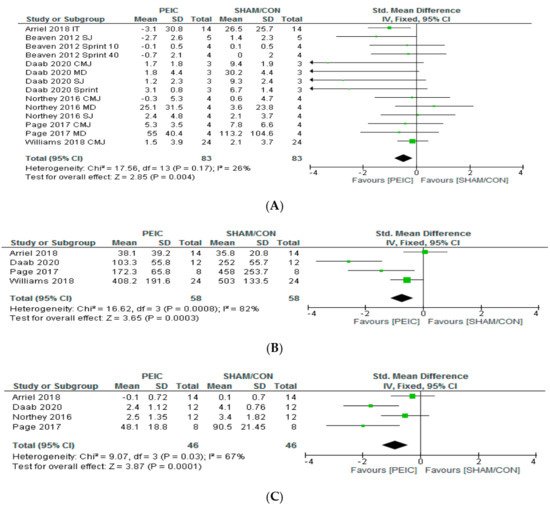
It has been demonstrated that brief cycles of ischemia followed by reperfusion (IR) applied before exercise can improve performance and, IR intervention, applied immediately after exercise (post-exercise ischemic conditioning—PEIC) exerts a potential ergogenic effect to accelerate recovery. Thus, the purpose of this systematic review with meta-analysis was to identify the effects of PEIC on exercise performance, recovery and the responses of associated physiological parameters, such as creatine kinase, perceived recovery and muscle soreness, over 24 h after its application. From 3281 studies, six involving 106 subjects fulfilled the inclusion criteria. Compared to sham (cuff administration with low pressure) and control interventions (no cuff administration), PEIC led to faster performance recovery (p = 0.004; ES = −0.49) and lower increase in creatine kinase (p < 0.001; effect size (ES) = −0.74) and muscle soreness (p < 0.001; ES = −0.88) over 24 h. The effectiveness of this intervention is more pronounced in subjects with low/moderate fitness level and at least a total time of 10 min of ischemia (e.g., two cycles of 5 min) is necessary to promote positive effects.
- intermittent occlusion
- blood flow occlusion
- sports
- ergogenic
- ischemic postconditioning
1. Introduction
2. Quality of the Papers
3. Participants Involved
4. Exercise Protocols to Induce Fatigue and Assess Performance
5. PEIC Effects on Performance Recovery, CK and MS
6. PEIC Protocols and Possible Mechanism
| PEIC Sets | Total PEIC and SHAM Time (min) | Ischemia Pressure (mm Hg) PEIC/SHAM/Limb | Time to Test | Groups | Were Subjects Informed about Effects of PEIC? | |
|---|---|---|---|---|---|---|
| Beaven et al. (2012) [24] | 2 × 3 min | 6 | 220/15/thigh | 5 min–24 h | PEIC/SHAM | No |
| Northey et al. (2016) [27] | 2 × 3 min | 6 | 220/#/thigh | 1–24 h | PEIC/CON | It was not exposed by authors |
| Page et al. (2017) [7] | 3 × 5 min | 15 | 220/20/thigh | 24–48–72 h | PEIC/SHAM | No |
| Williams et al. (2018) [28] | 2 × 3 min | 6 | 171−266/15/thigh | 2–24 h | PEIC/SHAM | Yes |
| Arriel et al. (2018) [6] | 2 × 5 min and 5 × 2 min | 10 and 10 | 50 > SAP/20/thigh | 24 h | PEIC/SHAM | Yes |
| Daab et al. (2020) [29] | 3 × 5 min | 15 | 50 > SAP/20/thigh | 0–24–48–72 h | PEIC/SHAM | It was not exposed by authors |
References
- Chapman, A.; Vicenzino, B.; Blanch, P.; Hodges, P. Do differences in muscle recruitment between novice and elite cyclists reflect different movement patterns or less skilled muscle recruitment? J. Sci. Med. Sport 2009, 12, 31–34.
- Rodríguez-Marroyo, J.A.; Pernía, R.; Villa, J.G.; Foster, C. Reliability and Seasonal Changes of Submaximal Variables to Evaluate Professional Cyclists. Int. J. Sports Physiol. Perform. 2017, 12, 1356–1362.
- Rowlands, D.S.; Pearce, E.; Aboud, A.; Gillen, J.B.; Gibala, M.J.; Donato, S.; Waddington, J.M.; Green, J.G.; Tarnopolsky, M.A. Oxidative stress, inflammation, and muscle soreness in an 894-km relay trail run. Eur. J. Appl. Physiol. 2012, 112, 1839–1848.
- Filho, E.; Di Fronso, S.; Forzini, F.; Murgia, M.; Agostini, T.; Bortoli, L.; Robazza, C.; Bertollo, M. Athletic performance and recovery-stress factors in cycling: An ever changing balance. Eur. J. Sport Sci. 2015, 15, 671–680.
- Barnett, A. Using recovery modalities between training sessions in elite athletes: Does it help? Sports Med. 2006, 36, 781–796.
