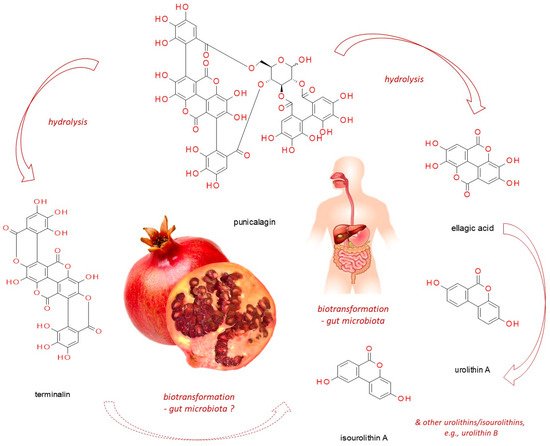
Punicalagin, present in pomegranate, myrobalan, yellow wood, tropical almond, and pink rock-rose, belongs to ellagitannins - a subgroup of hydrolyzable tannins. In vitro studies, based on cell line experiments, have demonstrated punicalagin anti-cancer actions on human cervical, ovarian, breast, lung, thyroid, colorectal, central nervous system, bone, as well as other cancer types. Punicalagin seems to work through a redirection of signal-transduction pathways from survival and proliferation into cell-cycle arrest, apoptosis, senescence or autophagy (thus compromising neoplastic progression). However, since it readily undergoes hydrolysis releasing e.g. ellagic acid, these might be its products which are responsible for the observed effects. Moreover, when practical application of punicalagin in chemoprevention is addressed, its biotransformation in the human organism should be considered (including both host and microbiome enzymatic impact). Therefore, however promising punicalagin antineoplastic properties seem to be, in vivo-based research should be conducted before translating the results obtained from in vitro studies into practice.
1. Pomegranate against Pathologies Development
Health profits of regular consumption of polyphenol-rich foods including fruits such as pomegranates, green tea or red wine, have been extensively studied in recent years
[1][2][3][4][5][6][7][8][1,2,3,4,5,6,7,8]. Their valuable health effects have been attributed not only to the activity of dietary fiber fractions, vitamins, and anthocyanins, but also to ellagitannins, which after intake are converted into ellagic acid, urolithins, and other low-molecular-weight phenols. The results of available epidemiological studies confirm that a diet rich in plant polyphenols reduces the risk of such chronic disorders as obesity
[3], associated with increased risk of metabolic syndrome and atherosclerosis, diabetes
[1][2][1,2], cardiovascular diseases
[4], as well as cancer
[5][6][5,6]. One of the most thoroughly examined plants, with respect to its broad-spectrum health-beneficial properties, is pomegranate (
Punica granatum L.), a fruit applied in medicine since ancient times
[7]. The chemical composition of pomegranate, with respect to the distribution of its components among different parts of the plant, as well as specific varieties, has been recently reviewed by Pirzadeh et al.
[8] and Ge et al.
[7]. Pomegranate is a rich source of hydrolyzable tannins and anthocyaninns which exhibit strong antioxidative and anti-inflammatory properties. Hydrolyzable tannins (ellagitannins and gallotannins), being the major class among pomegranate phenolic compounds, seem to be the main substances responsible for its observed favorable characteristics, and the major compounds present within this group comprise punicalagin, punicalin, granatins A and B, tellimagrandin I, pedunculagin, corilagin, as well as their hydrolytic products, namely gallagic acid, ellagic acid, and esters of glucose
[9]. The group of pomegranate anthocyanins includes delphinidin-3-glucoside, cyanidin-3-glucoside, delphinidin-3,5-diglucoside, cyanidin-3,5-diglucoside, pelargonidin-3,5-diglucoside, and pelargonidin-3-glucoside)
[8][10][11][8,10,11]. Pomegranate’s health-beneficial properties have been lately addressed with respect to its antimicrobial functions
[8]. Additionally, the scientific data elaborating pomegranate’s role in the prevention and therapy of inflammatory disorders
[12], as well as cancer diseases
[11][13][11,13], have been up-dated. Extracts derived from various
P. granatum parts, as well as purified components have been demonstrated to interfere with multiple signaling pathways involved in the development of pathological conditions.
2. Anti-Cancer Activities of Punicalagin (Pug)
Punicalagin (CAS No: 65995-63-3; (
Figure 1)) belongs to ellagitannins—a subgroup of hydrolyzable tannins. It is abundant in pomegranate juice, fruits, peel (pericarp), seeds, flowers, leaves and bark, as well as in the fruits of myrobalan (
Terminalia chebula Retz.), leaves of yellow wood (
Terminalia oblongata F. Muell.), tropical almond (
Terminalia catappa L.)
[9][14][9,14] and pink rock-rose (
Cistus ×
incanus L.)
[15]. In the aqueous environment, it readily undergoes hydrolysis releasing ellagic acid (EA, CAS No: 476-66-4), gallagic acid dilactone (terminalin, CAS No: 155144-63-1)
[16], and D-glucose.
Figure 1. The structure of punicalagin and its derivatives (pomegranate photograph has been created by Izabela Fecka).
Both punicalagin and its derivative—ellagic acid have been demonstrated to protect DNA from mutations
[17]. Zahin et al.
