Peptidoglycan recognition proteins (PGRPs) are ubiquitous among animals and play pivotal functions in insect immunity. Non-catalytic PGRPs are involved in the activation of immune pathways by binding to the peptidoglycan (PGN), whereas amidase PGRPs are capable of cleaving the PGN into non-immunogenic compounds. Drosophila PGRP-LB belongs to the amidase PGRPs and downregulates the immune deficiency (IMD) pathway by cleaving meso-2,6-diaminopimelic (meso-DAP or DAP)-type PGN. While the recognition process is well analyzed for the non-catalytic PGRPs, little is known about the enzymatic mechanism for the amidase PGRPs, despite their essential function in immune homeostasis. Here, we analyzed the specific activity of different isoforms of Drosophila PGRP-LB towards various PGN substrates to understand their specificity and role in Drosophila immunity.
Note: The following contents are extract from your paper. The entry will be online only after author check and submit it.
1. Introduction
Peptidoglycan (PGN) is a major bacterial cell wall component that allows the conservation of the cell shape and prevents the bursting of the bacteria
[1,2][1][2]. PGN is a polymer composed of linear glycan strands that are cross-linked by short peptides. The glycan strand is made of alternating β-1,4-connected N-acetylglucosamine (GlcNAc) and N-acetylmuramic acid (MurNAc) residues. The peptide stem varies among species but is usually a pentapeptide and contains L- and D-amino acids. The third residue can be either a
meso-2,6-diaminopimelic acid (
meso-DAP or DAP) for Gram-negative bacteria and Gram-positive bacilli or a L-lysine (L-Lys) for other Gram-positive bacteria
[1,2][1][2]. PGN, and its associated muropeptides like the tracheal cytotoxin (TCT), belong to the microbial-associated molecular patterns (MAMPs), which interact with the host pattern recognition receptors (PRRs) and activate the host innate immune system
[3,4,5][3][4][5]. Some of these PRRs are peptidoglycan recognition proteins (PGRPs) that are ubiquitous in most animals and play a pivotal role in the innate immune system.
The first PGRP was described in the silkworm
Bombyx mori [6]; further studies showed that PGRPs are widespread among invertebrates and vertebrates. However, this gene family has not been found in plants or in lower metazoan, such as nematodes, so far
[7]. Insect PGRPs were shown to be more diverse when compared to other species
[7]. The fruit fly
Drosophila melanogaster genome contains 13 PGRP genes encoding over 20 different proteins
[8,9][8][9] that are involved in triggering the two major antibacterial immune signaling pathways: the immune deficiency (IMD) and Toll pathways. Distinct PGRPs are specific to DAP-type or Lys-type PGN, leading to the activation of IMD or Toll pathways, respectively. Both pathways result in the synthesis and secretion of antimicrobial peptides (AMPs) that are directed against microbial intruders
[10]. In
Drosophila, PGRP-LC and PGRP-LE recognize DAP-type PGN and activate the IMD pathway
[11], while PGRP-SA recognizes Lys-type PGN and activates the Toll pathway
[12].
Other PGRPs, including
Drosophila PGRP-LB
[13] and PGRP-SB1
[14], belong to the N-acetylmuramoyl-L-alanine amidase and cleave PGN into non-immunogenic compounds. Amidase PGRPs are also found in mammals, for example, the human PGLYRP2
[15].
Drosophila PGRP-LB is specific to the DAP-type PGN. Its expression is regulated at the transcriptional level by the IMD pathway. This IMD-regulated expression of PGRP-LB in the gut avoids a constitutive local immune response to microbiota bacteria, and creates a threshold of infection for a systemic immune response activation
[13,16][13][16]. The
Drosophila PGRP-LB gene encodes three isoforms, two cytosolic (PGRP-LB
PA and PGRP-LB
PD) and one secreted (PGRP-LB
PC)
[17]. The secreted isoform PGRP-LB
PC and cytoplasmic isoform PGRP-LB
PA are the same protein (PGRP-LB
PA/PC), and following the signal peptide of PGRP-LB
PC, they share the same sequence (A). The second cytoplasmic isoform, PGRP-LB
PD, displays a longer N-terminal than PGRP-LB
PA/PC with no homology or known function (A). It was proposed that PGRP-LB
PC cleaves the PGN in the gut lumen, avoiding a constitutive and systemic immune response to gut microbiota, while PGRP-LB
PA and PGRP-LB
PD have a redundant role in degrading PGN in enterocytes
[17]. In insects coevolving with intracellular symbiotic bacteria (endosymbionts), the PGRP-LB protein was shown to downregulate the immune response against endosymbionts in the tsetse fly
[18[18][19],
19], and to maintain host homeostasis by cleaving endosymbiont TCT in the cereal weevils
[20].
