5. Discussion
This work proposes the valorization of an environmentally hazardous agro-industrial by-product into an extract with demonstrated potential as treatment for inflammatory and oxidative stress-related ocular surface diseases. Different extracts derived from OP, together with two of their major phenolic compounds, HT and OL, were tested in vitro on human corneal and conjunctival epithelial cells for their cytotoxicity, anti-inflammatory, and antioxidant effect.
Regarding the results of cell viability assay for both cell lines, CONV and OPT1 can be used up to 0.80 mg/mL, while OPT2 and OPT3 up to 0.40 mg/mL. HT is nontoxic up to 100 μM, OL up to 300 μM, and their mixture OL + HT up to 5 μM of OL + 50 μM of HT. These data agree with studies already published regarding cytotoxicity of the same or similar phenolic compounds on ocular cell lines. Granner et al.
[30], Zhu et al.
[32], and Liu et al.
[33] used safely HT up to 100 μM in retinal pigment epithelial cells, while Zou et al.
[31] proved the antioxidant effect of HT in the same cell line up to 200 μM. Abengózar-Vela et al.
[13] showed that resveratrol is nontoxic up to 300 μM for ocular surface epithelial cells, while for Paladino et al.
[39][42], resveratrol can be safely used up to 200 μM for retinal epithelial cells. Stoddard et al.
[40][43] studied the antioxidant activity of four different polyphenols (quercetin, n-propyl gallate, epigallocatechin gallate (EGCG), and gallic acid) in human corneal cells and demonstrated that their toxic concentrations could vary from 41.3 to 337.5 μM, depending on the compound. Regarding cytotoxicity studies of crude extracts on human corneal cells,
Aloe vera extract can be used up to 0.13 mg/mL
[41][44], Maple leaves extract up to 0.20 mg/mL
[42][45], and
Polygonum cuspidatum aqueous extract up to 100 mg/mL
[43][46]. Importantly, as also can be observed from the four different extracts produced from the same material in this study, the extract composition can vary, depending on the extraction method and conditions. This can affect the maximum allowable concentration of each extract.
