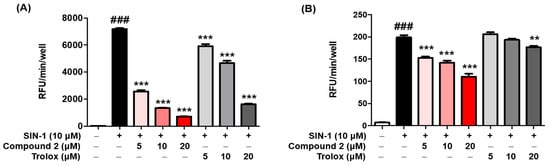
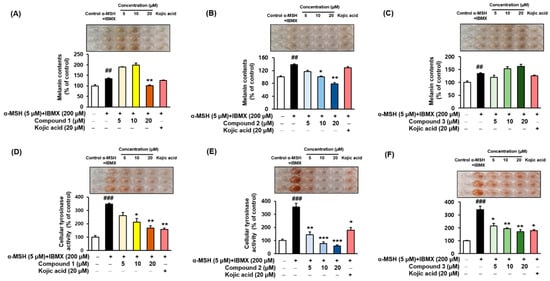
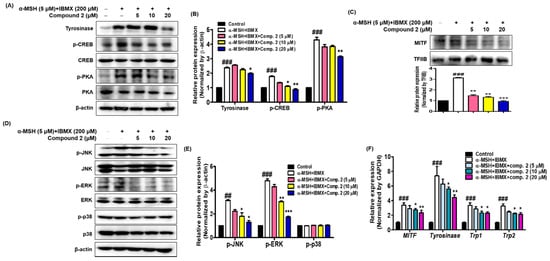
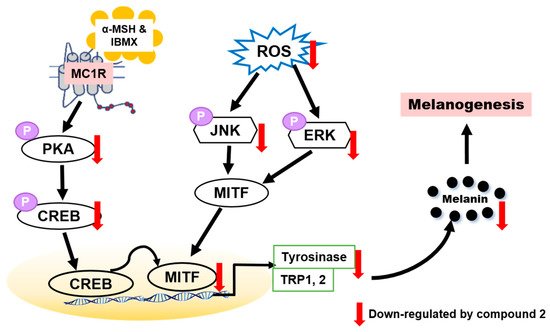
Thirteen (Z)-2-(substituted benzylidene)benzimidazothiazolone analogs were synthesized and evaluated for their inhibitory activity against mushroom tyrosinase. Among the compounds synthesized, compounds 1–3 showed greater inhibitory activity than kojic acid (IC50 = 18.27 ± 0.89 μM); IC50 = 3.70 ± 0.51 μM for 1; IC50 = 3.05 ± 0.95 μM for 2; and IC50 = 5.00 ± 0.38 μM for 3, and found to be competitive tyrosinase inhibitors. In silico molecular docking simulations demonstrated that compounds 1–3 could bind to the catalytic sites of tyrosinase. Compounds 1–3 inhibited melanin production and cellular tyrosinase activity in a concentration-dependent manner. Notably, compound 2 dose-dependently scavenged ROS in B16F10 cells. Furthermore, compound 2 downregulated the protein kinase A (PKA)/cAMP response element-binding protein (CREB) and mitogen-activated protein kinase (MAPK) signaling pathways, which led to a reduction in microphthalmia-associated transcription factor (MITF) expression, and decreased tyrosinase, tyrosinase related protein 1 (TRP1), and TRP2 expression, resulting in anti-melanogenesis activity. Hence, compound 2 may serve as an anti-melanogenic agent against hyperpigmentation diseases.
1. Introduction
Tyrosinase (polyphenol oxidase, EC 1.14.18.1), a binuclear copper-containing monooxygenase, is a critical rate-limiting melanogenic enzyme involved in melanogenesis, the process of melanin synthesis in the skin. Melanogenesis is initiated by the hydroxylation of tyrosine to
l-3,4-dihydroxy-phenylalanine (DOPA), which is catalyzed by tyrosinase. Tyrosinase also catalyzes the subsequent enzymatic conversion of DOPA to dopaquinone
[1][2][1,2]. Tyrosinase is the key factor involved in inducing dermatological disorders, including age spots, freckles, and melasma. Commercial tyrosinase inhibitors, such as hydroquinone, arbutin
[3], kojic acid
[4], ellagic acid
[5], and tranexamic acid, have been used as skin-whitening agents; however, they are associated with certain side effects, including carcinogenicity, chemical instability, and poor bioavailability
[6][7][6,7]. Thus, novel and efficient, anti-tyrosinase inhibitor with a favorable safety profile are necessary for anti-hyperpigmentation. There is a need to develop safe skin-whitening agents in order to overcome the limitations of established products.