[17] observed that these two compounds dose-dependently diminished the level of DNA damage caused by a variety of carcinogens. For instance, almost complete inhibition of DNA adducts formation by Pug (40 µM) was noted in the experimental model with benzo[a]pyren, DNA, and rat liver microsomes (containing cytochrome P-450 associated biotransformation system required for the conversion of benzopyren into a mutagen). Additionally, the authors noted a dose dependent cytotoxic/antiproliferative effect of both Pug and EA on human lung cancer cell lines A549 and H1299 (MTT assay). Seeram et al.
[18] demonstrated that pomegranate juice (PJ) and pomegranate tannin extract, both rich in punicalagin, inhibited the proliferation of human oral (KB, CAL27), colon (HT-29, HCT116, SW480, SW620), and prostate (RWPE-1, 22Rv1) tumor cells. Moreover, pure Pug as well as its hydrolytic product EA showed antiproliferative action on these cell lines. However, the effect of PJ was the strongest, suggesting the synergistic/additive accomplishment of other phytochemicals contained in PJ, as was hypothesized by the authors
[18].
2.1. Punicalagin against Cervical Cancer
Punicalagin has been shown to exert antiproliferative/proapoptotic impact on human cervical cancer cell lines
[19][20][19,20]. Zhang et al.
[19] observed the cytotoxic effect of Pug on ME-180 cells in a dose-dependent mode (up to 100 µM). Similarly, Tang et al.
[20] demonstrated a dose- (up to 200 µM) and time-dependent antiproliferative action of Pug on HeLa cells. Both scientific groups have noted the upregulation of proapoptotic Bax proteins and downregulation of antiapoptotic Bcl-2 factors resulting from Pug treatment. Additionally, Pug stimulated the expression of tumor suppressor p53 gene, as well as proapoptotic caspases 3 and 9
[19]. Moreover, the putative influence of Pug on signaling pathways, diverting cells into carcinogenesis/metastasis pathways has been investigated in those two studies. Tang et al.
[20] demonstrated a dose-dependent inhibitory effect of Pug on β-catenin, C-myc, and cyclin D1 expression at the protein level. β-catenin/Wnt signaling pathway has been implicated in the progression of many cancers including cervical cancer, so its attenuation by Pug might be associated with cancer growth limitation. Furthermore, the authors observed the inhibition of HeLa cell migration in a wound-healing test, which was in agreement with the downregulation of matrix metalloproteinases 2 and 9 (MMP-2 and 9), and upregulation of their inhibitors, namely tissue inhibitors of metalloproteinases 2 and 3 (TIMP-2 and 3), resulting from Pug treatment. The interplay between proteolytic enzymes such as MMPs and their inhibitors, TIMPs, affects extracellular matrix (ECM) remodeling/degradation, and shifting of the balance towards increased proteolysis of ECM components exerted by MMPs is associated with the augmentation of cancer cells invasive capabilities. Therefore, Pug seems to be a promising agent aimed at compromising cervical cancer cells invasion and metastasis. Zhang et al.
[19] found Pug to increase NF-κB-p65 protein expression in the cytosol but decrease its expression in the nucleus. Since the translocation of NF-κB to the nucleus is associated with its induction of target genes expression, their results suggest the attenuation of the signaling pathway associated with this component. NF-κB signaling route has been implicated in tumor initiation, progression and chemoresistance, including cervical cancer
[21]. This so-called canonical pathway can be triggered by a variety of stimuli, such as proinflammatory cytokines (e.g., TNFα or IL-1), and is associated with the survival promotion. However, an alternative non-canonical mode of this pathway activation seems to function in a reversed manner rather facilitating apoptosis and thus suppressing tumor growth
[21]. The direction of NF-κB action is context-dependent, related to multiple factors, such as cancer stage and type, as thoroughly discussed in the latest review publications
[22][23][24][22,23,24], also with respect to its targeting by polyphenols
[25]. Nevertheless, punicalagin effect on cervical cancer cells was evidently proapoptotic and antiproliferative, hence the canonical NF-κB route must have been silenced in the work of Zhang et al.
[19].
The main findings on the impact of punicalagin on cervical cancer, as well as other cancer types, discussed in further paragraphs, are summarized in
Table 1.
Table 1.
Modulatory punicalagin impact on signaling pathways in human cancer cell lines.
