Periodontitis, a major oral disease, affects a vast majority of the population but has been often ignored without realizing its long-fetched effects on overall human health. A realization in recent years of its association with severe diseases such as carditis, low birth weight babies, and preeclampsia has instigated dedicated research in this area. In the arena of periodontal medicines, the studies of past decades suggest a link between human periodontal afflictions and certain systemic disorders such as cardiovascular diseases, diabetes mellitus, respiratory disorders, preterm birth, autoimmune disorders, and cancer. Although, the disease appears as a locoregional infection, the periodontal pathogens, in addition their metabolic products and systemic mediators, receive access to the bloodstream, thereby contributing to the development of systemic disorders. Mechanism-based insights into the disease pathogenesis and association are highly relevant and shall be useful in avoiding any systemic complications.
- inflammation
- cardiovascular diseases
- diabetes mellitus
- preterm birth
- autoimmune disorders
- cancer
- periodonto-therapeutic
Note: The following contents are extract from your paper. The entry will be online only after author check and submit it.
1. Introduction
2. Possible Mechanism behind the Systemic Manifestations of Periodontitis
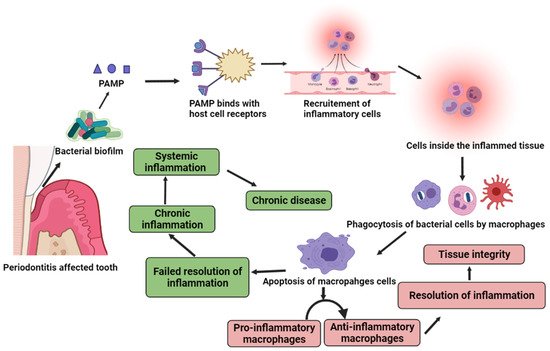
-
Oral-hematogenous migration of periodontal pathogens and its direct effects to target organs [14].
-
Transtracheal migration of periodontal pathogens and its direct effects to target organs.
-
Oral-hematogenous migration of inflammatory mediators such as cytokines and antibodies with their effects on distant organs.
3. Periodontitis and Systemic Complications
3.1. Periodontitis and Cardiovascular Diseases
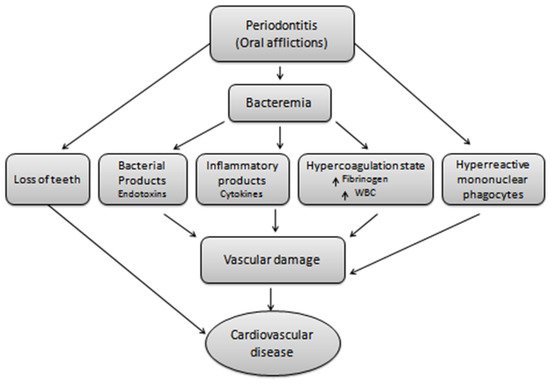
3.1.1. Atherosclerosis
3.1.2. Coronary Heart Disease
3.1.3. Stroke
3.2. Periodontitis and Autoimmune Disorders
3.3. Periodontitis and Respiratory Disorder
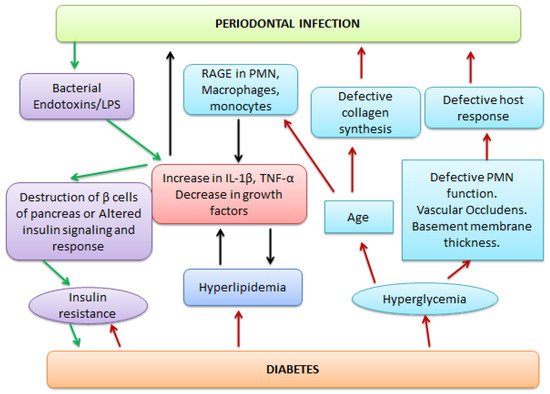
3.4. Periodontitis and Diabetes Mellitus
3.4.1. Periodontitis as a Consequence of Diabetes
3.4.2. Diabetes as a Complication of Periodontitis
3.5. Periodontitis and Pre-Term Low Birth Weight Babies

3.6. Periodontitis and Cancer
-
Periodontal pathogens induce chronic inflammation; this promotes already initiated cells, leading to uncontrolled cell growth and potential carcinogenesis.
-
Periodontopathic bacteria may also have a more direct role through local inflammatory responses and carcinogenic transformations. Helicobacter pylori infection is an example of this.
-
Chronic periodontal disease may suggest that an individual’s immune system is compromised, unable to clear the infection, and consequently deficient at surveillance for tumor growth.
-
Periodontal inflammation can lead to genetic alteration via the production of reactive oxygen and nitrogen species-type oxidizing compounds.
-
Carcinogenesis and wound healing shared several common biological processes and carcinogenesis can be considered as an unregulated form of wound healing.
-
Failure of the periodontal inflammation-resolving mechanism.
4. Current treatment modalities and advanced pharmaceutical approach
Although the clinical mainstay treatment of periodontitis revolves around the use of various types of antibiotics along with other symptomatic relief drugs like anti-inflammatory and others, however, the continuous efforts of pharmaceutical researchers have led to the design and development of certain advanced dento-therapeutics which are primarily focused on Spatio-temporal drug delivery for a prolonged period [56]. A recce of concerned literature has revealed the prevalence of various formulation designs focusing on locoregional effects. Table 1. summarizes the various pharmaceutical research designs with a primary objective of the formulation.
The conventional treatment modalities for periodontal infections primarily focus on dental plaque, an oral biofilm, responsible for causing various inflammatory reactions in periodontal tissues. Different available approaches to target dental plaque are plaque control, non-surgical, and surgical interventions. Adjuvant therapies including antibiotics or supplements are also included however, they face major limitations of antibiotic resistance, local inflammation in the periodontium, and host immune responses, thus, causing partial effectiveness [57]. Henceforth, advanced strategies to mitigate, treat and lessen periodontal infections have become a need of the present time. Recently, various modalities such as quorum sensing inhibitors, inflammasome targeting, host inflammatory substances, bone immune responses, and FDA-approved anabolic agents, namely, Teriparatide and sclerostin antibody have been introduced as a step forward against periodontal infections [58]. Quorum sensing inhibitors target molecules that modulate microbial signaling mechanisms, a primary step in biofilm formation, thus inhibiting plaque biofilm formation and controlling periodontal infections [59]. The inflammasome is a naturally produced cytosolic multiprotein oligomer of the innate immune system. Abnormal inflammasome activation is associated with various ailments including periodontal disease pathogenesis. Thus, the development of drugs that directly target and inhibit an inflammasome activation may hinder the progression of the periodontal infection [60]. The FDA-approved anabolic agents for bone regeneration could be incorporated with current treatment regimens as dental implants for severe cases of periodontitis [58]. Reportedly, the use of photodynamic therapy has also gained considerable attention against dental plaque for example the potential use of nano-based antibacterial photodynamic therapies to combat bacterial plaque-initiated oral diseases and adjunctive application of antimicrobial photodynamic therapy in nonsurgical periodontal treatment [61]. Various developing strategies such as bioactive-based dental polymers, nano-size building blocks, bio-implants are also being considered as new generation dental restoration tools and can also inhibit oral biofilms [57].
