Chitosan is a biopolymer with high added value, and its properties are related to its molecular weight. Thus, high molecular weight values provide low solubility of chitosan, presenting limitations in its use. Based on this, several studies have developed different hydrolysis methods to reduce the molecular weight of chitosan.
- hydrolysis
- chitosan
- molecular weight
- chitooligosaccharides
1. Introduction
Chitosan has received attention as a functional biopolymer due to its cationic nature, biocompatibility, biodegradability, non-toxicity, and adsorption properties [2][1]. Its main characteristics are the molecular weight (Mw) and the degree of acetylation (DA) or degree of deacetylation (DDA), which correspond to the molar fractions of GlcNAc and GlcN. Most commercial chitosans have molecular weights ranging from 50–2000 kDa, with an average DDA of 50–100% (commonly 80–90%) [3][2]. Based on molecular weight, chitosan can be grouped into low molecular weight (<100 kDa), medium molecular weight (100–1000 kDa), and high molecular weight (>1000 kDa) [4][3].
Chitooligosaccharides are the products of the hydrolysis of chitosan, and because they are soluble in water, they have several applications, such as antimicrobial, antioxidant, anti-tumor, and agricultural purposes [6,7,8,9,10,11,12][4][5][6][7][8][9][10].
Among hydrolysis techniques, acid hydrolysis has emerged as a convenient method for depolymerizing polysaccharides. However, acid hydrolysis generally requires severe treatment, as the rigid crystalline region in the chitosan granules inhibits acid penetration. For this reason, a high concentration of acid, ranging from 5 to 12 M, is used in most studies [9,23,24,25][7][11][12][13]. However, excessive acid loading can cause glucosamine degradation, which significantly reduces yield and generates major waste deposition problems [15,26][14][15]. In recent studies, a smaller amount of acid only made it possible to obtain medium molecular weight chitosan in a short hydrolysis time [4][3]. In high time, low molecular weight chitosan and chitooligosaccharides were obtained. However, yield decreased due to soluble products such as glucosamine monomers and dimers [15,27][14][16]. Therefore, current studies seek to develop the technique using different types and concentrations of acids associated with ionic liquids, induced electric fields, or other hydrolysis methods.
This review will present the main hydrolysis techniques of chitosan and analyze the main factors that influence the obtaining and characteristics of low molecular weight chitosan and chitooligosaccharides effectively at a lower cost.
2. Acid Hydrolysis
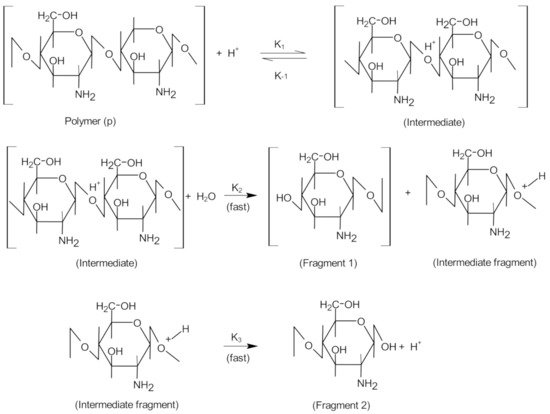
The acid hydrolysis of glycosidic bonds involves the following steps: (1) protonation of oxygen in the glycosidic bond through the connection of a proton (H 3O + ) to the glycosidic bond; (2) adding water to the final reducing sugar group; (3) decomposition of protonated glycosidic bonds ( Figure 31 ). In this case, catalytic protons may be present in the water contained in the samples, and the protonated amino group of chitosan is also likely to act as a proton donor in catalysis [24][12].
The concentration of the acid is also directly proportional to the rate of hydrolysis. Chitosan with a medium molecular weight (198.64 kDa) was obtained from hydrolysis with 5% acetic acid for 30 min. At concentrations of 1% and 3%, the molecular weight was 592.89 and 281.98 kDa, respectively [4][3]. In this case, it is observed that the lower acid concentration and short hydrolysis time enabled the production of only medium molecular weight chitosan.
Studies have shown that increasing the hydrolysis time decreases the molecular weight, resulting in low molecular weight chitosan and chitooligosaccharides in the first hours. Even so, it is necessary to use a high concentration of acid. When using chitosan with a molecular weight of 214 kDa (96% DDA), hydrolyzed in 85% phosphoric acid solution (60 °C), a high decrease was obtained in the first 4.0 h (74 kDa), and, after 15 h, molecular weight reached 19 kDa [36][17]. In another study with a 2 M HCl solution, the molecular weight of chitosan was reduced from 200 to 29, 17, and 3.0 kDa in 30 min, 1.0 h, and 5.0 h, respectively [27][16]. In chitosan 2038 kDa, hydrolyzed in 5 M HCl at 65 °C, the molecular weight after 5.0 h was 156 kDa, reaching 74 kDa in 36 h [24][12].
