Human neuroblastoma (NB), a pediatric tumor inclined to relapse, after an initial response to therapy, usually develops resistance. Since several chemotherapeutics, including the weel known etoposide (ETO), exert anticancer effect by increasing reactive oxygen species (ROS), NB cells overproduce antioxidant compounds becoming drugs-resistant. Moreover, ETO, although widely used, suffers from fast metabolism, poor solubility and systemic toxicity, that limit its administration dosage and its therapeutic efficiency. An appealing strategy to sensitize NB cells to chemotherapy involves the use of less toxic natural compounds able to reduce antioxidant defenses of NB cells and to induce ROS overproduction. In this contest, although affected by several issues as instability and poor absorbability, antioxidant/pro-oxidant polyphenols, such as gallic acid (GA), showed pro-oxidant anti-cancer effects and low toxicity for healthy cells, in several kind of tumors, not including NB. Herein, for the first time, free GA, two GA-dendrimers, and the dendrimer adopted as GA reservoir were tested on both sensitive and chemoresistant NB cells. Furthermore, the dendrimer adopted as carrier for GA was exploited also for entrapping and protecting ETO and for enhancing its solubility and effectiveness.The dendrimer device induced ROS-mediated death both in sensitive NB cells and also in chemoresistant ones. Free GA proved a dose-dependent ROS-mediated cytotoxicity on both cell populations. Intriguingly, when administered in dendrimer formulations at a dose not cytotoxic for NB cells, GA nullified any pro-oxidant activity of dendrimer. Unfortunately, due to GA, nanoformulations were inactive on NB cells, but GA resized in nanoparticles showed considerable ability in counteracting, at low dose, ROS production and oxidative stress, herein induced by the dendrimer. Interestingly, the ETO-dendrimer showed a synergistic action, controlled released over time with a significantly improved drug bioactivity, representing a novel biodegradable and promising device for the delivery of ETO into NB cells.
- Human neuroblastoma
- gallic acid (GA)
- polyester dendrimers
- dendrimer nanoformulations
- etoposide (ETO)
- ETO loaded dendrimer
1. Introduction
In the last decades, the correlation between oxidative stress (OS) and cancer onset has been highlighted by several pieces of evidence, but with contradictory findings. In fact, during the neoplastic transformation, there is an improvement in the production of reactive oxygen species (ROS), which supports tumor cell growth, by stimulating both redox-modulated signal transduction routes and transcription agents. In this regard, the use of ROS scavenging synthetic or natural compounds, counteracting the activation of redox sensitive pathways, could be employed as anti-carcinogenic for preventing the tumors onset and for treating the disease in the early stages.
When chemotherapeutic drugs induce an increase in intracellular ROS levels, the intrinsic antioxidant defense decreases and cancer cell death occurs. Therefore, in this perspective, antioxidants, contrasting the cytotoxic role of ROS, could favor neoplastic progression.
In this regard, an effective anti-tumor strategy could be to combine drugs capable of increasing ROS production with compounds able to reduce the intracellular antioxidant defenses. Human neuroblastoma (NB) is a solid tumor affecting young children, responsible for 15% of childhood cancer mortality [1][2][3].
NB is a heterogeneous tumor that origins from neural crest elements of the sympathetic nervous system and includes both low-risk forms, capable of regressing spontaneously or of differentiating into benign ganglioneuroblastoma, and high-risk (HR) forms, featured by metastatic disease and/or presence of MYCN proto-oncogene amplification. The amplification of MYCN is a biomarker still used today for early prognosticating risk and it is an indicative factor of poor prognosis [4].
Currently, HR patients undergo treatments, which include intensive and toxic chemotherapy followed by surgical resection, myeloablation and rescue of autologous stem cell, radiotherapy, and intensive immunotherapy [5].
Chemotherapeutic standard treatments consist of multi-drugs therapies, which combine several compounds including doxorubicin and etoposide (ETO). In particular, ETO is widely used [6][7][8] and induces a ROS-mediated anti-tumor effect. Unfortunately, its severe side effects [9][10] and chemoresistance limit its clinical success [11]. Moreover, poor water solubility, metabolic and non-metabolic inactivation, myelosuppression, and poor bioavailability are additional drawbacks related to ETO.