| Cancer Type |
Experimental Model |
Up-Regulation ↑ |
Down-Regulation ↓ |
Final Effect |
Ref |
| Cervical carcinoma |
ME-180 cervical cancer cell line |
Bax; Casp-3 and 9; p53; cytosolic NF-κB-p65 |
Bcl-2; nuclear NF-κB-p65 |
Inhibition of cell proliferation and induction of apoptosis via suppressing NF-κB signaling |
[19] |
| Cervix epitheloid carcinoma |
HeLa cervical cancer cell line |
Bax;
TIMP2/3 |
Bcl-2;
β-catenin, C-myc, cyclin D1;
MMP2/9 |
Inhibition of cell proliferation and migration
Cell cycle arrest at G1 phase
Suppression of β-catenin pathway
Induction of apoptosis |
[20] |
| Breast adenocarcinoma |
MCF-7 and MDA-MB-231 cell lines |
E-cadherin |
GOLPH3;
MMP2/9;
N-cadherin |
Suppression of cell viability, EMT and migration via the regulation of GOLPH3 |
[26] |
| Ovarian cancer |
A2780 ovarian cancer cells |
Bax;
TIMP2/3 |
Bcl-2;
β-catenin, cyclin D1, survivin;
MMP2/9 |
Inhibition of cell viability and migration
Cell cycle arrest at G1 phase
Suppression of β-catenin pathway
Induction of apoptosis |
[27] |
| Lung carcinoma |
Lung cancer A549 cell line |
Bax; Casp-3 and 9; cytochrome C; ROS; cytosolic STAT-3 |
Bcl-2; Jak-1; nuclear STAT-3 |
Inhibition of cell proliferation and induction of apoptosis via suppressing STAT-3 activation |
[28] |
| Lung carcinoma |
Lung cancer A549 cell line |
Casp-3, 8, and 9; PARP-1; mitochondrial ROS |
Cytosolic ROS |
Induction of apoptosis; cell cycle arrest at G1/S |
[29] |
| Osteosarcoma |
Cell lines U2OS, SaOS2 |
- |
Phosphorylated IκBα; nuclear NF-κB-p65; IL-6; IL-8 |
Inhibition of cell proliferation and induction of apoptosis possibly via suppressing NF-κB signaling
Reduction of invasion potential |
[30] |
| Colorectal carcinoma |
Cell line HCT116 |
Cytochrome C |
Annexin A1; caspases 3/7, 8 and 9 |
Induction of cell death via apoptosis and autophagy |
[31] |
| Colon adenocarcinoma |
Cell line HT-29 |
- |
COX-2 |
Suppression of inflammatory cell signaling |
[32] |
| Colon adenocarcinoma |
Cell line Caco-2 |
Casp-3 and 9; Cytochrome C; Cyclin E |
Bcl-XL; Cyclin A and B1 |
Cell cycle arrest at S phase; induction of apoptosis via the intrinsic-mitochondrial pathway stimulation |
[33] |
| Papillary thyroid carcinoma |
Cell line BCPAP |
LC3-II conversion, beclin-1; phosphorylated ERK 1/2 and p38 |
p62; phosphorylated p70, S6, and 4E-BP1 |
Induction of apoptosis-independent cell death via autophagy through the activation of MAPK and inhibition of mTOR signaling |
[34] |
| Papillary thyroid carcinoma |
Cell line BCPAP |
p-H2A.X; p-ATM |
- |
Induction of cells death via the ATM-mediated DNA damage response |
[35] |
| Papillary thyroid carcinoma |
Cell line BCPAP |
SA-beta-Gal; cyclin-dependent kinase inhibitor p21; IκBα; nuclear NF-κB-p65; IL-6; IL-1β |
- |
Induction of senescent growth arrest and senescence-associated secretory phenotype (SASP) through the activation of NF-κB |
[36] |
| Glioblastoma astrocytoma |
Cell line U87MG |
Casp-3 and 9; PARP; Cyclin E; LC3-II cleavage, AMPK-P, p27-P |
Bcl-2; Cyclin A and B |
Cell cycle arrest at G2/M phase; induction of cell death via apoptosis and autophagy |
[37] |
Abbreviations: AMPK-P = phosphorylated AMPK (AMP activated kinase); EMT = epithelial to mesenchymal transition; GOLPH3 = golgi phosphoprotein 3; LC3-II = microtubule-associated protein light chain 3 II; MMP2/9 = matrix metalloproteinases 2 and 9; PARP(−1) = poly(ADP-ribose) polymerase (1); p-H2A.X = phospohorylated histone 2A.X; p-ATM = phospohorylated ATM (ataxia telangiectasia mutated); p27-P = phosphorylated p27; SA-beta-Gal = senescence-associated beta-galactosidase; TIMP2/3 = tissue inhibitors of metalloproteinases 2 and 3.
2.2. Punicalagin against Ovarian Cancer
Tang et al.
[27] detected Pug effect against human ovarian cancer cells A2780 whose viability was dose- and time-dependently reduced, accompanied by cell cycle arrest and apoptosis induction. Similarly, as in their aforementioned studies on cervical cancer
[20], the authors found Pug to compromise ovarian cancer growth through the inhibition of β-catenin signal-transduction pathway, as well as to reduce the migration of cells by attenuating matrix metalloproteinases and boosting their inhibitors. The involvement of hyperactivated Wnt/β-catenin signaling in gynecological malignancies, especially ovarian neoplasia, with respect to its silencing as a therapeutic aim
, is addressed in Section 3.1.2.
2.3. Punicalagin against Breast Cancer
Pan et al.
[26] analyzed the impact of Pug on the factors associated with the stimulation of cancer cells motility (migration and invasion) through the mechanism termed epithelial to mesenchymal transition (EMT), which proceeds via the upregulation of N-cadherin paralleled by the down-regulation of E-cadherin. Pug pretreatment of two human breast cancer cell lines (MCF-7 and MDA-MB-231) resulted in the reversed pattern of the two cadherins expressions: E-cadherin protein level rose, whereas N-cadherin dropped, being indicative of EMT inhibition. Additionally, Pug dampened the expression of metalloproteinases (MMP2 and 9) associated with the acquirement of the invasive and metastatic cancer phenotype. Furthermore, the authors observed the participation and Pug-modulation of Golgi phosphoprotein 3 (GOLPH3) in the promotion of invasive potential in the investigated breast cancer cells. Whereas GOLPH3 overexpression enhanced the cells’ motility and up-regulated the pro-invasive factors mentioned above, Pug attenuated these effects. Oncogenic aspects of GOLPH3 in many malignancies including breast cancer, and the consequences of its upregulation have been recently discussed by Sechi et al.