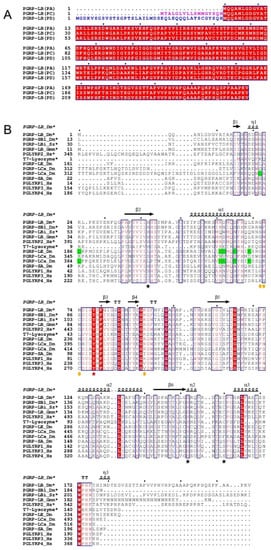
Figure 1. (
A) Multiple sequence alignment of different isoforms of
Drosophila melanogaster PGRP-LB. The signal peptide of PGRP-LB
PC is highlighted in pink while the N-terminal part of PGRP-LB
PD with no homology is highlighted in blue. The fully conserved residues are framed and highlighted in red. (
B) Multiple sequence alignment of catalytic and non-catalytic PGRPs. The residues indicated by black stars are responsible for the Zn
2+ interaction in
Drosophila PGRP-LB (H42, H152 and C160), the residue indicated by red star makes a fourth coordination with Zn
2+ through a water molecule in
Drosophila PGRP-LB (Y78). Orange circles indicate the residues responsible for the DAP/Lys specificity. Residues highlighted in green indicate residues responsible for the interaction with the sugar moiety of TCT at the dimer interface in complexed structure PGRP-LC and PGRP-LE. The conserved residues are framed and highlighted in red (fully conserved) or written in red (partially conserved). Only the
Drosophila PGRP-LB
PA/PC isoform is represented in this alignment. Dm =
Drosophila melanogaster, Hs =
Homo sapiens, Sz =
Sitophilus zeamais and Gmm =
Glossina morsitans morsitans. * for the catalytic proteins. Figures were made using ESPript3.0
[31][21].
Structural studies determined that amidase and non-catalytic PGRPs share the same structural domain involved in PGN recognition
[21,22,23,24,25,26,27,28,29][22][23][24][25][26][27][28][29][30]. The structure of the PGRP domain comprises three α-helices and one central β-sheet composed of six β-strands, similar to the bacteriophage T7 lysozyme, a zinc-dependent amidase
[21,30][22][31]. The main difference between amidase and non-catalytic PGRPs is the presence of Zn
2+ in only amidase PGRPs, which is coordinated by two histidines and one cysteine. Previous non-catalytic PGRP structures have provided a more comprehensive view on the residues responsible for the specificity towards the two types of PGN (B). For the analysis of DAP-type specificity, the TCT has been complexed to
Drosophila PGRP-LC
[23][24] or
Drosophila PGRP-LE
[24][25]. As for the Lys-type specificity, human PGLYRP3 has been complexed to muramyl tripeptide (MTP)
[26][27] or muramyl pentapeptide (MPP)
[27][28], and glucosamyl muramyl pentapeptide (GMPP) has been complexed to human PGLYRP4
[28][29]. However, the N-acetylmuramoyl-L-alanine amidase reaction mechanism remains unclear because of the lack of an amidase PGRP structure in interaction with a PGN compound. So far, only protein structure superposition and ligand docking have been used to propose a mechanism
[26][27].
2. Drosophila PGRP-LB Isoforms Have a Similar Amidase Activity
Drosophila PGRP-LB has a specific activity of cleaving DAP-type PGN and its muropeptides into non-immunogenic compounds
[13,16][13][16]. More recently, three isoforms of this protein have been described with different tissue localization and pattern of expression
[17]. The activity of these isoforms against muropeptides with varying sugar moieties has never been characterized. We hypothesized that the longer N-terminal of PGRP-LB
PD could be responsible for giving to the enzyme a specific activity towards some substrates compared to the PGRP-LB
PA/PC isoforms. In order to precisely define the substrate specificity of the PGRP-LB
PA/PC and PGRP-LB
PD isoforms, a series of polymeric and monomeric PGN substrates were tested using an in vitro enzymatic activity assay ( and
Figure S1). Both PGRP-LB
PA/PC and PGRP-LB
PD isoforms displayed the same, and the strongest activity on the GM(anh)-tetra
DAP (also called TCT) substrate while remaining very active against other muropeptides and polymeric DAP-type PGN. Therefore, the presence of the longer N-terminal in PGRP-LB
PD did not seem to induce any specificity for a particular compound leading to any influence in activity.
