 72 h | equine | No effect on sperm motility. Increased viability in certain cases. | 10 °C | [106] | [29] | ||||||||||||||
| 7–14 d | porcine | No effect on viability. | [43] | ||||||||||||||||||||
| Lycopene | Crt | 250–750 µg/ml | 500 µg/ml | A | 5 °C | CAT | 100–800 U/mL | 100/200 | A | 5 °C | 4 d | ovine | Increased sperm motility only on 4th day. | [ | |||||||||
| arginine | 77 | ] | 4–6 mM | [ | 20 | ] | |||||||||||||||||
| 72 h | canine | Increased motility, progressivity, viability and T-AOC and reduced MDA. | [ | 72 | ] | 4 mM | 5 °C | 5 d | caprine | Increased total motility, viability and reduced MDA. | |||||||||||||
| Melatonin | Idl | [ | 1 µM | 50 | A] | 17 °C | 7 d | porcine | Detrimental effect on sperm motility and viability on day 7. | [73] | CAT | 100–400 mM | 200–400 mM | A | 5 °C | 24 h | ovine | Slight effects on sperm motility. | [ | ||||
| carnitine | 50 mM | 111 | 50 mM | ] | [ | 20–25 °C30] | |||||||||||||||||
| 72 h | |||||||||||||||||||||||
| Melatonin | equine | Increased total motility and reduced ROS and lipid peroxidation. | [ | 51] | Idl | 1–4 mM | 3 mM | A | 4 °C | 30 h | bovine | Increased motility and decreased dead or abnormal spermatozoa, and acrosomal abnormalities. | [74] | GPx | 1–10 U/mL | 10 U/mL | A | 5 °C | 6 d | ovine | Improved sperm motility on 6th day. | [77] | [20] |
| carnitine | 0.5–2 mM | 5 °C | 72 h | equine | Increased sperm motility. No effect on ROS or viability. | [52 | |||||||||||||||||
| Melatonin | ] | Idl | 0.3 mM | A | 4 °C | 48 h | ovine | Increased motility, viability and T-AOC. Reduced MDA. | [75] | GPx | 1–10 U/mL | 10 U/mL | A | 25 °C | |||||||||
| carnitine | 12.5–100 mM | 6 d | ovine | Improved sperm motility on 6th day. | 50 mM | 17 °C | 5–10 d | porcine | Increased motility, viability, acrosome integrity, mitochondrial activity and T-AOC. Reduced MDA and ROS. | [53] | [77] | [20] | |||||||||||
| Melatonin | Idl | 0.05–0.4 mM | 0.1 mM | A | 4 °C | 5 d | ovine | Improved motility, viability, mitochondrial activity and T-AOC and reduced MDA. Toxic at High concentrations. | [76] | SOD | 50–150 U/mL | 100 | A | 4 °C | 30 h | bovine | Increased motility and decreased dead or abnormal spermatozoa, and sperm acrosomal abnormalities compared with control group. | [ | |||||
| carnitine | 87 | ] | 0.5–2 mM | [ | 21 | ] | |||||||||||||||||
| 2 mM | 5 °C | 24 h | rabbit | Increased total motility, viability and acrosomal abnormality. | [54] | ||||||||||||||||||
| Melatonin | Idl | 1–2 mM | 1.5 mM | A | 5 °C | 48 h | equine | Increased motility and viability. Reduced MDA | [77] | SOD | 100 U/mL | A | 4 °C | 96 h | canine | ||||||||
| Melatonin | Idl | 1 µM | No effect with respect to control group. | A | 5 °C | 6 h | equine | [101] | [22] | ||||||||||||||
| glutamine | 20–60 mM | 60 mM | 22 °C (24 h)/5 °C | 72 h | equine | Increased total and progressive motility. No effect or detrimental effect on sperm viability. | [55] | No effect on sperm motility, viability or ROS. Increased mitochondrial activity and intact acrosome. | [78] | SOD | 25–50 U/mL | A | 5 °C | 72 h | equine | Increased sperm motility and viability compared with control group. | |||||||
| Melatonin | Idl | [ | 0.1–3 mM | 102 | ] | [23] | |||||||||||||||||
| glutamine | 10–80 mM | 20 mM | 17 °C | 1 mM5 d | A | 5 °C | SOD | 100–800 U/mL | 800 U/mL | A | 5 °C | 6 d | ovine | Improved sperm motility. | [77] | [20] | |||||||
| porcine | Increased motility, velocities, viability and T-AOC. Reduced ROS production. Toxic at high concentrations. | [ | 56 | ] | |||||||||||||||||||
| 48 h | ovine | Improved progressive motility. | [ | 79 | ] | glutamine | 0.5–2 mM | 1–2 mM | 5 °C | 24 h | rabbit | Increased total motility, viability and acrosomal abnormality. | [54 | ||||||||||
| Melatonin | ] | Idl | 0.5–1.5 mM | 1 mM | A | 5 °C | 24 h | rabbit | Increased sperm motility, viability and reduced DNA fragmentation. | [ | Mix: GSH + CAT | 10 mM GSH + 100 IU/mL CAT | C | 4 °C | 72 h | ovine | No effect on viability or total motility. Reduced MDA. | [ | |||||
| Methionine | 80 | ] | 2–4 mM | [ | 31 | ] | |||||||||||||||||
| 80 | ] | 5 °C | 96 h | ovine | Increased motility and viability. | [57] | |||||||||||||||||