[38]. GOLPH3 overexpression has been correlated with the promotion of neoplastic phenotype, worsening patients’ prognosis and heightening resistance to chemotherapy
[39][40][41][39,40,41]. In normal cells, GOLPH3 is required for the proper functioning of the Golgi apparatus, whereas its disturbances such as abnormal vesicle trafficking contribute to the development of neoplastic phenotype. Overactivation of the GOLPH3/MYO18A pathway stimulates the exocytosis of growth factors and matrix metalloproteinases, enhancing cell invasive potential. GOLPH3 overexpression modifies integrin-mediated signaling, resulting in cytoskeleton reorganization and cell migration. GOLPH3 also responds to DNA damage leading to massive Golgi fragmentation which increases the neoplastic cell surveillance
[38]. Therefore, as observed by Pan et al.
[26], the GOLPH3-opposed Pug impact on breast cancer cells, paralleled by the reduction of EMT and invasive potential, seems promising with respect to anticancer Pug application.
2.4. Punicalagin against Colorectal Cancer
Ganesan et al.
[31] observed proapoptotic impact of Pug on human colorectal cancer cell line HCT116, reflected by the increase in early apoptotic cells (assessed by Annexin V/PI flow cytometry) and cytochrome C release from the mitochondrium. This finding is in accordance with the results obtained by Seeram et al.
[18] showing an antiapoptotic Pug effect in HCT116 as well as HT-29 (human colon adenocarcinoma) cell lines. However, Ganesan et al.
[31] observed that 72-h Pug treatment of HCT116 cells downregulated caspases 3/7, 8, and 9. Hence, the authors checked for the features indicative of autophagy stimulation by Pug, and with the application of autophagy flux assay via flow cytometry, noted autophagosomes degradation (reflecting autophagy involvement). Additionally, they analyzed Pug effect on annexin A1 expression and noted a significant downregulation of this protein. Annexins are ubiquitously occurring calcium-dependent proteins which recognize and bind membranous phospholipids. Annexin A1 is involved in a variety of physiological processes, including differentiation, proliferation, as well as apoptosis. However, its best described properties are associated with the attenuation of inflammatory processes (e.g., through the inhibition of phospholipase A2 and therefore reduction of arachidonic acid release, the main precursor of proinflammatory eicosanoids)
[42]. Annexin A1 faulty expression has been associated with cancer disease, but its actual role is unclear. As discussed by Fu et al.
[42], annexin A1 function in malignancies might be seen as a two-edged sword due to the fact that, in some cancers, it seems to act as a tumor suppressor, whereas in others as a tumor promoter. For instance, in prostate
[42][43][42,43] or oesophageal
[44] cancers, a downregulation of this protein has been observed, whereas in hepatocellular
[45] as well as colorectal cancers
[46][47][46,47] annexin A1 elevated expression has been detected. Therefore, in colorectal cancer, this protein seems to favor neoplastic development, hence its suppressing by Pug is favorable. Further, Ganesan et al.
[31] performed a proteome profiling analysis testing the impact of Pug (with annexin A1-route involvement) on 35 different proteins associated with apoptosis and autophagy processes. They observed four significantly altered proteins in HCT116 cells treated both with punicalagin and formyl peptide receptors (FPR) inhibitors (applied to block annexin A1 signaling). Three of them, i.e., heat shock proteins 27 and 60 (HSP27 and HSP60) and tumor necrosis factor receptor 1 (TNF RI), were down-regulated, whereas catalase was upregulated. Since the first two factors are associated with autophagy regulation (HSP27, HSP60), whereas TNF RI induction yields apoptotic events, the authors concluded that the stimulation of both pathways leading via annexin A1 suppression may be responsible for Pug-induced colorectal cells death in their research model. Adams et al.
[32] investigated the effect of Pug on human colon cancer cell line HT-29 stimulated with TNF-α. The authors observed a dose-dependent reduction in COX-2 expression in the cells pretreated with increasing Pug concentrations (up to 200 mg/L). However, this effect was stronger after the application of pomegranate juice (PJ) or total pomegranate tannin extract (TPT). Moreover, PJ treatment was shown to attenuate TNF-α-induced signaling pathway leading via AKT and NF-κB. Upregulation of this type of proinflammatory pathway, stimulating the expression of COX-2 and other inflammatory factors such as cytokines, has been observed in various cancers including colorectal cancer. Therefore, the anti-inflammatory Pug effect observed by the authors is promising with respect to its preventive application in cancer diseases. However, the authors underlined that many beneficial effects observed in theirs and aforementioned studies
[18] are enhanced when mixtures of tannins or pomegranate juice is applied, and not purified Pug. Larrosa et al.
[33] noted the Pug-induced arrest of cell cycle at S phase, and its proapoptotic effect (via the intrinsic-mitochondrial pathway stimulation) on human colon cancer cell line Caco-2. Nevertheless, the authors’ profound analysis demonstrated that it was rather ellagic acid released from hydrolyzed Pug molecule which exerted all of these effects in their experimental model. Therefore, it might be hypothesized that many effects interpreted as an impact of punicalagin are the consequences of its degradation products.
2.5. Punicalagin against Thyroid Cancer
Cheng et al.
