Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), an RNA virus of the family Coronaviridae, causes coronavirus disease 2019 (COVID-19), an influenza-like disease that chiefly infects the lungs through respiratory transmission. The spike protein of SARS-CoV-2, a transmembrane protein in its outer portion, targets angiotensin-converting enzyme 2 (ACE2) as the binding receptor for the cell entry. As ACE2 is highly expressed in the gut and pulmonary tissues, SARS-CoV-2 infections frequently result in gastrointestinal inflammation, with presentations ordinarily ranging from intestinal cramps to complications with intestinal perforations. However, the evidence detailing successful therapy for gastrointestinal involvement in COVID-19 patients is currently limited. A significant change in fecal microbiomes, namely dysbiosis, was characterized by the enrichment of opportunistic pathogens and the depletion of beneficial commensals and their crucial association to COVID-19 severity has been evidenced. Oral probiotics had been evidenced to improve gut health in achieving homeostasis by exhibiting their antiviral effects via the gut–lung axis.
- SARS-CoV-2
- COVID-19
- gut microbiome
- probiotics
- Lactobacillus
- Bifidobacteria
1. Introduction
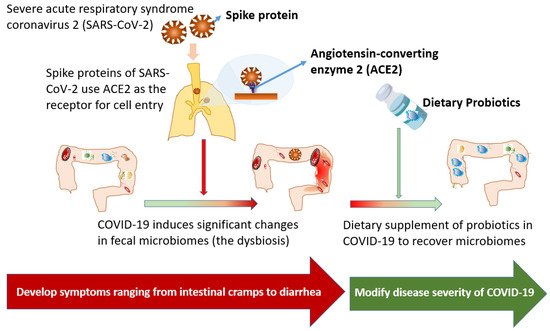
2. Gut Dysbiosis during COVID-19
Patients with COVID-19 had significant changes in fecal microbiomes, characterized by the enrichment of opportunistic pathogens and the depletion of beneficial commensals [12][22]. Dysbiosis has been vastly associated with COVID-19 severity [12][13][14][15][22,23,24,25], because the microbial diversity is regarded as a critical determinant of microbial ecosystem stability [16][26]. Among short-chain fatty acids (SCFAs), butyrate is not only responsible for energy requirements of the colonic epithelium, but also preserves tissues by mitigating chronic inflammatory responses through the regulation of pro- and anti-inflammatory cytokines [17][27]. Accordingly, decreases in the abundance of butyrate-producing bacteria (such as Faecalibacterium prausnitzii and Clostridium species), and the subsequent decline in SCFA availability have been correlated with severe COVID-19 [12][13][14][15][18][19][22,23,24,25,28,29]. Additionally, an increase in common pathogens in gut microbiota, such as Prevotella, Enterococcus, Enterobacteriaceae, or Campylobacter, were consistently associated with high infectivity, disease deterioration, or poor prognosis in COVID-19 patients [13][14][15][18][23,24,25,28]. The Prevotella species, for example, is associated with augmented T helper type 17 (Th17)-mediated mucosal inflammation, including activating TLR2 and Th17-polarizing cytokine production (such as IL-23 and IL-1), stimulating epithelial cells to produce IL-8, IL-6, and CCL20, and thus promoting neutrophil recruitment and inflammation [20][30]. The deterioration of the clinical course of patients with COVID-19 infection might be in part due to the activation of severe inflammation through disruption in gut microbiota and the out-growth of pathogenic bacteria. Patients with COVID-19 also had the increased proportion of opportunistic fungal pathogens, such as Aspergillus flavus and Aspergillus niger, detected in fecal samples [21][31]. In metagenomic sequencing analyses of fecal samples from COVID-19 patients, the baseline abundance of Coprobacillus, Clostridium ramosum, and Clostridium hathewayi was correlated with disease severity, and an inverse correlation between abundance of F. prausnitzii (an anti-inflammatory bacterium) and disease severity was disclosed [12][22]. Furthermore, Bacteroides dorei, Bacteroides thetaiotaomicron, Bacteroides massiliensis, and Bacteroides ovatus, which downregulated the expression of ACE2 in the gut, were correlated inversely with SARS-CoV-2 load [12][22]. The same study team also indicated that, in the cases of active SARS-CoV-2 infections, the gut microbiota presented a higher abundance of opportunistic pathogens, while increased nucleotide and amino acid biosynthesis, as well as carbohydrate metabolism, were evidenced [14][24]. In summary, these findings reasonably suggest that the development of therapeutic agents able to neutralize the SARS-CoV-2 activity in the gut, as well as to restore the physiological gut microbiota composition, may be warranted. A crucial association between the predominance of opportunistic pathogens in gut microbiomes and unfavorable outcomes of COVID-19 