Table 1. A recce of periodontal formulations and their applications
Formulation
Components/
Devices
Intervention/
condition
Description
Observations
Reference
Nanofibrous scaffolds
Silver nanoparticles, AgNPs and hydroxyapatite nanoparticles, HANPs electrospun to prepare nanofibrous composites based on Polylactic acid/cellulose acetate (PLA/CA) or polycaprolactone (PCL) polymers.
Periodontal tissue and bone regeneration
Biodegradable electrospun nanoparticles-in-nanofibers-based based scaffolds for Guided tissue regeneration (GTR) and guided bone regeneration (GBR) with enhanced mechanical properties, cell adhesion, biocompatibility, and antibacterial properties.
In-vitro studies of nanofibrous films cut into 10 × 10 mm2 samples showed that the addition of HANPs improved the cell viability by about 50 and AgNPs provided sustained antibacterial activity (40 mm zone of inhibition diameter) for 32 days. Additionally, the nanofibrous scaffolds offer optimum mechanical properties, tensile modulus (20-38 MPa), and a desirable degradation profile (40-70% of its mass in 8 weeks).
[62]
Mineralized Nanofiber
PLGA-collagen-gelatin
coupled with calcium-binding bone morphogenetic protein 2 (BMP-2)
Alveolar bone regeneration
Mineralized nanofiber segments (20 μm) coupled with BMP-2 mimicking peptides for periodontal bone regeneration.
In animal studies, the mineralized nanofibers were implanted into critical-sized maxillary defects of 2 mm diameter 2 mm depth in rats and a sustained release profile was recorded for over 4 weeks. X-ray microcomputed tomography (μ-CT) analysis revealed ~3 times greater new bone volume and mineral density.
[63]
Polymeric Fibers
Electrospun nanofibers encapsulating BAR using Poly (lactic-co-glycolic acid) (PLGA), poly (L-lactic acid) (PLLA), and polycaprolactone (PCL), either as a single solution or as blended with polyethylene oxide (PEO).
Inhibition of Porphyromonas gingivalis and adherence to Streptococcus gordonii
Rapid-release polymeric electrospun nanofibers against P. gingivalis/S. gordonii biofilms in vitro.
The most promising formulation, 10:90 PLGA: PEO of electrospun nanofibers has demonstrated a 95% BAR release after 4 h, dose-dependent inhibition of biofilm formation (IC50 = 1.3 μM), disruption of established dual-species biofilms (IC50 =2 μM), and maintenance of high cell viability.
[64]
Nanofibrous membrane
PLLA/gelatin
Periodontal tissue regeneration
Biodegradable multifunctional nanofibrous membrane prepared by electrospinning biodegradable polymers with magnesium oxide nanoparticles (nMgO) for periodontal tissue regeneration and high antibacterial capacity.
In vitro results showed that incorporating nMgO into poly (L-lactic acid) (PLA)/gelatin elevates the tensile strength to maintain structural stability and adjust the degradation rate for periodontal regeneration. Considerable antibacterial and osteogenic properties were also observed. The in-vivo investigations in a rat periodontal defect model demonstrated effective periodontal tissue regeneration guided via nMgO-incorporated membranes.
[65]
Polymeric films
PLLA/PCL blends containing propolis
Guided periodontal tissue regeneration
Biodegradable composite membranes produced from PCL/PLLA blends with a natural antibacterial extract (propolis) as novel periodontal barrier membranes.
The in-vitro antibacterial studies have revealed remarkable activities against Staphylococcus aureus (17mm zone of inhibition). The prepared films also showed faster degradation in physiological conditions.
[66]
Biopolymer composite film
Curcumin
Topical patches for wound care, periodontitis, and oral cancer treatment
Multifunctional biopolymer composites based on curcumin-loaded bacterial cellulose/alginate/gelatin.
The in-vitro studies have shown substantial antibacterial activity against E. coli and S. aureus infection. The purported composite films exhibited cytotoxicity to human keratinocytes and human gingival fibroblasts and also show potent anticancer activity in oral cancer cells.
[67]
Regenerative scaffolds
Cellulose hydrogels and biopolymers derived from plants. Larrea tridentate
Periodontal tissue regeneration
Cellulose hydrogel films enriched with LT for biomedical application in wound healing and as regenerative scaffolds.
For in-vitro studies, NIH3T3 mouse embryonic cells were used for the measurements of cell viability and morphology assays. For in-vivo assay hydrogel films were implanted intramuscularly into female Wistar rats (250 g weight; 2 months), to analyze their cytocompatibility and biocompatibility.
[68]
Nanofibrous membrane
PLGA/gelatin, Dexamethasone (osteogenic), Doxycycline hyclate (anti-bacterial agent).
Guided bone regeneration
Bi-layered electrospun composite nano-membrane with combined osteogenic and antibacterial properties for guided bone regeneration.
In vitro studies indicated that both dexamethasone and doxycycline hyclate have followed a favorable sustained drug release profile. The cell viability evaluation suggested good cytocompatibility. The osteogenesis analyses demonstrated an enhanced osteoinductive capacity for rat bone marrow stem cells, increased alkaline phosphatase activity, enhanced calcium deposition, and upregulated osteocalcin expression. Further, the antimicrobial experiments revealed effective antibacterial potency.
[69,70]
Hydrogel
Polyacrylic acid (PAA) hydrogel containing metronidazole
Therapeutic dressing
Gamma-ray irradiation targeted metronidazole-loaded PAA hydrogel.
The in-vitro cytocompatibility test was performed according to ISO 10993-5 and the formulation exhibited no cytotoxicity. The antibacterial activity against E. coli (ATCC 43895), S. aureus (ATCC 14458), and S. mutans (ATCC 25175) yielded satisfactory results. In release studies, metronidazole from the PAA hydrogel was consistently released and reached approximately 80% at 120 min.
[71]
Hydrogel
Doxycycline/
Lipoxin, Poly isocyano peptide (PIC)
Periodontal
Antimicrobial and anti-inflammatory thermo-reversible hydrogel for improved gingival clinical attachment and periodontal drug delivery.