Chemoresistance is a multifactorial phenomenon and, recently, it has been demonstrated that ETO-resistant NB cells had high levels of glutathione (GSH) [12], which is the most important intracellular antioxidant thiol crucially involved in both cancer progression and chemoresistance [13]. In this context, the current trend of research, in the field of cancer treatment, increasingly focuses on developing alternative preventive and/or therapeutic strategies, based on the use of less toxic natural bioactive compounds, such as polyphenols, having both pro-oxidant and antioxidant activities, usually correlated to the dose [14].
Polyphenols, due to their antioxidant properties, have the possibility to act as preventive anti-cancer compounds [15], and, thanks to their pro-oxidant effects, they can work as mimics of chemotherapeutic drugs, inducing ROS-mediated cancer cell death [16].
It is the case of gallic acid (GA, Figure 1), which is the 3,4,5-tri-hydroxyls derivative of benzoic acid and represents one of the major phenolic acids present in various edible natural products, such as green tea, gallnuts, oak bark, apple peels, grapes, strawberries, pineapples, bananas, and many other fruits [17].
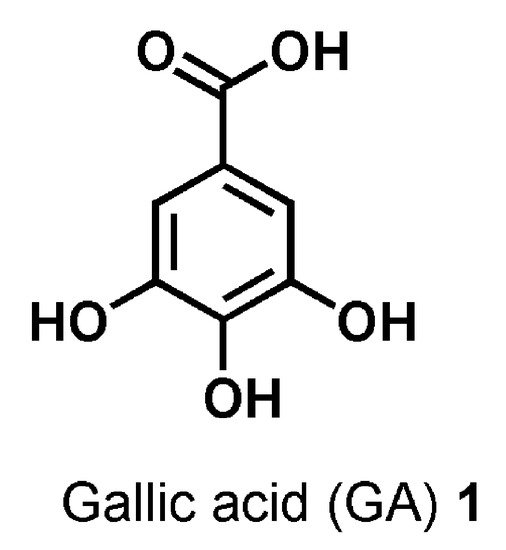
Figure 1. Chemical structure of gallic acid (GA) 1.
The ingestion of these foods is justifiably correlated to beneficial outcomes for the human health, such as reduced risk of cardiovascular diseases and myocardial infarction, reduced predisposition to tumor diseases, and improvement of the quality of life in people suffering from neurodegenerative diseases or early stage tumors. Inside plants, GA is one of the secondary metabolites involved in the formation of the galatotannin-hydrolysable tannins, but, in the biomedical sector, it has long attracted the interest of scientists for its ambivalent antioxidant/pro-oxidant behavior [18] and its capacity in counteracting diseases correlated to OS, through its anti-bacterial, anti-viral, anti-inflammatory, anti-neurodegenerative, and anticancer activities [15][16][17][19][20][21][22][23][24][25][26][27][28][29].
Furthermore, GA finds applications in several other areas, including food and cosmetic industry, as natural preservative in food, beverages, beauty products, and essential oils, because of its free radicals scavenging activity (RSA) [30].
Several studies report that GA can counteract cancer growth and progression, thanks to its anti-invasive and anti-metastatic activities and that it could be useful both to treat neoplasia, inducing ROS production such as ETO, and to prevent malignant transformation, exerting a protective antioxidant effect on healthy cells [31][32][33][34].
Based on these findings and considering that, to our knowledge, no evidence proving significant GA activity against human NB cells has yet been reported yet, this compound, for the first time, was investigated as novel alternative therapeutic approach to treat this pediatric tumor. Unfortunately, GA is sparingly soluble in water and alcohols [35], practically insoluble in hydrophobic solvents, and its activity and clinical applications are hampered by additional drawbacks such as high instability and low bioavailability.
Luckily, the recent advances in the field of nanomedicine and in the use of nanoparticles (NPs), as convenient drug carrier systems capable of improving drug solubility, half-life, and bioavailability and of lessening the metabolism and systemic toxicity of several problematic bioactive compounds, including polyphenols, increasingly allow for further expansion of the possibilities of anticancer treatments by employing natural compounds.
In the last decades, dendrimers have arisen as the most talented NP carrier systems endowed with the possibility of revolutionizing cancer treatments. Dendrimers can be employed to efficiently deliver anti-neoplastic drugs and typically are used as scaffolding with a well-defined architecture or as nanovehicles to conjugate, complex, or entrap therapeutic drugs.
