The lung extracellular matrix (ECM) comprises of a network of protein–protein and protein–proteoglycan interactions that modulate the biophysical environment of cells through ECM stiffness, porosity, topography and insolubility. In response, the lung ECM is remodelled by cells, whereby its components are deposited, degraded or modified in a reciprocal relationship. Changes in the ECM dynamics is associated with chronic fibrotic lung diseases such as asthma, chronic obstructive pulmonary disease and idiopathic pulmonary fibrosis.
- extracellular matrix
- lung
- miRNA
- asthma
- chronic obstructive pulmonary disease
- idiopathic pulmonary disease
1. Introduction
The lung extracellular matrix (ECM) plays a key role in the normal architecture of the lung, from embryonic lung development, alveolarization at birth, to mechanical stability and elastic recoil of the breathing adult lung [1][2]. The lung ECM provides both biochemical and biophysical cues, which direct cellular functions and differentiation. The lung ECM can also alter the biochemical environment of surrounding cells, by storing and sequestering growth factors and cytokines to regulate spatially and temporally their bioavailability. The lung ECM can modulate the biophysical environment of cells through ECM stiffness, porosity, topography (spatial arrangement and orientation) and insolubility. Lung ECM molecules connect to cells via integrins, syndecans and other receptors to influence cell signalling, migration and proliferation. In response, the lung ECM is remodelled by cells, whereby its components are deposited, degraded or modified in a reciprocal relationship [3][4].
2. The Lung Extracellular Matrix
3. How Cells Modify the Lung Extracellular Matrix
The lung ECM functions as a physical barrier, an anchorage site and a migration highway for cells within the organ. The ECM dynamics within each cellular niche are tightly regulated to ensure normal development, physiology and homeostasis of organ systems. Alterations in the lung ECM environment result from the synthesis, degradation or altered organization of ECM components by surrounding structural and immune cells. This is achieved by redundancy in the mechanisms used to modulate the expression and function of the ECM and ECM-modifying enzymes. When such control mechanisms are corrupted, ECM dynamics become deregulated, leading to various congenital defects or disease. To maintain homeostasis, the lung ECM undergoes remodelling, a process whereby old or damaged ECM proteins undergo a series of proteolytic events and are replaced by newly synthesized proteins. Lung ECM remodeling is modulated by the synthesis rate of new ECM molecules and the surfeit of proteases released by specialized mesenchymal cells, predominantly fibroblasts and, to a lesser extent, airway smooth muscle, immune cells and epithelial cells of the lungs [4][10]. The remodelling process is directed by the interaction between the different cell types, cytokines, growth factors and enzymes within the lung ECM. The levels and ratios of ECM proteins during the repair response must be maintained to avoid disrupting the lung ECM characteristics, including tensile strength and elastic recoil. Upon lung injury, activation of the coagulation process initiates damage control and a provisional matrix (fibronectin), which is then followed by the development of an acute inflammatory response that recruits polymorphonuclear leukocytes (PMNs) and macrophages to protect against pathogens and remove debris from dead and dying cells. These important early steps prepare the site for activation of local fibroblasts, migrating myofibroblasts and recruited fibrocytes that remodel collagenous and noncollagenous tissues into scar tissue. Following a single injury, inflammatory cells usually leave the site before the resident fibroblasts and migrating myofibroblasts, and fibrocytes are activated to drive the tissue repair process. Over time, the provisional ECM is degraded via cell-mediated or proteolytic pathways and replaced with a restorative ECM consisting mainly of collagen I and collagen III, before these cells then undergo apoptosis [11]. In sharp contrast, repetitive injury is associated with the persistence of inflammatory immune cell infiltration during the remodelling of the ECM, which increases the potential for the development of an abnormal repair process creating further injury with abnormal scar formation, remodelling or permanent destruction of damaged tissue. Repeated lung injury and repair can trigger a vicious cycle of aberrant ECM protein deposition, accompanied by elevated ECM stiffness, further resulting in changes in cell phenotype and function, and this has a lasting effect on tissue function and ultimately disease progression [12]. Although the processes governing the resolution of injury repair are regulated by several pathways, in chronic fibrotic lung diseases the processes are compromised, thus resulting in impaired fibroblast proliferation, apoptosis and aberrant ECM remodelling [3][11]. Disruption of this