[34] in their studies on human papillary thyroid carcinoma cell line (BCPAP) observed a reduction of the cell viability upon Pug treatment in a concentration and time-dependent mode (after 24-, 36-, and 48-h incubation with doses up to 100 μM). However, the closer analysis of this effect demonstrated apoptosis-independent mechanism associated rather with the induction of autophagy (type II programmed cell death). The authors did not detect characteristic features indicative of apoptotic events (e.g., neither the cleavage of caspase-3 nor poly(ADP-ribose) polymerase (PARP) were noted). Reversely, Pug stimulated-cancer cells expressed the profile of markers associated with autophagy; increase in autophagic vacuoles, increase in microtubule-associated protein light chain 3 II (LC3-II) conversion, beclin-1 expression, and p62 degradation. Additionally, the authors observed the stimulation of MAPK signaling pathway (increase in ERK 1/2 and p38 phosphorylation) and inhibition of mTOR route (drop in phosphorylated p70, S6, and 4E-BP1). These findings allowed them to conclude that the reversed mode of the two signal-transduction pathways modification exerted by Pug led to the final lethal effect via autophagy in the studied thyroid cancer cells. In the further studies of the same team, Yao et al.
[35] analyzed the effect of Pug on DNA damage in BCPAP cells, as well as its modulation of DNA damage response (DDR) pathway. The authors observed that Pug-induced over 5-fold reduction in the cell viability was accompanied by the comparable degree of phosphorylated-H2A.X amplification. H2A.X is a form of 2A histone being a sensitive marker of DNA damage, and its phosphorylation indicates the initiation of DDR pathway. Moreover, 24-h treatment with 100 μM of Pug augmented the phosphorylation of ATM kinase (ataxia telangiectasia mutated) but did not affect ATR (ATM and rad3-related) kinase phosphorylation. Finally, the authors suggested that Pug contributed to the studied cells’ death via the induction of double strand breaks (DSB) in DNA helix, followed by the stimulation of DDR pathway associated with ATM signaling. In their ensuing experiments, the authors reported the Pug-induced acquirement of the senescent phenotype of the BCPAP thyroid cancer cells
[36]. Pug altered the cells’ morphological features, increased their granularity, and upregulated both senescence-associated beta-galactosidase (SA-beta-Gal) as well as cyclin-dependent kinase inhibitor p21. The observed effects seem to have been mediated by NF-κB signaling pathway, which activation by Pug was found in this study. Namely, Pug was shown to induce the translocation of NF-κB-p65 to the nucleus. However, there is a discrepancy between the authors comment on Pug effect on IκBα and the results presented in Figure 4A
[36] included in their paper. The authors concluded that Pug induced the phosphorylation and degradation of IκBα. However, judging from this figure, Pug seems to have stimulated only the total IκBα factor, and not its phosphorylated form
[36].
2.6. Punicalagin against Lung Cancer
Fang et al.
[28] observed punicalagin to exert concentration-dependent (up to 30 μM) pro-apoptotic effect on human lung cancer A549 cells. The authors demonstrated Pug-induction of proapoptotic factors (Bax; caspases 3 and 9; cytochrome C), and inhibition of anti-apoptotic Bcl-2. Similar results have been obtained by Berkoz et al.
[29] in the same type of cancer cells which underwent apoptosis upon Pug treatment (dose-dependently at 50 and 75 μmolar levels), associated with the increase in caspases 3, 8, and 9 expression. Moreover, the induction of reactive oxygen species (ROS) by Pug was noted in both studies, however Berkoz et al.
[29] observed ROS stimulation only in the mitochondria. Additionally, Pug was found to inhibit JAK/STAT signaling pathway in the lung cancer cells. Pug treatment reduced Jak-1 protein expression, and affected the STAT-3 protein expression profile, inducing its expression in the cytosolic fraction and inhibiting the expression of STAT-3 in the nucleus
[28]. Therefore, as the authors suggested, Pug seemed to be able to inhibit this transcription factor translocation to the nucleus. Since Stat3 has been shown to inhibit apoptosis by the induction of Bcl-2 and inhibition of Bax expression
[48], it might be imagined that in this experiment punicalagin inhibited JAK/STAT pathway followed by the reversed expression profile of Bcl-2/Bax, which eventually induced the process of apoptosis.
2.7. Punicalagin against Osteosarcoma
Huang et al.
[30] demonstrated Pug to induce apoptosis in three human osteosarcoma cell lines (U2OS, MG63, and SaOS2); 48-h treatment with 100 μM Pug led to the substantial increase in early and late apoptotic cells in all three cell lines. Furthermore, 24-h Pug treatment of the three cell lines reduced the cells invasiveness in Matrigel assay. Additionally, the authors investigated Pug effect on the NF-κB signaling route and found the inhibition of active (phosphorylated) form of IκBα as well as the prevention of NF-κB-p65 translocation to the nucleus in two cell lines (U2OS and SaOS2). However, no impact on mTOR signaling has been noted. Moreover, antiangiogenic and anticancer effects of Pug have been observed by the authors on xenografted mice model, where the injection of Pug attenuated the growth of osteosarcoma, as well as reduced the tumor neoangiogenesis.
2.8. Punicalagin against Glioma
Wang et al.
[37] demonstrated that 24- and 48-h Pug-treatment of human U87MG glioma cells significantly decreased cell viability in a dose-dependent manner (in the concentrations up to 30 μg/mL), arresting cell cycle at G2/M phase (reflected by Pug downregulation of cyclins A and B). Additionally, the authors analyzed Pug impact on chosen factors associated with the stimulation of cell lethality through two mechanisms, namely apoptosis and autophagy. Their findings led them to conclusions considering the involvement of both pathways in Pug-stimulated attenuation of cancer progression. Namely, Pug induced caspases 3 and 9 levels, activated PARP cleavage, as well as decreased Bcl-2 expression, all being characteristic apoptotic features. On the other hand, the application of caspases inhibitor only partly reversed Pug lethal effect exerted on the cells, which indicated the participation of an alternative route. Therefore, the authors checked for the autophagy markers, and noted the Pug-induced accumulation of microtubule-associated protein light chain 3 II (LC3-II), as well as punicalagin-stimulated phosphorylation of AMP activated kinase (AMPK) and p27
Kip1. Therefore, the authors suggested the Pug-induction of autophagy through the upregulation of LKB1-AMPK pathway.