| HT and DHPG | Phl | 5–100 µg/mL for each one | A/C | 15 °C | 96 h | ovine | No effect in general, only affected VLC. | [81] | Mix: Vit EP, SOD + Cat and GPx | ||||||||||||||
| MethionineVit EP 12.5 μmol/L, SOD 37 μmol/L+ CAT 500 IU/mL, and GPx 20 IU/ml | C | 5 °C | 72 h | ovine | No effect on the studied sperm parameters (viability, acrosome). | 1–12 mM | 5 °C[9] | ||||||||||||||||
| 96 h | Rabbit | No effect on studied parameters. | [58 | ||||||||||||||||||||
| HT and DHPG | ] | Phl | 5–100 µg/mL for each one | A/C | 5 °C | 96 h | ovine | Only affected some kinetic parameters of motility. | [81] | SOD, CAT and GPx | 15 IU/mL each of one | C | 4 °C | 10 d | canine | Increased total and progressive sperm motility, reduced DNA fragmentation mainly in hypofertile males. | |||||||
| Methionine | 1–12 mM | 15 °C | [ | 96 h | Rabbit | 103 | No effect on studied parameters. | [58] | ] | [24] | |||||||||||||
| polyphenol- murtilla | Phl | 0.0315 μg GAE mL | A | 17 °C | 7 d | porcine | Increased motility, viability. Reduced ROS production. | [82] | SOD, CAT and GPx | 15 IU/mL each of one | C | 5 °C | 72 h | equine | Increased motility and viability, and reduced DNA damage of spermatozoa compared with control group after 72 h storage. | [104 | |||||||
| proline | 20–60 mM | 60 mM | ] | 22 °C (24 h)/5 °C | |||||||||||||||||||
| procyanidin extract | 72 h | Phl | 10–70 mg/L | 30 mg/L | Aequine | Increased total and progressive motility. No effect on viability. | [ | 5 °C55 | 120 h][ | caprine25] | |||||||||||||
| Increased motility, viability, acrosome integrity, mitochondrial activity and T-AOC. Reduced MDA. | [ | 11 | ] | proline | 25–125 mM | 75 | 17 °C | 5 d | porcine | Increased total and progressive motility, GSH levels and activities of CAT and SOD. Improved viability, MMP and ATP levels. Reduced ROS. | |||||||||||||
| Resveratrol | Phl | 10–80 µM | [59] | ||||||||||||||||||||
| A | 10 °C/4 °C | 24 h | equine | Detrimental effects at high concentration. | [83] | GSH | 5–10 mM | 5 mM | 5 °C | 48 h | equine | Increased total motility and viability, reduced MDA. Toxic at high concentrations. | |||||||||||
| Resveratrol | Phl | 200–400 uM | A | 5 °C | 168 h[44] | ||||||||||||||||||
| ovine | Improved motility, kinematic parameters and in vitro fertility, antioxidant activities and reduced oxidative stress | [ | 84 | ] | GSH | ||||||||||||||||||
| Resveratrol0.5–3.0 mM | 0.5 mM | 4–8 °C | 5 d | Phlbovine | Increased motility. Reduced acrosomal damage. Toxic at high concentrations. | [39] | |||||||||||||||||
| 0.01–1 mM | A | 17 °C | 72 h | porcine | No positive effect on motility or kinetics, viability. Reduced SOD. Toxic at high concentrations. | [85] | GSH | 0.2–5 mM | 1–1.5 mM | 25 °C | 24 h | bovine | Slightly improved progressive motility (depending on extender). | [38] | |||||||||
| GSH | |||||||||||||||||||||||
| Resveratrol | Phl | 10–100 µM | A | 17 °C | 4–7 d | porcine | No positive effect on motility or kinetics. Toxic at high concentrations. | [86] | 0.2–5 mM | 5 °C | 24 h | bovine | No effect on studied parameters. | [38] | |||||||||
| Resveratrol | Phl | 25–150 uM | 50 uM | A | 17 °C | 5 d | porcine | Increased sperm motility, membrane integrity and mitochondrial activity and T-AOC levels. Decreased ROS and MDA. | [87] | GSH | 1 mM | 5 °C | 72 h | ||||||||||
| Quercetin | caprine | Phl | 25–200 µM | AIncreased progressive motility and viability. Reduced lipid peroxidation. | 5 °C[40] | ||||||||||||||||||
| 96 h | rabbit | No effect on motility, kinetics or DNA fragmentation. Reduced intracellular H | 2 | O | 2. Toxic at high concentrations. | [58] | GSH | 0.2–5 mM | 5 °C/15 °C | ||||||||||||||
| Quercetin | Phl | 25–200 µM96 h | ovine | A | 15 °CNone or detrimental effect on motility and viability (improved mitochondrial activity at 5 °C) (depending on extender). | 96 h[45] | |||||||||||||||||
| rabbit | No effect on motility, kinetics or DNA fragmentation. Reduced intracellular H | 2 | O | 2 | . Toxic at high concentrations. | [58 | GSH | 50–200 mM | 200 mM | 5 °C | 72 h | ovine | Increased motility and kinetics, viability, T-AOC, and MMP. | [42] | |||||||||
| GSH | 100–400 mM | 5 °C | 24 h | ovine | Detrimental effects at high concentration. | [30] | |||||||||||||||||