The formulations were characterized in-vitro and in dogs with naturally occurring periodontitis. The results showed that the prepared hydrogel could be easily injected into periodontal pockets owing to the thermo-reversible nature of the material. The formulation yielded significant release with no local or systemic adverse effects. The reduced subgingival bacterial load, pro-inflammatory interleukin-8 level and an improved gingival clinical attachment by 0.6 mm was also observed.
[72]
Liposomal gel
Lidocaine/
Prilocaine
Periodontal
A randomized, double-blind, cross-over, placebo-controlled clinical trial of liposomal gel (intra pocket) for non-invasive anesthesia in scaling and root during periodontal therapy.
The sample size calculation was based on pain intensity (primary outcome) using visual analogue scale (VAS) data. The study reported no difference between intervention groups concerning pain frequency/intensity (primary outcome). The anesthetic gel did not interfere with the hemodynamic parameters (secondary outcome). However, the above observations have few limitations, first, there is no ideal scale for measuring pain, and hence further clarification is needed. Second, periodontal procedures usually cause low or moderate pain. Third, low patient compliance as many does not prefer local anesthesia.
[73]
Gel
Doxycycline encapsulated in β-cyclodextrin
Periodontitis
A randomized, blinded clinical trial to compare the effects of 10% doxycycline gel with doxycycline encapsulated in β-cyclodextrin gel on thirty-three subjects with periodontitis for 30 days.
The adjunctive topical agents (doxycycline encapsulated in β-cyclodextrin gel) + scaling and root planning resulted in significant improvements in clinical periodontal parameters such as visible plaque index, measurement of periodontal probing depth, clinical attachment level, and bleeding on probing.
[74]
Hybrid hydrogels
Mesoporous silica, Minocycline, Silver, Gelatin methacrylate
Periodontal infection
Near-infrared light (NIR)-activated hybrid hydrogels.
The hybrid hydrogels showed controllable minocycline delivery with increased release rates (in-vitro). The hydrogels also exhibited synergistic antibacterial activity (90%) against Porphyromonas gingivalis. The photothermal treatment was as high as 66.7% against P. gingivalis as well to rapidly eliminate and maintains low bacterial retention in periodontal pockets. Further, the in vitro cytotoxicity studies revealed an 80% cell viability.
[75]
Hydrogel nanoparticles
Minocycline, Zinc oxide, Serum albumin
Periodontitis
Broad-spectrum hydrogel-based minocycline and zinc oxide-loaded serum albumin nanoparticles for periodontitis application with low toxicity and high antimicrobial and antibacterial activity.
In in-vitro analysis, a slow-release time was observed. Encapsulation efficiency was 99.99%. The in-vitro skin adhesion experiment showed a bioadhesive force of 0.35 N. Broad-spectrum antimicrobial, antibacterial ability, and high cell survival rates with low toxicity.
[76]
Microspheres
PLGA, PIC, Doxycycline, Lipoxin
Periodontal infection
A tunable and injectable localized system based on PLGA microspheres, containing doxycycline and lipoxin, dispersed into thermo-reversible PIC hydrogel for personalized periodontal application.
The in-vitro efficacy and bioactivity of the released doxycycline
has presented a comparable zone of inhibition with respect to fresh or unbound drug against gram-negative anaerobic bacteria Porphyromonas gingivalis (ATCC 33277). The fluorescent bead internalization assay of lipoxin has revealed that more fluorescent beads were internalized that may stimulate RAW264.7 macrophage (Gibco) phagocytosis. The in-vivo test on ten 8-week-old male Wistar rats (~250 g) has exhibited no obvious inflammatory responses.
[77]
Combination gel
Chlorhexidine, Metronidazole
Gingivitis
A triple-blind, randomized clinical trial on 90 subjects to compare and assess 0.8% Metronidazole gel, 0.2% Chlorhexidine gel, and alternate application of the two gels against dental plaque and gingivitis for 14 days.
The primary outcome measures are the bleeding index. The secondary outcome measures are oral hygiene index, probing depth, and gingival index. The aforementioned outcomes were compared after 2 weeks and 6 weeks.
[78]
Chitosan templates
Chitosan, Glutaraldehyde, Doxycycline Hyclate
Periodontal tissue regeneration
Cross-linking doxycycline-loaded freeze gelated chitosan templates for periodontitis.
The in-vitro analysis of chitosan templates through a conventional dialysis sac method showed a 40 μg/mL of release after 24 h. Such a suitable drug release rate will also limit the toxicological effect of the cross-linking agent.
[79]
Retraction gels
Epinephrine, Tetrahydrozoline, Oxymetazoline, Phenylephrine
Gingival retraction
In-vitro vaso-constrictive retraction agents against primary human gingival fibroblasts in periodontal tissues.
Immunocytochemical analysis revealed the biological effect of retraction gels on the expression of collagen types I and III. The generation of reactive oxygen species triggered by the retraction gels indicated oxidative stress similar to the control cells using the dichlorofluorescein (DCF) fluorescent probe.
[80]
In-situ gel
Doxycycline Hyclate, Shellac, Ethocel, Eudragit RS
Periodontitis
In situ forming gels for localized periodontal pocket delivery.
The in-vitro release study through a dialysis membrane follows a sustained release pattern. Also, it exhibited in-vitro degradability and antimicrobial effect against S. aureus, S mutans, E. coli, P. gingivalis, and C. albicans.
[81]
Nanofiber based hydrogel
Cellulose, κ-carrageenan oligosaccharide, Surfactin, Herbmedotcin
Periodontitis
Anti-microbial loaded cellulose nanofiber and κ-carrageenan oligosaccharide composite hydrogels for strong antibacterial activity against periodontal pathogens such as Streptococcus mutans, Porphyromonas gingivalis, Fusobacterium nucleatum, and Pseudomonas aeruginosa in periodontitis treatment.
Purportedly, they reduce the reactive oxygen species (ROS) generation, transcription factor, and cytokine production in human gingival fibroblast cells (HGF) under inflammatory conditions.
[82]
Hydrosilver gel
Silver
Chronic periodontitis
A prospective longitudinal pilot study using polymerase chain reaction analysis of hydrosilver gel against dental plaque in chronic periodontitis.
The in-vivo model of chronic periodontitis was used for 15 days. The LAB®-Test (LAB s.r.l.®, Ferrara, Italy) detected and quantify the presence and the level of the most involved periodontitis pathogens that constitute the ‘red complex’, P. gingivalis, Tannerella forsythia, and Treponema denticola. Other bacteria of the ‘orange complex’ were also monitored, such as Fusobacterium nucleatum, Campylobacter rectus, Aggregatibacter actinomycetemcomitans, Atopobium rimae, Eubacterium saphenum, Porphyromonas endodontalis, and Treponema lecithinolyticum, as the main component of microbiological shift.