balance changes the dynamics of the lung ECM with characteristics such as stiffness and elastance, as seen in asthma, chronic obstructive pulmonary disease (COPD) and idiopathic pulmonary fibrosis (IPF) [13][14][15]. Compared to the normal lung ECM (0.5–5 kPa), studies have shown that a stiff ECM (15–100 kPA) [16][17][18][19] can exacerbate TGF-β activation leading to enhanced pro-fibrotic signalling. Disruption of homeostasis within the lung ECM can also lead to the dysregulated synthesis of ECM proteins, such as fibrillar collagen I, by activated fibroblasts, leading to tissue fibrosis regardless of the inflammatory response [20]. The interplay between several diverse factors (cell types, genes, cytokines, growth factors, enzymes and epigenetics) is essential for the normal repair and remodeling processes within the lung [21]. Studies have shown that deviations in the posttranslational modification of ECM proteins, including enzymatic cross-linking, glycation and oxidation, impact the tensile strength, biomechanics and cell–ECM interactions and are contributors to lung diseases [22][23]. As shown in Figure 1, compared to a terminal bronchiole (last generation of conducting airways within the lung) from a donor control, there is extensive fibrosis surrounding the terminal bronchiole airway walls in the lungs from patients with IPF, COPD and asthma.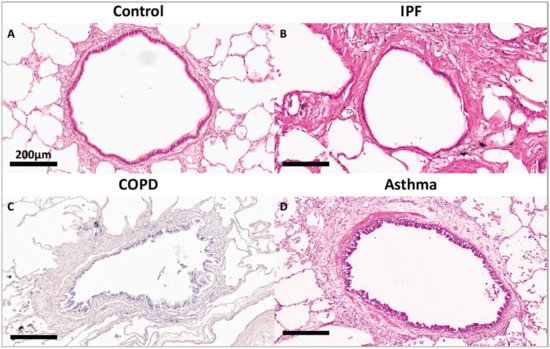
3.1. Asthma
3.2. Chronic Obstructive Pulmonary Disease
COPD is one of the most common lung diseases worldwide, and the third leading cause of death[35] due to exposure of tobacco smoke and/or environmental pollutants[36]. COPD is characterized by chronic lung inflammation and irreversible airflow limitation. COPD is caused by the chronic inhalation of cigarette smoke or other harmful particles, which cause pulmonary injury, leading to chronic airway inflammation and tissue remodelling. The major histopathological changes observed within the lung include chronic bronchitis, small airway disease and emphysema. Recent studies have shown that small airway disease precedes emphysematous tissue destruction, suggesting a temporal pattern of ECM deposition and destruction in the disease progression [37]. Using a large cross-sectional study, Hogg and colleagues showed that the progression of COPD from GOLD (Global Initiative for Obstructive Lung Disease) stage 0 to GOLD stage 4 is strongly associated with thickening of the airway wall and each of its compartments by remodelling of the ECM, which affects airway wall elasticity, thickness and resistance [38][39]. In addition, loss of elastic recoil from the destruction of the ECM within the parenchyma (emphysematous tissue destruction) is a well-described feature of COPD [40], with several studies showing decreased protein expression and volume fraction of elastin in both the conducting airways and the parenchyma of patients with mild, moderate or severe COPD [41][42].3.3. Idiopathic Pulmonary Fibrosis
IPF is the most common and progressive type of idiopathic interstitial pneumonia that is unresponsive to treatment, leading to a median survival of 3–5 years [43][44][45][46]. The pathology of IPF is characterized by heterogeneous interstitial fibrosis, honeycomb cysts and fibrotic foci associated with excessive deposition of ECM proteins resulting in aberrant matrix metalloproteinases, connective tissues, morphogens and impaired signalling of growth factors [47][48][49][50][51]. More recently the early pathology of IPF has been shown to involve the loss of small airways[52].The current hypothesis for tissue destruction in IPF involves continuous damage and senescence of the alveolar epithelium, leading to the destruction of the basement membrane and activation of myofibroblasts [53]. As the disease progresses, the composition of the ECM has been shown to vary, with an accumulation of versican in the onset of IPF, whereas collagen I and collagen III accumulation is observed in both the early and late stages of IPF [54]. The severity of fibrosis in IPF has also been shown to correlate with the number of elastic fibers, with a higher elastic fiber score being related to worse disease outcomes [55]. The diseased ECM is a critical linchpin in IPF, and it serves as a causal link for alteration in cell gene expression patterns at the translational level [3].4. Conclusions
The initiation and progression of chronic lung diseases are modulated by complex environmental and epigenetic factors. In the initiation and progression of these diseases, there is an intricate interaction between cells and the ECM through various molecules and signalling pathways. Alterations in the lung ECM are driven by synthesis, degradation, and changes in topography by multiple cell types in the lung, but more importantly, dysregulation of the ECM seems to provide a positive feedback loop to drive fibrosis progression. The development of complex models and screening of human tissues is essential to understand how ECM regulation is abnormal in lung disease and how it could be targeted therapeutically.