| GSH | 5–10 mM | 5–10 mM | 5 °C | 30 h | ovine | No effect on motility, increased viability. | [46] | ||||||||||||||||
| GSH | 5 mM | 10 °C | 7–14 d | porcine | Increased viability at 14 d | [43] | |||||||||||||||||
| GSH | 1–15 mmol/L | 1 mM | 17 °C | 5 d | porcine | Increased motility, viability, T-AOC. | [41] | ||||||||||||||||
| GSH | 0.5–1.5 mM | 5 °C | 24 h | tigrina | No significant effect on studied parameters. | [47] | |||||||||||||||||
| Taurine | 20–60 mM | 60 mM | 22 °C (24 h)/5 °C | 72 h | equine | Increased total and progressive motility. No effect on viability. | [55] | ||||||||||||||||
| Taurine | 25–100 mM | 50 mM | 5 °C | No data | bovine | Improved sperm motility, viability, acrosome integrity. | [60] | ||||||||||||||||
| Taurine | 0.2 mM | 4 °C | 72 h | canine | Increased motility and viability. | [27] | |||||||||||||||||
| Taurine | 100 mM | 5 °C | 5 d | equine | Increased total motility. | [61] | |||||||||||||||||
| Taurine | 50–100 mM | 5 °C | 30 h | ovine | No effect on motility or viability. | [46] | |||||||||||||||||
| Taurine | 0.5–10 mmol/L | 5 mM | 17 °C | 72 h | porcine | Increased motility, viability, acrosome integrity and T-AOC. Reduced MDA. | [62] |
2.2.2. Vitamins, Carotenoids and Polyphenols
| Antioxidant | T | Concentr | Opt | A/C | Temp | Time | Species | In Vitro Effects | Ref. |
|---|---|---|---|---|---|---|---|---|---|
| Vit C + GSH | 0.5–2 mg/mL + 1 mM | C | 5 °C | 24 h | bovine | No effect on studied parameters. | [38] | ||
| ] | |||||||||
| Vit C | |||||||||
| Vit | |||||||||
| 0.5 mM | |||||||||
| A | |||||||||
| 4 °C | |||||||||
| 72 h | |||||||||
| canine | |||||||||
| No effect on sperm motility, increased sperm viability. | |||||||||
| [ | 27 | ] | |||||||
| Vit C | Vit | 0.5–2 mg/mL | A | 5 °C | 24 h | bovine | No effect. | [38] | |
| Vit C | Vit | 0.45–0.9 mg/mL | A | 5 °C | 72 h | equine | Increased sperm viability, at high concentrations reduced motility. | [28] | |
| Vit C | Vit | 1–4 mM | A | 5 °C | 72 h | equine | No significant effect. | [29] | |
| Vit E | Vit | 0.1 mM | A | 4 °C | 72 h | canine | Reduced total ROS, increased motility and viability. | [27] | |
| Vit E: trolox | Vit | 2 mM | A | 5 °C | 72 h | equine | No effect on motility. | [29] | |
| Vit E: Trolox | Vit | 0.2–5 mM | A | 5 °C/15 °C | 96 h | ovine | None or detrimental effect on motility and viability (improved mitochondrial activity at 5ºC); depending on extender. | [45] | |
| Vit E: α-tocopherol | Vit | 3 mM | A | 5 °C | 72 h | caprine | Increased progressive motility and viability, reduced lipid peroxidation. | [40] | |
| Vit E: α-tocopherol | Vit | 5–10 mM | A | 5 °C | 48 h | equine | No effect on motility or viability or MDA. | [68] | |
| Vit E: α-tocopherol | Vit | 1–4 mM | A | 5 °C | 72 h | equine | No effect on motility. | [29] | |
| Vit E: α-tocopherol | Vit | 200 µM | A | 17–15 °C | 72 h | porcine | No effect on motility and host (in non-dialyzed samples). | [69] | |
| Vit E: α-tocopherol | Vit | 0.2 mg/mL | A | 19 °C | 5 d | porcine | Improved sperm viability. | [70] |
2.2.3. Other Antioxidant Substances
3. Treatments with Antioxidants in Livestock Species
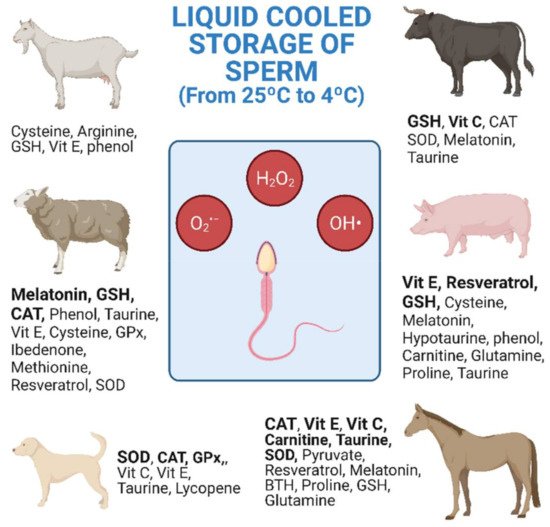
References
- Nijs, M.; Ombelet, W. Cryopreservation of human sperm. Hum. Fertil. 2001, 4, 158–163.
- Del-Pozo-Lérida, S.; Salvador, C.; Martínez-Soler, F.; Tortosa, A.; Perucho, M.; Giménez-Bonafé, P. Preservation of fertility in patients with cancer (Review). Oncol. Rep. 2019, 41, 2607–2614.
- Sadeghi, S.; Del Gallego, R.; García-Colomer, B.; Gómez, E.A.; Yániz, J.L.; Gosálvez, J.; López-Fernández, C.; Silvestre, M.A. Effect of Sperm Concentration and Storage Temperature on Goat Spermatozoa during Liquid Storage. Biology 2020, 9, 300.
- Allan, I.W.; Irvine, D.S.; Macnamee, M.; Aitken, R.J. Field trial of a diluent for the transportation of human semen at ambient temperatures. Fertil. Steril. 1997, 67, 348–354.
- Hezavehei, M.; Sharafi, M.; Kouchesfahani, H.M.; Henkel, R.; Agarwal, A.; Esmaeili, V.; Shahverdi, A. Sperm cryopreservation: A review on current molecular cryobiology and advanced approaches. Reprod. Biomed. Online 2018, 37, 327–339.