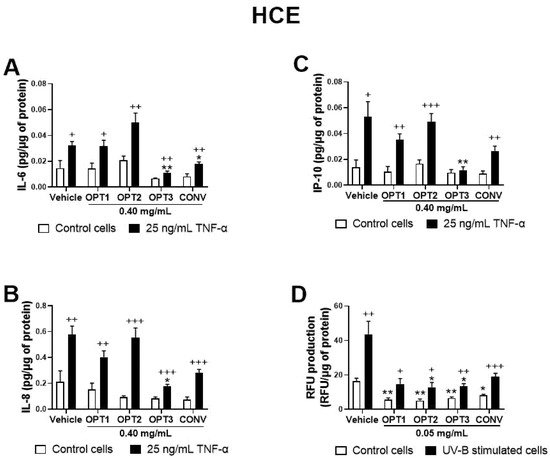
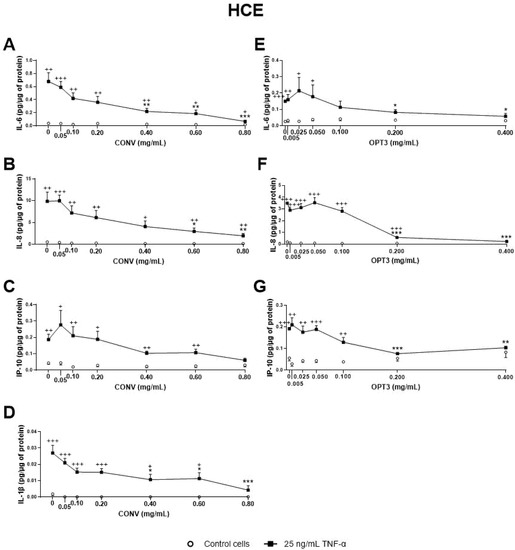
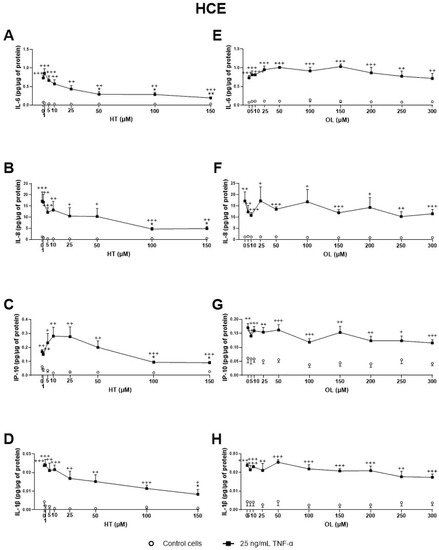
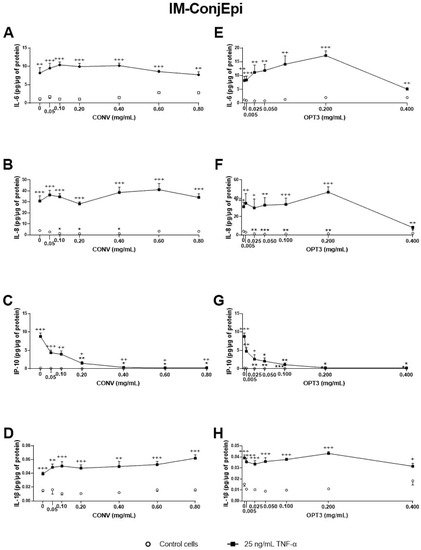
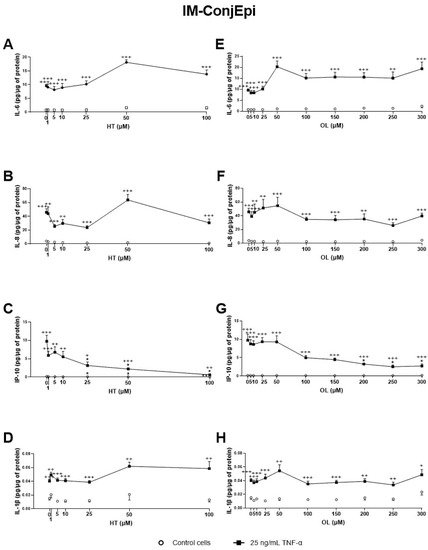
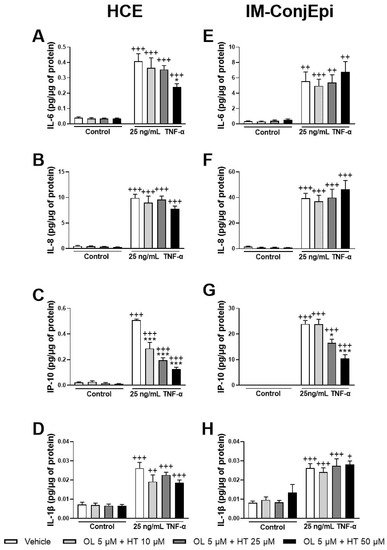
Our results also showed that, among the four extracts derived from OP tested, CONV (produced by conventional solid–liquid extraction) and OPT3 (generated by sequential scCO
2 de-oiling, followed by PLE optimization) demonstrated a strong anti-inflammatory activity in a dose-dependent manner. Both inhibited TNF-α -stimulated IL-6, IL-8, IL-1β, and IP-10 secretion by ocular surface corneal and conjunctival epithelial cells. HT was also able to decrease the levels of the aforementioned interleukins in both cell types. OL demonstrated significantly less anti-inflammatory effect than HT or OP extracts, inhibiting only IP-10 stimulated production in conjunctival cells. However, when OL is combined with HT, the mixture can inhibit IP-10 secretion by HCE cells at very low concentrations, at which each compound alone would have no significant activity. This fact proves that the compounds can demonstrate a remarkable synergistic anti-inflammatory effect when combined. Increased levels of the measured cytokines/chemokines have been related to several inflammatory ocular surface diseases, such as DES
[44][45][46][47][48][49][50][51][52][53][54][47,48,49,50,51,52,53,54,55,56,57] and ocular allergy
[55][56][57][58][58,59,60,61]. More recent findings reveal altered levels of IL-17A in conjunctiva of DES patients, proving that IL-17A plays an important role in the conjunctival epithelial disruption
[59][62]. High levels of IL-17A were also detected in mice stimulated by desiccating stress
[46][49]. However, in our study, TNF-α failed to stimulate IL-17A production in both ocular surface cell lines selected; additionally, in some of our experiments on HCE cells, IL-1β secretion upon TNF-α-stimulation was not sufficient. Another proinflammatory stimulus should be tried in the future either alone or in combination with TNF-α.