References
- Rozario, T.; DeSimone, D.W. The extracellular matrix in development and morphogenesis: A dynamic view. Dev. Biol. 2010, 341, 126–140.
- Gattazzo, F.; Urciuolo, A.; Bonaldo, P. Extracellular matrix: A dynamic microenvironment for stem cell niche. Biochim. Acta 2014, 1840, 2506–2519.
- Parker, M.W.; Rossi, D.; Peterson, M.; Smith, K.; Sikström, K.; White, E.; Connett, J.E.; Henke, C.A.; Larsson, O.; Bitterman, P. Fibrotic extracellular matrix activates a profibrotic positive feedback loop. J. Clin. Investig. 2014, 124, 1622–1635.
- Zhou, Y.; Horowitz, J.C.; Naba, A.; Ambalavanan, N.; Atabai, K.; Balestrini, J.; Bitterman, P.B.; Corley, R.A.; Ding, B.-S.; Engler, A.J.; et al. Extracellular matrix in lung development, homeostasis and disease. Matrix Biol. 2018, 73, 77–104.
- Hynes, R.O.; Naba, A. Overview of the Matrisome--An Inventory of Extracellular Matrix Constituents and Functions. Cold Spring Harb. Perspect. Biol. 2011, 4, a004903.
- Halfter, W.; Oertle, P.; Monnier, C.; Camenzind, L.; Reyes-Lua, M.; Hu, H.; Candiello, J.; Labilloy, A.; Balasubramani, M.; Henrich, P.B.; et al. New concepts in basement membrane biology. FEBS J. 2015, 282, 4466–4479.
- LeBleu, V.S.; Macdonald, B.; Kalluri, R. Structure and Function of Basement Membranes. Exp. Biol. Med. 2007, 232, 1121–1129.
- Raghu, G.; Striker, L.J.; Hudson, L.D.; Striker, G.E. Extracellular matrix in normal and fibrotic human lungs. Am. Rev. Respir. Dis. 1985, 131, 281–289.
- Faffe, D.S.; Zin, W.A. Lung Parenchymal Mechanics in Health and Disease. Physiol. Rev. 2009, 89, 759–775.
- Brandsma, C.; Berge, M.V.D.; Hackett, T.; Brusselle, G.; Timens, W. Recent advances in chronic obstructive pulmonary disease pathogenesis: From disease mechanisms to precision medicine. J. Pathol. 2020, 250, 624–635.
- Friedman, S.L.; Sheppard, D.; Duffield, J.S.; Violette, S. Therapy for Fibrotic Diseases: Nearing the Starting Line. Sci. Transl. Med. 2013, 5, 167sr1.
- Burgstaller, G.; Oehrle, B.; Gerckens, M.; White, E.S.; Schiller, H.B.; Eickelberg, O. The instructive extracellular matrix of the lung: Basic composition and alterations in chronic lung disease. Eur. Respir. J. 2017, 50, 1601805.
- Polio, S.R.; Stasiak, S.E.; Jamieson, R.R.; Balestrini, J.L.; Krishnan, R.; Parameswaran, H. Extracellular matrix stiffness regulates human airway smooth muscle contraction by altering the cell-cell coupling. Sci. Rep. 2019, 9, 1–12.
- McKleroy, W.; Lee, T.-H.; Atabai, K. Always cleave up your mess: Targeting collagen degradation to treat tissue fibrosis. Am. J. Physiol. Cell. Mol. Physiol. 2013, 304, L709–L721.