Structurally, dendrimers are symmetric monodisperse [36] tree-like macromolecules, with both internal cavities for guest molecule encapsulation [37] and several peripheral chemical groups for further functionalization by covalent bond.
Interestingly, they possess an unusually low intrinsic viscosity that makes easy their transport in the blood [38][39].
They possess the capability of controlling molecular weight, hydrophilicity, solubility [39][40][41], bioavailability, and pharmacokinetic behavior of transported drugs.
Thanks to dendrimer’s ability in establishing strong interactions with several drugs, their loading results facilitated and their systemic toxicity, often due to the initial massive drug release (burst release), is minimized [39][40][41].
Among dendrimers, positively charged and commercially available poly(amidoamine) (PAMAMs) and polypropyleneimine (PPIs) are well-functioning for several biomedical applications, including gene therapy and drug delivery, but their clinical application is hampered by high non-selective cytotoxicity, hemolytic toxicity, genotoxicity, low biodegradability, fast removal from circulatory system, and high uptake in the reticuloendothelial system [42].
On the contrary, uncharged polyester dendrimer scaffolds appear more attractive and suitable for biomedical applications, because respectful of physiologic membranes and characterized by a good biodegradability [36][43][44].
Concerning ETO, inorganic nanohybrid constructs, consisting of layered double hydroxides (LDHs) intercalated with VP16 (ETO phosphate), have been successfully synthesized and characterized, that showed a high cytotoxic effect on cancer cells while being less toxic for healthy cells. However, although the results obtained from the employment of LDHs are promising, further improvements could be important in order to enhance some physicochemical properties, which are pivotal for a well-functioning nanosized drug delivery system, such as Z-potential and an increase in DL%. Among the strategies adopted, the use of dendrimer offers interesting perspectives . Concerning GA-enriched dendrimers, PAMAM dendrimers have been adopted both for covalently linking and for encapsulating GA [45][46], while PPI dendrimers have been used to solubilize GA and to control its release profile [47].
In addition, examples of biodegradable polyester-based dendrimers in which GA is either the repeated monomeric unity [48][49] or makes part of the dendrimer backbone [50] are reported.
In this work, thinking about a strategy to improve the outcomes from ETO treatments and about a future clinical application of GA, in order to ameliorate their solubility and stability, to slow down their metabolism, and to minimize the active dosage, a lab-made biodegradable dendrimer (namely, 4) [51][52][53][54][55][56] was adopted for formulating both ETO and GA in NPs (Figure 2).
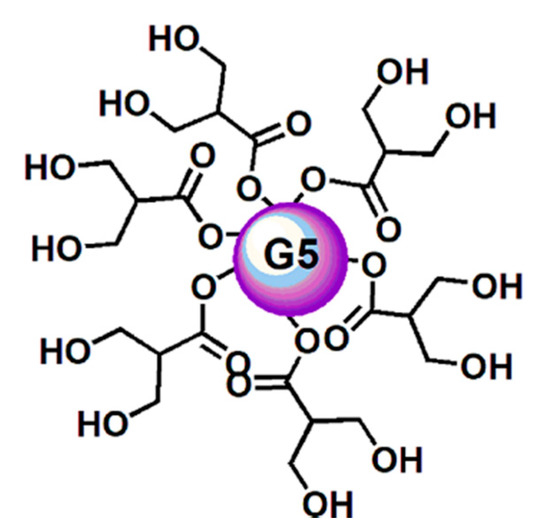
Figure 2. Simplified and intuitive representation of the structure of the dendrimer (4) having in reality a fifth-generation polyester-based inner matrix based on the tree-like repetition of units of the 2,2-bis(hydroxymethyl) propionic acid and 64 peripheral hydroxyl groups. The image shows only a partial number of the real hydroxyl groups and an inner sphere to facilitate the drawing and for reasons of space. This way to represent dendrimer 4 was previously used and accepted as valid [57].
Dendrimer 4 was selected for the serie of reasons following.
It has been reported [58] that the internal construction of a dendrimer, depending on its chemical structure, is suitable for complexing both hydrophobic and hydrophilic compounds [59].
Hence, in addition to being chosen for its intrinsic cytotoxic activity, the polyester-based dendrimer 4, having hydrophilic characteristics, was considered suitable to host a hydrophilic molecule such as GA, with which it can establish hydrogen bonds.
