Fyn is a non-receptor or cytoplasmatic tyrosine kinase (TK) belonging to the Src family kinases (SFKs) involved in multiple transduction pathways in the central nervous system (CNS) including synaptic transmission, myelination, axon guidance, and oligodendrocyte formation. Almost one hundred years after the original description of Fyn, this protein continues to attract extreme interest because of its multiplicity of actions in the molecular signaling pathways underlying neurodevelopmental as well as neuropathologic events. Fyn is a common factor in healthy and diseased brains that targets different proteins and shapes different transduction signals according to the neurological conditions. In particular, Fyn mediates signaling pathways involved in neuronal differentiation and plasticity that have been subjected to considerable attention lately, opening the fascinating scenario to target Fyn TK for the development of potential therapeutic interventions for the treatment of CNS injuries and certain neurodegenerative disorders like Alzheimer's disease.
Fyn is a non-receptor or cytoplasmatic tyrosine kinase (TK) belonging to the Src family kinases (SFKs) involved in multiple transduction pathways in the central nervous system (CNS) including synaptic transmission, myelination, axon guidance, and oligodendrocyte formation. Almost one hundred years after the original description of Fyn, this protein continues to attract extreme interest because of its multiplicity of actions in the molecular signaling pathways underlying neurodevelopmental as well as neuropathologic events. Fyn is a common factor in healthy and diseased brains that targets different proteins and shapes different transduction signals according to the neurological conditions. In particular, Fyn mediates signaling pathways involved in neuronal differentiation and plasticity that have been subjected to considerable attention lately, opening the fascinating scenario to target Fyn TK for the development of potential therapeutic interventions for the treatment of CNS injuries and certain neurodegenerative disorders like Alzheimer's disease.
- Fyn
- Tyrosine Kinase
- Neurodegeneration
- Src-Family kinases
Definition
Fyn is a non-receptor or cytoplasmatic tyrosine kinase belonging to the Src family kinases (SFKs) consisting of 11 members (Blk, Brk, Fgr, Frk, Hck, Lck, Lyn, c-Src, Srm, and c-Yes) in humans [1] .
1. Introduction
Fyn is primarily involved in several transduction pathways in the central nervous system (CNS) including myelination, axon guidance, and oligodendrocytes formation. Indeed, when disrupted these pathways, Fyn can contribute to the development of severe brain pathologies, such as Alzheimer’s disease (AD) and multiple sclerosis (MS)[2][3][4].
In the peripheral immune system, Fyn plays an important role in the regulation and functions of T-cell and B-cell receptor signaling and in the differentiation of natural killer cells [1][5][6]. A number of additional biological functions in which Fyn activity is involved has been extensively reported and includes growth factor and cytokine receptor signaling, ion channel function, platelet activation, fertilization, entry into mitosis [7][8].
On the other side, Fyn upregulation and genetic alterations have been associated with some malignancies as well as to several neuronal dysfunctions [4][9].
2. Fyn Structure
Fyn is 59kDa non-receptor protein tyrosine-kinase (TK) member of Src family [1][10] comprising 537 amino acids encoded by the Fyn gene, located on chromosome 6q21 [11]. Three isoforms of Fyn have been shown to arise via alternative splicing of exon 7[10]: the isoform 1 (Fyn[B], “canonical sequence”) is the first identified; the isoform 2 (Fyn[T]) that tends to be expressed in T-cells and differs from the isoform 1 in the linker region between the SH2 and the SH1 domain[7]; the isoform 3 has been found in the blood cells and differs from the isoform type 1 as missing the sequence 233-287[11]. FynT contains exon 7B (159 bp) and is expressed in thymocytes, splenocytes, and some hematolymphoid cell lines, while FynB (168 bp) includes exon 7A and it is more ubiquitous in its expression, although it accumulates principally in the brain [10].
Fyn, similarly, to the other Src members, is composed of several functional parts connected in a single protein chain (Figure 1). Beside the catalytic domain (SH1), Fyn contains two small, mutually unrelated, non-catalytic domains called SH2 and SH3 (Src-homology regions 2 and 3), of about 100 and 60 amino acids, respectively. SH2 and SH3 domains interact with other proteins, and these interactions regulate the TK activity. [9]
Fyn exists in two conformations: active and inactive. Fyn activation (as for the other Src members) depends on the ligand binding to the SH2 and/or SH3 domains and on the phosphorylation/dephosphorylation of two critical tyrosine residues, Tyr528 in the isoform 2, FynT, (corresponding to Tyr531 in isoform 1, FynB) and Tyr417 (the activating Tyr residue) of the isoform 2, FynT, (corresponding to Tyr420 in isoform 1, FynB) [9][5].
Multiple regulator mechanisms have been proposed leading to Fyn activation. Indeed, the dynamic control of Tyr420 and Tyr531 phosphorylation in the brain provides an important level of regulation of Fyn activity and its ability to interact with other proteins. When Tyr531 is not phosphorylated, Fyn adopts an “open” conformation that allows the interaction of the catalytic domain and the SH2 domain with their specific substrates (Figure 1). Conversely, when Tyr531 is phosphorylated, Fyn folds such that the tail and the SH2 domain interact to each other, preventing the two domains to bind their substrates and making Fyn inactive [5][9][12]. This suggests that increasing the local concentration of either an SH2 or an SH3 ligand would be expected to shift the equilibrium toward the “on” state. Particularly, potent activators would have binding sites for both the SH2 and SH3 domains, and consequently, decreased levels of such ligands would allow Fyn to revert to “the off state”[4]. This destabilization of SH2-SH3 interaction has been suggested as one of the most important mechanisms of Fyn activity regulation[7].