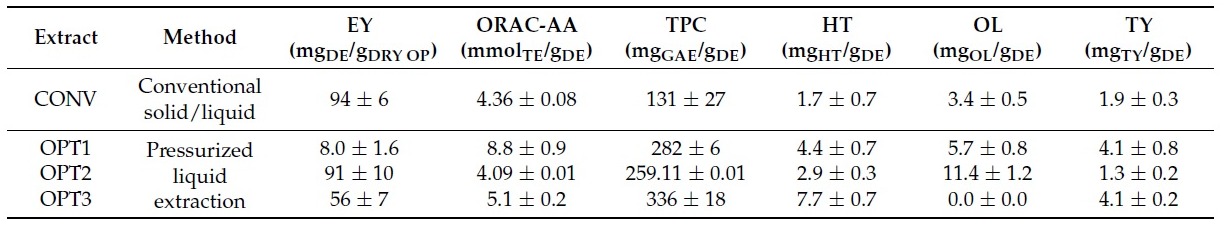
In terms of extracts’ composition, as already described in
Table 1, CONV contained 1.7 mg of HT/g of DE and 3.4 mg of OL/g of DE, OPT1: 4.4 mg
HT/g and 5.7 mg
OL/g, OPT2: 2.9 mg
HT/g and 11.4 mg
OL/g, and OPT3: 7.7 mg
HT/g and 0.0 mg
OL/g. Hence, at a concentration of 0.40 mg/mL (used for the first screening of anti-inflammatory activity), 4.4 μM of HT and 2.5 μM of OL were tested for CONV, 11.4 μM of HT and 4.2 of μM of OL for OPT1, 7.5 μM of HT and 8.4 μM of OL for OPT2, and 20.0 μM of HT and 0 μM of OL for OPT3. As already mentioned, HT demonstrated a much stronger inhibition of the secretion of the selected interleukins/chemokines than OL on both cell lines. Therefore, because OPT3 had the strongest anti-inflammatory activity, this can be probably related more to HT (or similar compounds) than OL. OPT2 had the highest concentration of OL; however, it demonstrated very poor inhibition of the selected cytokine/chemokines. This fact can also support this hypothesis. On the other hand, OPT3 comprises many different phenolic compounds apart from HT. As can be observed from
Table 1, OPT3 is the extract with the highest total phenolic content (2.6 times more than the CONV) and simple phenols’ concentration (like HT)
[38]. For OPT3, 0.200 mg/mL comprise 10.0 μM HT and 0 μM OL, while 0.050 mg/mL contains 2.5 μM HT and 0 μM OL. For CONV, 0.20 mg/mL comprises 2.2 μM HT and 1.3 μM OL, 0.40 mg/mL includes 4.4 μM of HT and 2.5 μM of OL, while 0.60 mg/mL contains 6.6 μM of HT and 3.8 μM of OL. Comparing the effective concentrations of OL and HT alone with those included in the concentrations of the extracts tested, a clear synergistic effect can be observed in the extracts. This fact has already been proven by the combination of OL + HT. All these data can explain why OPT3 demonstrate a stronger anti-inflammatory activity compared with CONV, confirming our hypothesis.