- White, E. Lung Extracellular Matrix and Fibroblast Function. Ann. Am. Thorac. Soc. 2015, 12, S30–S33.
- Southern, B.D.; Grove, L.M.; Rahaman, S.O.; Abraham, S.; Scheraga, R.; Niese, K.A.; Sun, H.; Herzog, E.L.; Liu, F.; Tschumperlin, D.J.; et al. Matrix-driven Myosin II Mediates the Pro-fibrotic Fibroblast Phenotype. J. Biol. Chem. 2016, 291, 6083–6095.
- Hinz, B. Mechanical aspects of lung fibrosis: A spotlight on the myofibroblast. Proc. Am. Thorac. Soc. 2012, 9, 137–147.
- Booth, A.J.; Hadley, R.; Cornett, A.M.; Dreffs, A.A.; Matthes, S.A.; Tsui, J.L.; Weiss, K.; Horowitz, J.C.; Fiore, V.F.; Barker, T.H.; et al. Acellular Normal and Fibrotic Human Lung Matrices as a Culture System forIn VitroInvestigation. Am. J. Respir. Crit. Care Med. 2012, 186, 866–876.
- Liu, F.; Mih, J.D.; Shea, B.S.; Kho, A.T.; Sharif, A.S.; Tager, A.M.; Tschumperlin, D.J. Feedback amplification of fibrosis through matrix stiffening and COX-2 suppression. J. Cell Biol. 2010, 190, 693–706.
- Karsdal, M.A.; Nielsen, M.J.; Sand, J.M.; Henriksen, K.; Genovese, F.; Bay-Jensen, A.-C.; Smith, V.; Adamkewicz, J.I.; Christiansen, C.; Leeming, D.J. Extracellular Matrix Remodeling: The Common Denominator in Connective Tissue DiseasesPossibilities for Evaluation and Current Understanding of the Matrix as More Than a Passive Architecture, but a Key Player in Tissue Failure. ASSAY Drug Dev. Technol. 2013, 11, 70–92.
- Cox, T.R.; Erler, J.T. Remodeling and homeostasis of the extracellular matrix: Implications for fibrotic diseases and cancer. Dis. Model. Mech. 2011, 4, 165–178.
- Erler, J.T.; Weaver, V.M. Three-dimensional context regulation of metastasis. Clin. Exp. Metastasis 2008, 26, 35–49.
- Cheng, D.T.; Kim, D.K.; Cockayne, D.A.; Belousov, A.; Bitter, H.; Cho, M.H.; Duvoix, A.; Edwards, L.; Lomas, D.A.; Miller, B.E.; et al. Systemic Soluble Receptor for Advanced Glycation Endproducts Is a Biomarker of Emphysema and Associated with AGER Genetic Variants in Patients with Chronic Obstructive Pulmonary Disease. Am. J. Respir. Crit. Care Med. 2013, 188, 948–957.