In addition, being uncharged, it has been thought to also be well compatible with the phenyl hydrophobic portion of GA. Moreover, dendrimer 4 was considered a proper candidate carrier to entrap GA because, thanks to its high generation, it could offer more space to host drugs, thus allowing a higher loading [60].
Furthermore, the large chemical architectures, with wide surface and high molecular weight, achievable with high generation dendrimers such as 4, characteristically remain in the blood circle for longer periods [61].
Finally, dendrimer 4 was preferred because its uncharged polyester-based hydrolysable architecture matches the requirements of low toxicity and high biodegradability desirable for biomedical applications [36][43][44].
Starting from these assumptions, dendrimer 4 was used for entrapping ETO in its inner cavities, thus proving ETO with a protective shell. The dendrimeric ETO-loaded NPs (ETOD) showed an highly improved solubility in hydrophilic and biocompatible solvents such as ethanol and water (by 37–390 times). Concerning GA, by using dendrimer 4, firstly a GA-enriched dendrimer (GAD 6, Figure 3a), in which GA is covalently linked on the dendrimer surface by ester type bonds, was synthetized according to a reported procedure [54].
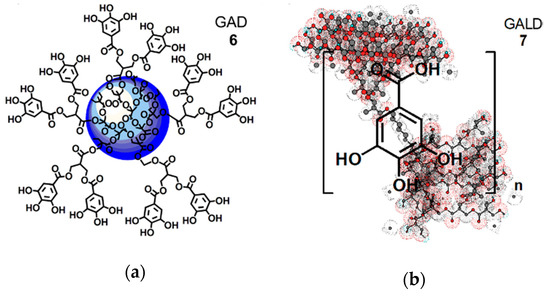
Figure 3. Simplified and intuitive representation of the GA-enriched dendrimers’ structure: (a) structure of the GAD nanoconjugate (6); (b) structure of the GALD nanocomposite (7) encompassing the tri-dimensional representation of dendrimer 4 obtained by Chem3D Pro 7.0 software with included n molecules of GA. Concerning GAD, the blue sphere represents the inner polyester-based scaffold of dendrimer 4; the five linkers, connecting two GA molecules each, are a stylized representation of the groups actually present on the surface of 4 that in reality possesses 32 analogous structures, for a total of 64 hydroxyl groups bearing 64 GA units.
Secondly, a GA-loaded dendrimer (GALD 7, Figure 3b), in which GA is physically complexed with dendrimer 4, through both inside entrapment and surface absorption, was prepared and totally characterized.
After investigations concerning the dose- and time-dependent GA effects on two NB cell lines differently sensitive to ETO [12][62], the biological activities of dendrimer 4, free GA, and GA-enriched dendrimers 6 and 7 at specific selected doses were investigated as well.
2. Experimental
2.1. Materials
FTIR and NMR spectra of commercially available ETO are observable in Figure S3 accessible in Supplementary Materials (SM) accessible on line. Details concerning the characterization of commercial GA as copies of FTIR (lab-made) and 1H and 13C NMR spectra (database of Aldrich, Darmstad, Germany) are accessible in Section S3 (Figures S13–S15) in the SM available on line. Synthetized dendron intermediates (D4BnA, D4BnOH, D5BnA, and D5ACOOH) necessary to achieve dendrimer 4, adopted as scaffold-carrier to link and entrap GA (1), were prepared according to what reported previously [51][52][53]. Their chemical structures are observable in Figure S1. Dendrimer 4 and GA-enriched dendrimer GAD 6 were prepared according to procedures previously reported [54][55][56].
Characterization data of dendrimer 4 and GAD 6, counting copies of FTIR, 1H and 13C NMR spectra are accessible in SM (Section S1, Figures S3–S8, and Table S1). The protected/activated GA derivative (GA-TBDMS-Cl) necessary to esterify peripheral hydroxyls of dendrimer 4 was prepared reproducing the synthetic pathway shown in SM (Section S1, Scheme S1) [54]. Synthetic procedures for preparing ETOD and its detailed characterization data are accessible on line at https://doi.org/10.3390/antiox9010050.