Table 1. In vitro enzymatic activity of Drosophila PGRP-LB isoforms and their mutants toward different substrates.
| Specific Activity (nmol.min | −1 | .mg of Protein | −1 | ) |
| Protein |
Substrate |
| E. coli | PGN (DAP Type) |
GM(anh)-
tetra | DAP | (TCT) |
GM(anh)-
tetra | DAP |
dimer * |
GM(anh)-
tetra | DAP |
dimer ** |
GM(anh)-penta | Lys |
GM-tetra | DAP |
M(anh)-
tetra | DAP |
M-tetra | DAP |
GM(anh)-
tetra | DAP |
(No Zn | 2+ | ) |
| PGRP-LB | PA/PC |
1570 ± 40 |
, along with a multiple sequence alignment of PGRPs from different organisms, reveal the highly conserved residues of
Drosophila PGRP-LB
PA/PC histidine 42 (H42), histidine 152 (H152), tyrosine 78 (Y78) and cysteine 160 (C160) in coordination with Zn
2+ in the catalytic site (B and A). Y78 residue is invariant among amidase and even non-catalytic PGRP sequences from different organisms (B). In the T7 Lysozyme, mutation of the conserved tyrosine residue to a phenylalanine (Y46F), corresponding to Y78 in
Drosophila, results in a loss of the amidase activity
[30][31]. This mutation highlights the crucial role played by the conserved tyrosine in the enzymatic process. The H42, H152 and C160 residues in
Drosophila PGRP-LB
PA/PC sequence are conserved only in the catalytic PGRPs (B).
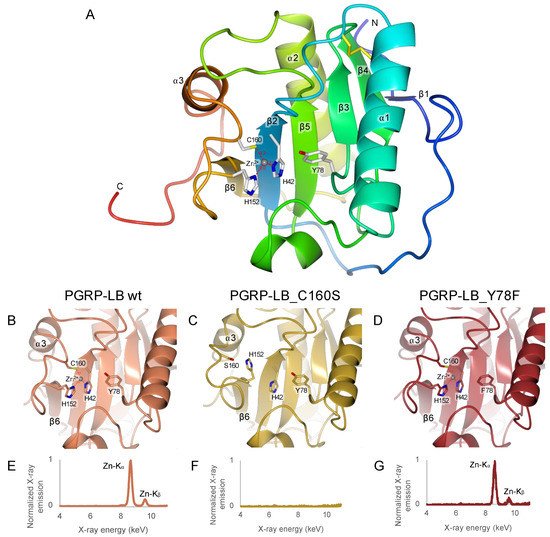
Figure 2. Analysis of Zn
2+ interaction in
Drosophila PGRP-LB
PA/PC wild-type and its mutants. (
A) Overall structure of
Drosophila PGRP-LB
PA/PC wild-type (PDB 7NSX). (
B–
D) Focus of the X-ray structures of the
Drosophila PGRP-LB
PA/PC wild-type and C160S and Y78F mutants in the Zn
2+ binding pocket. The C160S mutant lacks Zn
2+ leading to a RMSD of 2.37 Å on the loop between the sheet β6 and the helix α3. (
E–
G) Emission fluorescence spectrum on crystals of the
Drosophila PGRP-LB
PA/PC wild-type and C160S and Y78F mutants. The structure figures were made using CCP4mg
[33] and the X-ray emission fluorescence spectrum was analyzed with pyMCA
[34].
Because no other residues seemed close enough to be involved the catalytic reaction or in the stabilization of the transition state during the enzymatic reaction, we raised two alternative hypotheses: (a) one of the Zn
2+ chelating histidines is also involved in the stabilization of the transition state, or (b) only the tyrosine is involved in the amidase reaction and no stabilization of the transition state is required.
To tackle these two scenarios, we designed and analyzed four different mutations of the conserved residues in the catalytic
Drosophila PRGP-LB
PA/PC: H42A, Y78F, H152A and C160S.