- Theo Vos; Stephen S Lim; Cristiana Abbafati; Kaja M Abbas; Mohammad Abbasi; Mitra Abbasifard; Mohsen Abbasi-Kangevari; Hedayat Abbastabar; Foad Abd-Allah; Ahmed Abdelalim; et al.Mohammad AbdollahiIbrahim AbdollahpourHassan AbolhassaniVictor AboyansElissa M AbramsLucas Guimarães AbreuMichael R M AbrigoLaith Jamal Abu-RaddadAbdelrahman I AbushoukAlyssa AcebedoIlana N AckermanMaryam AdabiAbdu A AdamuOladimeji M AdebayoVictor AdekanmbiJaimie D AdelsonOlatunji O AdetokunbohDavoud AdhamMahdi AfshariAshkan AfshinEmilie E AgardhGina AgarwalKareha M AgesaMohammad AghaaliSeyed Mohammad Kazem AghamirAnurag AgrawalTauseef AhmadAlireza AhmadiMehdi AhmadiHamid AhmadiehEhsan AhmadpourTemesgen Yihunie AkaluRufus Olusola AkinyemiTomi AkinyemijuBlessing AkombiZiyad Al-AlyKhurshid AlamNoore AlamSamiah AlamTahiya AlamTurki M AlanziSamuel B AlbertsonJacqueline Elizabeth Alcalde-RabanalNiguse Meles AlemaMuhammad AliSaqib AliGianfranco AlicandroMehran AlijanzadehCyrus AliniaVahid AlipourSyed Mohamed AljunidFrançois AllaPeter AllebeckAmir Almasi-HashianiJordi AlonsoRajaa M Al-RaddadiKhalid A AltirkawiNelson Alvis-GuzmanNelson J Alvis-ZakzukSaeed AminiMostafa Amini-RaraniArya AminorroayaFatemeh AmiriArianna Maever L AmitDickson A AmugsiGianna Gayle Herrera AmulDeanna AnderliniCatalina Liliana AndreiTudorel AndreiMina AnjomshoaFereshteh AnsariIman AnsariAlireza Ansari-MoghaddamCarl Abelardo T AntonioCatherine M AntonyErnoiz AntriyandartiDavood AnvariRazique AnwerJalal ArablooMorteza Arab-ZozaniAleksandr Y AravkinFilippo ArianiJohan ÄrnlövKrishna K AryalAfsaneh ArzaniMehran Asadi-AliabadiAli A Asadi-PooyaBabak AsghariCharlie AshbaughDesta Debalkie AtnafuSachin R AtreFloriane AusloosMarcel AusloosBeatriz Paulina Ayala QuintanillaGetinet AyanoMartin Amogre AyanoreYared Asmare AynalemSamad AzariGhasem AzarianZelalem Nigussie AzeneEbrahim BabaeeAlaa BadawiMojtaba BagherzadehMohammad Hossein BakhshaeiAhad BakhtiariSenthilkumar BalakrishnanShivanthi BalallaShelly BalassyanoMaciej BanachPalash Chandra BanikMarlena S BannickAgegnehu Bante BanteAdhanom Gebreegziabher BarakiMiguel A BarbozaSuzanne Lyn Barker-ColloCeline M BarthelemyLingkan BaruaAkbar BarzegarSanjay BasuBernhard T BauneMohsen BayatiGholamreza BazmandeganNeeraj BediEttore BeghiYannick BéjotAminu K BelloRose G BenderDerrick A BennettFiona B BennittIsabela M BensenorCatherine P BenzigerKidanemaryam BerheEduardo BernabeGregory J BertolacciReshmi BhageerathyNeeraj BhalaDinesh BhandariPankaj BhardwajKrittika BhattacharyyaZulfiqar A BhuttaSadia BibiMolly H BiehlBoris BikbovMuhammad Shahdaat Bin SayeedAntonio BiondiBinyam Minuye BirihaneDonal BisanzioCatherine BisignanoRaaj Kishore BiswasSomayeh BohlouliMehdi BohluliSrinivasa Rao Rao BollaArchith BoloorAlexandra S Boon-DooleyGuilherme BorgesAntonio Maria BorzìRupert BourneOliver J BradyMichael BrauerCarol BrayneNicholas J K BreitbordeHermann BrennerPaul Svitil BriantAndrew M BriggsNikolay Ivanovich BrikoGabrielle B BrittonDana BryazkaRachelle BuchbinderBlair R BumgarnerReinhard BusseZahid A ButtFlorentino Luciano Caetano dos SantosLuis La Alberto CámeraIsmael R Campos-NonatoJosip CarRosario CárdenasGiulia CarrerasJuan J CarreroFelix CarvalhoJoao Mauricio Castaldelli-MaiaCarlos A Castañeda-OrjuelaGiulio CastelpietraChris D CastleFranz CastroFerrán Catalá-LópezKate CauseyChristopher R CederrothKelly M CercyEster CerinJoht Singh ChandanAlex R ChangFiona