[83]
Microporous annealed particle (MAP) hydrogels
Poly(ethylene) glycol
Tissue engineering and regeneration
Versatile new platform for the delivery of human periodontal ligament stem cells and periodontal tissue regeneration.
In-vitro characterization revealed excellent retention, proliferation, and spreading of platelet-derived growth factors and human periodontal ligament stem cells within hydrogels.
[84]
Exosomal nanoparticles
Ginger phosphatidic acid
Oral biofilms
Plant-derived nanoparticles to inhibit P. gingivalis biofilm.
Demonstrated inhibition of P. gingivalis induced bone loss and pathogenicity in an in-vivo mouse model of chronic periodontitis
[85]
Mesoporous nanospheres
Ipriflavone
Periodontal infection
Ipriflavone-loaded mesoporous nanospheres for periodontal augmentation
Periodontal augmentation in an in-vitro osteogenesis model (MC3T3-E1 osteoprogenitor cells).
[86]
Nanocomposites
Chlorin e6
Periodontal diseases
A Photodynamic therapy guided bioactive nanocomposites containing chlorin e6 as photosensitizer against biofilms on dentin squares. The dentin samples were prepared from extracted caries-free human molars which serve as the substrates for biofilm formation.
Photosensitizer effect on Porphyromonas gingivalis, Prevotella intermedia, and Fusobacterium nucleatum and their corresponding biofilms on dentin squares 5 × 5 mm (thickness of about 1 mm).
[87]
Nanocomplexes
Bovine serum albumin
Periodontitis
Nanocomplexes for enhanced the osteogenic differentiation of inflammatory periodontal ligament stem cells.
The in-vitro hemolysis assay and in-vivo cytocompatibility assay using BALB/c mice (8 weeks old) revealed the high transfection efficiency and biocompatibility of the prepared nano complexes.
[88]
Carbon Quantum Dots
Tinidazole, Metronidazole
Oral biofilms
Periodontitis treatment by penetrating the P. gingivalis biofilm and destroying its related genes.
An in-vitro biofilm penetration assay revealed that nanoscale tinidazole carbon quantum dots can penetrate through the biofilm to induce significant inhibition of P. gingivalis under the biofilm. Also, as exhibited in the in-vitro antibacterial assay, tinidazole carbon quantum dots impairs toxicity, inhibits the major virulence factors and related genes involved in the biofilm formation of P. gingivalis.
[89]
Nanoplatelets
Fluoride
Periodontal bone tissue regeneration.
Osteogenic differentiation of human dental follicle stem cells for tissue regeneration.
MTS assay and cellular morphology analysis demonstrated low cytotoxicity of prepared nanoplatelets at low concentrations.
[90]
5. Conclusion
Periodontitis is no more a standalone disease and transcends its effect beyond the site of its infection in the oral cavity. The unravelling of the underpinnings of its association with plethora of systemic diseases have put on alert both the medical practitioners as well as researchers. There seems to be an unmet need of collating scientifically derived data in the aforementioned domain so as to support the therapeutic decision making of the physicians. As a part of therapeutic strategy, it is advisable to carry out a discernible assessment of the periodontal infections while diagnosing apparently unrelated diseases like atherosclerosis, diabetes and even cancer. There has been a remarkable progression in devising various novel dental therapeutics adding both volume and quality to the dentists’ armamentarium which if used judiciously would lead to faster and earliest amelioration of these site-specific diseases. The novel pharmaceutical interventions are majorly laden with attributes of reduced drug doses and sustained effect which make them economically viable. Additionally, an early recognition, treatment of the disease shall be a great stepping stone towards limiting or precluding its adverse effect(s)on other systemic disorders and would substantially improvise patient outcomes.
References- Divaris, K. Searching Deep and Wide: Advances in the Molecular Understanding of Dental Caries and Periodontal Disease. Adv. Dent. Res. 2019, 30, 40–44.
- Jain, P.; Mirza, M.A.; Iqbal, Z. Unraveling the Etiology of Periodontitis. Int. J. Biomed. Investig. 2021, 4, 1–4.
- Jain, P.; Mirza, M.A.; Iqbal, Z. A 4-D Approach for Amelioration of Periodontitis. Med. Hypotheses 2019, 133, 109392, doi:10.1016/j.mehy.2019.109392.
- Bui, F.Q.; Almeida-da-Silva, C.L.C.; Huynh, B.; Trinh, A.; Liu, J.; Woodward, J.; Asadi, H.; Ojcius, D.M. Association between Periodontal Pathogens and Systemic Disease. Biomed. J. 2019, 42, 27–35, doi:10.1016/j.bj.2018.12.001.
- Falcao, A.; Bullón, P. A Review of the Influence of Periodontal Treatment in Systemic Diseases. Periodontol. 2000 2019, 79, 117–128.
- Abusleme, L.; Dupuy, A.K.; Dutzan, N.; Silva, N.; Burleson, J.A.; Strausbaugh, L.D.; Gamonal, J.; Diaz, P.I. The Subgingival Microbiome in Health and Periodontitis and Its Relationship with Community Biomass and Inflammation. ISME J. 2013, 7, 1016–1025.
- Rodger, S.F.A.E.L.; Haslett, D.C.; Rossi, A.G. Neutrophil Apoptosis: Relevance to the Innate Immune Response and Inflammatory Disease. 2010.
- Paul, O.; Arora, P.; Mayer, M.; Chatterjee, S. Inflammation in Periodontal Disease: Possible Link to Vascular Disease. Front. Physiol. 2021, 11, 1818.
- Cecoro, G.; Annunziata, M.; Iuorio, M.T.; Nastri, L.; Guida, L. Periodontitis, Low-Grade Inflammation and Systemic Health: A Scoping Review. Medicina (Mex.) 2020, 56, 272.
- Hajishengallis, G.; Chavakis, T. Local and Systemic Mechanisms Linking Periodontal Disease and Inflammatory Comorbidities. Nat. Rev. Immunol. 2021, 1–15, doi:10.1038/s41577-020-00488-6.
- Preshaw, P.M.; Taylor, J.J.; Jaedicke, K.M.; De Jager, M.; Bikker, J.W.; Selten, W.; Bissett, S.M.; Whall, K.M.; van de Merwe, R.; Areibi, A. Treatment of Periodontitis Reduces Systemic Inflammation in Type 2 Diabetes. J. Clin. Periodontol. 2020, 47, 737–746.