Chitosan of initial molecular weight of 726 kDa (medium molecular weight) and 28 kDa (low molecular weight) hydrolyzed in lactic acid (1%) at temperatures of 8, 22, and 37 °C also showed an increase in the rate of hydrolysis with the increasing temperature. However, there was an influence of the initial molecular weight of chitosan on the hydrolysis rate in this case. This rate increased from 25% to 50% with the increase in temperature in medium molecular weight chitosan. The effect of temperature was less on low molecular weight chitosan. In these conditions, rates of 5% to 10% were observed [35][18].
3. Oxidative Hydrolysis with Hydrogen Peroxide
This type of hydrolysis is mainly influenced by the concentration of hydrogen peroxide, time, and temperature of hydrolysis, Table 41 .
| Reference | Chitosan | Solvent | Solvent Concentration | Temperature | Time | Mw/DP |
|---|---|---|---|---|---|---|
| [10] | 1% | H2O2; CH3COOH | 0.5–3%; 1% | 50–75 °C | 1–6 h | 2–7 DP |
| [19] | 1% | H2O2; CH3COOH; H3PW12O40 | 0.5–3%; 1%; 0.04–0.14% | 50–75 °C | 10–60 min | 7 DP |
| [20] | 3% | H2O2; CH3COOH | 3%; 2% | 80 °C | 180 min | 1.36 kDa |
| [21] | 1% | H2O2; CH3COOH | 5.7, 2.8, 1.9, and 1.7 (molar ratio) | 50 °C | 8 h | 6.61–9.97 kDa |
| [22] | 2% | H2O2; CH3COOH | 2%; 1% | 40 °C | 30–180 min | - |
| [23] | 2% | H2O2; HCl | (0.5, 1.0, 1.5, 2.0 M)/0.5% | 25, 40, 50, 70 °C | 1, 2, 3 h | 11–1200 kDa |
| [24] | 7.5% | H2O2; H3PW12O40 | 4.5%; 0.1% | 70 °C | 30–120 min | 4.3–4.7 kDa |
| [25] | - | H2O2; HCl | 0–5%; 0–9% | 10–90 °C | 0.5–8 h | 5–200 kDa |
In addition to these methods, chitosan is also effectively hydrolyzed by hydrogen peroxide under ultraviolet irradiation. In this case, when chitosan is hydrolyzed only in the presence of hydrogen peroxide, viscosity decreases by 20% and 63% at 30 and 180 min, respectively. However, when hydrogen peroxide is combined with ultraviolet irradiation, viscosity decreases by 84% and 92% for the same analyzed times [48][22].
When associated with acetic acid, its molar ratios influence the recovery of chitooligosaccharides. Therefore, the higher the concentration of hydrogen peroxide about acetic acid, the higher the yield of chitooligosaccharides (dimers–decamers), with a yield of 36%, 22%, 18%, and 14%, for molar ratios of 5.7, 2.8, 1.9, and 1.7 respectively, in addition to obtaining chitosan with lower molecular weight, 6.61, 7.7, 9.29, and 9.03 kDa, respectively [46][21].
As with other types of hydrolysis, time will also influence when using hydrogen peroxide. In chitosan hydrolyzed by H 2O 2 in HCl solution (0.9%) for 0.5–8 h, there was a reduction in molecular weight with the increase in hydrolysis time. A rapid decrease from 480 to 50 kDa in 0.5 h has been shown, with a 95% yield [51][25]. In the study using hydrogen peroxide and acetic acid, the yields of chitooligosaccharides increased considerably with increasing hydrolysis time, reaching an optimal yield of 62.42% in 4 h [12][10].
4. Enzymatic Hydrolysis
The temperature on enzyme activity is related to two established thermal parameters. The first is the Arrhenius activation energy, which relates to the effect of temperature on the catalytic reaction. The second is thermal stability, which shows the effect of temperature on thermal inactivation [62][26].
The ideal value in the hydrolysis rate depends on the type of enzyme. In the study using the enzymatic complex Celuzyme ® XB, formed by cellulase, xylanase, and β-glucanase, the highest hydrolysis rate was reached at 51 °C, reaching a relative activity of 90%. Additionally, the increase in temperature tends to decrease its catalytic activity. This occurs due to the modification of the tertiary structure that may lose its active and functional conformation [31][27]. This behavior was also observed when using pepsin, which showed an increase in hydrolysis activity by increasing the temperature to 45 °C, with a relative enzymatic activity of 75% [32][28].