patients has been comprehensively reported [13][23]. In a Chinese cohort of COVID-19 patients with different disease severity, the abundance of butyrate-producing bacteria decreased significantly, which may help discriminate critically ill patients from general and severe patients. The increased proportion of opportunistic pathogens, such as Enterococcus and Enterobacteriaceae, in critically ill patients might be associated with a poor prognosis [13][23]. In another study, a higher abundance of opportunistic pathogens, such as Streptococcus, Rothia, Veillonella, and Actinomyces species, and a lower abundance of beneficial symbionts, could be noted in the gut microbiota of COVID-19 patients [15][25]. In the American cohort, the specific alteration in the gut microbiome, particularly Peptoniphilus, Corynebacterium, and Campylobacter, was also noticed [18][28]. Nevertheless, opportunistic pathogens were prevalent in the COVID-19 cases, particularly among critically individuals, but the causal effect of the predominance of opportunistic pathogens, and a grave outcome remains to be determined. The recovery of dysbiosis after active SARS-CoV-2 infections exhibited geographical and demographic differences [12][18][22][22,28,32]. After the clearance of SARS-CoV-2 and resolution of respiratory symptoms, depleted symbionts and gut dysbiosis were usually persistent among recovered COVID-19 patients, because microbiota richness did not yield to normal levels after 6-month recovery [12][22]. In contrast, in an American cohort including recovered COVID-19 cases, the dysbiosis could rapidly recover with a return of the human gut microbiota to an uninfected status [18][28]. Although the great diversity in the ability of the microbiota return was disclosed, it was evident that the recovery of gut microbiota could be regarded as an indicator of the favorable prognosis among patients with COVID-19.3. Therapeutic Effects of Dietary Supplement of Probiotics for COVID-19
Oral probiotics had been proven to exhibit antiviral effects and thereby to improve gut health for achieving homeostasis [23][24][33,34]. To take the influenza infection as an example, Lactococcus lactis JCM 5805 demonstrated the activity against influenza virus through the activation of anti-viral immunity [24][34]. The oral administration of Bacteroides breve YIT4064 can enhance antigen-specific IgG against influenza virus [23][33]. Moreover, a meta-analysis report indicated the administration of these probiotics significantly reduced the incidence of ventilator-associated pneumonia, possibly through reducing the overgrowth of potentially opportunistic pathogens and stimulating immune responses [25][35]. However, such a promotion of oral probiotics in treating critically ill patients experiencing COVID-19 should be further explored. In COVID-19 patients, the excessive production of pro-inflammatory cytokines, a so-called “cytokine storm”, is pathologically related to acute respiratory distress syndrome and extensive tissue injury, multi-organ failure, or eventually death [26][36]. With COVID-19 progression, critically ill patients had higher plasma levels of many cytokines, in terms of IL-2, IL-7, IL-10, granulocyte colony-stimulating factor, IFN-γ-inducible protein-10, monocyte chemoattractant protein-1, macrophage inflammatory protein-1A, and TNF-α [27][37]. Therefore, therapeutic targeting on cytokines in COVID-19 treatment was evidenced to increase survival [26][36]. Fecal levels of IL-8 and IL-23 and intestinal specific IgA responses were vastly associated with severe COVID-19 disease, which indicated the co-existence of systemic and local intestine inflammation in critically ill patients [28][38]. One of the commercial probiotics, Lactobacillus rhamnosus HDB1258, might be effective in treating COVID-19 by modulating both microbiota-mediated immunity in gut and systemic inflammation induced by lipopolysaccharide [29][39]. Accordingly, concomitant targeting on local and systemic inflammatory responses by probiotics is reasonably believed to be valuable to counteract COVID-19-related gut and systemic inflammation. Numerous probiotics and by-probiotic products exhibiting direct and indirect antiviral effects have been reported in the scientific literature. Lactic acid-producing bacteria such as lactobacilli can exert their antiviral activity by direct probiotic–virus interaction, the production of antiviral inhibitory metabolites, preventing secondary