- Rapone, B.; Ferrara, E.; Corsalini, M.; Qorri, E.; Converti, I.; Lorusso, F.; Delvecchio, M.; Gnoni, A.; Scacco, S.; Scarano, A. Inflammatory Status and Glycemic Control Level of Patients with Type 2 Diabetes and Periodontitis: A Randomized Clinical Trial. Int. J. Environ. Res. Public. Health 2021, 18, 3018.
- Igari, K.; Kudo, T.; Toyofuku, T.; Inoue, Y.; Iwai, T. Association between Periodontitis and the Development of Systemic Diseases. Oral Biol Dent 2014, 2, 4.
- Tonetti, M.S.; Van Dyke, T.E.; working group 1 of the joint EFP/AAP workshop* Periodontitis and Atherosclerotic Cardiovascular Disease: Consensus Report of the Joint EFP/AAPWorkshop on Periodontitis and Systemic Diseases. J. Periodontol. 2013, 84, S24–S29.
- Blaizot, A.; Vergnes, J.-N.; Nuwwareh, S.; Amar, J.; Sixou, M. Periodontal Diseases and Cardiovascular Events: Meta-Analysis of Observational Studies. Int. Dent. J. 2009, 59, 197–209.
- Janket, S.-J.; Baird, A.E.; Chuang, S.-K.; Jones, J.A. Meta-Analysis of Periodontal Disease and Risk of Coronary Heart Disease and Stroke. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endodontology 2003, 95, 559–569.
- Bäck, M.; Yurdagul, A.; Tabas, I.; Öörni, K.; Kovanen, P.T. Inflammation and Its Resolution in Atherosclerosis: Mediators and Therapeutic Opportunities. Nat. Rev. Cardiol. 2019, 16, 389–406.
- Sanz, M.; Marco del Castillo, A.; Jepsen, S.; Gonzalez-Juanatey, J.R.; D’Aiuto, F.; Bouchard, P.; Chapple, I.; Dietrich, T.; Gotsman, I.; Graziani, F. Periodontitis and Cardiovascular Diseases: Consensus Report. J. Clin. Periodontol. 2020, 47, 268–288.
- Jiménez-Sánchez, M.C.; Cabanillas-Balsera, D.; Areal-Quecuty, V.; Velasco-Ortega, E.; Martín-González, J.; Segura-Egea, J.J. Cardiovascular Diseases and Apical Periodontitis: Association Not Always Implies Causality. Med. Oral Patol. Oral Cirugia Bucal 2020, 25, e652.
- Miricescu, D.; Totan, A.; Stanescu, I.-I.; Radulescu, R.; Stefani, C.; Alexandra Stanescu, A.M.; Greabu, M. PERIODONTAL DISEASE AND SYSTEMIC HEALTH. Romanian Med. J. 2019, 66.
- Zardawi, F.; Gul, S.; Abdulkareem, A.; Sha, A.; Yates, J. Association between Periodontal Disease and Atherosclerotic Cardiovascular Diseases: Revisited. Front. Cardiovasc. Med. 2020, 7.
- Bourgeois, D.; Inquimbert, C.; Ottolenghi, L.; Carrouel, F. Periodontal Pathogens as Risk Factors of Cardiovascular Diseases, Diabetes, Rheumatoid Arthritis, Cancer, and Chronic Obstructive Pulmonary Disease—Is There Cause for Consideration? Microorganisms 2019, 7, 424.
- Ji, S.; Choi, Y. Microbial and Host Factors That Affect Bacterial Invasion of the Gingiva. J. Dent. Res. 2020, 99, 1013–1020.
- Yamamoto, T.; Eguchi, T. Heat Shock Proteins and Periodontitis–Cross-Reaction Between Bacterial and Human HSP in Periodontal Infection Linking with Cardiovascular Diseases. 2020.
- Esteves-Lima, R.-P.; Reis, C.-S.; Santirocchi-Júnior, F.; Abreu, L.-G.; Costa, F.-O. Association between Periodontitis and Serum C-Reactive Protein Levels. J. Clin. Exp. Dent. 2020, 12, e838.
- Montero, E.; López, M.; Vidal, H.; Martínez, M.; Virto, L.; Marrero, J.; Herrera, D.; Zapatero, A.; Sanz, M. Impact of Periodontal Therapy on Systemic Markers of Inflammation in Patients with Metabolic Syndrome: A Randomized Clinical Trial. Diabetes Obes. Metab. 2020, 22, 2120–2132.
- Degasperi, G.R.; Ossick, M.V.; Pinheiro, S.L.; Etchegaray, A. Autoimmunity and Periodontal Disease: Arguing a Possible Correlation. Indian J. Dent. Res. 2020, 31, 615.
- Möller, B.; Kollert, F.; Sculean, A.; Villiger, P.M. Infectious Triggers in Periodontitis and the Gut in Rheumatoid Arthritis (RA): A Complex Story about Association and Causality. Front. Immunol. 2020, 11, 1108.
- Ballini, A.; Dipalma, G.; Isacco, C.G.; Boccellino, M.; Di Domenico, M.; Santacroce, L.; Nguyễn, K.C.; Scacco, S.; Calvani, M.; Boddi, A. Oral Microbiota and Immune System Crosstalk: A Translational Research. Biology 2020, 9, 131.
- Lamont, R.J.; Hajishengallis, G.N.; Koo, H.M.; Jenkinson, H.F. Oral Microbiology and Immunology; John Wiley & Sons, 2020;
- Xu, W.; Zhou, W.; Wang, H.; Liang, S. Roles of Porphyromonas Gingivalis and Its Virulence Factors in Periodontitis. Adv. Protein Chem. Struct. Biol. 2020, 120, 45–84.
- Chopra, A.; Bhat, S.G.; Sivaraman, K. Porphyromonas Gingivalis Adopts Intricate and Unique Molecular Mechanisms to Survive and Persist within the Host: A Critical Update. J. Oral Microbiol. 2020, 12, 1801090.
- Hočevar, K.; Vizovišek, M.; Wong, A.; Kozie\l, J.; Fonović, M.; Potempa, B.; Lamont, R.J.; Potempa, J.; Turk, B. Proteolysis of Gingival Keratinocyte Cell Surface Proteins by Gingipains Secreted From Porphyromonas Gingivalis–Proteomic Insights Into Mechanisms Behind Tissue Damage in the Diseased Gingiva. Front. Microbiol. 2020, 11, 722.
- Farrugia, C.; Stafford, G.P.; Potempa, J.; Wilkinson, R.N.; Chen, Y.; Murdoch, C.; Widziolek, M. Mechanisms of Vascular Damage by Systemic Dissemination of the Oral Pathogen Porphyromonas Gingivalis. FEBS J. 2021, 288, 1479–1495.