Several studies have analyzed the effect of chitosan’s molecular weight on antibacterial activity. Some studies concluded that antibacterial activity increased when there was a reduction in molecular weight. This was observed in 12 kDa low molecular weight chitosan compared to 228 kDa medium molecular weight chitosan against Bacillus cereus , E. coli , Staphylococcus aureus , Pseudomonas aeruginosa , Salmonella sp, and Saccharomyces cerevisiae [58][29]. It was also observed in 64 kDa chitosan compared to 152 kDa chitosan against Staphylococcus aureus , Pantoea ananatis , Pseudomonas aeruginosa , Raoultella planticola , and Escherichia coli [31][27].
Some reports have shown that chitosan of medium molecular weight (194 kDa) has more significant antibacterial activity than chitosan of lower molecular weight (41, 14, and 5 kDa) against Gram-positive microorganisms due to its greater thickness of the peptidoglycan layer, preventing the access of the chitooligosaccharides to the cell. Although the peptidoglycan layer prevents the chitooligosaccharides from accessing the cell, the antimicrobial effect of the higher molecular weight chitosan is due to the formation of a chitosan film. This film can inhibit the absorption of nutrients by the microorganism [34][30]. Another study, with chitosan from 300 to 3.3 kDa, showed that the pH could influence differently according to the molecular weight of the chitosan, being observed that under acid pH conditions, the chitosan activity increased with the increase of the molecular weight, regardless of the temperature and the bacteria tested. However, at neutral pH, chitosan activity increased as the molecular weight decreased [52][31].
References
- Thakur, V.K.; Thakur, M.K. Recent advances in graft copolymerization and applications of chitosan: A review. ACS Sustain. Chem. Eng. 2014, 2, 2637–2652.
- Mourya, V.K.; Inamdar, N.N.; Choudhari, Y.M. Chitooligosaccharides: Synthesis, characterization and applications. Polym. Sci. Ser. A 2011, 53, 583–612.
- Santoso, J.; Adiputra, K.C.; Soerdirga, L.C.; Tarman, K. Effect of acetic acid hydrolysis on the characteristics of water soluble chitosan. IOP Conf. Ser. Earth Environ. Sci. 2020, 414, 012021.
- Chang, S.-H.; Wu, C.-H.; Tsai, G.-J. Effects of chitosan molecular weight on its antioxidant and antimutagenic properties. Carbohydr. Polym. 2018, 181, 1026–1032.
- El-Sawy, N.M.; El-Rehim, H.A.A.; Elbarbary, A.M.; Hegazy, E.-S.A. Radiation-induced degradation of chitosan for possible use as a growth promoter in agricultural purposes. Carbohydr. Polym. 2010, 79, 555–562.
- Khan, A.; Mehmood, S.; Shafiq, M.; Yasin, T.; Akhter, Z.; Ahmad, S. Structural and antimicrobial properties of irradiated chitosan and its complexes with zinc. Radiat. Phys. Chem. 2013, 91, 138–142.
- Li, K.; Xing, R.; Liu, S.; Li, R.; Qin, Y.; Meng, X.; Li, P. Separation of chito-oligomers with several degrees of polymerization and study of their antioxidant activity. Carbohydr. Polym. 2012, 88, 896–903.
- Liang, T.-W.; Chen, Y.-J.; Yen, Y.-H.; Wang, S.-L. The antitumor activity of the hydrolysates of chitinous materials hydrolyzed by crude enzyme from Bacillus amyloliquefaciens V656. Process. Biochem. 2007, 42, 527–534.
- Muley, A.B.; Shingote, P.R.; Patil, A.P.; Dalvi, S.G.; Suprasanna, P. Gamma radiation degradation of chitosan for application in growth promotion and induction of stress tolerance in potato (Solanum tuberosum L.). Carbohydr. Polym. 2019, 210, 289–301.
- Wu, S.-J.; Pan, S.-K.; Wang, H.-B.; Wu, J.-H. Preparation of chitooligosaccharides from cicada slough and their antibacterial activity. Int. J. Biol. Macromol. 2013, 62, 348–351.
- Benhabiles, M.; Salah, R.; Lounici, H.; Drouiche, N.; Goosen, M.; Mameri, N. Antibacterial activity of chitin, chitosan and its oligomers prepared from shrimp shell waste. Food Hydrocoll. 2012, 29, 48–56.
- Kasaai, M.R.; Arul, J.; Charlet, G. Fragmentation of chitosan by acids. Sci. World J. 2013, 2013, 1–11.