infection, and eliciting anti-viral immunity [30][31][32][33][34][35][36][37][40,41,42,43,44,45,46,47]. Nisin, one of the well-characterized bacteriocins from probiotics, contributes to probiotic antiviral effects against influenza A virus and other respiratory viruses [31][33][41,43]. A peptide, P18, produced by the probiotic Bacillus subtilis strain, was regarded as an antiviral compound against influenza virus [32][42]. Probiotics capsules containing live B. subtilis and E. faecalis (Medilac-S) can lower the acquisition of the gut colonization of potentially pathogenic microorganisms [34][44]. L. rhamnosus GG have been reported to prevent ventilator-associated pneumonia [35][45]. The heat-killed L. casei DK128 strain has been active against different subtypes of influenza viruses by an increasing proportion of alveolar macrophages in lungs and airways, the early induction of virus-specific antibodies, and reduced levels of pro-inflammatory cytokines and innate immune cells [36][46]. S. salivarius 24SMB and S. oralis 89a were able to inhibit the biofilm formation capacity of airway bacterial pathogens and even to disperse their pre-formed biofilms [37][47]. The S. salivarius strain K12 may stimulate IFN-γ release and suppress bronchial inflammation, and its colonization in the oral cavity and upper respiratory tract will actively interfere with the growth of pathogenic microbes [38][48]. Although these probiotics and their products provide the favorable antiviral interaction with immune composition in the gut, the feasibility and health effect of dietary probiotics to improve the dysbiosis in COVID-19 patients remains to be studied. Numerous probiotics had been proposed to be beneficial in coronaviral infections, but the evidence detailing their efficacies in treating COVID-19 infection is limited [39][49]. L. plantarum Probio-38 and L. salivarius Probio-37 could inhibit transmissible gastroenteritis coronavirus [40][50]. The probiotic, E. faecium NCIMB 10415, has been approved as a feed additive for young piglets in the European Union for treating the transmissible coronavirus gastroenteritis [41][51]. The recombinant IFN-λ3-anchored L. plantarum can in vitro inhibit porcine gastroenteritis caused by coronavirus [42][52]. However, the clinical utility of probiotics in human infections caused by SARS-CoV-2 warrants further evaluations [43][44][45][46][47][53,54,55,56,57]. Another important issue regarding probiotics for COVID-19 cases is the patient safety. For an example, B. longum bacteremia had been reported in preterm infants receiving probiotics [48][49][58,59]. Since gastrointestinal SARS-CoV-2 involvement has been reported, the possibility of increased intestinal permeability should be expected and the risk of secondary bacterial infections in the gut is substantial if high-dosage steroid and other immunomodulation agents are administrated to treat the cytokine storm associated with COVID-19 [50][51][60,61]. The oral formulation Sivomixx®, which was a mixture of probiotics, was independently associated with a reduced risk for death in a retrospective, observational cohort study that included 200 adults with severe COVID-19 pneumonia [52][62]. In another study, nearly all COVID-19 patients treated with Sivomixx® showed remission of diarrhea and other symptoms within 72 h, in contrast to less than half in the control group [53][63]. However, the clinical application of probiotics in COVID-19 patients requires more evidence. In ClinicalTrials.gov, 22 trials of probiotics for the prevention or adjuvant therapy of COVID-19 were registered since April 2020, including one aiming to study the effect of oxygen-ozone therapy, one studying intranasal probiotics, and the other using throat spray-containing probiotic [54][64]. Of the remaining 19 trials, 8 common probiotic strains include Lactobacillus (7 trials), a mixture of Bifidobacteria and Lactobacillus (5), and Saccharomyces species (2) (Table 1). The major outcome was greatly diverse in these trials, including disease prevention, symptom relief, antibody titers, disease progression, changes of viral load, microbiome effects, and mortality. Based on these trials, the role of dietary supplement probiotics for COVID-19 can be more evident in the near future.| ClinicalTrials.gov Identifier | Study Title | First Posted | Study Design | Probiotic Strain | Location | Outcome Measures | Status |
|---|---|---|---|---|---|---|---|