4. Only Y78 Residue Is Necessary for the Amidase Reaction
We performed enzymatic activity assays on the four selected mutants, using the same compounds as for the isoforms of
Drosophila PGRP-LB (). The activity of PGRP-LB
PA/PC_C160S was firstly described as abolished
[13]. Our results show, however, that the activity of the H42A, H152A and C160S PGRP-LB
PA/PC mutants depends on the substrate type and is either significantly reduced or completely abolished. Only the Y78F mutation caused complete loss of the enzymatic activity for every compounds (). To rule out the possibility that the reduced activity of the mutants is caused by impaired recognition of the PGN substrate, we performed binding assays of H42A, Y78F, H152A and C160S mutants as well as the wild-type PGRP-LB
PA/PC with a DAP-type polymeric PGN. The introduced mutations did not affect the ability to recognize PGN, attesting that Y78 is the only residue taking part in the amidase mechanism.
5. C160S Is the Most Essential Residue for Zn2+ Chelation
The structure of the
Drosophila PGRP-LB
PA/PC wild-type was solved by X-ray crystallography (PDB 7NSX) (). The resolution was slightly improved (1.90 Å) from the previous publication (PDB 1OHT, 2.00 Å)
[21][22]. These two structures are almost identical with a calculated RMSD on the Cα of 0.90 Å and obtained in different space groups, P6
122 for 1OHT and C222
1 for 7NSX. In both structures, the Zn
2+ is coordinated by H42, H152 and C160. Yet, the water molecule involved in the interaction between the residue Y78 and Zn
2+ is replaced by a residue of the C-terminal belonging to a symmetry-related molecule, E182 and D180 respectively in 1OHT and in 7NSX. This interaction is non-physiological and can be considered as an artefact of crystallization between the C-terminal residues of a symmetry related molecule and the active site. In solution, one should expect that a water molecule would interact with both the residue Y78 and Zn
2+ like in the T7 Lysozyme structure (Y46 and Zn
2+)
[30][31].
Table 2. Data collection and structure refinement statistics.
| |
PGRP-LB | PA/PC |
PGRP-LB | PA/PC | _C160S |
PGRP-LB | PA/PC | _Y78F |
PGRP-LB | PA/PC | _Y78F + TCT |
| PDB code |
7NSX | 4525 ± 682 |
1807 ± 341 |
992 ± 671 |
167 ± 34 |
7NSY1132 ± 230 |
1456 ± 542 |
669 ± 215 |
3468 ± 624 |
| 7NSZ |
7NT0 |
PGRP-LB | PD |
660 ± 27 |
3730 ± 597 |
1055 ± 220 |
330 ± 312 |
126 ± 28 |
832 ± 279 |
1441 ± 235 |
266 ± 84 |
4908 ± 1351 |
| Data collection |
|
|
|
|
PGRP-LB | PA/PC | _H42A |
NA |
13 ± 4 |
11 ± 2 |
| Space Group |
C222 | 1 |
P1 |
P6NA |
NA |
15 ± 6 |
NA |
NA |
10 ± 4 |
| 1 | 22 |
P1 |
PGRP-LB | PA/PC | _Y78F |
NA |
| Cell parameters |
| NA |
NA |
NA |
|
NA |
NA |
NA |
NA |
NA |
| PGRP-LB | PA/PC | _H152A |
NA |
7 ± 2 |
4 ± 1 |
NA |
| a, b, c (Å) | NA |
39.87, 70.55, 112.83 |
41.02, 49.19, 52.14 |
40.50, 40.50, 338.94 |
38.59, 52.17, 55.58 | NA |
NA |
NA |
7 ± 3 |
| PGRP-LB | PA/PC | _C160S |
NA |
26 ± 5 |
28 ± 8 |
7 ± 9 |
NA |
NA |
10 ± 4 |
NA |
NA |
This extra sequence could bring a supplementary domain complementing the protein activity of PGN degradation, similar to the PGRP3 from
Branchiostoma belcheri tsingtauense, where the PGRP domain is fused with a chitin binding domain
[32]. However, this N-terminal does not show any homology and its function could not be predicted. This is why, in the rest of the article, the research will focus on the PGRP-LB
PA/PC isoform, described as PGRP-LB in previous papers
[13,21][13][22].