- Yan, X.; Evenocheck, H.M. Chitosan analysis using acid hydrolysis and HPLC/UV. Carbohydr. Polym. 2012, 87, 1774–1778.
- Li, D.-D.; Tao, Y.; Shi, Y.-N.; Han, Y.-B.; Yang, N.; Xu, X.-M. Effect of re-acetylation on the acid hydrolysis of chitosan under an induced electric field. Food Chem. 2020, 309, 125767.
- Zhang, Z.; Li, C.; Wang, Q.; Zhao, Z.K. Efficient hydrolysis of chitosan in ionic liquids. Carbohydr. Polym. 2009, 78, 685–689.
- Aljbour, N.D.; Beg, M.D.; Gimbun, J. Acid hydrolysis of chitosan to oligomers using hydrochloric acid. Chem. Eng. Technol. 2019, 42, 1741–1746.
- Jia, Z.; Shen, D. Effect of reaction temperature and reaction time on the preparation of low-molecular-weight chitosan using phosphoric acid. Carbohydr. Polym. 2002, 49, 393–396.
- Il’Ina, A.V.; Varlamov, V.P. Hydrolysis of chitosan in lactic acid. Appl. Biochem. Microbiol. 2004, 40, 300–303.
- Xia, Z.; Wu, S.; Chen, J. Preparation of water soluble chitosan by hydrolysis using hydrogen peroxide. Int. J. Biol. Macromol. 2013, 59, 242–245.
- Xing, R.; Liu, Y.; Li, K.; Yu, H.; Liu, S.; Yang, Y.; Chen, X.; Li, P. Monomer composition of chitooligosaccharides obtained by different degradation methods and their effects on immunomodulatory activities. Carbohydr. Polym. 2017, 157, 1288–1297.
- Tishchenko, G.; Šimůnek, J.; Brus, J.; Netopilík, M.; Pekárek, M.; Walterová, Z.; Koppová, I.; Lenfeld, J. Low-molecular-weight chitosans: Preparation and characterization. Carbohydr. Polym. 2011, 86, 1077–1081.
- Wang, S.-M.; Huang, Q.-Z.; Wang, Q.-S. Study on the synergetic degradation of chitosan with ultraviolet light and hydrogen peroxide. Carbohydr. Res. 2005, 340, 1143–1147.
- Tian, F.; Liu, Y.; Hu, K.; Zhao, B. Study of the depolymerization behavior of chitosan by hydrogen peroxide. Carbohydr. Polym. 2004, 57, 31–37.
- Huang, Q.Z.; Wang, S.M.; Huang, J.F.; Zhuo, L.H.; Guo, Y.C. Study on the heterogeneous degradation of chitosan with hydrogen peroxide under the catalysis of phosphotungstic acid. Carbohydr. Polym. 2007, 68, 761–765.
- Qin, C.; Du, Y.; Xiao, L. Effect of hydrogen peroxide treatment on the molecular weight and structure of chitosan. Polym. Degrad. Stab. 2002, 76, 211–218.
- Peterson, M.; Daniel, R.M.; Danson, M.J.; Eisenthal, R. The dependence of enzyme activity on temperature: Determination and validation of parameters. Biochem. J. 2007, 402, 331–337.
- Águila-Almanza, E.; Salgado-Delgado, R.; Vargas-Galarza, Z.; García-Hernández, E.; Hernández-Cocoletzi, H. Enzymatic depolimerization of chitosan for the preparation of functional membranes. J. Chem. 2019, 2019, 1–8.
- Roncal, T.; Oviedo, A.; Armentia, I.L.; Fernández, L.; Villarán, M.C. High yield production of monomer-free chitosan oligo-saccharides by pepsin catalyzed hydrolysis of a high deacetylation degree chitosan. Carbohydr. Res. 2007, 342, 2750–2756.
- Tsai, G.-J.; Zhang, S.-L.; Shieh, P.-L. Antimicrobial activity of a low-molecular-weight chitosan obtained from cellulase digestion of chitosan. J. Food Prot. 2004, 67, 396–398.
- Laokuldilok, T.; Potivas, T.; Kanha, N.; Surawang, S.; Seesuriyachan, P.; Wangtueai, S.; Phimolsiripol, Y.; Regenstein, J.M. Physicochemical, antioxidant, and antimicrobial properties of chitooligosaccharides produced using three different enzyme treatments. Food Biosci. 2017, 18, 28–33.
- Chang, S.-H.; Lin, H.-T.V.; Wu, G.-J.; Tsai, G.J. pH Effects on solubility, zeta potential, and correlation between antibacterial activity and molecular weight of chitosan. Carbohydr. Polym. 2015, 134, 74–81.