- Amidi, F.; Pazhohan, A.; Shabani Nashtaei, M.; Khodarahmian, M.; Nekoonam, S. The role of antioxidants in sperm freezing: A review. Cell Tissue Bank. 2016, 17, 745–756.
- Li, Y.R.; Trush, M. Defining ROS in Biology and Medicine. React. Oxyg. Species 2016, 1, 9.
- Phaniendra, A.; Jestadi, D.B.; Periyasamy, L. Free Radicals: Properties, Sources, Targets, and Their Implication in Various Diseases. Indian J. Clin. Biochem. 2015, 30, 11–26.
- Falchi, L.; Galleri, G.; Zedda, M.T.; Pau, S.; Bogliolo, L.; Ariu, F.; Ledda, S. Liquid storage of ram semen for 96 h: Effects on kinematic parameters, membranes and DNA integrity, and ROS production. Livest. Sci. 2018, 207, 1–6.
- Liu, T.; Han, Y.; Zhou, T.; Zhang, R.; Chen, H.; Chen, S.; Zhao, H. Mechanisms of ROS-induced mitochondria-dependent apoptosis underlying liquid storage of goat spermatozoa. Aging 2019, 11, 7880–7898.
- Wen, F.; Li, Y.; Feng, T.; Du, Y.; Ren, F.; Zhang, L.; Han, N.; Ma, S.; Li, F.; Wang, P.; et al. Grape Seed Procyanidin Extract (GSPE) Improves Goat Sperm Quality When Preserved at 4 °C. Animals 2019, 9, 810.
- Rodríguez-Varela, C.; Labarta, E. Clinical Application of Antioxidants to Improve Human Oocyte Mitochondrial Function: A Review. Antioxidants 2020, 9, 1197.
- Amorini, A.M.; Listorti, I.; Bilotta, G.; Pallisco, R.; Saab, M.W.; Mangione, R.; Manca, B.; Lazzarino, G.; Tavazzi, B.; Lazzarino, G.; et al. Antioxidant-Based Therapies in Male Infertility: Do We Have Sufficient Evidence Supporting Their Effectiveness? Antioxidants 2021, 10, 220.
- Suleiman, S.A.; Elamin Ali, M.; Zaki, Z.M.S.; El-Malik, E.M.A.; Nasr, M.A. Lipid peroxidation and human sperm motility: Protective role of vitamin E. J. Androl. 1996, 17, 530–537.
- Allai, L.; Benmoula, A.; Marciane da Silva, M.; Nasser, B.; El Amiri, B. Supplementation of ram semen extender to improve seminal quality and fertility rate. Anim. Reprod. Sci. 2018, 192, 6–17.
- Aitken, R.J.; Drevet, J.R. The Importance of Oxidative Stress in Determining the Functionality of Mammalian Spermatozoa: A Two-Edged Sword. Antioxidants 2020, 9, 111.
- Pezo, F.; Yeste, M.; Zambrano, F.; Uribe, P.; Risopatrón, J.; Sánchez, R. Antioxidants and their effect on the oxidative/nitrosative stress of frozen-thawed boar sperm. Cryobiology 2021, 98, 5–11.
- Barranco, I.; Padilla, L.; Tvarijonaviciute, A.; Parrilla, I.; Martínez, E.A.; Rodriguez-Martinez, H.; Yeste, M.; Roca, J. Levels of activity of superoxide dismutase in seminal plasma do not predict fertility of pig AI-semen doses. Theriogenology 2019, 140, 18–24.
- Kehrer, J.P. The Haber-Weiss reaction and mechanisms of toxicity. Toxicology 2000, 149, 43–50.
- Maxwell, W.; Stojanov, T. Liquid storage of ram semen in the absence or presence of some antioxidants. Reprod. Fertil. Dev. 1996, 8, 1013.
- Perumal, P. Effect of Superoxide Dismutase on Semen Parameters and Antioxidant Enzyme Activities of Liquid Stored (5 °C) Mithun (Bos frontalis) Semen. J. Anim. 2014, 2014, 1–9.
- Chatdarong, K.; Chaivechakarn, A.; Thuwanut, P.; Ponglowhapan, S. Effects of Cold Storage Prior to Freezing on Superoxide Dismutase, Glutathione Peroxidase Activities, Level of Total Reactive Oxygen Species and Sperm Quality in Dogs. Reprod. Domest. Anim. 2012, 47, 274–277.
- Cocchia, N.; Pasolini, M.P.; Mancini, R.; Petrazzuolo, O.; Cristofaro, I.; Rosapane, I.; Sica, A.; Tortora, G.; Lorizio, R.; Paraggio, G.; et al. Effect of sod (superoxide dismutase) protein supplementation in semen extenders on motility, viability, acrosome status and ERK (extracellular signal-regulated kinase) protein phosphorylation of chilled stallion spermatozoa. Theriogenology 2011, 75, 1201–1210.
- Del Prete, C.; Ciani, F.; Tafuri, S.; Pasolini, M.P.; Della Valle, G.; Palumbo, V.; Abbondante, L.; Calamo, A.; Barbato, V.; Gualtieri, R.; et al. Effect of superoxide dismutase, catalase, and glutathione peroxidase supplementation in the extender on chilled semen of fertile and hypofertile dogs. J. Vet. Sci. 2018, 19, 667.
- Del Prete, C.; Stout, T.; Montagnaro, S.; Pagnini, U.; Uccello, M.; Florio, P.; Ciani, F.; Tafuri, S.; Palumbo, V.; Pasolini, M.P.; et al. Combined addition of superoxide dismutase, catalase and glutathione peroxidase improves quality of cooled stored stallion semen. Anim. Reprod. Sci. 2019, 210, 106195.