- Sapey, E.; Yonel, Z.; Edgar, R.; Parmar, S.; Hobbins, S.; Newby, P.; Crossley, D.; Usher, A.; Johnson, S.; Walton, G.M. The Clinical and Inflammatory Relationships between Periodontitis and Chronic Obstructive Pulmonary Disease. J. Clin. Periodontol. 2020, 47, 1040–1052.
- Gomes-Filho, I.S.; Cruz, S.S. da; Trindade, S.C.; Passos-Soares, J. de S.; Carvalho-Filho, P.C.; Figueiredo, A.C.M.G.; Lyrio, A.O.; Hintz, A.M.; Pereira, M.G.; Scannapieco, F. Periodontitis and Respiratory Diseases: A Systematic Review with Meta-Analysis. Oral Dis. 2020, 26, 439–446.
- Shehri, I.; Nissar, S.; Anis, B.A.; Singh, S. Evaluation of Prevalence of Periodontitis in Patients with Pulmonary Disease. J. Adv. Med. Dent. Sci. Res. 2020, 8, 72–75.
- Apessos, I.; Voulgaris, A.; Agrafiotis, M.; Andreadis, D.; Steiropoulos, P. Effect of Periodontal Therapy on COPD Outcomes: A Systematic Review. BMC Pulm. Med. 2021, 21, 1–16.
- Genco, R.J.; Graziani, F.; Hasturk, H. Effects of Periodontal Disease on Glycemic Control, Complications, and Incidence of Diabetes Mellitus. Periodontol. 2000 2020, 83, 59–65, doi:10.1111/prd.12271.
- Nguyen, A.T.M.; Akhter, R.; Garde, S.; Scott, C.; Twigg, S.M.; Colagiuri, S.; Ajwani, S.; Eberhard, J. The Association of Periodontal Disease with the Complications of Diabetes Mellitus. A Systematic Review. Diabetes Res. Clin. Pract. 2020, 165, 108244, doi:10.1016/j.diabres.2020.108244.
- Graves, D.T.; Ding, Z.; Yang, Y. The Impact of Diabetes on Periodontal Diseases. Periodontol. 2000 2020, 82, 214–224, doi:10.1111/prd.12318.
- AlQranei, M.S.; Chellaiah, M.A. Osteoclastogenesis in Periodontal Diseases: Possible Mediators and Mechanisms. J. Oral Biosci. 2020, 62, 123–130, doi:10.1016/j.job.2020.02.002.
- Polak, D.; Sanui, T.; Nishimura, F.; Shapira, L. Diabetes as a Risk Factor for Periodontal Disease—Plausible Mechanisms. Periodontol. 2000 2020, 83, 46–58, doi:10.1111/prd.12298.
- Pérez-Losada, F. de L.; Estrugo-Devesa, A.; Castellanos-Cosano, L.; Segura-Egea, J.J.; López-López, J.; Velasco-Ortega, E. Apical Periodontitis and Diabetes Mellitus Type 2: A Systematic Review and Meta-Analysis. J. Clin. Med. 2020, 9, 540, doi:10.3390/jcm9020540.
- Wu, C.; Yuan, Y.; Liu, H.; Li, S.; Zhang, B.; Chen, W.; An, Z.; Chen, S.; Wu, Y.; Han, B.; et al. Epidemiologic Relationship between Periodontitis and Type 2 Diabetes Mellitus. BMC Oral Health 2020, 20, 204, doi:10.1186/s12903-020-01180-w.
- Jinnat, M.A.; Ahmed, S.; Monira, S.; Haque, M.M.; Musharraf, M.; Hasan, M.R. Maternal Risk Factors, Clinical Profile and Short-Term Outcome of Pre-Term Low Birth Weight Babies. KYAMC J. 2020, 11, 77–82, doi:10.3329/kyamcj.v11i2.48419.
- Green, E.S.; Arck, P.C. Pathogenesis of Preterm Birth: Bidirectional Inflammation in Mother and Fetus. Semin. Immunopathol. 2020, 42, 413–429, doi:10.1007/s00281-020-00807-y.
- Parris, K.M.; Amabebe, E.; Cohen, M.C.; Anumba, D.O. Placental Microbial–Metabolite Profiles and Inflammatory Mechanisms Associated with Preterm Birth. J. Clin. Pathol. 2021, 74, 10–18, doi:10.1136/jclinpath-2020-206536.
- Boggess, K.A. Choosing the Left Fork: Steven Offenbacher and Understanding Maternal Periodontal Disease and Adverse Pregnancy Outcomes. J. Periodontol. 2020, 91, doi:10.1002/JPER.20-0090.
- Manrique‐Corredor, E.J.; Orozco‐Beltran, D.; Lopez‐Pineda, A.; Quesada, J.A.; Gil‐Guillen, V.F.; Carratala‐Munuera, C. Maternal Periodontitis and Preterm Birth: Systematic Review and Meta‐analysis. Community Dent. Oral Epidemiol. 2019, 47, 243–251, doi:10.1111/cdoe.12450.
- Madianos, P.; Lieff, S.; Murtha, A.; Boggess, K.; Auten Jr, R.; Beck, J.; Offenbacher, S. Maternal Periodontitis and Prematurity. Part II: Maternal Infection and Fetal Exposure. Ann. Periodontol. 2001, 6, 175–182.
- Ferlay, J.; Colombet, M.; Soerjomataram, I.; Parkin, D.M.; Piñeros, M.; Znaor, A.; Bray, F. Cancer Statistics for the Year 2020: An Overview. Int. J. Cancer 2021, ijc.33588, doi:10.1002/ijc.33588.
- Piotrowski, I.; Kulcenty, K.; Suchorska, W. Interplay between Inflammation and Cancer. Rep. Pract. Oncol. Radiother. 2020, 25, 422–427, doi:10.1016/j.rpor.2020.04.004.
- Irani, S.; Barati, I.; Badiei, M. Periodontitis and Oral Cancer - Current Concepts of the Etiopathogenesis. Oncol. Rev. 2020, 14, 465, doi:10.4081/oncol.2020.465.
- Chung, P.-C.; Chan, T.-C. Association between Periodontitis and All-Cause and Cancer Mortality: Retrospective Elderly Community Cohort Study. BMC Oral Health 2020, 20, 168, doi:10.1186/s12903-020-01156-w.
- Jain, P.; Mirza, M.A.; Talegaonkar, S.; Nandy, S.; Dudeja, M.; Sharma, N.; Anwer, M.K.; Alshahrani, S.M.; Iqbal, Z. Design and in Vitro/in Vivo Evaluations of a Multiple-Drug-Containing Gingiva Disc for Periodontotherapy. RSC Adv. 2020, 10, 8530–8538.
