Euphorbia genus (Euphorbiaceae family), which is the third largest genus of angiosperm plants comprising ca. 2000 recognized species, is used all over the world in traditional medicine, especially in the traditional Chinese medicine. Members of this taxa are promptly recognizable by their specialized inflorescences and latex. In this review, an overview of Euphorbia-derived natural products such as essential oils, extracts, and pure compounds, active in a broad range of biological activities, and with potential usages in health maintenance, is described. The chemical composition of essential oils from Euphorbia species revealed the presence of more than 80 phytochemicals, mainly oxygenated sesquiterpenes and sesquiterpenes hydrocarbons, while Euphorbia extracts contain secondary metabolites such as sesquiterpenes, diterpenes, sterols, flavonoids, and other polyphenols.
- Euphorbia
- essential oils
- extracts
- phytochemicals
- terpenoids
- bioactivity
- antimicrobial
- anti-inflammation
- anticancer
- antioxidant
- phytoconstituents
1. Introduction
2. Traditional Medicine Uses of Euphorbia Plants
The Euphorbia genus is well-known to involve several plants used in folk medicine in different parts of the world, especially in traditional Chinese medicine [5][7][9][5,7,9]. Moreover, a recent study discriminated the global geographical distribution regarding uses of Euphorbia plants in traditional medicine [6]. In this regard, three particular uses were most often detected, such as (1) treatments of digestive system disorders (very globally frequent excepting Australasia); (2) as remedies for infections/infestations (mainly in Southern Africa and America, Pacific, Asia-tropical, and Asia-temperate); and (3) for treating skin/subcutaneous cellular tissue disorders (particularly in Australasia, Europe, Asia, and Northern America). On the other hand, within the 33 species with citations in folk practices worldwide, the three most-referenced plants used as traditional medicines were E. hirta L., E. thymifolia L., and E. lathyris [6]. Euphorbia hirta whole plant has been employed in Burundi, China, Philippines, and Nigeria to manage diarrhoea [35][36][37][38][35,36,37,38], while E. hirta decoction is used in Vietnam, India, and Mozambique to treat dysentery [39][40][41][39,40,41] and to treat bronchitis/asthma/coughs in Nepal, Australia, the South Western United States, and Hawaii [6][39][42][6,39,42]. Additionally, the latex from E. hirta is also applied to treat skin diseases and fever mostly in Asia [6] and to treat gonorrhoea in Malaysia [43] and other conditions such as malaria, candidiasis, and ringworm infections [6]. Populations aroundthe Vellore District of Tamil Nadu, India, use decoction of the E. hirta whole plant to treat poisonous snakebites (topically and orally administration) [44]. Despite the registered abortifacient properties of E. thymifolia decoctions in Chile, its latex or leaf decoctions have been recorded as lactation stimulants in different continents [45]. In the case of E. lathyris, emetic and purgative actions have been described in Europe as well as its seeds used to treat snakebites, ascites, schistosomiasis, and hydropsy [38][46][38,46]. Euphorbia maculata L. in Northern America is used for the treatment of corneal opacities and warts [47], while in China, it is used to treat blood disorders (e.g., haematuria, haemoptysis, epistaxis, and hemafecia), carbuncles, and