- Peruma, P.; Chamuah, J.K.; Rajkhowa, C. Effect of catalase on the liquid storage of mithun (Bos frontalis) semen. Asian Pac. J. Reprod. 2013, 2, 209–214.
- Michael, A.J.; Alexopoulos, C.; Pontiki, E.A.; Hadjipavlou-Litina, D.J.; Saratsis, P.; Ververidis, H.N.; Boscos, C.M. Effect of antioxidant supplementation in semen extenders on semen quality and reactive oxygen species of chilled canine spermatozoa. Anim. Reprod. Sci. 2009, 112, 119–135.
- Aurich, J.E.; Schönherr, U.; Hoppe, H.; Aurich, C. Effects of antioxidants on motility and membrane integrity of chilled-stored stallion semen. Theriogenology 1997, 48, 185–192.
- Ball, B.A.; Medina, V.; Gravance, C.G.; Baumber, J. Effect of antioxidants on preservation of motility, viability and acrosomal integrity of equine spermatozoa during storage at 5 °C. Theriogenology 2001, 56, 577–589.
- Câmara, D.R.; Mello-Pinto, M.M.C.; Pinto, L.C.; Brasil, O.O.; Nunes, J.F.; Guerra, M.M.P. Effects of reduced glutathione and catalase on the kinematics and membrane functionality of sperm during liquid storage of ram semen. Small Rumin. Res. 2011, 100, 44–49.
- Paul, R.; Kumar, D.; Naqvi, S. Antioxidants protect proteins’ anchorage to the bilayer by improving plasma membrane integrity of ram spermatozoa during liquid preservation in a soya lecithin-based diluent. Reprod. Domest. Anim. 2017, 52, 1052–1060.
- Pisoschi, A.M.; Pop, A. The role of antioxidants in the chemistry of oxidative stress: A review. Eur. J. Med. Chem. 2015, 97, 55–74.
- Agarwal, A.; Aponte-Mellado, A.; Premkumar, B.J.; Shaman, A.; Gupta, S. The effects of oxidative stress on female reproduction: A review. Reprod. Biol. Endocrinol. 2012, 10, 1.
- Angrimani, D.S.R.; Silva, R.O.C.; Losano, J.D.A.; Dalmazzo, A.; Tsunoda, R.H.; Perez, E.G.A.; Góes, P.A.A.; Barnabe, V.H.; Nichi, M. Extender Supplementation with Antioxidants Selected after the Evaluation of Sperm Susceptibility to Oxidative Challenges in Goats. Anim. Biotechnol. 2019, 30, 21–29.
- Thuwanut, P.; Chatdarong, K.; Johannisson, A.; Bergqvist, A.S.; Söderquist, L.; Axnér, E. Cryopreservation of epididymal cat spermatozoa: Effects of in vitro antioxidative enzymes supplementation and lipid peroxidation induction. Theriogenology 2010, 73, 1076–1087.
- Mousavi, S.M.; Towhidi, A.; Zhandi, M.; Amoabediny, G.; Mohammadi-Sangcheshmeh, A.; Sharafi, M.; Hussaini, S.M.H. Comparison of two different antioxidants in a nano lecithin-based extender for bull sperm cryopreservation. Anim. Reprod. Sci. 2019, 209, 106171.
- Mirończuk-Chodakowska, I.; Witkowska, A.M.; Zujko, M.E. Endogenous non-enzymatic antioxidants in the human body. Adv. Med. Sci. 2018, 63, 68–78.
- Foote, R.H.; Brockett, C.C.; Kaproth, M.T. Motility and fertility of bull sperm in whole milk extender containing antioxidants. Anim. Reprod. Sci. 2002, 71, 13–23.
- Munsi, M.; Bhuiyan, M.; Majumder, S.; Alam, M. Effects of Exogenous Glutathione on the Quality of Chilled Bull Semen. Reprod. Domest. Anim. 2007, 42, 358–362.
- Sarangi, A.; Singh, P.; Virmani, M.; Yadav, A.S.; Sahu, S.; Ajithakumar, H.M.; Kumari, A.; Rath, A.P. Effect of antioxidants supplementation on the quality of Beetal buck semen stored at 4 °C. Vet. World 2017, 10, 1184–1188.
- Zhang, X.-G.; Liu, Q.; Wang, L.-Q.; Yang, G.-S.; Hu, J.-H. Effects of glutathione on sperm quality during liquid storage in boars. Anim. Sci. J. 2016, 87, 1195–1201.
- Shi, L.; Jin, T.; Hu, Y.; Ma, Z.; Niu, H.; Ren, Y. Effects of reduced glutathione on ram sperm parameters, antioxidant status, mitochondrial activity and the abundance of hexose transporters during liquid storage at 5 °C. Small Rumin. Res. 2020, 189, 106139.
- Funahashi, H.; Sano, T. Select antioxidants improve the function of extended boar semen stored at 10 °C. Theriogenology 2005, 63, 1605–1616.
- Zhandi, M.; Ghadimi, V. Effect of Glutathione-Supplemented INRA82 Extender on Miniature Caspian Stallion Sperm Quality during Storage at 5 °C. J. Equine Vet. Sci. 2014, 34, 606–610.
- Mata-Campuzano, M.; Álvarez-Rodríguez, M.; Tamayo-Canul, J.; López-Urueña, E.; de Paz, P.; Anel, L.; Martínez-Pastor, F.; Álvarez, M. Refrigerated storage of ram sperm in presence of Trolox and GSH antioxidants: Effect of temperature, extender and storage time. Anim. Reprod. Sci. 2014, 151, 137–147.
