Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 1 by Joana Maria Ramis and Version 2 by Rita Xu.
Platelet-derived extracellular vesicles (pEVs) are nanosized membranous subcellular structures released by platelets, which comprise different subpopulations that differ on morphology, size, composition and cellular origin. Extracellular vesicles (EVs) work as intercellular communicators exerting their function by transporting their cargo that includes nucleic acids, proteins and lipids. pEVs have shown to mediate same functions as platelets, presenting a great potential for the development of new treatments in the biomedical field.
- extracellular vesicles
- exosomes
- platelets
1. Introduction
In recent years, extracellular vesicles (EVs) have emerged as potential therapeutic effectors in the regenerative biomedical field. EVs are membranous subcellular structures released by any cell type, which comprise different subpopulations that differ on morphology, size, composition and cellular origin [1]. In the past, EVs had been referred to by many different names such as microvesicles, exosomes, microparticles, apoptotic bodies, ectosomes or oncosomes, among others; according to their size, their tissue or cell origin, their claimed function or even their presence outside the cell [2]. However, these EV subgroups presented a great diversity on the biomechanism behind their formation and the functions they perform, thus distinguishing them has not been proven to be easy [3]. Therefore, a consensus has been reached and the most accepted classification is performed according to the characterization and the isolation methodology used [1].
In general, EVs present a significant interest for the development of new treatments. EVs enable cell to cell communication, which can prevent the development of diseases by promoting homeostatic physiology or lead to pathological states, depending on the nature of the producing cell and the stimuli that activated the EV production [4]. Different cellular mechanisms for EVs secretion and uptake exist, crucial for intercellular communication, which are still unknown [5]. For this reason, some research focuses on the use of naturally produced EVs while other research aims to understand the molecular functionality of EVs to design new bioengineered carriers for enhanced cell delivery treatments or the addition of alternative cargos [6][7][6,7].
Today, EVs are thought to be secreted by all cell types, being stem cells and immune cells, some of the most studied EV sources for therapeutical approaches [8][9][8,9]. Nevertheless, clinical translation of cell cultured derived EVs has been hindered due to the high regulation requirements for ex vivo cell expansion [10]. On the contrary, the use of platelets presents some advantages, mainly related to safety and regulatory concerns. On one hand, clinical-grade allogenic platelets can be obtained from whole-blood donations as a byproduct from red blood cells obtention. On the other hand, compared to other cell sources, ex vivo cell expansion is avoided, and the human origin and the lack of growth medium components diminishes concerns over contamination or immunological safety [10]. Thereby, although relatively little attention has been paid so far to the therapeutic use of platelet EVs (pEVs), platelets and its concentrates are emerging as a potential source that overcome the limitations of other EV sources for regenerative medicine.
Platelet concentrates, such as platelet rich plasma (PRP) or platelet lysate (PL), are biological samples that have already been widely evaluated in regenerative medicine [11]. Thus, the use of platelet concentrates in regenerative approaches has already been reviewed elsewhere [12][13][12,13]. Some of the main fields in which platelet concentrates are being used include dermatology, aesthetic medicine, musculoskeletal regeneration, cardiovascular diseases, or neural regeneration among others [14]. The therapeutical applicability of PRP was first associated to the biomolecules released by platelets, mainly attributed to growth factors. In fact, platelets can release growth factors, cytokines and extracellular matrix modulators that promote revascularization, restoration of damaged tissue and activation of mesenchymal stem cells [15][16][15,16]. However, it has not been until relatively recently that pEVs have also emerged as a potential effector of platelet concentrates and platelets themselves, involved in their regenerative and therapeutical application [17].
While until recently the use of pEVs in therapeutics have not been explored, already in 1967, Peter Wolf described the release by platelets of minute lipid-rich particulate material, which could be separated by ultracentrifugation, distinguishable from intact platelets and showing coagulant properties, terming this minute particulate as “platelet dust” [18]. Later on, future studies identified platelet-released particles again, which were observed in electron microscopy samples. This further characterization and description allowed a renaming of “platelet dust” for a more accurate term: microparticles [19]. Further on, the particle release was observed in many other cell types, thus microparticles were joined up with what are now called EVs [1]. The aggrupation the different historical names under the common label of EVs aims to lead to a more comprehensive and accurate report of the activity and functionality of EVs bringing consensus among the different disciplines [1].