2.2. Experimental procedure
Detailed experimental procedures are available in Section 2 of the original article available online at https://www.mdpi.com/2079-4991/10/6/1243/s1 and in Section 2 of the paper accessible on line at https://doi.org/10.3390/antiox9010050.
3. Results and Discussion
3.1. Preparation of ETO-Dendrimer Nanoformulation (ETOD)
A detailed discussion concerning the physicochemical properties of ETOD are accesible on line at https://doi.org/10.3390/antiox9010050, while a discussion on its bioactivity on sensitive NB cells (HTLA-230) is reported in the following sections of the present entry.
3.2. Preparation of GA-Dendrimer Nanoformulations GAD (6) and GALD (7)
Dendrimer NPs have proved to be particularly efficient in improving drugs therapeutic efficacy at lower dosages by behaving as “excipients” or enhancers of permeability, by altering the barrier properties of the intestinal epithelium, thus enhancing the permeation ability of the transported drug [61].
Following this current and successful trend, GA was previously formulated in NPs, by its covalent bond to a fifth-generation, biodegradable dendrimer matrix (4) obtaining a peripherally GA-decorated dendrimer (GAD), with appealing results from several points of view [54][55][56].
In the present study, GAD was prepared according to the above cited procedure , and, in addition, novel GA dendrimer NPs were achieved by physical complexation of GA with the architecture of dendrimer 4. A drug delivery system of GA (GALD, 7) was realized, in which, differently from GAD, GA units are not chemically linked on the surface, but are located inside the dendrimer matrix cavities or absorbed onto its surface, where they are withheld mainly by hydrogen bond interactions.
Concerning GAD, both the structure of dendrimer scaffold and of GA have been chemically modified by the mutual covalent bond. Differently, in GALD the structure of GA and dendrimer 4 were not modified during the synthetic pathway.
3.3. Chemistry
Dendrimer 4, herein adopted as nanocarrier to complex GA, and the GA-enriched dendrimer GAD (6) were prepared as previously described .
Concerning the novel GA delivery system (GALD, 7), it has been achieved by dissolving dendrimer 4 in MeOH and by subjecting it to vigorously stir in the presence of a strong excess of GA (42.8 equiv.) for 48 h at room temperature in the dark (Scheme 1).
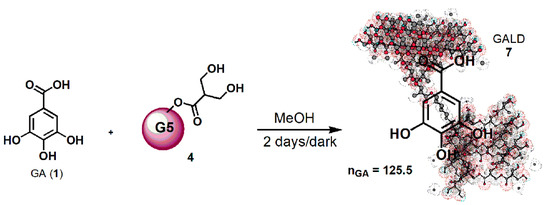
Scheme 1. Synthetic procedure for entrapping GA in dendrimer 4 (GALD 7).
Since, as reported in the literature and Merck Index 2001, GA can be isolated by recrystallization from methanol, it was thought to purify the crude product, by removing the not entrapped free GA, through its precipitation from the final clear methanol solution. Briefly, the solution was concentrated until it became cloudy and a white solid started to separate. The precipitation was completed by placing the fine suspension at 4 °C overnight, and the obtained solid was recovered from the GALD solution by performing a centrifugation at 3500 rpm for 15 min.
GALD 7, having the intuitive structure shown in Scheme 1, was achieved as off-white glassy solid (188.2 mg) and was stored on P2O5 in a dryer at r.t.
In regard to Scheme 1 and Figure 3b, the 3D structure of the dendrimer 4 was gained, using the 3D Chem Draw software (Chem 3D Pro 7.0) and the mode of representing the complex host-molecule/dendrimer herein used, was accepted as valid in a recent publication, where dendrimer 4 was employed to complex ETO [57].
3.4. Physicochemical Characterization of Dendrimer 4 and GA-Enriched Dendrimers 6 and 7
The physicochemical characterization of dendrimer 4, not including the DLS analysis, and the complete characterization of GAD 6 have been previously reported , and the salient data are provided in SM in Figures S2,S6–S8 and Table S1. Additional experiments to determine particle size, Z-potential, and the respective PDI value of dendrimer 4, lacking in the previous study , were performed and the results are reported in Section 3.2.9.
GALD 7 was instead qualitatively characterized, by performing the colorimetric FeCl3 test for detection of phenol’s presence and by Fourier Transform Infrared (FTIR) spectroscopy.