- Golub, L.M.; Lee, H.-M. Periodontal Therapeutics: Current Host-Modulation Agents and Future Directions. Periodontol. 2000 2020, 82, 186–204.
- Kim, W.J.; Soh, Y.; Heo, S.-M. Recent Advances of Therapeutic Targets for the Treatment of Periodontal Disease. Biomol. Ther. 2021, 29, 263–267, doi:10.4062/biomolther.2021.001.
- Muras, A.; Mallo, N.; Otero‐Casal, P.; Pose‐Rodríguez, J.M.; Otero, A. Quorum Sensing Systems as a New Target to Prevent Biofilm‐related Oral Diseases. Oral Dis. 2020, odi.13689, doi:10.1111/odi.13689.
- Marchesan, J.T.; Girnary, M.S.; Moss, K.; Monaghan, E.T.; Egnatz, G.J.; Jiao, Y.; Zhang, S.; Beck, J.; Swanson, K.V. Role of Inflammasomes in the Pathogenesis of Periodontal Disease and Therapeutics. Periodontol. 2000 2020, 82, 93–114, doi:10.1111/prd.12269.
- Qi, M.; Chi, M.; Sun, X.; Xie, X.; Weir, M.D.; Oates, T.W.; Zhou, Y.; Wang, L.; Bai, Y.; Xu, H.H. Novel Nanomaterial-Based Antibacterial Photodynamic Therapies to Combat Oral Bacterial Biofilms and Infectious Diseases. Int. J. Nanomedicine 2019, 14, 6937–6956, doi:10.2147/IJN.S212807.
- Abdelaziz, D.; Hefnawy, A.; Al-Wakeel, E.; El-Fallal, A.; El-Sherbiny, I.M. New Biodegradable Nanoparticles-in-Nanofibers Based Membranes for Guided Periodontal Tissue and Bone Regeneration with Enhanced Antibacterial Activity. J. Adv. Res. 2021, 28, 51–62, doi:10.1016/j.jare.2020.06.014.
- Boda, S.K.; Almoshari, Y.; Wang, H.; Wang, X.; Reinhardt, R.A.; Duan, B.; Wang, D.; Xie, J. Mineralized Nanofiber Segments Coupled with Calcium-Binding BMP-2 Peptides for Alveolar Bone Regeneration. Acta Biomater. 2019, 85, 282–293, doi:10.1016/j.actbio.2018.12.051.
- Mahmoud, M.Y.; Sapare, S.; Curry, K.C.; Demuth, D.R.; Steinbach-Rankins, J.M. Rapid Release Polymeric Fibers for Inhibition of Porphyromonas Gingivalis Adherence to Streptococcus Gordonii. Front. Chem. 2019, 7, 926, doi:10.3389/fchem.2019.00926.
- Liu, X.; He, X.; Jin, D.; Wu, S.; Wang, H.; Yin, M.; Aldalbahi, A.; El-Newehy, M.; Mo, X.; Wu, J. A Biodegradable Multifunctional Nanofibrous Membrane for Periodontal Tissue Regeneration. Acta Biomater. 2020, 108, 207–222, doi:10.1016/j.actbio.2020.03.044.
- Ahi, Z.B.; Renkler, N.Z.; Gul Seker, M.; Tuzlakoglu, K. Biodegradable Polymer Films with a Natural Antibacterial Extract as Novel Periodontal Barrier Membranes. Int. J. Biomater. 2019, 2019, 7932470, doi:10.1155/2019/7932470.
- Chiaoprakobkij, N.; Suwanmajo, T.; Sanchavanakit, N.; Phisalaphong, M. Curcumin-Loaded Bacterial Cellulose/Alginate/Gelatin as A Multifunctional Biopolymer Composite Film. Mol. Basel Switz. 2020, 25, doi:10.3390/molecules25173800.
- Tovar-Carrillo, K.L.; Saucedo-Acuña, R.A.; Ríos-Arana, J.; Tamayo, G.; Guzmán-Gastellum, D.A.; Díaz-Torres, B.A.; Nava-Martínez, S.D.; Espinosa-Cristóbal, L.F.; Cuevas-González, J.C. Synthesis, Characterization, and In Vitro and In Vivo Evaluations of Cellulose Hydrogels Enriched with Larrea Tridentata for Regenerative Applications. BioMed Res. Int. 2020, 2020, 1425402, doi:10.1155/2020/1425402.
- Jain, P.; Garg, A.; Farooq, U.; Panda, A.K.; Mirza, M.A.; Noureldeen, A.; Darwish, H.; Iqbal, Z. Preparation and Quality by Design Assisted (Qb-d) Optimization of Bioceramic Loaded Microspheres for Periodontal Delivery of Doxycycline Hyclate. Saudi J. Biol. Sci. 2021, 28, 2677–2685.
- Lian, M.; Sun, B.; Qiao, Z.; Zhao, K.; Zhou, X.; Zhang, Q.; Zou, D.; He, C.; Zhang, X. Bi-Layered Electrospun Nanofibrous Membrane with Osteogenic and Antibacterial Properties for Guided Bone Regeneration. Colloids Surf. B Biointerfaces 2019, 176, 219–229, doi:10.1016/j.colsurfb.2018.12.071.
- Jeong, J.-O.; Park, J.-S.; Kim, E.J.; Jeong, S.-I.; Lee, J.Y.; Lim, Y.-M. Preparation of Radiation Cross-Linked Poly(Acrylic Acid) Hydrogel Containing Metronidazole with Enhanced Antibacterial Activity. Int. J. Mol. Sci. 2019, 21, doi:10.3390/ijms21010187.
- Wang, B.; Booij-Vrieling, H.E.; Bronkhorst, E.M.; Shao, J.; Kouwer, P.H.J.; Jansen, J.A.; Walboomers, X.F.; Yang, F. Antimicrobial and Anti-Inflammatory Thermo-Reversible Hydrogel for Periodontal Delivery. Acta Biomater. 2020, 116, 259–267, doi:10.1016/j.actbio.2020.09.018.
- Moraes, G.S.; Santos, I.B.D.; Pinto, S.C.S.; Pochapski, M.T.; Farago, P.V.; Pilatti, G.L.; Santos, F.A. Liposomal Anesthetic Gel for Pain Control during Periodontal Therapy in Adults: A Placebo-Controlled RCT. J. Appl. Oral Sci. Rev. FOB 2020, 28, e20190025, doi:10.1590/1678-7757-2019-0025.