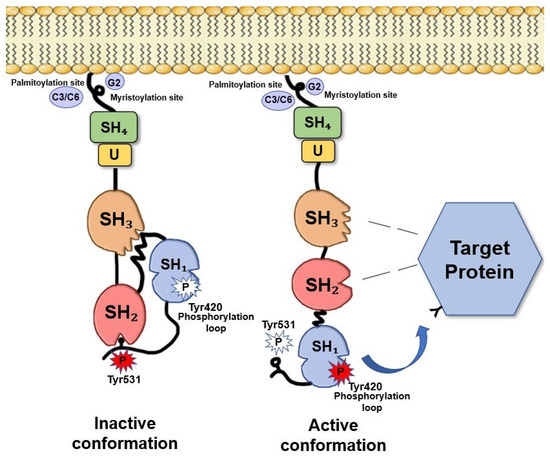
Figure 1. Fyn structure. The SH4 domain, is a membrane-targeting domain containing signals for the appropriate subcellular localization and membrane attachment of Fyn [13]. This domain consists of an extreme N-terminal ‘Met-Gly-Cys’ motif followed by a polar region [5][14]. The N-terminal Gly2 seems to be an absolute requirement for myristylation. Cys3 and Cys6, are both palmitoylated via a thioester linkage [15], thus, allowing Fyn anchors to the plasma membrane [16]. Between the SH4 and the SH3 motifs, there is a short region called “the unique region” that it is likely to be required for the subcellular localization of Fyn[17]. The SH3 motif constitutes a small domain of 50 amino acids containing a consensus sequence XPXXPPPXXP [18] that allows the binding to amino acid sequences rich in proline residues [19]. The SH2 domain consists of about 100 amino acids and appears to facilitate the binding to phosphotyrosine residues and to hydrophobic sequences within the cytoplasmic tails of growth factor receptors (i.e., PDGF-R, CSF-1 R)[20]. The catalytic domain SH1 is responsible of TK specific activity. This domain is highly conserved among Src family members [21]. The kinase domain, that catalyses the transfer of the terminal ATP phosphate group to a tyrosine residue of a target protein, shows a typical bilobed structure consisting in a small N-terminal lobe, involved in the binding with ATP, and a larger C-terminal lobe, where an activation loop (A-loop) is present. The conserved Tyr420, crucial for Src activity, is included in this domain [22]. However, the major site of phosphorylation and regulation of Fyn TK activity is the short negative regulatory tail where Tyr531 is located [21]. Adapted from Krämer-Albers et al. (2011)[23].
References
- Src-related protein tyrosine kinases and their surface receptors. . Science Direct. Retrieved 2020-7-3
- The Y682ENPTY687 motif of APP: Progress and insights toward a targeted therapy for Alzheimer’s disease patients . Science Direct. Retrieved 2020-7-3
- Remyelination in the CNS: from biology to therapy . Nature Neuroscience. Retrieved 2020-7-3
- Fyn Kinase in Brain Diseases and Cancer: The Search for Inhibitors . Bentham Science. Retrieved 2020-7-3
- Fyn, a Src family tyrosine kinase . Science Direct. Retrieved 2020-7-3
- Fyn, a novel molecular target in cancer . ACS Journals. Retrieved 2020-7-3
- CELLULAR FUNCTIONS REGULATED BY SRC FAMILY KINASES . Annual Review. Retrieved 2020-7-3
- Structures of Src-family tyrosine kinases . Science Direct. Retrieved 2020-7-3
- Fyn in Neurodevelopment and Ischemic Brain Injury . Developmental Neuroscience. Retrieved 2020-7-3
- Expression of a Novel Form of the Fyn Proto-Oncogene in Hematopoietic Cells . National Library of Medicine. Retrieved 2020-7-3
- Identification of an alternatively spliced isoform of the fyn tyrosine kinase . Science Direct. Retrieved 2020-7-3
- Crosstalk between the catalytic and regulatory domains allows bidirectional regulation of Src . Nature Structural and Molecular Biology. Retrieved 2020-7-3
- The first seven amino acids encoded by the v-src oncogene act as a myristylation signal: lysine 7 is a critical determinant. . American Society of Microbiology. Retrieved 2020-7-3
- Interaction of tyrosine kinase oncoproteins with cellular membranes . Science Direct. Retrieved 2020-7-3
- Determinants of membrane association in the SH4 domain of Fyn: Roles of N-terminus myristoylation and side-chain thioacylation . Science Direct. Retrieved 2020-7-3
- Myristoylation and Membrane Binding Regulate c-Src Stability and Kinase Activity . American Society of Microbiology. Retrieved 2020-7-3
- SH2 and SH3 domains: elements that control interactions of cytoplasmic signaling proteins . Science. Retrieved 2020-7-3
- Signalling through SH2 and SH3 domains . Science Direct. Retrieved 2020-7-3
- Modular Binding Domains in Signal Transduction Proteins . National Library of Medicine. Retrieved 2020-7-3
- Protein modules and signalling networks . Nature. Retrieved 2020-7-3
- Regulation, substrates and functions of src . Science Direct. Retrieved 2020-7-3
- An Allosteric Cross-Talk Between the Activation Loop and the ATP Binding Site Regulates the Activation of Src Kinase . Scientific Reports. Retrieved 2020-7-3
- From axon–glial signalling to myelination: the integrating role of oligodendroglial Fyn kinase . Springer Link. Retrieved 2020-7-3