3. Identification of Potential Key Residues in PGRP-LB for Amidase Reaction
Structural analysis of
Drosophila PGRP-LB
[21][22] and T7 Lysozyme
[30][31]
| α, β, γ (°) |
| 90, 90, 90 |
| 71.82, 79, 66.84 |
90, 90, 120 |
92.11, 105.12, 111.51 |
| Resolution range |
56.48–1.90 (1.94–1.90) |
49.39–1.40 (1.40–1.42) |
56.55–1.30 (1.32–1.30) |
53.07–1.80 (1.84–1.80) |
| Unique reflection |
12,942 (808) |
67,209 (3218) |
42,802 (1995) |
34,852 (2038) |
| R | meas |
0.154 (1.406) |
0.043 (0.420) |
0.099 (0.476) |
0.099 (0.564) |
| Completeness (%) |
99.8 (99.2) |
96.1 (92.2) |
99.9 (98.9) |
97.7 (97.2) |
| Multiplicity |
7.9 (7.2) |
3.4 (2.6) |
31.5 (13.0) |
6.9 (6.8) |
| I/σ(I) |
8.5 (1.4) |
14.1 (2.5) |
20.8 (2.8) |
11.9 (3.3) |
| CC1/2 |
0.993 (0.562) |
0.999 (0.875) |
0.998 (0 0.955) |
0.998 (0.891) |
| Refinement |
|
|
|
|
| R | work | /R | free |
0.180/0.213 |
0.167/0.188 |
0.186/0.221 |
0.145/0.180 |
| RMSD |
|
|
|
|
| Bond length (Å) |
0.0098 |
0.0145 |
0.0143 |
0.0131 |
| Bond angle (°) |
1.597 |
1.906 |
1.904 |
1.684 |
| B factors (Å | 2 | ) |
|
|
|
|
| Protein |
29.09 |
18.05 |
14.32 |
20.44 |
| Ion |
19.64 |
- |
11.35 |
14.68 |
| Ligand |
- |
- |
- |
25.35 |
| Water |
33.55 |
30.97 |
26.97 |
30.78 |
| Ramachandran (%) |
|
|
|
|
| Favored |
94.64 |
94.22 |
94.71 |
93.96 |
| Allowed |
4.17 |
4.56 |
4.71 |
5.44 |
| Outliers |
1.19 |
1.22 |
0.59 |
0.60 |
The structures of the mutants C160S and Y78F PGRP-LB
PA/PC were solved by X-ray crystallography (respectively, PDB 7NSY and PDB 7NSZ) (). Unfortunately, we were unable to crystallize H42A and H152A PGRP-LB
PA/PC mutants. Y78F and C160S PGRP-LB
PA/PC mutant structures were elucidated to 1.30 Å and 1.40 Å resolution, respectively. Both mutants Y78F and C160S PGRP-LB
PA/PC share a similar fold with PGRP-LB
PA/PC wild type with a RMSD on Cα of 0.49 Å and 1.12 Å, respectively, but in different space groups, P6
122 for PGRP-LB
PA/PC_Y78F and P1 for PGRP-LB
PA/PC_C160S (B–D). PGRP-LB
PA/PC_Y78F also presents a crystal packing artefact where E182 from the C-terminal of a symmetry-related molecule makes a coordination with Zn
2+ (
Figure S3B). However, major structural changes were observed in PGRP-LB
PA/PC_C160S structure around the mutation. The loop between the β6 sheet and the α3 helix is drifting apart from the active site with a RMSD of 2.37 Å, calculated on the Cα from the residues 148 to 172. This deviation is due to the loss of interaction between C160 and Zn
2+, where the cysteine is replaced by a serine (C). Additionally, the presence or absence of Zn
2+ was assessed by X-ray emission fluorescence on PGRP-LB
PA/PC wild type, Y78F and C160S crystals (E–G). We showed that Zn
2+ is totally absent in PGRP-LB
PA/PC_C160S mutant as already observed in the structure resolution (C), while it is still present in PGRP-LB
PA/PC_Y78A mutant and PGRP-LB
PA/PC wild type. Noticeably, no Zn
2+ was added to the buffer during purification and crystallization steps, meaning that the Zn
2+ present in the active site of PGRP-LB
PA/PC_Y78A and PGRP-LB
PA/PC proteins comes from the expression cell system.