Data obtained from FTIR spectroscopy were also handled by performing Principal Component Analysis (PCA). Finally, Nuclear Magnetic Resonance (NMR) and UV–Vis analysis were carried out. UV–Vis spectrophotometric analysis associated to the Folin–Ciocalteu method was selected to evaluate the GA concentration in GALD 7 and to determine the drug loading (DL%) and the entrapment efficiency (EE%) of GALD.
In addition, UV–Vis analysis was used to investigate the GA release profile while GALD’s particles were examined by Dynamic Light Scattering (DLS) analysis.
3.5. Colorimetric FeCl3 Essay
To confirm the success of the complexation reaction, the FeCl3 test for the detection of phenols was performed by comparing the coloration obtained on an ethanol solution of GALD (Figure S16a, Section S4 in SM) with that of a GA ethanol solution.
As shown in Figure S16b (Section S4 in SM), the strong dark blue color of the GALD solution was completely analogous to that found in the GA solution.
3.6. FTIR Characterization
An exemplificative copy of the FTIR spectrum of GALD is shown in SM Figure S17 (Section S4).
The FTIR spectra of dendrimer 4 and GA were acquired in the same conditions, for allowing the comparison between the spectra of the original components (4 and GA) and that one of the complex (GALD). Copies of these spectra are available in SM in Figure S3 (Section S1) and Figure S13 (Section S3). FTIR analysis confirmed the success of the reaction of complexation of GA with dendrimer 4.
In the FTIR spectrum of GA herein acquired, available in SM in Figure S13, Section S3, bands detected at 3496 and 3228 cm−1 (OH groups), at 3061 cm−1 (stretching of C–H in aromatics), at 1688 cm−1 (conjugated carboxyl absorption), and at 1610 cm−1 (C=C stretching) were considered as significant.
As shown in Figure 4 in the FTIR spectrum of GALD 7, several bands belonging to GA and not observable in the spectrum of 4 are instead well visible, thus confirming the presence of GA in the cavities of hosting dendrimer 4.
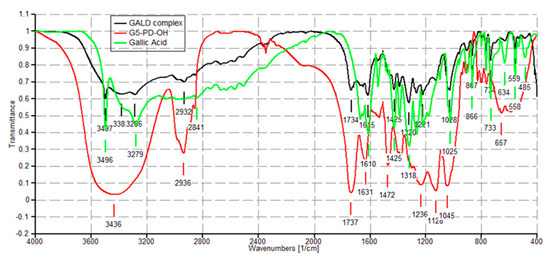
Figure 4. FTIR spectra of dendrimer 4 (red), GA (green) and GALD 7 (black).
3.7. NMR Characterization
The successful complexation of GA with dendrimer 4 was further validated by 1H NMR spectroscopy. Since GA is a tetra-substituted phenyl derivative, in the 1H NMR spectrum, it presents a single signal in the aromatic protons zone (7–8 ppm) belonging to the two equivalent protons in ortho position to the carboxyl group. Differently, dendrimer 4, adopted to entrap GA, does not have signals in this region of the spectrum because it does not encompass aromatic rings in its structure, as obserbable in SM in Figure S4. The 1H NMR spectrum of GALD 7 (Figure 5) presents both signals in the region 0–5 ppm, corresponding to the signals belonging to dendrimer 4 and a singlet signal at 7.15 ppm, matching the signal belonging to GA.
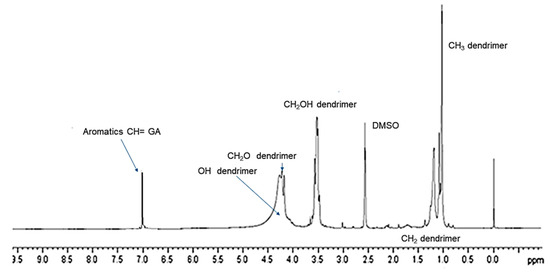
Figure 5. 1H NMR (DMSO-d6, 300 MHz) spectrum of GALD 7.
3.8. PCA on FTIR Spectral Data
Concerning the FTIR prediction of chemical composition of GA-enriched formulation 7, more reliable information was achieved, by performing the PCA on data from FTIR spectra of dendrimer 4, GA and GALD.
Briefly, PCA is able to evidence similarities or differences among the samples under study by clustering or separating them within a square of two components identified for being Information carried out by PCs is expressed in terms of percentage of explained variance. By definition, PC1 has the largest percent explained variance, followed by PC2, PC3, etc. .