J CharlsonVijay Kumar ChattuSarika ChaturvediOdgerel Chimed-OchirKen Lee ChinDaniel Youngwhan ChoHanne ChristensenDinh-Toi ChuMichael T ChungFlavia M CicuttiniLiliana G CiobanuMassimo CirilloEmma L CollinsKelly ComptonSara ContiPaolo Angelo CortesiVera Marisa CostaEwerton CousinRichard G CowdenBenjamin C CowieElizabeth A CromwellDi H CrossChristopher Stephen CroweJessica A CruzMatthew CunninghamSaad M A DahlawiGiovanni DamianiLalit DandonaRakhi DandonaAso Mohammad DarweshAhmad DaryaniJai K DasRajat Das GuptaJosé das NevesClaudio Alberto Dávila-CervantesKairat DavletovDiego De LeoFrances E DeanNicole K DeCleeneAmanda DeenLouisa DegenhardtRobert Paul DellavalleFeleke Mekonnen DemekeDesalegn Getnet DemsieEdgar Denova-GutiérrezNebiyu Dereje DerejeNikolaos DervenisRupak DesaiAssefa DesalewGetenet Ayalew DessieSamath Dhamminda DharmaratneGovinda Prasad DhunganaMostafa DianatinasabDaniel DiazZahra Sadat Dibaji ForooshaniZachary V DingelsM Ashworth DiracShirin DjalaliniaHoa Thi DoKlara DokovaFariba DorostkarChirag P DoshiLeila DoshmangirAbdel DouiriMatthew C DoxeyTim Robert DriscollSusanna J DunachieBruce B DuncanAndre Rodrigues DuraesArielle Wilder EaganMohammad Ebrahimi KalanDavid EdvardssonJoshua R EhrlichNevine El NahasIman El SayedMaha El TantawiIffat ElbaraziIslam Y ElgendyHala Rashad ElhabashyShaimaa I El-JaafaryIqbal Rf Elya Global burden of 369 diseases and injuries in 204 countries and territories, 1990–2019: a systematic analysis for the Global Burden of Disease Study 2019. The Lancet 2020, 396, 1204-1222, 10.1016/s0140-6736(20)30925-9.
- Harry L. Huber; THE PATHOLOGY OF BRONCHIAL ASTHMA. Archives of Internal Medicine 1922, 30, 689, 10.1001/archinte.1922.00110120002001.
- Emmanuel T. Osei; Leila B. Mostaço-Guidolin; Aileen Hsieh; Stephanie M. Warner; May Al-Fouadi; Mary Wang; Darren J. Cole; Geoffrey N. Maksym; Teal S. Hallstrand; Wim Timens; et al.Corry-Anke BrandsmaIrene H. HeijinkTillie-Louise. Hackett Epithelial-interleukin-1 inhibits collagen formation by airway fibroblasts: Implications for asthma. Scientific Reports 2020, 10, 1-14, 10.1038/s41598-020-65567-z.
- Stephen T. Holgate; Pathogenesis of Asthma. Clinical & Experimental Allergy 2008, 38, 872-897, 10.1111/j.1365-2222.2008.02971.x.
- Mostaço-Guidolin, L.; Osei, E.; Ullah, J.; Hajimohammadi, S.; Fouadi, M.; Li, X.; Li, V.; Shaheen, F.; Yang, C.X.; Chu, F.; et al. Defective Fibrillar Collagen Organization by Fibroblasts Contributes to Airway Remodeling in Asthma. Am. J. Respir. Crit. Care Med. 2019, 200, 431–443.
- Roche, W.; Williams, J.; Beasley, R.; Holgate, S. Subepithelial fibrosis in the bronchi of asthmatics. Lancet 1989, 333, 520–524.
- Dunnill, M.S. The pathology of asthma, with special reference to changes in the bronchial mucosa. J. Clin. Pathol. 1960, 13, 27–33.
- Carroll, N.; Elliot, J.; Morton, A.; James, A. The Structure of Large and Small Airways in Nonfatal and Fatal Asthma. Am. Rev. Respir. Dis. 1993, 147, 405–410.
- James, A.L.; Paré, P.D.; Hogg, J.C. The Mechanics of Airway Narrowing in Asthma. Am. Rev. Respir. Dis. 1989, 139, 242–246.
- Mostaco-Guidolin, L.B.; Loube, J.; Barlow, A.; Osei, E.T.; Vasilescu, D.M.; Hsieh, A.; Fouadi, M.; Young, C.; Scott, A.L.; Mitzner, W.; et al. Second harmonic generation imaging of collagen scaffolds within the alveolar ducts of healthy and emphysematous mouse lungs. Histochem. Cell Biol. 2021, 155, 279–289.