- Arriel, R.A.; De Souza, H.L.R.; Da Mota, G.R.; Marocolo, M. Declines in exercise performance are prevented 24 hours after post-exercise ischemic conditioning in amateur cyclists. PLoS ONE 2018, 13, e0207053.
- Page, W.; Swan, R.; Patterson, S.D. The effect of intermittent lower limb occlusion on recovery following exercise-induced muscle damage: A randomized controlled trial. J. Sci. Med. Sport 2017, 20, 729–733.
- Garcia, C.A.; Da Mota, G.R.; Marocolo, M. Cold Water Immersion is Acutely Detrimental but Increases Performance Post-12 h in Rugby Players. Int. J. Sports Med. 2016, 37, 619–624.
- Lanferdini, F.J.; Bini, R.R.; Baroni, B.M.; Klein, K.D.; Carpes, F.P.; Vaz, M.A. Low-Level Laser Therapy Improves Performance and Reduces Fatigue in Competitive Cyclists. Int. J. Sports Physiol. Perform. 2017, 1–27.
- Marocolo, M.; Billaut, F.; Da Mota, G.R. Ischemic Preconditioning and Exercise Performance: An Ergogenic Aid for Whom? Front. Physiol. 2018, 9, 1874.
- Incognito, A.V.; Burr, J.F.; Millar, P.J. The Effects of Ischemic Preconditioning on Human Exercise Performance. Sports Med. Auckl. N. Z. 2016, 46, 531–544.
- Marocolo, M.; Da Mota, G.R.; Simim, M.M.; Appell Coriolano, H.-J. Myths and Facts about the Effects of Ischemic Preconditioning on Performance. Int. J. Sports Med. 2016, 37, 87–96.
- Murry, C.E.; Jennings, R.B.; Reimer, K.A. Preconditioning with ischemia: A delay of lethal cell injury in ischemic myocardium. Circulation 1986, 74, 1124–1136.
- Zhao, Z.-Q.; Corvera, J.S.; Halkos, M.E.; Kerendi, F.; Wang, N.-P.; Guyton, R.A.; Vinten-Johansen, J. Inhibition of myocardial injury by ischemic postconditioning during reperfusion: Comparison with ischemic preconditioning. Am. J. Physiol. Heart Circ. Physiol. 2003, 285, H579–H588.
- Crisafulli, A.; Melis, F.; Tocco, F.; Santoboni, U.M.; Lai, C.; Angioy, G.; Lorrai, L.; Pittau, G.; Concu, A.; Pagliaro, P. Exercise-induced and nitroglycerin-induced myocardial preconditioning improves hemodynamics in patients with angina. Am. J. Physiol. Heart Circ. Physiol. 2004, 287, H235–H242.
- Marongiu, E.; Crisafulli, A. Cardioprotection acquired through exercise: The role of ischemic preconditioning. Curr. Cardiol. Rev. 2014, 10, 336–348.
- Jin, C.; Wu, J.; Watanabe, M.; Okada, T.; Iesaki, T. Mitochondrial K+ channels are involved in ischemic postconditioning in rat hearts. J. Physiol. Sci. 2012, 62, 325–332.
- Libonati, J.R.; Cox, M.; Incanno, N.; Melville, S.K.; Musante, F.C.; Glassberg, H.L.; Guazzi, M. Brief periods of occlusion and reperfusion increase skeletal muscle force output in humans. Cardiologia 1998, 43, 1355–1360.
- Jean-St-Michel, E.; Manlhiot, C.; Li, J.; Tropak, M.; Michelsen, M.M.; Schmidt, M.R.; McCrindle, B.W.; Wells, G.D.; Redington, A.N. Remote preconditioning improves maximal performance in highly trained athletes. Med. Sci. Sports Exerc. 2011, 43, 1280–1286.
- Bailey, T.G.; Jones, H.; Gregson, W.; Atkinson, G.; Cable, N.T.; Thijssen, D.H.J. Effect of ischemic preconditioning on lactate accumulation and running performance. Med. Sci. Sports Exerc. 2012, 44, 2084–2089.
- Arriel, R.A.; Meireles, A.; Hohl, R.; Marocolo, M. Ischemic preconditioning improves performance and accelerates the heart rate recovery. J. Sports Med. Phys. Fitness 2020.
- Burt, D.G.; Twist, C. The effects of exercise-induced muscle damage on cycling time-trial performance. J. Strength Cond. Res. 2011, 25, 2185–2192.
- Reid, M.B. Reactive Oxygen Species as Agents of Fatigue. Med. Sci. Sports Exerc. 2016, 48, 2239–2246.
- Beaven, C.M.; Cook, C.J.; Kilduff, L.; Drawer, S.; Gill, N. Intermittent lower-limb occlusion enhances recovery after strenuous exercise. Appl. Physiol. Nutr. Metab. 2012, 37, 1132–1139.