- Bucak, M.N.; Tekin, N. Protective effect of taurine, glutathione and trehalose on the liquid storage of ram semen. Small Rumin. Res. 2007, 73, 103–108.
- Angrimani, D.S.R.; Barros, P.M.H.; Losano, J.D.A.; Cortada, C.N.M.; Bertolla, R.P.; Guimarães, M.A.B.V.; Correa, S.H.R.; Barnabe, V.H.; Nichi, M. Effect of different semen extenders for the storage of chilled sperm in Tigrina (Leopardus tigrinus). Theriogenology 2017, 89, 146–154.
- Salvador, I.; Yániz, J.; Viudes-de-Castro, M.P.; Gómez, E.A.; Silvestre, M.A. Effect of solid storage on caprine semen conservation at 5 °C. Theriogenology 2006, 66, 974–981.
- Gungor, S.; Ozturk, C.; Omur, A. Positive effects of trehalose and cysteine on ram sperm parameters. Vet. Med. 2017, 62, 245–252.
- Susilowati, S.; Triana, I.N.; Wurlina, W.; Arimbi, A.; Srianto, P.; Mustofa, I. Addition of L-arginine in skim milk extender maintains goat spermatozoa quality in chilled temperature for five days. Vet. World 2019, 12, 1784–1789.
- Gibb, Z.; Lambourne, S.R.; Quadrelli, J.; Smith, N.D.; Aitken, R.J. L-carnitine and pyruvate are prosurvival factors during the storage of stallion spermatozoa at room temperature. Biol. Reprod. 2015, 93, 1–9.
- Nery, I.H.A.V.; Araújo Silva, R.A.J.; Souza, H.M.; Arruda, L.C.P.; Monteiro, M.M.; Seal, D.C.M.; Silva, G.R.; Silva, T.M.S.; Carneiro, G.F.; Batista, A.M.; et al. Effects of L-Carnitine on Equine Semen Quality During Liquid Storage. Biopreserv. Biobank. 2020, 18, 403–408.
- Yang, K.; Wang, N.; Guo, H.T.; Wang, J.R.; Sun, H.H.; Sun, L.Z.; Yue, S.-L.; Zhou, J. B Effect of L-carnitine on sperm quality during liquid storage of boar semen. Asian Australas. J. Anim. Sci. 2020, 33, 1763–1769.
- Sarıözkan, S.; Özdamar, S.; Türk, G.; Cantürk, F.; Yay, A. In vitro effects of l-carnitine and glutamine on motility, acrosomal abnormality, and plasma membrane integrity of rabbit sperm during liquid-storage. Cryobiology 2014, 68, 349–353.
- Dorado, J.; Acha, D.; Ortiz, I.; Gálvez, M.J.; Carrasco, J.J.; Gómez-Arrones, V.; Calero-Carretero, R.; Hidalgo, M. Effect of extender and amino acid supplementation on sperm quality of cooled-preserved Andalusian donkey (Equus asinus) spermatozoa. Anim. Reprod. Sci. 2014, 146, 79–88.
- Wang, S.; Sun, M.; Wang, N.; Yang, K.; Guo, H.; Wang, J.; Zhang, Y.; Yue, S.; Zhou, J. Effects of L-glutamine on boar sperm quality during liquid storage at 17°C. Anim. Reprod. Sci. 2018, 191, 76–84.
- Bucak, M.N.; Çoyan, K.; Öztürk, C.; Güngör, Ş.; Ömür, A.D. Methionine supplementation improves ram sperm parameters during liquid storage at 5 °C. Cryobiology 2012, 65, 335–337.
- Johinke, D.; de Graaf, S.P.; Bathgate, R. Quercetin reduces the in vitro production of H2O2 during chilled storage of rabbit spermatozoa. Anim. Reprod. Sci. 2014, 151, 208–219.
- Feng, C.; Zhu, Z.; Bai, W.; Li, R.; Zheng, Y.; Tian, X.; Wu, D.; Lu, H.; Wang, Y.; Zeng, W. Proline Protects Boar Sperm against Oxidative Stress through Proline Dehydrogenase-Mediated Metabolism and the Amine Structure of Pyrrolidine. Animals 2020, 10, 1549.
- Perumal, P.; Vupru, K.; Rajkhowa, C. Effect of Addition of Taurine on the Liquid Storage (5 °C) of Mithun (Bos frontalis) Semen. Vet. Med. Int. 2013, 2013, 1–7.
- Ijaz, A.; Ducharme, R. Effect of various extenders and taurine on survival of stallion sperm cooled to 5 °C. Theriogenology 1995, 44, 1039–1050.
- Li, H.; Zhang, X.G.; Fang, Q.; Liu, Q.; Du, R.R.; Yang, G.S.; Wang, L.Q.; Hu, J.H. Supplemental effect of different levels of taurine in Modena on boar semen quality during liquid preservation at 17 °C. Anim. Sci. J. 2017, 88, 1692–1699.
- Ortiz-Rodriguez, J.M.; Martín-Cano, F.E.; Ortega-Ferrusola, C.; Masot, J.; Redondo, E.; Gázquez, A.; Gil, M.C.; Aparicio, I.M.; Rojo-Domínguez, P.; Tapia, J.A.; et al. The incorporation of cystine by the soluble carrier family 7 member 11 (SLC7A11) is a component of the redox regulatory mechanism in stallion spermatozoa. Biol. Reprod. 2019, 101, 208–222.
- Zhai, W.; Neuman, S.L.; Latour, M.A.; Hester, P.Y. The effect of dietary L-carnitine on semen traits of white leghorns. Poult. Sci. 2007, 86, 2228–2235.