- Cañas, J.A.; Rodrigo-Muñoz, J.M.; Sastre, B.; Gil-Martinez, M.; Redondo, N.; Del Pozo, V. MicroRNAs as Potential Regulators of Immune Response Networks in Asthma and Chronic Obstructive Pulmonary Disease. Front. Immunol. 2021, 11.
- Organization WH. World Health Organization (2012). Top 10 Causes of Death. 2012.
- Klaus F. Rabe; Suzanne Hurd; Antonio Anzueto; Peter J. Barnes; Sonia A. Buist; Peter Calverley; Yoshinosuke Fukuchi; Christine Jenkins; Roberto Rodriguez-Roisin; Chris van Weel; et al.Jan Zielinski Global Strategy for the Diagnosis, Management, and Prevention of Chronic Obstructive Pulmonary Disease. American Journal of Respiratory and Critical Care Medicine 2007, 176, 532-555, 10.1164/rccm.200703-456so.
- Hogg, J.C.; Timens, W. The Pathology of Chronic Obstructive Pulmonary Disease. Annu. Rev. Pathol. Mech. Dis. 2009, 4, 435–459.
- Agustí, A.; Hogg, J.C. Update on the Pathogenesis of Chronic Obstructive Pulmonary Disease. N. Engl. J. Med. 2019, 381, 1248–1256.
- Williams, M.H. Chronic Obstructive Pulmonary Disease [Internet]. N. Y. State J. Med. 1979, 79, 919–921.
- Black, P.N.; Ching, P.S.T.; Beaumont, B.; Ranasinghe, S.; Taylor, G.; Merrilees, M.J. Changes in elastic fibres in the small airways and alveoli in COPD. Eur. Respir. J. 2008, 31, 998–1004.
- Merrilees, M.J.; Ching, P.S.; Beaumont, B.; Hinek, A.; Wight, T.N.; Black, P.N. Changes in elastin, elastin binding protein and versican in alveoli in chronic obstructive pulmonary disease. Respir. Res. 2008, 9, 41.
- Tan, B.W.; Sim, W.L.; Cheong, J.K.; Kuan, W.S.; Tran, T.; Lim, H.F. MicroRNAs in chronic airway diseases: Clinical correlation and translational applications. Pharmacol. Res. 2020, 160, 105045.
- Richeldi, L.; Du Bois, R.M.; Raghu, G.; Azuma, A.; Brown, K.K.; Costabel, U.; Cottin, V.; Flaherty, K.R.; Hansell, D.M.; Inoue, Y.; et al. Efficacy and Safety of Nintedanib in Idiopathic Pulmonary Fibrosis. N. Engl. J. Med. 2014, 370, 2071–2082.
- Hughes, G.; Toellner, H.; Morris, H.; Leonard, C.; Chaudhuri, N. Real World Experiences: Pirfenidone and Nintedanib are Effective and Well Tolerated Treatments for Idiopathic Pulmonary Fibrosis. J. Clin. Med. 2016, 5, 78.
- Raghu, G.; Remy-Jardin, M.; Myers, J.L.; Richeldi, L.; Ryerson, C.J.; Lederer, D.J.; Behr, J.; Cottin, V.; Danoff, S.K.; Morell, F.; et al. Diagnosis of Idiopathic Pulmonary Fibrosis. An Official ATS/ERS/JRS/ALAT Clinical Practice Guideline. Am. J. Respir. Crit. Care Med. 2018, 198, e44–e68.
- Takagi, K.; Yamakuchi, M.; Hashiguchi, T.; Inoue, H. The Role of miRNAs in Idiopathic Pulmonary Fibrosis. Interstitial Lung Dis. 2019, 2, 5–22.
- Jones, M.G.; Fabre, A.; Schneider, P.; Cinetto, F.; Sgalla, G.; Mavrogordato, M.; Jogai, S.; Alzetani, A.; Marshall, B.; O’Reilly, K.M.; et al. Three-dimensional characterization of fibroblast foci in idiopathic pulmonary fibrosis. JCI Insight 2016, 1.
- King, T.E.; Pardo, A.; Selman, M. Idiopathic pulmonary fibrosis. Lancet 2011, 378, 1949–1961.