After the initial studies performed on the functionality of “platelet dust” or platelet microparticles, it seems now clear that pEVs appear to be important effectors not only for coagulation but also for platelet regenerative function along with the rest of the biomolecules released by platelets [10].
Even more, the clinical use of platelet concentrates is still limited due to its main drawbacks that are the lack of reproducibility, mainly due to the non-standardized separation methods, the variability among donors or the storage conditions [20][21][22][20,21,22]. Moreover, the use of autologous concentrates limits the total obtained volume and needs programming of its obtention to arrange a proper treatment [15][23][15,23]. Even more, some patients may not be suited for this sort of interventions due to their medical record, e.g., cancer patients or tobacco users [24]. Together, along with the lack of quality controls, this leads to high heterogeneity of the obtained concentrates. Therefore, pEVs are a promising alternative to surpass PRP and other platelet concentrates limitations [11], due to providing off-the-shelf controlled product methods [25][26][25,26].
On the other hand, pEVs may surpass the platelet concentrate limitations and even present some desirable advantages that could improve the benefits of their clinical use. For instance, not only do pEVs share platelet function but they are more powerful, in terms of coagulation [27] or osteogenic [25][26][25,26] capacity. In addition, pEVs in contrast to platelets, can cross tissue barriers, extending their abilities beyond the blood [17]. In fact, pEVs have been identified in some spatial contexts where platelets are rarely found, such as the synovial fluid, the lymph or the bone marrow, and like other types of EVs they are expected to be able to cross other tissue barriers including the blood-brain barrier [17].
2. Regenerative Effects of pEVs
Recently, pEVs have been postulated to play a key role in homeostatic processes [28]. In fact, platelets and pEVs are natural mediators of different physiological processes and contribute to the immune system response functions and regenerative process [29]. However, only a few articles have evaluated the potential of pEVs as therapeutic regenerative tools (Figure 1). The main fields in which pEVs have been evaluated include injuries, neurogenesis, muscle regeneration, angiogenesis, biomaterials, bone regeneration, and osteoarthritis. Therefore, in this review, we aim to detail the main advances in the different regenerative approaches evaluated until now in order to assemble their applicability and realize of the main limitations that still hinder pEVs clinical use.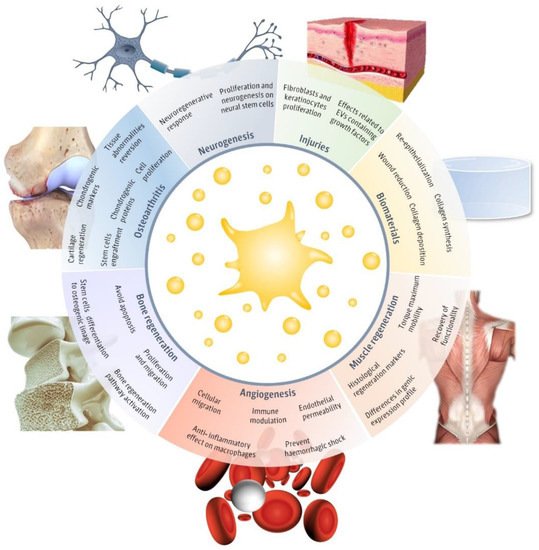
Figure 1. Regenerative applications of platelet-derived extracellular vesicles (pEVs). Main regenerative effects reported for pEVs in regenerative fields, including injuries [30][31][32][33][34][35][36][30,31,32,33,34,35,36], biomaterials [30][31][30,31], neurogenesis [37][38][37,38], muscle regeneration [39], angiogenesis [37][38][40][41][42][37,38,40,41,42], bone regeneration [25][26][43][44][45][25,26,43,44,45] and osteoarthritis [46][47][48][49][46,47,48,49] and the major reported therapeutical effects. This figure was created using Freepik images.