In this regard, the data of nine spectra obtained by the FTIR analysis made in triplicate on dendrimer 4, GALD 7, and GA, were organized in a large matrix of variables, which were processed by PCA.
The results are reported as bi-plot in SM in Figure S21 (Section S6). The wanted information was unequivocally observable on PC2.
In this regard, by observing the relative positions of dendrimer 4, GALD 7 and GA 1 on PC2, it appears that 7 is located in the first left square as GA, while the empty dendrimer 4 is located in a different square. Consequently, GALD, in terms of both location in PCA results and chemical composition, outcomes much closer to GA rather than to dendrimer 4, suggesting that in the chemical structure of GALD 7 there is an unequivocal contribution of GA. By extrapolating the vectors up to intercept the PC2 axis, the GA loading can be estimated around 69%, i.e., higher than the expected 50%. The so high DL%, mathematically not allowed by the amounts of material employed in the complexation reaction, was confirmed by UV determination and relative explanations are discussed in the relative section.
3.9. UV–Vis Spectrophotometric Analysis of GALD
The presence of GA in GALD formulation was confirmed also by UV–Vis spectrophotometric analysis. The UV–Vis spectrum of free GA in methanol shows absorbance maxima at λ = 218 and 273 nm (Figure 6, yellow plot), while the UV–Vis spectrum of empty dendrimer 4 complexing GA, in methanol, shows no peck of absorbance in the λ range considered (not presented results).
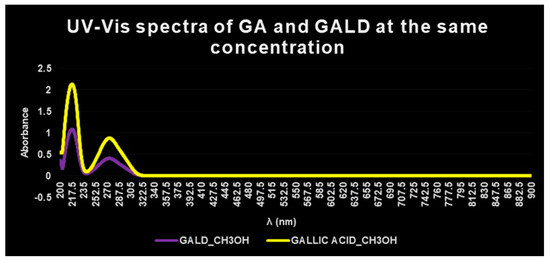
Figure 6. UV–Vis spectra of GALD 7 (violet plot) and GA (yellow plot) in methanol solution and equal concentrations.
The UV–Vis spectrum of GALD, acquired in the same solvent and at the same concentration, after dissolution and release of GA from the dendrimer scaffold, showed two absorption peaks, obviously less intense, at the same λ = 218 and 273 nm (Figure 6, violet plot).
In a study concerning GA complexes with iron[63], it was reported that, after complex formation, a bathochromic shift of the absorbance maxima of GA was observed, probably provoked by GA structural changes and/or by partial oxidation of GA caused by iron action.
Since in this case, after complex formation between GA and dendrimer 4, no bathochromic shift in λmax values was observed, it can be asserted that no structural change or oxidation processes have been occurred.
3.10. GA Content Determination
For the estimation of polyphenols such as GA, the most reported method is the well-known colorimetric Folin–Ciocalteu method based on the UV determination of the colored compounds deriving from oxidation of phenols, by the aqueous solution of phosphomolybdate and phosphotungstate (Folin–Ciocalteu reagent).
Drug Loading (DL%) was found to be 74.1% w/w.
Once known DL%, it was possible to compute both the Molecular Weight (MW) and the Encapsulation Efficiency (EE%) of GALD.
Both EE%, DL% and estimated MW of GALD resulted very high and according to what is reported , a high MW characteristically extends retention time in systemic circle and affects positively the bio-efficiency of delivering nanodevices.
3.11. In Vitro GA Release Profile from GALD
The in vitro release profile of GA from GALD 7 NPs into phosphate buffer saline solution at 37 °C and pH 7.4, mimicking human blood conditions, evaluated also to predict the GALD in vivo stability and its half-life, in reported Figure 7 as Cumulative GA release percentage in function of time. No action to stabilize GALD 7 or to influence GA release, as well as the polyester construction of dendrimer degradation, was performed.