- Liebow, A.A. Definition and Classification of Interstitial Pneumonias in Human Pathology1. Clin. Exerc. Test. 2015, 8, 1–33.
- Wolters, P.J.; Collard, H.R.; Jones, K.D. Pathogenesis of Idiopathic Pulmonary Fibrosis. Annu. Rev. Pathol. Mech. Dis. 2014, 9, 157–179.
- Le Saux, C.J.; Chapman, H.A. Idiopathic Pulmonary Fibrosis: Cell Death and Inflammation Revisited. Am. J. Respir. Cell Mol. Biol. 2018, 59, 137–138.
- Stijn Verleden; Naoya Tanabe; John McDonough; Dragoş M Vasilescu; Feng Xu; Wim A Wuyts; Davide Piloni; Laurens De Sadeleer; Stijn Willems; Cindy Mai; et al.Jeroen HostensJoel D CooperErik K VerbekenJohny VerschakelenCraig J GalbanDirk Van RaemdonckThomas V ColbyMarc DecramerGeert M VerledenNaftali KaminskiTillie-Louise HackettBart VanaudenaerdeJames C Hogg Small airways pathology in idiopathic pulmonary fibrosis: a retrospective cohort study. The Lancet Respiratory Medicine 2020, 8, 573-584, 10.1016/s2213-2600(19)30356-x.
- Burgess, J.K.; Mauad, T.; Tjin, G.; Karlsson, J.C.; Westergren-Thorsson, G. The extracellular matrix–The under-recognized element in lung disease? J. Pathol. 2016, 240, 397–409.
- Enomoto, N.; Suda, T.; Kono, M.; Kaida, Y.; Hashimoto, D.; Fujisawa, T.; Nakamura, Y.; Inui, N.; Imokawa, S.; Chida, K. Amounts of Elastic Fibers Predict Prognosis of Idiopathic Pulmonary Fibrosis. Respir. Med. 2013, 107, 1608–1616.
- Zhang, H.; Song, M.; Guo, J.; Ma, J.; Qiu, M.; Yang, Z. The function of non-coding RNAs in idiopathic pulmonary fibrosis. Open Med. 2021, 16, 481–490.
- Stephen T. Holgate; Pathogenesis of Asthma. Clinical & Experimental Allergy 2008, 38, 872-897, 10.1111/j.1365-2222.2008.02971.x.
- Emmanuel T. Osei; Leila B. Mostaço-Guidolin; Aileen Hsieh; Stephanie M. Warner; May Al-Fouadi; Mary Wang; Darren J. Cole; Geoffrey N. Maksym; Teal S. Hallstrand; Wim Timens; et al.Corry-Anke BrandsmaIrene H. HeijinkTillie-Louise. Hackett Epithelial-interleukin-1 inhibits collagen formation by airway fibroblasts: Implications for asthma. Scientific Reports 2020, 10, 1-14, 10.1038/s41598-020-65567-z.
- Organization WH. World Health Organization (2012). Top 10 Causes of Death. 2012.
- Klaus F. Rabe; Suzanne Hurd; Antonio Anzueto; Peter J. Barnes; Sonia A. Buist; Peter Calverley; Yoshinosuke Fukuchi; Christine Jenkins; Roberto Rodriguez-Roisin; Chris van Weel; et al.Jan Zielinski Global Strategy for the Diagnosis, Management, and Prevention of Chronic Obstructive Pulmonary Disease. American Journal of Respiratory and Critical Care Medicine 2007, 176, 532-555, 10.1164/rccm.200703-456so.
- Stijn Verleden; Naoya Tanabe; John McDonough; Dragoş M Vasilescu; Feng Xu; Wim A Wuyts; Davide Piloni; Laurens De Sadeleer; Stijn Willems; Cindy Mai; et al.Jeroen HostensJoel D CooperErik K VerbekenJohny VerschakelenCraig J GalbanDirk Van RaemdonckThomas V ColbyMarc DecramerGeert M VerledenNaftali KaminskiTillie-Louise HackettBart VanaudenaerdeJames C Hogg Small airways pathology in idiopathic pulmonary fibrosis: a retrospective cohort study. The Lancet Respiratory Medicine 2020, 8, 573-584, 10.1016/s2213-2600(19)30356-x.