α-Melanocyte-stimulating hormone (α-MSH) and 3-isobutyl-1-methylxanthine (IBMX) are the main physiological inducers of melanogenesis and accelerate tyrosinase activity through the cyclic adenosine monophosphate (cAMP) signaling pathway
[8]. Furthermore, α-MSH binds to melanocortin-1 receptors (MC1R) on the surface of melanocytes to activate the protein kinase A (PKA) pathway, phosphorylate the cAMP response element-binding protein (CREB) transcription factor; and induce the expression of microphthalmia-associated transcription factor (MITF), thus promoting melanogenesis
[9][10][9,10]. Activated MITF induces the expression of melanogenic enzyme genes, such as tyrosinase-related protein 1 (TRP1) and dopachrome tautomerase (TRP2; DCT)
[11][12][13][11,12,13]. The PKA/CREB and mitogen-activated protein kinase (MAPK) signaling pathways are important in melanogenesis; therefore, we wondered if chemicals or agents that block or activate this axis could have anti- or pro-melanogenic activity and thus serve as potential therapeutic agents for hyperpigmentation disorders
[14][15][16][14,15,16].
Reactive oxygen species (ROS) have been linked to several disorders, including aging and age-related diseases
[17][18][17,18]. Importantly, ROS levels can accelerate skin aging and increase both melanocyte proliferation and hyperpigmentation
[19][20][19,20]. Among the reactive species (RS) derived from melanocytes and keratinocytes, nitric oxide (∙NO) and superoxide anion radical (O
2˙
−) stimulate melanin synthesis by enhancing the protein expression of tyrosinase and TRP1
[21][22][23][24][21,22,23,24]. Therefore, antioxidants, inhibitors of tyrosinase, and scavengers of ROS may suppress melanogenesis in the epidermis.
Benzimidazothiazoles, as core rings, are polycyclic isosteres whose derivatives have been reported to exert anticancer
[25] and anti-inflammatory
[26][27][26,27] activities. However, no study has yet described its anti-tyrosinase and hyperpigmenting effects in vitro model. Previously, we reported that (
Z)-β-phenyl-α,β-unsaturated carbonyl scaffolds inhibit tyrosinase and melanogenesis both in vitro
[28][29][30][31][28,29,30,31]. As our continuous efforts to find potent tyrosinase inhibitors using (
Z)-β-phenyl-α,β-unsaturated carbonyl scaffolds, we integrated the benzimidazothiazolone template with other benzaldehydes to synthesize a (
Z)-β-phenyl-α,β-unsaturated carbonyl scaffold.
2. Results and Discussion on New Benzimidazothiazolone Derivatives
2.1. Synthesis of (Z)-2-(Substituted benzylidene)benzimidazothiazolone Derivatives 1–13
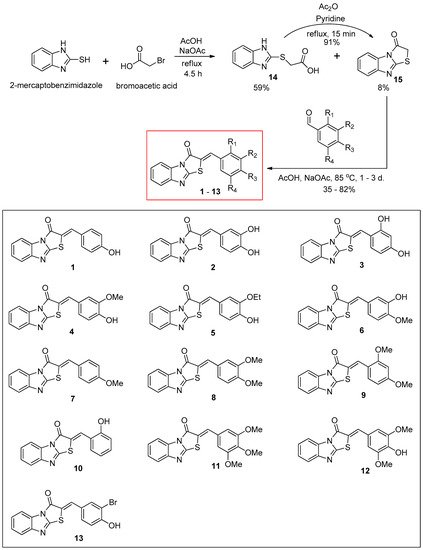
The synthetic route for the benzimidazothiazolone derivatives
1–
13 is illustrated in
Scheme 1. To synthesize the core template benzimidazothiazolone
15, commercially available 2-mercaptobenzimidazole was first condensed with bromoacetic acid. The reaction provided the desired compound
15 at a yield of 8%, with an intermediate
14 (59%). In the presence of acetic anhydride and pyridine, the intermediate
14 could be converted to the core template
15 rapidly and in high yield (91%) under reflux. Heating
15 and various benzaldehydes in the presence of acetic acid and sodium acetate for 15 h to 3 days produced 13 benzimidazothiazolone derivatives
1–
13 as solids in yields of 35–82%. The configuration of the newly formed double bond was determined using the vicinal
1H and
13C-coupling constants (
3J) in the proton-coupled
13C spectra.
Scheme 1. Synthesis scheme of (Z)-2-(substituted benzylidene)benzimidazothiazolone derivatives 1–13.
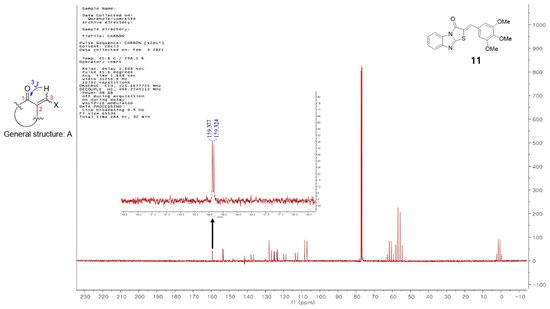
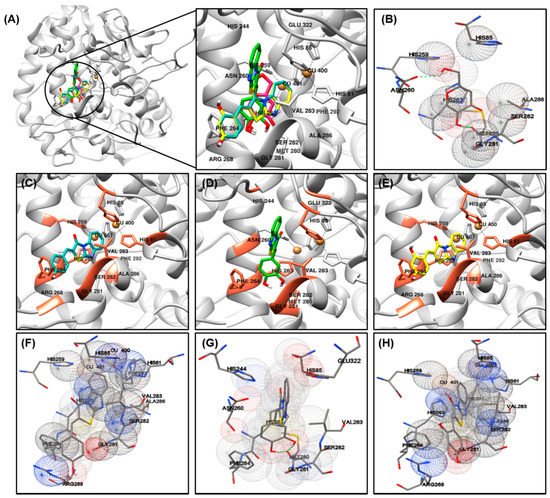
Vögeli et al.