- Gülçin, I. Antioxidant and antiradical activities of L-carnitine. Life Sci. 2006, 78, 803–811.
- Levine, R.L.; Berlett, B.S.; Moskovitz, J.; Mosoni, L.; Stadtman, E.R. Methionine residues may protect proteins from critical oxidative damage. Mech. Ageing Dev. 1999, 107, 323–332.
- Rietjens, I.M.C.; Boersma, M.G.; de Haan, L.; Spenkelink, B.; Awad, H.M.; Cnubben, N.H.P.; van Zanden, J.J.; Van Der Woude, H.; Alink, G.M.; Koeman, J.H. The pro-oxidant chemistry of the natural antioxidants vitamin C, vitamin E, carotenoids and flavonoids. Environ. Toxicol. Pharmacol. 2002, 11, 321–333.
- Yousefian, I.; Zare-Shahneh, A.; Zhandi, M. The Effect of Coenzyme Q10 and α-Tocopherol in Skim Milk–Based Extender for Preservation of Caspian Stallion Semen in Cool Condition. J. Equine Vet. Sci. 2014, 34, 949–954.
- Zakošek Pipan, M.; Mrkun, J.; Nemec Svete, A.; Zrimšek, P. Improvement of liquid stored boar semen quality by removing low molecular weight proteins and supplementation with α-tocopherol. Anim. Reprod. Sci. 2017, 186, 52–61.
- Cerolini, S.; Maldjian, A.; Surai, P.; Noble, R. Viability, susceptibility to peroxidation and fatty acid composition of boar semen during liquid storage. Anim. Reprod. Sci. 2000, 58, 99–111.
- Eslami, M.; Jahan-Roshan, N.; Farrokhi-Ardabili, F. Influence of idebenone on ram semen quality stored at 4 °C. Reprod. Domest. Anim. 2019, 54, 486–497.
- Sheikholeslami, S.A.; Soleimanzadeh, A.; Rakhshanpour, A.; Shirani, D. The evaluation of lycopene and cysteamine supplementation effects on sperm and oxidative stress parameters during chilled storage of canine semen. Reprod. Domest. Anim. 2020, 55, 1229–1239.
- Martín-Hidalgo, D.; Barón, F.J.; Bragado, M.J.; Carmona, P.; Robina, A.; García-Marín, L.J.; Gil, M.C. The effect of melatonin on the quality of extended boar semen after long-term storage at 17 °C. Theriogenology 2011, 75, 1550–1560.
- Perumal, P.; Vupru, K.; Khate, K. Effect of Addition of Melatonin on the Liquid Storage (5 °C) of Mithun (Bos frontalis) Semen. Int. J. Zool. 2013, 2013.
- Rateb, S.A.; Khalifa, M.A.; Abd El-Hamid, I.S.; Shedeed, H.A. Enhancing liquid-chilled storage and cryopreservation capacities of ram spermatozoa by supplementing the diluent with different additives. Asian Australas. J. Anim. Sci. 2020, 33, 1068–1076.
- Dai, G.; Meng, Y.; Zhang, L.; Du, Y.; Wen, F.; Feng, T.; Hu, J. Effect of addition of melatonin on liquid storage of ram semen at 4 °C. Andrologia 2019, 51, e13236.
- Izadpanah, G.; Zare-Shahneh, A.; Zhandi, M.; Yousefian, I.; Emamverdi, M. Melatonin Has a Beneficial Effect on Stallion Sperm Quality in Cool Condition. J. Equine Vet. Sci. 2015, 35, 555–559.
- Affonso, F.J.; Carvalho, H.F.; Lançoni, R.; Lemes, K.M.; Leite, T.G.; Oliveira, L.Z.; Celeghini, E.C.C.; de Arruda, R.P. Addition of Antioxidants Myoinositol, Ferulic Acid, and Melatonin and Their Effects on Sperm Motility, Membrane Integrity, and Reactive Oxygen Species Production in Cooled Equine Semen. J. Equine Vet. Sci. 2017, 59, 57–63.
- Ashrafi, I.; Kohram, H.; Naijian, H.; Bahreini, M.; Poorhamdollah, M. Protective effect of melatonin on sperm motility parameters on liquid storage of ram semen at 5 °C. Afr. J. Biotechnol. 2011, 10, 6670–6674.
- Fadl, A.M.; Ghallab, A.R.M.; Abou-Ahmed, M.M.; Moawad, A.R. Melatonin can improve viability and functional integrity of cooled and frozen/thawed rabbit spermatozoa. Reprod. Domest. Anim. 2021, 56, 103–111.
- Arando, A.; Delgado, J.V.; Bermúdez-Oria, A.; León, J.M.; Fernández-Prior, Á.; Nogales, S.; Pérez-Marín, C.C. Effect of olive-derived antioxidants (3,4-dihydroxyphenylethanol and 3,4 dihydroxyphenylglycol) on sperm motility and fertility in liquid ram sperm stored at 15 °C or 5 °C. Reprod. Domest. Anim. 2020, 55, 325–332.
- Jofré, I.; Cuevas, M.; De Castro, L.S.; De Agostini Losano, J.D.; Torres, M.A.; Alvear, M.; Scheuermann, E.; Cesar Andrade, A.F.; Nichi, M.; Ortiz Assumpção, M.E.; et al. Antioxidant Effect of a Polyphenol-Rich Murtilla (Ugni molinae Turcz.) extract and its effect on the regulation of metabolism in refrigerated boar sperm. Oxid. Med. Cell. Longev. 2019, 2019.