References
- Pham, N.M.; Do, V.V.; Lee, A.H. Polyphenol-rich foods and risk of gestational diabetes: a systematic review and meta-analysis. J. Clin. Nutr.2019, 73, 647–656. [DOI: 10.1038/s41430-018-0218-7].
- Palma-Duran, S.A.; Vlassopoulos, A.; Lean, M; Govan, L.; Combet, E. Nutritional intervention and impact of polyphenol on glycohemoglobin (HbA1c) in non-diabetic and type 2 diabetic subjects: Systematic review and meta-analysis. Rev. Food Sci. Nutr.2017, 57, 975-986. [DOI: 10.1080/10408398.2014.973932].
- Guo, X.; Tresserra-Rimbau, A.; Estruch, R.; Martínez-González, M.A.; Medina-Remón, A.; Fitó, M.; Corella, D.; Salas-Salvadó, J.; Portillo, M.P.; Moreno, J.J.; Pi-Sunyer, X.; Lamuela-Raventós, R.M. Polyphenol Levels Are Inversely Correlated with Body Weight and Obesity in an Elderly Population after 5 Years of Follow Up (The Randomised PREDIMED Study). Nutrients 2017, 9, 452. [DOI: 10.3390/nu9050452].
- Yamagata, K. Polyphenols Regulate Endothelial Functions and Reduce the Risk of Cardiovascular Disease. Pharm. Des. 2019, 25, 2443-2458. [DOI: 10.2174/1381612825666190722100504].
- Gu, H.F.; Mao, X.Y.; Du, M. Prevention of breast cancer by dietary polyphenols-role of cancer stem cells. Rev. Food Sci. Nutr. 2020, 60, 810-825. [DOI: 10.1080/10408398.2018.1551778].
- Miyata, Y.; Shida, Y.; Hakariya, T.; Sakai, H. Anti-Cancer Effects of Green Tea Polyphenols Against Prostate Cancer. Molecules 2019, 24, [DOI: 10.3390/molecules24010193].
- Ge, S.; Duo, L.; Wang, J.; GegenZhula; Yang, J.; Li, Z.; Tu, Y. A unique understanding of traditional medicine of pomegranate, Punica granatum L. and its current research status. Ethnopharmacol. 2021, 271, 113877. [DOI: 10.1016/j.jep.2021.113877].
- Pirzadeh, M.; Caporaso, N.; Rauf, A.; Shariati, M.A.; Yessimbekov, Z.; Khan, M.U.; Imran, M.; Mubarak, M.S. Pomegranate as a source of bioactive constituents: a review on their characterization, properties and applications. Rev. Food Sci. Nutr. 2021, 61, 982-999. [DOI: 10.1080/10408398.2020.1749825].
- Satomi, H.; Umemura, K.; Ueno, A.; Hatano, T.; Okuda, T.; Noro, T. Carbonic anhydrase inhibitors from the pericarps of Punica granatum L. Pharm. Bull. 1993, 16, 787-790. [DOI: 10.1248/bpb.16.787].
- Viuda-Martos, M.; Fernández-Lóaez, J.; Pérez-álvarez, J.A. Pomegranate and its Many Functional Components as Related to Human Health: A Review. Rev. Food Sci. Food Saf. 2010, 9, 635-654. [DOI: 10.1111/j.1541-4337.2010.00131.x].
- Sharma, P.; McClees, S.F.; Afaq, F. Pomegranate for prevention and treatment of cancer: An update. Molecules 2017, 22, 177. [DOI: 10.3390/molecules22010177].
- Danesi, F.; Ferguson, L.R. Could pomegranate juice help in the control of inflammatory diseases? Nutrients 2017, 9, 958. [DOI: 10.3390/nu9090958].
- Khwairakpam, A.D.; Bordoloi, D.; Thakur, K.K.; Monisha, J.; Arfuso, F.; Sethi, G.; Mishra, S.; Kumar, A.P.; Kunnumakkara, A.B. Possible use of Punica granatum (Pomegranate) in cancer therapy. Res. 2018, 133, 53-64. [DOI: 10.1016/j.phrs.2018.04.021].
- Yoshida, T.; Amakura, Y.; Yoshimura, M. Structural features and biological properties of ellagitannins in some plant families of the order Myrtales. J. Mol. Sci. 2010, 11, 79-106. [DOI: 10.3390/ijms11010079].
- Fecka, I.; Włodarczyk, M.; Starzec, A. Isolation and structure elucidation of cistusin: A new ellagitannin from Cistus × incanus L. leaves. crops prod. 2020, 158. [DOI: 10.1016/j.indcrop.2020.112971].
- Oelrichs, P.B.; Pearce, C.M.; Zhu, J.; Filippich, L.J. Isolation and structure determination of terminalin A toxic condensed tannin from Terminalia oblongata. Toxins 1994, 2, 144-150. [DOI: 10.1002/nt.2620020311].
- Zahin, M.; Ahmad, I.; Gupta, R.C.; Aqil, F. Punicalagin and ellagic acid demonstrate anti-mutagenic activity and inhibition of benzo[a]pyrene induced DNA adducts. Biomed Res. Int. 2014, 2014, 467465. [DOI: 10.1155/2014/467465].