- Trajano, V.C. da C.; Brasileiro, C.B.; Henriques, J.A. de S.; Cota, L. de M.; Lanza, C.R.; Cortés, M.E. Doxycycline Encapsulated in β-Cyclodextrin for Periodontitis: A Clinical Trial. Braz. Oral Res. 2020, 33, e112, doi:10.1590/1807-3107bor-2019.vol33.0112.
- Lin, J.; He, Z.; Liu, F.; Feng, J.; Huang, C.; Sun, X.; Deng, H. Hybrid Hydrogels for Synergistic Periodontal Antibacterial Treatment with Sustained Drug Release and NIR-Responsive Photothermal Effect. Int. J. Nanomedicine 2020, 15, 5377–5387, doi:10.2147/IJN.S248538.
- Mou, J.; Liu, Z.; Liu, J.; Lu, J.; Zhu, W.; Pei, D. Hydrogel Containing Minocycline and Zinc Oxide-Loaded Serum Albumin Nanopartical for Periodontitis Application: Preparation, Characterization and Evaluation. Drug Deliv. 2019, 26, 179–187, doi:10.1080/10717544.2019.1571121.
- Wang, B.; Wang, J.; Shao, J.; Kouwer, P.H.J.; Bronkhorst, E.M.; Jansen, J.A.; Walboomers, X.F.; Yang, F. A Tunable and Injectable Local Drug Delivery System for Personalized Periodontal Application. J. Control. Release Off. J. Control. Release Soc. 2020, 324, 134–145, doi:10.1016/j.jconrel.2020.05.004.
- Badar, S.B.; Zafar, K.; Ghafoor, R.; Khan, F.R. Comparative Evaluation of Chlorhexidine, Metronidazole and Combination Gels on Gingivitis: A Randomized Clinical Trial. Int. J. Surg. Protoc. 2019, 14, 30–33, doi:10.1016/j.isjp.2019.04.001.
- Qasim, S.S.B.; Nogueria, L.P.; Fawzy, A.S.; Daood, U. The Effect of Cross-Linking Efficiency of Drug-Loaded Novel Freeze Gelated Chitosan Templates for Periodontal Tissue Regeneration. AAPS PharmSciTech 2020, 21, 173, doi:10.1208/s12249-020-01708-x.
- Nowakowska, D.; Saczko, J.; Szewczyk, A.; Michel, O.; Ziętek, M.; Weżgowiec, J.; Więckiewicz, W.; Kulbacka, J. In Vitro Effects of Vasoconstrictive Retraction Agents on Primary Human Gingival Fibroblasts. Exp. Ther. Med. 2020, 19, 2037–2044, doi:10.3892/etm.2020.8462.
- Senarat, S.; Wai Lwin, W.; Mahadlek, J.; Phaechamud, T. Doxycycline Hyclate-Loaded in Situ Forming Gels Composed from Bleached Shellac, Ethocel, and Eudragit RS for Periodontal Pocket Delivery. Saudi Pharm. J. SPJ Off. Publ. Saudi Pharm. Soc. 2021, 29, 252–263, doi:10.1016/j.jsps.2021.01.009.
- Johnson, A.; Kong, F.; Miao, S.; Lin, H.-T.V.; Thomas, S.; Huang, Y.-C.; Kong, Z.-L. Therapeutic Effects of Antibiotics Loaded Cellulose Nanofiber and κ-Carrageenan Oligosaccharide Composite Hydrogels for Periodontitis Treatment. Sci. Rep. 2020, 10, 18037, doi:10.1038/s41598-020-74845-9.
- Lauritano, D.; Nota, A.; Martinelli, M.; Severino, M.; Romano, M.; Rossi, D.; Caruso, S. A Hydrosilver Gel for Plaque Control in Adults Affected by Chronic Periodontitis: Effects on the “red Complex” Bacterial Load. A Prospective Longitudinal Pilot Study Using Polymerase Chain Reaction Analysis. Int. J. Immunopathol. Pharmacol. 2019, 33, 2058738418825212, doi:10.1177/2058738418825212.
- Isaac, A.; Jivan, F.; Xin, S.; Hardin, J.; Luan, X.; Pandya, M.; Diekwisch, T.G.H.; Alge, D.L. Microporous Bio-Orthogonally Annealed Particle Hydrogels for Tissue Engineering and Regenerative Medicine. ACS Biomater. Sci. Eng. 2019, 5, 6395–6404, doi:10.1021/acsbiomaterials.9b01205.
- Sundaram, K.; Miller, D.P.; Kumar, A.; Teng, Y.; Sayed, M.; Mu, J.; Lei, C.; Sriwastva, M.K.; Zhang, L.; Yan, J.; et al. Plant-Derived Exosomal Nanoparticles Inhibit Pathogenicity of Porphyromonas Gingivalis. iScience 2020, 23, 100869, doi:10.1016/j.isci.2020.100869.
- Casarrubios, L.; Gómez-Cerezo, N.; Feito, M.J.; Vallet-Regí, M.; Arcos, D.; Portolés, M.T. Ipriflavone-Loaded Mesoporous Nanospheres with Potential Applications for Periodontal Treatment. Nanomater. Basel Switz. 2020, 10, doi:10.3390/nano10122573.
- Zhang, T.; Ying, D.; Qi, M.; Li, X.; Fu, L.; Sun, X.; Wang, L.; Zhou, Y. Anti-Biofilm Property of Bioactive Upconversion Nanocomposites Containing Chlorin E6 against Periodontal Pathogens. Mol. Basel Switz. 2019, 24, doi:10.3390/molecules24152692.
- Wang, Y.; Song, W.; Cui, Y.; Zhang, Y.; Mei, S.; Wang, Q. Calcium-SiRNA Nanocomplexes Optimized by Bovine Serum Albumin Coating Can Achieve Convenient and Efficient SiRNA Delivery for Periodontitis Therapy. Int. J. Nanomedicine 2020, 15, 9241–9253, doi:10.2147/IJN.S278103.
- Liang, G.; Shi, H.; Qi, Y.; Li, J.; Jing, A.; Liu, Q.; Feng, W.; Li, G.; Gao, S. Specific Anti-Biofilm Activity of Carbon Quantum Dots by Destroying P. Gingivalis Biofilm Related Genes. Int. J. Nanomedicine 2020, 15, 5473–5489, doi:10.2147/IJN.S253416.
- Veernala, I.; Giri, J.; Pradhan, A.; Polley, P.; Singh, R.; Yadava, S.K. Effect of Fluoride Doping in Laponite Nanoplatelets on Osteogenic Differentiation of Human Dental Follicle Stem Cells (HDFSCs). Sci. Rep. 2019, 9, 915, doi:10.1038/s41598-018-37327-7.