wounds [38]. Euphorbia denticulata Lam. and E. macrocarpa Boiss. & Buhse are also used for wound healing in Turkey [48], and a similar use is reported in Ethiopia for E. heterophylla L. and E. prostrata Aiton [49]. The decoction, unguent, or hot steam of other Euphorbia species are used on inflammation conditions, such as E. corollata L. (for dropsy), E. marginata Pursh, and E. antiquorum (for swellings) [6]. Similarly, E. antiquorum is utilized in Vietnam to alleviate toothache events [41] as well as for treating cutaneous dropsy, cutaneous infections, cancer, and liver ailment [50]. E. tirucalli L. and E. ingens E.Mey. ex Boiss. like E. lathyris, can be used as an emetic against snakebites [39][51][39,51]. A recent review has been published showing that E. tirucalli (whole plant and its parts individually separated) has some records in South America, India, the Middle East, and Africa regarding beneficial effects on leprosy, syphilis, cancer, asthma, and intestinal parasites [51]. The same research group [52] also published a review where they report the various applications in traditional medicine of E. neriifolia L. Its latex is used as a carminative and expectorant, as well as in the treatment of tumours, abdominal and skin problems, leprosy, asthma, and kidney stones, while the roots are used in the treatment of scorpion stings and snake bites. The leaves can also be used as carminative and in the treatment of pain, inflammation, bronchial infections and lack of appetite [52]. Euphorbia helioscopia L. is used in the traditional Chinese medicine in situations of bacillary dysentery, osteomyelitis, and malaria [53]. In Uyghur medicine, China, E. resinifera O.Berg is recurrently employed to suppress tuberculosis, toothache, and chronic pain [54], while E. fischeriana have been used as a remedy for cancer, ascites, and oedema [55], and E. granulata Forssk. is utilized against intestine worms, oedema, cough, blood impurities, and renal diseases [56][57][56,57]. However, some Euphorbia plants, especially their latex or milky sap (e.g., E. hirta, E. helioscopia, E. royleana Boiss. among others), are considered as irritating materials for skin, mouth, and throat, causing burning sensation, acute inflammation (even blisters), and nausea [58]. In veterinary medicine, E. milii Des Moul. and E. nivulia is used to treat diarrhoea and wounds in livestock, respectively, but other Euphorbia species can produce irritations [6].3. Euphorbia Plants: Essential Oil Composition and Activities
Researchers from various countries worldwide have studied the chemical composition of essential oils (EOs) from different Euphorbia species. An overview of their most abundant components (the content higher than 5%) along with the most relevant biological activities to health maintenance (when available, and when the biological activity of a positive standard compound was also presented) is given in Table 1. The chemical structure of the major constituents of EOs from Euphorbia species whose content is higher than 25% is depicted in Figure 1.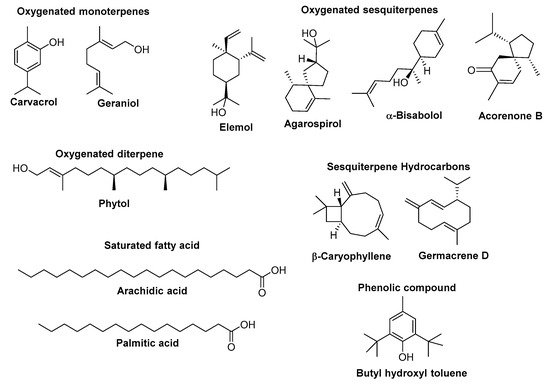
| Species | Origin | Raw Material | Extraction Method | Main Components a (%) | Most Relevant Biological Activities | Ref. |
|---|---|---|---|---|---|---|
| E. acanthothamnos Heldr. & Sart. ex Boiss. | Greece | Inflorescences | Steam distillation | Phytol (28.3), phytol acetate (9.3), β-caryophyllene (7.5) | not evaluated | [59] |