- Giaretta, E.; Bucci, D.; Mari, G.; Galeati, G.; Love, C.C.; Tamanini, C.; Spinaci, M. Is Resveratrol Effective in Protecting Stallion Cooled Semen? J. Equine Vet. Sci. 2014, 34, 1307–1312.
- Al-Mutary, M.G.; Al-Ghadi, M.Q.; Ammari, A.A.; Al-Himadi, A.R.; Al-Jolimeed, A.H.; Arafah, M.W.; Amran, R.A.; Aleissa, M.S.; Swelum, A.A.-A. Effect of different concentrations of resveratrol on the quality and in vitro fertilizing ability of ram semen stored at 5 °C for up to 168 h. Theriogenology 2020, 152, 139–146.
- Torres, M.A.; Rigo, V.H.B.; Leal, D.F.; Pavaneli, A.P.P.; Muro, B.B.D.; de Agostini Losano, J.D.; Kawai, G.K.V.; del Collado, M.; Perecin, F.; Nichi, M.; et al. The use of resveratrol decreases liquid-extend boar semen fertility, even in concentrations that do not alter semen quality. Res. Vet. Sci. 2021, 136, 360–368.
- Martín-Hidalgo, D.; Hurtado de Llera, A.; Henning, H.; Wallner, U.; Waberski, D.; Bragado, M.J.; Gil, M.C.; García-Marín, L.J. The Effect of Resveratrol on the Quality of Extended Boar Semen during Storage at 17 °C. J. Agric. Sci. 2013, 5, p231.
- Sun, L.; Fan, X.; Zeng, Y.; Wang, L.; Zhu, Z.; Li, R.; Tian, X.; Wang, Y.; Lin, Y.; Wu, D.; et al. Resveratrol protects boar sperm in vitro via its antioxidant capacity. Zygote 2020, 28, 417–424.
- Kojo, S. Vitamin C: Basic Metabolism and Its Function as an Index of Oxidative Stress. Curr. Med. Chem. 2005, 11, 1041–1064.
- De Mello Andrade, J.M.; Fasolo, D. Polyphenol Antioxidants from Natural Sources and Contribution to Health Promotion; Elsevier Inc.: Amsterdam, The Netherlands, 2013; Volume 1, ISBN 9780123984562.
- Chainy, G.B.N.; Sahoo, D.K. Hormones and oxidative stress: An overview. Free Radic. Res. 2020, 54, 1–26.
- Ortiz, A.; Espino, J.; Bejarano, I.; Lozano, G.M.; Monllor, F.; García, J.F.; Pariente, J.A.; Rodríguez, A.B. High endogenous melatonin concentrations enhance sperm quality and short-term in vitro exposure to melatonin improves aspects of sperm motility. J. Pineal Res. 2011, 50, 132–139.
- Cebrián-Pérez, J.; Casao, A.; González-Arto, M.; dos Santos Hamilton, T.; Pérez-Pé, R.; Muiño-Blanco, T. Melatonin in Sperm Biology: Breaking Paradigms. Reprod. Domest. Anim. 2014, 49, 11–21.
- Fujinoki, M. Melatonin-enhanced hyperactivation of hamster sperm. Reproduction 2008, 136, 533–541.
- Balao Da Silva, C.M.; MacÍas-García, B.; Miró-Morán, A.; González-Fernández, L.; Morillo-Rodriguez, A.; Ortega-Ferrusola, C.; Gallardo-Bolaños, J.M.; Stilwell, G.; Tapia, J.A.; Peña, F.J. Melatonin reduces lipid peroxidation and apoptotic-like changes in stallion spermatozoa. J. Pineal Res. 2011, 51, 172–179.
- Poeggeler, B.; Saarela, S.; Reiter, R.J.; Tan, D.X.; Chen, L.D.; Manchester, L.C.; Barlow-Walden, L.R. Melatonin—A Highly Potent Endogenous Radical Scavenger and Electron Donor: New Aspects of the Oxidation Chemistry of this Indole Accessed in vitro. Ann. N. Y. Acad. Sci. 1994, 738, 419–420.
- Jang, H.Y.; Kim, Y.H.; Kim, B.W.; Park, I.C.; Cheong, H.T.; Kim, J.T.; Park, C.K.; Kong, H.S.; Lee, H.K.; Yang, B.K. Ameliorative effects of melatonin against hydrogen peroxide-induced oxidative stress on boar sperm characteristics and subsequent in vitro embryo development. Reprod. Domest. Anim. 2010, 45, 943–950.
- Rao, A.V.; Ray, M.R.; Rao, L.G. Lycopene. Adv. Food Nutr. Res. 2006, 51, 99–164.
- Muzandu, K.; Ishizuka, M.; Sakamoto, K.Q.; Shaban, Z.; El Bohi, K.; Kazusaka, A.; Fujita, S. Effect of lycopene and β-carotene on peroxynitrite-mediated cellular modifications. Toxicol. Appl. Pharmacol. 2006, 215, 330–340.
- Sieme, H.; Schäfer, T.; Stout, T.A.; Klug, E.; Waberski, D. The effects of different insemination regimes on fertility in mares. Theriogenology 2003, 60, 1153–1164.
- Loomis, P. The equine frozen semen industry. Anim. Reprod. Sci. 2001, 68, 191–200.
- Samper, J.C.; Plough, T. Techniques for the Insemination of Low Doses of Stallion Sperm. Reprod. Domest. Anim. 2010, 45, 35–39.
- Didion, B.A.; Braun, G.D.; Duggan, M.V. Field fertility of frozen boar semen: A retrospective report comprising over 2600 AI services spanning a four year period. Anim. Reprod. Sci. 2013, 137, 189–196.