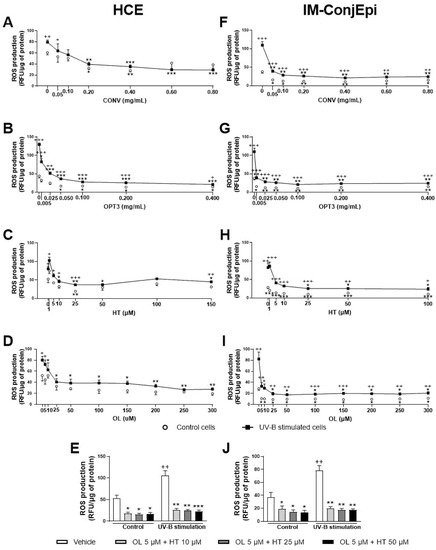
In addition, all extracts and compounds tested demonstrated a significant antioxidant activity in a dose-dependent way, starting from very low concentrations, inhibiting UV-B induced ROS production in corneal and conjunctival cells. The combination of OL + HT demonstrated a strong synergistic antioxidant effect in both cell lines either in the stimulated (for IM-ConjEpi) or in the baseline (for HCE) cells. ROS are produced during oxidative metabolism as by-products and are formed by partial reduction of oxygen
[60][61][63,64]. Oxidative stress is the disruption of the balance between the antioxidant and the pro-oxidant system of the cells, many times leading to oxidative damage
[62][65]. The oxidative damage has been involved in the pathophysiology of many chronic systematic diseases, like cancer, inflammation, and neuro-degenerative disorders
[2]. Ocular surface epithelial tissues are exposed to atmospheric oxygen and UV rays of the sunlight
[63][66], also being vulnerable to oxidative stress-induced ocular diseases. Accumulated ROS in tears and Meibomian gland have been related to ocular tissue inflammation and damage
[64][67]. Oxidative damage has been proven to be involved in the pathophysiology of several ocular surface diseases, such as conjunctivochalasis
[6], atopic keratoconjunctivitis
[7], and Sjogren and non-Sjogren DES
[7][9][65][66][7,9,68,69]. Further, Birkedal-Hansen et al.
[67][70] proved that chronic exposure to environmental stress upregulates oxidative biomarkers, causing loss of the regenerative ability of the corneal cells.
At the concentration used during the first screening of antioxidant activity (0.05 mg/mL), 0.6 μM of HT and 0.3 μM of OL were tested for CONV, 0.18 μM of HT and 0.5 of μM OL for OPT1, 0.9 μM of HT and 1.1 μM of OL for OPT2, and 2.5 μM HT and 0 μM OL for OPT3. OPT3 at 0.005 mg/mL comprises 0.3 μM HT and 0 μM OL. CONV at 0.20 mg/mL contains 2.2 μM HT and 1.3 μM OL, while at 0.05 mg/mL, it contains 0.3 μM of HT and 0.6 μM of OL. HT and OL alone can inhibit ROS production by HCE cells at 10 μM and by IM-ConjEpi at 25 μM. From the combination of OL + HT, a synergistic antioxidant effect was observed. As already explained above, both extracts comprise several phenolic compounds and different total phenolic content (
Table 1)
[38]. In addition, from the results of both cell lines, it can be observed that OPT3 demonstrates a strong inhibition of the ROS production in 10 to 40 times lower concentrations compared with CONV. Therefore, the synergistic antioxidant activity of several phenolic compounds present in the extracts can be confirmed.
The beneficial anti-inflammatory and antioxidant effects of other pure phenolic compounds on the ocular surface have already been reported. For instance, previous studies from our group demonstrated that quercetin and resveratrol, either alone or in combination, inhibited IL-6, IL-8, and IP-10 secretion by TNF-α–stimulated human corneal and conjunctival cells
[13]. They also reduced IL-4, IL-1α, IL-2, and TNF-α tear production in mice exposed to desiccating stress and decreased corneal staining when administrated as eye drops
[14]. CD4 T-cell conjunctival infiltration was also decreased in adoptively transferred mice with T cells from desiccating stress-exposed animals treated with quercetin and resveratrol. In addition, both compounds were able to inhibit the UV-B induced oxidative stress in human corneal and conjunctival cells, in a dose-dependent manner
[13]. Further, several studies have demonstrated the in vitro anti-inflammatory activity on human corneal cells of curcumin
[68][71], DA-6034 (7-carboxymethyloxy-3′4′,5-trimethoxy flavone)
[69][72], Daidzin
[70][73], EGCG
[71][72][73][74][74,75,76,77], Pterostilbene
[75][78], and a mixture of ferulic acid with kaempferol
[76][79]. Catechin was also able to reduce Prostaglandin E2 production on rabbit cornea cells
[77][80]. A strong antioxidant activity on human corneal cells has been reported only for Pterostiblene
[75][78], Daidzin
[70][73], and EGCG
[71][74]. However, there are few studies exploring and establishing the relation between the antioxidant and the anti-inflammatory activity of natural phenolic compounds on human ocular surface cells. In addition, all these studies examined the effect of the molecules only on corneal cells and not on any additional ocular surface cell line. Our work presents an in vitro comparative study for the olive phenolic compounds between two different parts of the ocular surface (conjunctiva and cornea) exclusively of the human epithelium. Thus, it proposes a promising therapy for oxidative- and inflammatory-related diseases of the entire human ocular surface.