[32][42] reported that the configuration of trisubstituted exocyclic C, C-double bonds in a general structure A (
Figure 1) differentiated the C, H-spin coupling constants over three bonds. The
3J of C(1) of compounds in which the oxygen of carbonyl and the 3-hydrogen were on the same side was 3.6 to 6.4 Hz, whereas that of the compounds in which the oxygen of carbonyl and 3-hydrogen were on the same side was roughly twice as large (generally >10 Hz). The
3J of C(1) of compound
11 was 5.3 Hz, confirming that
11 is a (
Z)-isomer. Structures of the 13 final compounds were confirmed with
1H and
13C NMR spectroscopy and mass spectroscopy.
Figure 1. General structure of derivatives with trisubstituted exocyclic C, C-double bond, and the proton-coupled 13C spectrum of compound 11.
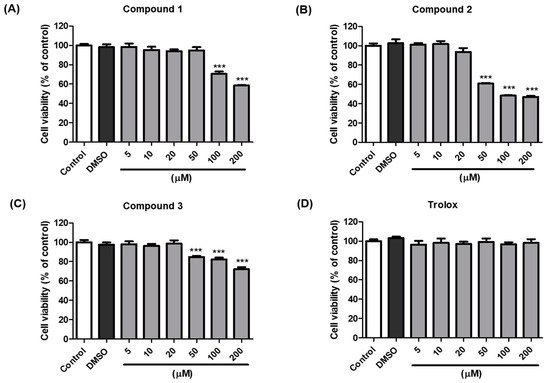
2.2. Inhibitory Activities of (Z)-2-(Substituted Benzylidene)benzimidazothiazolone Derivatives 1–13 against Mushroom Tyrosinase
To choose potent derivatives for cell-based in vitro of anti-tyrosinase and melanogenic-inhibitory activity, we synthesized 13 (
Z)-2-(substituted benzylidene)benzimidazothiazolone derivatives
1–
13, which were determined using mushroom tyrosinase and kojic acid (as a potent tyrosinase inhibitor)
[33][34][43,44]. The inhibitory potential of compounds
1–
13 against tyrosinase was assessed using
l-tyrosine as the substrate, and the results were described as the IC
50 values determined with linear regression analysis (
Table 12). Of the 13 synthesized compounds, three exhibited potent tyrosinase inhibitor;
1 (IC
50 = 3.70 µM) with a 4-hydroxy substituent;
2 (IC
50 = 3.05 µM) with a 3,4-dihydroxy substituent; and
3 (IC
50 = 5.00 µM) with a 2,4-dihydroxy substituent. Specifically, the inhibitory potencies of compounds
1,
2, and
3 were 4.9-, 6.0-, and 3.7-fold greater, respectively, than those of kojic acid (IC
50 = 18.27 µM). Compounds
4,
5,
8,
10,
11, and
13 showed moderate inhibitory activities, with IC
50 values of 42.06 µM, 21.03 µM, 41.40 µM, 27.96 µM, 34.10 µM, and 34.60 µM, respectively. Compounds
6,
7,
9, and
12 were inactive at the tested concentrations.
Table 12. Mushroom tyrosinase inhibitory activity of (Z)-2-(substituted benzylidene) benzimidazothiazolone derivatives 1–13.
| Compounds |
R1 |
R2 |
R3 |
R4 |
IC50 Values a (µM) |
| 1 |
H |
H |
OH |
H |
3.70 ± 0.51 |
| 2 |
H |
OH |
OH |
H |
3.05 ± 0.95 |
| 3 |
OH |
H |
OH |
H |
5.00 ± 0.38 |
| 4 |
H |
OMe |
OH |
H |
42.06 ± 4.28 |
| 5 |
H |
OEt |
OH |
H |
21.03 ± 0.82 |
| 6 |
H |
OH |
OMe |
H |
NI b |
| 7 |
H |
H |
OMe |
H |
NI |
| 8 |
H |
OMe |
OMe |
H |
41.40 ± 0.68 |
| 9 |
OMe |
H |
OMe |
H |
NI |
| 10 |
OH |
H |
H |
H |
27.96 ± 1.32 |
| 11 |
H |
OMe |
OMe |
OMe |
34.10 ± 4.06 |
| 12 |
H |
OMe |
OH |
OMe |
53.55 ± 4.71 |
| 13 |
H |
Br |
OH |
H |
34.60 ± 2.32 |
| Kojic acid c |
|
|
|
|
18.27 ± 0.89 |