- De Souza, H.L.R.; Arriel, R.A.; Hohl, R.; Da Mota, G.R.; Marocolo, M. Is Ischemic Preconditioning Intervention Occlusion-Dependent to Enhance Resistance Exercise Performance? Strength Cond. Res. 2019.
- Tocco, F.; Marongiu, E.; Ghiani, G.; Sanna, I.; Palazzolo, G.; Olla, S.; Pusceddu, M.; Sanna, P.; Corona, F.; Concu, A.; et al. Muscle ischemic preconditioning does not improve performance during self-paced exercise. Int. J. Sports Med. 2015, 36, 9–15.
- Northey, J.M.; Rattray, B.; Argus, C.K.; Etxebarria, N.; Driller, M.W. Vascular Occlusion and Sequential Compression for Recovery After Resistance Exercise. J. Strength Cond. Res. 2016, 30, 533–539.
- Williams, N.; Russell, M.; Cook, C.J.; Kilduff, L.P. The effect of lower limb occlusion on recovery following sprint exercise in academy rugby players. J. Sci. Med. Sport 2018, 21, 1095–1099.
- Daab, W.; Bouzid, M.A.; Lajri, M.; Bouchiba, M.; Rebai, H. Brief cycles of lower-limb occlusion accelerate recovery kinetics in soccer players. Phys. Sports Med. 2020, 1–8.
- Arriel, R.A.; De Souza, H.L.R.; Da Silva, B.V.C.; Marocolo, M. Ischemic preconditioning delays the time of exhaustion in cycling performance during the early but not in the late phase. Mot. Rev. Educ. Fís. 2019, 25.
- Richard, P.; Billaut, F. Time-Trial Performance in Elite Speed Skaters after Remote Ischemic Preconditioning. Int. J. Sports Physiol. Perform. 2018, 1–9.
- Gathercole, R.J.; Sporer, B.C.; Stellingwerff, T.; Sleivert, G.G. Comparison of the Capacity of Different Jump and Sprint Field Tests to Detect Neuromuscular Fatigue. J. Strength Cond. Res. 2015, 29, 2522–2531.
- Córdova, A.; Sureda, A.; Albina, M.L.; Linares, V.; Bellés, M.; Sánchez, D.J. Oxidative stress markers after a race in professional cyclists. Int. J. Sport Nutr. Exerc. Metab. 2015, 25, 171–178.
- Córdova Martínez, A.; Martorell Pons, M.; Sureda Gomila, A.; Tur Marí, J.A.; Pons Biescas, A. Changes in circulating cytokines and markers of muscle damage in elite cyclists during a multi-stage competition. Clin. Physiol. Funct. Imaging 2015, 35, 351–358.
- Halson, S.L.; Bridge, M.W.; Meeusen, R.; Busschaert, B.; Gleeson, M.; Jones, D.A.; Jeukendrup, A.E. Time course of performance changes and fatigue markers during intensified training in trained cyclists. J. Appl. Physiol. 2002, 93, 947–956.
- Dankel, S.J.; Buckner, S.L.; Jessee, M.B.; Mattocks, K.T.; Mouser, J.G.; Counts, B.R.; Laurentino, G.C.; Abe, T.; Loenneke, J.P. Post-exercise blood flow restriction attenuates muscle hypertrophy. Eur. J. Appl. Physiol. 2016, 116, 1955–1963.
- Powers, S.K.; Jackson, M.J. Exercise-induced oxidative stress: Cellular mechanisms and impact on muscle force production. Physiol. Rev. 2008, 88, 1243–1276.
- Raat, N.J.H.; Shiva, S.; Gladwin, M.T. Effects of nitrite on modulating ROS generation following ischemia and reperfusion. Adv. Drug Deliv. Rev. 2009, 61, 339–350.
- Sun, H.-Y.; Wang, N.-P.; Kerendi, F.; Halkos, M.; Kin, H.; Guyton, R.A.; Vinten-Johansen, J.; Zhao, Z.-Q. Hypoxic postconditioning reduces cardiomyocyte loss by inhibiting ROS generation and intracellular Ca2+ overload. Am. J. Physiol. Heart Circ. Physiol. 2005, 288, H1900–H1908.
- Johnsen, J.; Pryds, K.; Salman, R.; Løfgren, B.; Kristiansen, S.B.; Bøtker, H.E. The remote ischemic preconditioning algorithm: Effect of number of cycles, cycle duration and effector organ mass on efficacy of protection. Basic Res. Cardiol. 2016, 111, 10.
- Wang, G.; Li, X.; Wang, H.; Wang, Y.; Zhang, L.; Zhang, L.; Liu, B.; Zhang, M. Later phase cardioprotection of ischemic post-conditioning against ischemia/reperfusion injury depends on iNOS and PI3K-Akt pathway. Am. J. Transl. Res. 2015, 7, 2603–2611.
- Powers, S.K.; Ji, L.L.; Kavazis, A.N.; Jackson, M.J. Reactive oxygen species: Impact on skeletal muscle. Compr. Physiol. 2011, 1, 941–969.