The antioxidant activity of the phenolic compounds has been attributed to the presence of the quinone/semiquinone/hydroquinone triad equilibrium, the chain-breaking scavenging effect of aromatic H atom donors, and the hydroxyl substitution of the aromatic ring
[78][79][80][81,82,83]. Regarding the latest, the position and number of the aromatic hydroxyl groups in the structure of the phenolic molecule can affect its final ROS scavenging capacity
[81][84]. The anti-inflammatory activity of these molecules has also been related to the position of these groups. In our study, HT was the molecule with the highest inhibitory activity of the measured ILs. For HT, the hydroxyl groups have been found to demonstrate strong interaction with the ASP348 protein of sirtuin 1 enzyme, upregulating its expression, and hence downregulating inflammatory response by vascular endothelial cells
[82][85]. More recently, hydrogen bond interactions have been confirmed between the hydroxyl groups of HT and the Ser-530 residue of cyclooxygenase-2, inhibiting the expression of the enzyme
[83][86]. It is also important to observe that TY, which does not have the 3-hydroxyl of HT, does not have any affinity with the enzyme, confirming the importance of the position of these groups
[83][86].
Apart from individual phenolic compounds, numerous crude polyphenolic-rich extracts have also demonstrated strong in vitro antioxidant and/or anti-inflammatory activity on the ocular surface. Preliminary studies on fractions of olive polyphenolic extracts have already demonstrated interesting inhibition of ROS and TNF-α production by UV-B-induced and LPS-induced rabbit epithelial corneal cells, respectively
[37]. However, our work consists of a complete pharmacological study of four different interleukins/chemokines secretion by two different cell lines of the human epithelium not only for olive extracts, but also for their two major phenolic compounds. Further, it compares extracts with different composition obtained from the same material, explaining their distinct activities. Regarding different plant materials, extracts from
Camellia japonica [84][87],
Chamaecyparis obtuse [85][88],
Aloe vera [41][44],
Euphrasia officinalis [86][89],
Lamium album [87][90],
Polygonum cuspidatum [43][46], and Maple leaves
[42][45] have also been found to reduce inflammatory biomarkers in human corneal cells. Hence, the potential of naturally derived polyphenols and polyphenols-rich extract is becoming an emerging issue in the field.
In summary, our results have demonstrated that a convectional and an HT- enriched optimized extract derived from OP, along with the major OP pure phenolic compounds HT and OL, can inhibit inflammation and oxidative damage in human corneal and conjunctival epithelial cells. However, there are some limitations in this study. Although TNF-α acted good as inflammatory stimulus, different inducers could be used to achieve the stimulation and measurement of IL-17A, TNF-α itself, and/or any other interleukin/chemokine not stimulated by TNF-α. Further, although HCE has been proven to demonstrate high correspondence to the human tissues of origin
[88][39], IM-ConjEpi is a relatively new commercial SV-40 Large T antigen immortalized cell line from primary human conjunctival epithelial. However, according to the manufacturer, it has a 99% purity and maintains expression of epithelial specific markers such as CK18 and CK19. Finally, it is also true that in vivo models are much more complex. For many of the compounds
[69][70][73][74][76][89][90][91][72,73,76,77,79,91,92,93] and extracts
[43][84][85][92][93][94][95][46,87,88,94,95,96,97] mentioned in the bibliography, the anti-inflammatory activity has also been proven in vivo. For EGCG
[96][98] and Daidzin
[70][73], the in vivo antioxidant effect has also been studied. Hence, in vivo studies in a desiccating stress mice model are currently being performed for selected extracts and compounds, to support our in vitro findings.
In conclusion, extracts derived from OP and their major phenolic compounds, particularly HT, seem to be promising potential treatment for ocular surface inflammatory diseases, such as DES. The use of these type of OP extracts is of chief importance for the green development of related industries, as they propose a high value application of an agro-industrial by-product. The results of this study illustrate how the sustainable and intensified extraction techniques are proved to be competitive compared to the conventional ones, while a high selectivity towards biomarkers related to dry eye is established.