| E. apios L. | Greece | Inflorescences | Steam distillation | Germacrene D (30.0), heptacosane (12.7), β-caryophyllene (10.0), tricosane (6.5), pentacosane (6.0) | not evaluated | [59] |
| E. characias L. | Greece | Inflorescences | Steam distillation | Nonanal (22.8), phytol (13.5), pentacosane (8.5), heptacosane (7.4), palmitic acid (5.7), nonacosane (5.6) | not evaluated | [59] |
| E. cotinifolia L. (syn. E. caracasana (Klotzsch & Garcke) Boiss.) | Venezuela | Leaves | Hydro-distillation | β-Caryophyllene (39.3), germacrene-D (21.5%), α-copaene (9.3), α-humulene (5.2) | not evaluated | [60] |
| E. dendroides L. | Greece | Inflorescences | Steam distillation | Heptacosane (10.5), pentacosane (6.0), 4-terpineol (5.5), tricosane (5.0) | not evaluated | [59] |
| E. densa Schrenk | Syria | Aerial parts | Hydro-distillation | 1,8-Cineole (18.87), linalool (13.61), carvacrol (13.32), (E)-caryophyllene (10.29) | Radical scavenging activity (EC50 = 0.35 µg/mL) lower than BHA (EC50 = 0.135 µg/mL) | [61] |
| E. fischeriana Steud. | China | Roots | Steam distillation | Eudesmol (18.22), p-menth-8-en-2-ol (9.36), caryophyllene oxide (8.61), selinenol (6.83) | Radical scavenging activity (IC50 = 57.2 µg/mL) similar to ascorbic acid (IC50 = 63.1 µg/mL) but lower than BHT (IC50 = 26.1 µg/mL) | [62] |
| E. fragifera Jan | Italy | Inflorescences | Steam distillation | Carvacrol (61.55), carvon (9.22), β-caryophyllene (5.80)/geraniol (59.65), β-caryophyllene (9.05) | not evaluated | [63] |
| E. gaillardotii Boiss. & Blanche | Turkey | Aerial parts | Hydro-distillation | Arachidic acid (32), hexatriacontane (8.7), mint furanone (8.4), palmitic acid (8.0), tetratetracontane (6.2), octadecane (5.6), α-silenene (5.2) | Anti-lipid peroxidation activity (IC50 = 14.8 µg/mL) similar to α-tocopherol, but much lower radical scavenging activity than BHT. | [64] |
| E. golondrina L.C.Wheeler | Cameroon | Leaves | Steam distillation | Caryophyllene oxide (14.16), 2-pentadecanone (13.78), camphor (9.41), phytol (5.75) | not evaluated | [65] |
| E. hebecarpa Boiss. | Iran | Aerial parts | Hydro-distillation | α-Bisabolol (31.2), cis-cadin-4-en-7-ol (20.1), trans-piperitol (8.6), cis-p-menth-2-en-1-ol (6.4), trans-p-menth-2-en-1-ol (6.2) | not evaluated | [66] |
| E. helioscopia L. | Greece | Inflorescences | Steam distillation | Phytol (21.2), β-caryophyllene (10.0), behenic acid methyl ester (8.1), myristic acid methyl ester (5.5) | not evaluated | [59] |
| E. helioscopia L. | Turkey | Aerial parts | Hydro-distillation | β-Cubebene (19.3), palmitic acid (12.2), caryophyllene oxide (11.7), τ-elemene (9.3), spathulenol (9.3), phytol (6.9), hexahydrofarnesly acetone (5.3) | Low antioxidant and antiacetylcholinesterase activity, moderate butyrylcholinesterase and similar anti-urease activity to thiourea. | [67] |
| E. heterophylla L. | Nigeria | Leaves | Hydro-distillation | 3,7,12,15-Tetramethyl-2-hexadecen-1-ol (12.30), stearic acid (11.21), oleic acid (10.42), linoleic acid (8.97), 1,2-epoxy-cyclododecane (7.91), 13-tetradece-11-yn-1-ol (7.83), 7,10-hexadecadienal (7.62), 1,2,15,16-diepoxyhexadecane (6.37), phytol (6.32), 2-monopalmitin (5.43) | Toxic to brine shrimp larvae (LC50 = 21.7 µg/mL). Radical scavenging activity similar to ascorbic acid, lower than BHA but higher than α-tocopherol at 250 µg/mL. | [68] |