Finally, the addition of Zn
2+ to the reaction buffer used for activity assays () allowed a small residual amidase activity of PGRP-LB
PA/PC_C160S, whereas the absence of Zn
2+ in the buffer totally abolished the amidase activity of PGRP-LB
PA/PC_C160S, which is consistent with X-ray structural data (C,F). These data suggest that the C160 residue is needed to retain Zn
2+ in the active site but not essential for residual amidase activity if Zn
2+ is present in the media. For the H42A and H152A PGRP-LB
PA/PC mutants, they remain active even without any Zn
2+ added to the reaction buffer, meaning that Zn
2+ could be still partially present in the active site. Regarding PGRP-LB
PA/PC_Y78F, the amidase activity is also abolished without Zn
2+ in the buffer. Of note, in the wild-type PGRP-LB
PA/PC and PGRP-LB
PD even without any Zn
2+ in the media, the activity remains the same confirming the strong chelation of Zn
2+ by the protein.
6. TCT Makes Two Important Interactions with R92 and Zn2+
Initial X-ray and enzymatic analyses showed that the Y78F mutation does not destabilize the protein structure and prevents PGN substrate from being degraded. Hence, we used this mutation to study the interaction between the PGRP-LB
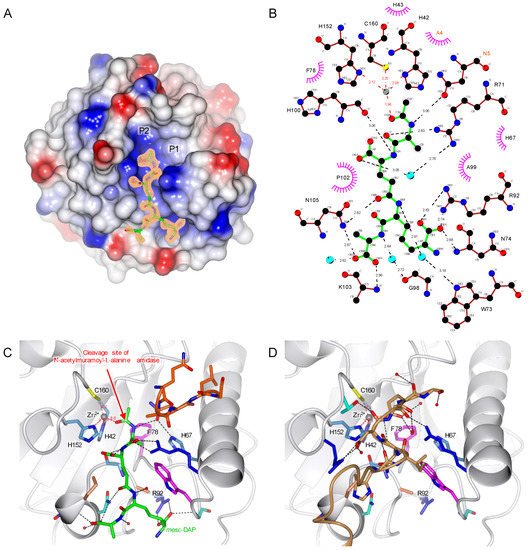
PA/PC and one of the muropeptides, the TCT with X-ray crystallography (PDB 7NT0) (). In this structure elucidated to 1.80 Å resolution, the ligand is buried in a mostly positively charged pocket. Whereas the tetra-peptide is well defined, the carbohydrate moiety of TCT is not visible in the electron density (A).
Figure 3. TCT binding to
Drosophila PGRP-LB
PA/PC. (
A) Overall structure of
Drosophila PGRP-LB
PA/PC complexed with TCT (PDB 7NT0). The surface of the protein represents its electrostatic potential, positive potential is represented in blue and negative in red. The TCT is represented in green, and the initial F
o-F
c electron density at 3σ is shown around the TCT. P1 and P2 are possible pockets for the sugar moiety. (
B) TCT hydrogen bonding network. TCT is represented in green, hydrogen bonds are represented in black dotted lines, red dotted lines show the Zn
2+ (grey ball) chelation and hydrophobic interactions in the pink circle. The residue belonging to the symmetry-related molecule are annotated in orange. (
C) Interaction of the TCT within the binding pocket. TCT is shown in green, and the protein is in ribbon representation with side chains of the TCT-interacting residues shown according to their residue property. Hydrogen bonds are represented in black dotted lines; red dotted lines show the Zn
2+ (grey ball) chelation. The symmetry related chain of 5 residues is colored in orange. The red arrow indicates the cleavage site of the amidase PGRPs (
D) TCT binding pocket in the apo protein, the symmetry-related molecule represented in brown is filling the site of the TCT. The structure figures were made using CCP4mg
[33] and the interaction diagram with LigPlot+
[35].
The specificity of the PGRP-LB
PA/PC to the PGN is ensured by the recognition of the
meso-DAP residue, interacting via hydrogen bonds with R92 (B,C). Previous studies pointed out this interaction between the DAP residue and an arginine as observed in PGRP-LE
[24][25] and PGRP-LC
[23][24], and by comparing different DAP-type PGRP sequences, the arginine residue appears well conserved (B). For the Lys-type specific PGRPs, this residue is replaced by a threonine in
Drosophila PGRP-SA
[29][30] and a valine in human PGLYRP3
[26,27][27][28] and PGLYRP4
[28][29] (B). This specificity of
Drosophila PGRP-LB
PA/PC for the DAP-type PGN is confirmed with an activity around 30 times higher for the TCT compared to the GM(anh)-penta
Lys ().