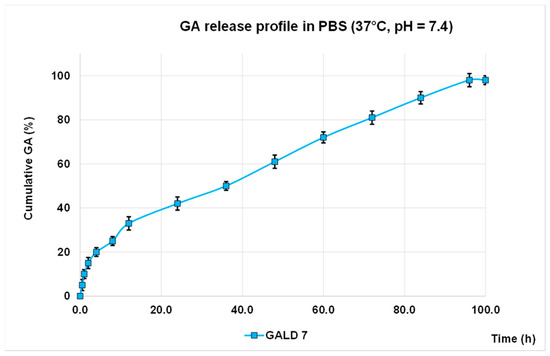
Figure 7. GA release profile in PBS, at 37 °C and pH = 7.4 expressed as cumulative percentage of release
As shown in Figure 8, an initial slight burst release of GA can be observed and only 42% of GA was released in 24 h.
The significantly softened burst release asserts a higher stability of GALD if compared to other reported cases[64][65][66] and this is a great improvement, in terms of minor systemic toxicity, over most micellar carriers, such as PEGylated PLGA-based NPs that generally have a high initial drug release [6564][66].
The secondary slow and protracted release phase observed for GALD can be explained hypothesizing a degradation-controlled release and a slow degradation rate of the polyester-based scaffold of the dendrimer carrier at pH = 7.4 and at 37 °C, as reported [6765].
The rather total GA release (98%) achieved suggests a complete disintegration of the carrier into small monomer compounds as previously described [6765], whcih can more readily be removed from the body, allowing to minimize long-term side effects.
Finally, the initial faster release of GA, followed by a secondary slow and protracted release phase support the above-postulated presence of a part of GA associated at the surface and another part included inside of dendrimer.
3.12. Kinetics Release of GA from GALD Nanocomposite
The kinetic release of GA from the GALD nanocomposite was fitted using a number of different kinetic models, such as zero-order, first-order, Higuchi, Hixoson–Crowel, and Korsmeyer–Peppas models (Figure 8).
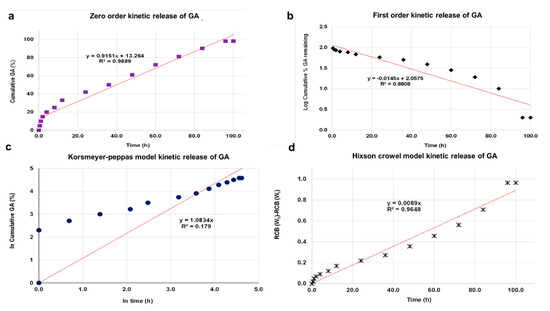
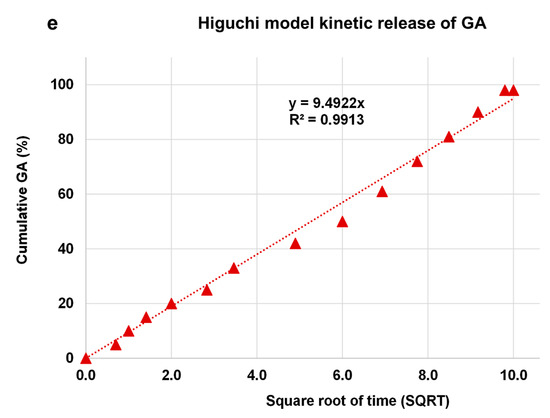
Figure 8. Fitting the data of GA release from GALD nanocomposite at pH 7.4 for kinetic models: (a) zero-order model; (b) first-order model; (c) Korsmeyer–Peppas model; (d) Hixson–Crowel model and (e) Higuchi model.
The results in Figure 8 establish that the release of GA from GALD 7 fits the Higuchi kinetic Model (Figure 9e).
3.13. Particle Size, Z-Potential and PDI of Dendrimer 4 and GALD
Particle sizes of dendrimer 4 and GALD were determined by DLS analysis and were expressed as Z-AVE size (nm).
Dendrimer 4 particles showed an average size of 45 nm with a satisfying PDI of 0.2, which does not denote possible formation of aggregates, confirmed by a sufficiently high Z-potential negative, assuring stability for dendrimer 4 aqueous solutions. On the other hand, NPs bearing too high surface charges, either positive or negative, attract easier macrophages.
Consequently, a Z-potential around 20 mV, as in the present case, may be promising for avoiding opsonins adsorption and subsequent clearance from the body by phagocytic cells. In addition, a Z-potential negative, probably due to the preferential absorption of hydroxyl ions on the uncharged surface of the dendrimer, hampers the binding with plasma proteins, which in turn may impede or reduce cellular up-take.