| NCT04366180 | Evaluation of probiotic Lactobacillus coryniformis K8 on COVID-19 prevention in healthcare workers | 28 April 2020 | Randomized | L. coryniformis K8 | Granada, Spain | Incidence of COVID-19 infection in healthcare workers | Recruiting |
| NCT04390477 | Study to evaluate the effect of a probiotic in COVID-19 | 15 May 2020 | Randomized | Not revealed | Alicante, Spain | ICU admission rate | Recruiting |
| NCT04399252 | Effect of Lactobacillus on the microbiome of household contacts exposed to COVID-19 | 22 May 2020 | Randomized | L. rhamnosus GG | North Carolina, United States | Incidence of symptoms of COVID-19 | Active, not recruiting |
| NCT04420676 | Synbiotic therapy of gastrointestinal symptoms during COVID-19 infection (SynCov) | 9 June 2020 | Randomized | Omni-Biotic® 10 AAD (chiefly Lactobacillus and Bifidobacterium) | Graz, Austria | Stool calprotectin | Recruiting |
| NCT04462627 | Reduction of COVID 19 transmission to health care professionals | 8 July 2020 | Non-randomized | Metagenics Probactiol plus (chiefly Lactobacillus and Bifidobacterium) | Brussels, Belgium | Antibody concentration | Recruiting |
| NCT04507867 | Effect of a NSS to reduce complications in patients with COVID-19 and comorbidities in stage III | 11 August 2020 | Randomized | Saccharomyces bourllardii with nutritional support system (NSS) | Mexico | Oxygen saturation | Not yet recruiting |
| NCT04517422 | Efficacy of L. plantarum and P. acidilactici in adults with SARS-CoV-2 and COVID-19 | 18 August 2020 | RCT | L. plantarum and P. acidilactici | Mexico City, Mexico | Severity progression of COVID-19 | Completed |
| NCT04621071 | Efficacy of probiotics in reducing duration and symptoms of COVID-19 (PROVID-19) | 9 November 2020 | RCT | Not revealed | Canada, Quebec | Duration of symptoms of the COVID-19 | Recruiting |
| NCT04666116 | Changes in viral load in COVID-19 after probiotics | 14 December 2020 | Randomized, single blind | GASTEEL PLUS (mixture of Bifidobacteria and Lactobacillus) | Valencia, Spain | Viral load in nasopharyngeal smear | Recruiting |
| NCT04734886 | The effect of probiotic supplementation on SARS-CoV-2 antibody response after COVID-19 | 2 February 2021 | Randomized | L. reuteri DSM 17938 + vitamin D | Örebro Län, Sweden | SARS-CoV-2 specific antibodies | Recruiting |
| NCT04756466 | Effect of the consumption of a Lactobacillus strain on the incidence of COVID-19 in the elderly | 16 February 2021 | RCT | Lactobacillus strain | A Coruña, Spain | Incidence of SARS CoV-2 infection | Active, not recruiting |
| NCT04798677 | Efficacy and tolerability of ABBC1 in volunteers receiving the influenza or COVID-19 Vaccine | 15 March 2021 | Non-randomized | S. cerevisiae, rich in selenium and zinc | Barcelona, Spain | Change in acute immune response to influenza vaccine after supplementation | Recruiting |
| NCT04813718 | Post COVID-19 syndrome and the gut-lung axis | 24 March 2021 | Randomized | Omni-Biotic Pro Vi 5 (chiefly Lactobacillus) | Graz, Austria | Microbiome composition | Recruiting |
| NCT04847349 | Live microbials to boost anti-severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) immunity clinical trial | 19 April 2021 | RCT | OL-1 (Content not revealed) | New Jersey, United States | Change in serum titer of anti-SARS-CoV-2 IgG | Recruiting |
| NCT04854941 | Efficacy of probiotics in the treatment of hospitalized patients with novel coronavirus infection | 22 April 2021 | Randomized | L. rhamnosus, B. bifidum, B. longum subsp. infantis and B. longum | Moscow, Russian | Mortality | Completed |
| NCT04877704 | Symprove (Probiotic) as an add-on to COVID-19 management | 7 May 2021 | Randomized | Symprove ( L. rhamnosus, E. faecium, L. acidophilus and L. plantarum) | London, United Kingdom | Length of hospital stay | Not yet recruiting |
| NCT04884776 | Modulation of gut microbiota to enhance health and immunity | 13 May 2021 | RCT | Probiotics blend (3 Bifidobacteria) | Hong Kong | Restoration of gut dysbiosis | Not yet recruiting |
| NCT04907877 | Bifidobacteria and Lactobacillus in symptomatic adult COVID-19 outpatients (ProCOVID) | 1 June 2021 | Randomized | NordBiotic ImmunoVir (mixture of Bifidobacteria and Lactobacillus) | Not revealed | Global symptom score | Not yet recruiting |
| NCT04922918 | Ligilactobacillus salivarius MP101 for elderly in a nursing home (PROBELDERLY) | 11 June 2021 | Single group | Ligilactobacillus salivarius MP101 | Madrid, Spain | Barthel index, functional status score | Recruiting |