The structure obtained here also shows another interesting interaction with a fourth coordination of Zn
2+ made with the oxygen from the amide group of the TCT, which highlights the important role of Zn
2+ in the enzymatic reaction (B,C). This interaction would increase the electrophilic character of the carbonyl group of the amide function where the amidase activity is occurring.
By comparing the apo and complexed PGRP-LB
PA/PC_Y78F structures, the C-terminal symmetry-related molecule is filling the binding pocket of the peptide moiety from the PGN attesting the non-physiological nature of this interaction (C,D).
7. PGRP-LC and PGRP-LE Helix α1 Allows a Stronger Recognition of the Sugar Moiety
The absence of the electron density for the carbohydrate moiety of the TCT in the PGRP-LB
PA/PC complexed structure is most probably due to its high flexibility. There are two possible pockets that can accept the sugar moiety of the ligand (annotated P1 and P2 in A). The first pocket (P1) has a well-defined electron density corresponding to 5 residues of the N-terminal from a symmetry-related molecule (B). This rearrangement, different from the apo structure, is also probably due to the crystal packing and may not be biologically relevant. The second pocket (P2) could accommodate the sugar moiety of the TCT. The structure of PGN recognition PGRPs, PGRP-PGRP-LE
[24][25] and PGRP-LC
[23][24], in complex with TCT, have been solved previously. In these structures, the electron density is well defined throughout the entire ligand and the sugar is located in the same pocket as the pocket (P2) in the PGRP-LB
PA/PC_Y78F. We can then assume that the pocket (P2) is accommodating the sugar moiety even though no interpretable electron density is visible in the vicinity.
This PGRP-LB
PA/PC _Y78F structure complexed with TCT allows to better understand the PGN binding and multimerization event in the non-catalytic PGRPs in
Drosophila. PGRP-LC and PGRP-LE are PGN receptors involved in the activation of the IMD pathway
[11]. To activate the IMD pathway, these proteins form an amyloidal signaling complex through their RHIM motif
[36,37][36][37]. In the formation of amyloids, PGRP-LC is organized in a heterodimer
[23][24], while PGRP-LE in a homomultimer
[24][25]. The dimer interface of these PGRPs is interacting with TCT and is located at the carbohydrate moiety of the muropeptide (A,B). To better analyze the interaction of the sugar part of the TCT with the PGRP-LB, we have modelled the missing atoms in the pocket (P2) in the PGRP-LB
PA/PC_Y78F + TCT structure (C).
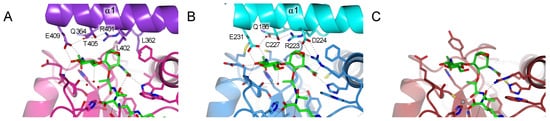
Figure 4. Interaction between carbohydrate moiety of the TCT represented in green and
Drosophila PGRPs. Only the residues from the helix α1 are responsible for the dimerization and interactions with TCT are annotated. (
A) Dimer interface of PGRP-LCx in pink and PGRP-LCa in violet (PDB 2F2L)
[23][24]. (
B) Dimer interface of PGRP-LE, the main interacting chain is in blue and the chain at the dimer interface is in cyan (PDB 2CB3)
[24][25]. (
C) Model for the interaction of the PGRP-LB
PA/PC with the sugar moiety of the TCT. The missing sugar moiety has been modelled using Maestro and Prime (Schrödinger, LLC, New York, NY, 2020) and colored in darker green. Hydrogen bonds are represented by black dotted lines; red dotted lines show the Zn
2+ (grey ball) chelation. The structure figures were made using CCP4mg
[33].
The majority of the residues from PGRP-LCa and PGRP-LE, responsible for the dimerization in the complexed structures, directed to the anhydro bond and the GlcNAc part of the TCT, are located in the helix α1 (B). In the X-ray structure of PGRP-LB
PA/PC, this dimerization involving the helix α1 was not observed. Moreover, in the model no residue was able to balance the missing hydrogen bond network stabilizing the sugar moiety explaining the absence of electron density (C).