- Seeram, N.P.; Adams, L.S.; Henning, S.M.; Niu, Y.; Zhang, Y.; Nair, M.G.; Heber, D. In vitro antiproliferative, apoptotic and antioxidant activities of punicalagin, ellagic acid and a total pomegranate tannin extract are enhanced in combination with other polyphenols as found in pomegranate juice. Nutr. Biochem. 2005, 16, 360-367. [DOI: 10.1016/j.jnutbio.2005.01.006].
- Zhang, L.; Chinnathambi, A.; Alharbi, S.A.; Veeraraghavan, V.P.; Mohan, S.K.; Zhang, G. Punicalagin promotes the apoptosis in human cervical cancer (ME-180) cells through mitochondrial pathway and by inhibiting the NF-kB signaling pathway. Saudi J. Biol. Sci. 2020, 27, 1100-1106. [DOI: 10.1016/j.sjbs.2020.02.015].
- Tang, J.; Li, B.; Hong, S.; Liu, C.; Min, J.; Hu, M.; Li, Y.; Liu, Y.; Hong, L. Punicalagin suppresses the proliferation and invasion of cervical cancer cells through inhibition of the β-catenin pathway. Med. Rep. 2017, 16, 1439-1444. [DOI: 10.3892/mmr.2017.6687].
- Tilborghs, S.; Corthouts, J.; Verhoeven, Y.; Arias, D,; Rolfo, C.; Trinh.; X.B.; van Dam, P.A. The role of Nuclear Factor-kappa B signaling in human cervical cancer. Rev. Oncol. Hematol. 2017, 120, 141-150. [DOI: 10.1016/j.critrevonc.2017.11.001].
- Lalle, G.; Twardowski, J.; Grinberg-Bleyer, Y. NF-κB in Cancer Immunity: Friend or Foe. Cells 2021, 10, 355. [DOI: 10.3390/cells10020355]
- Puar, Y.R.; Shanmugam, M.K.; Fan, L.; Arfuso, F.; Sethi, G.; Tergaonkar, V. Evidence for the Involvement of the Master Transcription Factor NF-κB in Cancer Initiation and Progression. Biomedicines 2018, 6, 82. [DOI: 10.3390/biomedicines6030082].
- Tegowski, M.; Baldwin, A. Noncanonical NF-κB in Cancer. Biomedicines 2018, 6, 66. [DOI: 10.3390/biomedicines6020066].
- Khan, H,; Ullah, H.; Castilho, P.C.M.F.; Gomila, A.S.; D'Onofrio, G.; Filosa, R.; Wang, F.; Nabavi, S.M.; Daglia, M.; Silva, A.S.; Rengasamy, K.R.R.; Ou, J.; Zou, X.; Xiao, J.; Cao, H. Targeting NF-κB signaling pathway in cancer by dietary polyphenols. Rev. Food Sci. Nutr. 2020, 60, 2790-2800. [DOI: 10.1080/10408398.2019.1661827].
- Pan, L.; Duan, Y.; Ma, F.; Lou, L. Punicalagin inhibits the viability, migration, invasion, and EMT by regulating GOLPH3 in breast cancer cells. Recept. Signal Transduct. 2020, 40, 173-180. [DOI: 10.1080/10799893.2020.1719152].
- Tang, J.M.; Min, J.; Li, B.S.; Hong, S.S.; Liu, C.; Hu, M.; Li, Y.; Yang, J.; Hong, L. Therapeutic Effects of Punicalagin Against Ovarian Carcinoma Cells in Association with β-Catenin Signaling Inhibition. J. Gynecol. Cancer 2016, 26, 1557-1563. [DOI: 10.1097/IGC.0000000000000805].
- Fang, L.; Wang, H.; Zhang, J.; Fang, X. Punicalagin induces ROS-mediated apoptotic cell death through inhibiting STAT3 translocation in lung cancer A549 cells. Biochem. Mol. Toxicol. 2021, 35, 1-10. [DOI: 10.1002/jbt.22771].
- Berköz, M.; Krośniak, M. Punicalagin induces apoptosis in a549 cell line through mitochondria-mediated pathway. Physiol. Biophys. 2020, 39, 557-567. [DOI: 10.4149/gpb_2020024].
- Huang, T.; Zhang, X.; Wang, H. Punicalagin inhibited proliferation, invasion and angiogenesis of osteosarcoma through suppression of NF-κB signaling. Med. Rep. 2020, 22, 2386-2394. [DOI: 10.3892/mmr.2020.11304].
- Ganesan, T.; Sinniah, A.; Chik, Z.; Alshawsh, M.A. Punicalagin regulates apoptosis-autophagy switch via modulation of annexin a1 in colorectal cancer. Nutrients 2020, 12, 2430. [DOI: 10.3390/nu12082430].
- Adams, L.S.; Seeram, N.P.; Aggarwal, B.B.; Takada, Y.; Sand, D.; Heber, D. Pomegranate juice, total pomegranate ellagitannins, and punicalagin suppress inflammatory cell signaling in colon cancer cells. Agric. Food Chem. 2006, 54, 980-985. [DOI: 10.1021/jf052005r].
- Larrosa, M.; Tomás-Barberán, F.A.; Espín, J.C. The dietary hydrolysable tannin punicalagin releases ellagic acid that induces apoptosis in human colon adenocarcinoma Caco-2 cells by using the mitochondrial pathway. Nutr. Biochem. 2006, 17, 611-625. [DOI: 10.1016/j.jnutbio.2005.09.004].