| E. heterophylla L. | Nigeria | Stems | Hydro-distillation | Stearic acid (11.21), oleic acid (10.42), linoleic acid (8.97), 1,2-epoxy-cyclododecane (7.91), 13-tetradece-11-yn-1-ol (7.83), 7,10-hexadecadienal (7.62), 1,2,15,16-diepoxyhexadecane (6.37), phytol (6.32), 2-monopalmitin (5.43), 2-aminoethoxyethynediyl methyl ester (5.40) | Very toxic to brine shrimp larvae (LC50 = 8.94 µg/mL). Radical scavenging activity similar to ascorbic acid, lower than BHA but higher than α-tocopherol at 250 µg/mL. | [68] |
| E. heterophylla L. | Egypt | Aerial parts | Hydro-distillation | 1,8-Cineole (32.0), camphor (16.5), β-elemene (5.9 ) | Radical scavenging activity (IC50 325.3 µL/L) lower than ascorbic acid (204.4 µL/L). | [69] |
| E. hirta L. | Lagos | Leaves | Hydro-distillation | Phytol and its isomeric forms (34.8), 6,10,14-trimethyl-2-pentadecanone (12.37), hexadecanal (7.63), palmitic acid (6.26) | not evaluated | [70] |
| E. macroclada Boiss. | Turkey | Aerial parts | Hydro-distillation | Tetratetracontane (42.7), hexatriacontane (12), mint furanone (6.0) | Anti-lipid peroxidation activity (IC50 = 14.8 µg/mL) similar to α-tocopherol. Lower radical scavenging activity than BHT but higher than E. gaillardotii essential oil. | [64] |
| E. macrorrhiza C.A.Mey. ex Ledeb. | China | Aerial parts | Hydro-distillation | Acorenone B (16.72), (+)-cycloisosativene (14.94), 3β-hydroxy-5α-androstane (10.62), β-cedrene (8.40), copaene (7.37), palmitic acid (5.68) | Cytotoxic activity against Caco-2 cell line (IC50 = 78.32 µg/mL), antibacterial activity against Staphyloccocus aureus (MIC = 5.6 µg/mL) but lower than ampicillin (MIC = 0.25 µg/mL) | [71] |
| E. macrorrhiza C.A.Mey. ex Ledeb. | China | Roots | Hydro-distillation | Acorenone B (25.80), (+)-cycloisosativene (12.40), β-cedrene (7.98), copaene (6.29), 3β-hydroxy-5α-androstane (5.52) | Cytotoxic activity against Caco-2 cell line (IC50 = 11.86 µg/mL), antibacterial activity against Staphyloccocus aureus (MIC = 2.8 µg/mL) but lower than ampicillin (MIC = 0.25 µg/mL) | [71] |
| E. pekinensis Rupr. | China | Roots | Steam distillation | Agarospirol (49.23), hedycargol (20.66) | not evaluated | [72] |
| E. pilosa L. | India | Aerial parts | Hydro-distillation | Phytol (5.75), n-pentadecanal (5.12) | not evaluated | [73] |
| E. rigida M.Bieb. | Greece | Inflorescences | Steam distillation | Heneicosane (13.8), heptacosane (12.7), β-caryophyllene (9.4), linalool (6.7), pentacosane (6.5) | not evaluated | [59] |
| E. sanctae-caterinae Fayed | Egypt | Aerial parts | Hydro-distillation | Valencene (16.01), (+) spathulenol (15.41), (-)-caryophyllene oxide (10.50), limonene (7.66) | not evaluated | [74] |
| E. sanctae-caterinae Fayed | Egypt | Aerial parts | Microwave-assisted | Butyl hydroxyl toluene (25.58), β-eudesmol (13.67), 6-epi-shyobunol (11.83), (+) spathulenol (10.32), thymol (7.00) | not evaluated | [74] |
| E. teheranica Boiss. | Iran | Aerial parts | Hydro-distillation | Elemol (57.5), β-caryophyllene (8.1%), caryophyllene oxide (7.8%) | not evaluated | [75] |
| E. thymifolia L. | India | Aerial parts | Steam distillation | Palmitic acid (33.03), phytol (10.367), myristic acid (6.58) | not evaluated | [76] |
| E. tithymaloides L. | Bangladesh | Aerial parts | Steam distillation | Eugenol (22.52), phenyl ethyl alcohol (14.63), 3-pentanol (9.22), caryophyllene oxide (7.73), isoeugenol (7.32), pentadecanol (5.14), spathulenol (5.11) | Radical scavenging activity (DPPH IC50 = 13.67 µg/mL) higher than BHA (IC50 = 18.26 µg/mL). | [77] |