- Cheng, X.; Gao, Y.; Yao, X.; Yu, H.; Bao, J.; Guan, H.; Sun, Y.; Zhang, L. Punicalagin induces apoptosis-independent autophagic cell death in human papillary thyroid carcinoma BCPAP cells. RSC Adv. 2016, 6, 68485-68493. [DOI: 1039/C6RA13431A].
- Yao, X.; Cheng, X.; Zhang, L.; Yu, H.; Bao, J.; Guan, H.; Lu, R. Punicalagin from pomegranate promotes human papillary thyroid carcinoma BCPAP cell death by triggering ATM-mediated DNA damage response. Res. 2017, 47, 63-71. [DOI: 10.1016/j.nutres.2017.09.001].
- Cheng, X.; Yao, X.; Xu, S.; Pan, J.; Yu, H.; Bao, J.; Guan, H.; Lu, R.; Zhang, L. Punicalagin induces senescent growth arrest in human papillary thyroid carcinoma BCPAP cells via NF-κB signaling pathway. Pharmacother. 2018, 103, 490-498. [DOI: 10.1016/j.biopha.2018.04.074].
- Wang, S.G.; Huang, M.H.; Li, J.H.; Lai, F.I.; Lee, H.M.; Hsu, Y.N. Punicalagin induces apoptotic and autophagic cell death in human U87MG glioma cells. Acta Pharmacol. Sin. 2013, 34, 1411-1419. [DOI: 10.1038/aps.2013.98].
- Sechi, S.; Frappaolo, A.; Karimpour-Ghahnavieh, A.; Piergentili, R.; Giansanti, M.G. Onco-genic roles of GOLPH3 in the physiopathology of cancer. J. Mol. Sci. 2020, 21, 933. [DOI: 10.3390/ijms21030933].
- Zeng, Z.; Lin, H.; Zhao, X.; Liu, G.; Wang, X.; Xu, R.; Chen, K.; Li, J.; Song, L. Overexpression of GOLPH3 promotes proliferation and tumorigenicity in breast cancer via suppression of the FOXO1 transcription factor. Cancer Res. 2012, 18, 4059-4069. [DOI: 10.1158/1078-0432.CCR-11-3156].
- Tokuda, E.; Itoh, T.; Hasegawa, J.; Ijuin, T.; Takeuchi, Y.; Irino, Y.; Fukumoto, M.; Takenawa, T. Phosphatidylinositol 4-phosphate in the golgi apparatus regulates cell-cell adhesion and invasive cell migration in human breast cancer. Cancer Res. 2014, 74, 3054-3066. [DOI: 10.1158/0008-5472.CAN-13-2441].
- Tang, S.; Pan, H.; Wei, W.; Yang, H.; Liu, J.; Yang, R. GOLPH3: A novel biomarker that correlates with poor survival and resistance to chemotherapy in breast cancer. Oncotarget 2017, 8, 105155-105169. [DOI: 10.18632/oncotarget.21927].
- Fu, Z.; Zhang, S.; Wang, B.; Huang, W.; Zheng, L.; Cheng, A. Annexin A1: A double-edged sword as novel cancer biomarker. Chim. Acta 2020, 504, 36-42. [DOI: 10.1016/j.cca.2020.01.022].
- Patton, K.T.; Chen, H.M.; Joseph, L.; Yang, X.J. Decreased annexin I expression in prostatic adenocarcinoma and in high-grade prostatic intraepithelial neoplasia. Histopathology 2005, 47, 597-601. [DOI: 10.1111/j.1365-2559.2005.02300.x].
- Kang, J.S.; Calvo, B.F.; Maygarden, S.J.; Caskey, L.S.; Mohler, J.L.; Ornstein, D.K. Dysregulation of annexin I protein expression in high-grade prostatic intraepithelial neoplasia and prostate cancer. Clin Cancer Res. 2002, 8, 117-123. PMID: 11801547. DOI: Published January 2002
- Moghanibashi, M.; Jazii, F.R.; Soheili, Z.S.; Zare, M.; Karkhane, A.; Parivar, K.; Mohamadynejad, P. Proteomics of a new esophageal cancer cell line established from Persian patient. Gene 2012, 500, 124-133. [DOI: 10.1016/j.gene.2012.03.038].
- Suo, A.; Zhang, M.; Yao, Y.; Zhang, L.; Huang, C.; Nan, K.; Zhang, W. Proteome analysis of the effects of sorafenib on human hepatocellular carcinoma cell line HepG2. Oncol. 2012, 29, 1827-1836. [DOI: 10.1007/s12032-011-0013-y].
- Roth, U.; Razawi, H.; Hommer, J.; Engelmann, K.; Schwientek, T.; Uller, S.M.; Baldus, S.E.; Patsos, G.; Corfield, A.P.; Paraskeva, C.; et al. Differential expression proteomics of human colorectal cancer based on a syngeneic cellular model for the progression of adenoma to carcinoma. Proteomics 2010, 10, 194-202. [DOI: 10.1002/pmic.200900614].
- Lecona, E.; Barrasa, J.I.; Olmo, N.; Llorente, B.; Turnay, J.; Lizarbe, M.A. Upregulation of Annexin A1 Expression by Butyrate in Human Colon Adenocarcinoma Cells: Role of p53, NF-Y, and p38 Mitogen-Activated Protein Kinase. Cell. Biol. 2008, 28, 4665-4674. [DOI: 10.1128/MCB.00650-07].