The sugar moiety of TCT is involved in stabilizing the dimerization via a strong hydrogen bond network (A,B). A shorter sugar will make fewer interactions and will result in a weaker dimerization, which could explain why other muropeptides other than TCT can still activate the IMD pathway to a lesser extent
[3]. As for PGRP-LB, its scavenger role only needs the degradation towards all PGN substrates and therefore the dimerization to increase the specificity is not necessary.
8. Amidase PGRP Reaction Mechanism Needs the Dynamic Role of Y78
Zn
2+ is one of the most important cofactors, allowing metalloenzymes interacting with this ion to catalyze essential reactions
[38]. Here, we demonstrate the essential role played by Zn
2+ in
Drosophila PGRP-LB
PA/PC and in N-acetylmuramoyl-L-alanine amidase in general. Our findings also highlight the pivotal role and major implication of the Y78 residue in the catalytic process of PGRP-LB.
During the analysis of the structure of PGRP-LB
PA/PC_Y78F + TCT, the residue H67 seemed worthy of further investigation as it was previously suggested to be responsible for the transition-state stabilization
[21][22]. It interacts with the nitrogen of the amide group between the sugar moiety and the peptide stem through an Asn of the N-terminal brought by the crystal packing (C). In the PDB 1OHT structure, H67 forms a hydrogen bond with a water molecule, raising the question of its participation in the mechanism
[21][22]. We tested the enzymatic activity of a mutant H67A against the
E. coli polymeric PGN and the specific activity was 1041 ± 34 nmol.min
−1.mg of protein
−1, which indicates that the mutation does not change the enzyme activity and binding affinity to PGN. This result shows that H67 is not involved in the reaction mechanism.
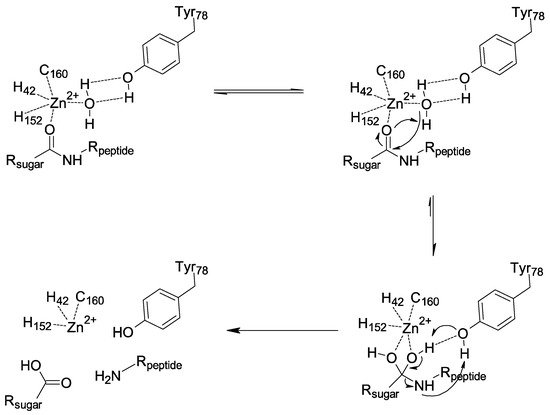
Enzymatic assays and structural data suggest that the catalytic amidase PGPRP-LB proceeds through the chemical mechanism displayed in . First, a nucleophilic attack of the water stabilized by the Y78 occurs. It is the interaction between the substrate and Zn
2+ that increases the electrophilic character of the carbonyl group of the amide function, which allows this attack. We then have a formation of a tetrahedral intermediate stabilized by Y78, leading to a concerted rearrangement assisted by Y78, which results in the formation of two products. Our analysis points to a dynamic role of this tyrosine in the amidase mechanism from substrate binding to product release, in contrast with the previous mechanism proposed, which was based only on a model without experimental data
[26][27], and did not consider enough the crucial and dynamic role of Y78. This analysis also shows how the mechanism involved with PGRP-LB differs from bacterial N-acetylmuramoyl-L-alanine amidases such as AmiD
[39] and AmiA
[40].
Figure 5. Proposed mechanism for Drosophila PGRP-LB.
In conclusion, we have shown the redundant activity of the three isoforms of Drosophila PGRP-LB, all presenting a similar activity towards the polymeric PGN and its associated muropeptides. In addition to this observation, PGRP-LB presents a wide range of activity towards various DAP-type PGN compounds. It is capable of degrading into non-immunogenic compounds both E. coli polymeric PGN and muropeptides with variation on their sugar moiety, with a preference for the TCT. This result indicates that the sugar moiety is not as important as the DAP residue in the recognition and degradation of PGN by the Drosophila PGRP-LB. Moreover, we have characterized the pivotal role of the residue Y78 in the reaction mechanism of PGRP-LB and by extension in all amidase PGRPs. The tyrosine makes a nucleophilic attack through a water molecule to the carbonyl group of the amide function destabilized by a Zn2+ interaction. It is noteworthy that the three residues responsible for the Zn2+ chelation are not involved in the amidase reaction itself. This work provides a clearer view on the N-acetylmuramoyl-L-alanine amidase activity harbored by the catalytic PGRPs and the T7 Lysozyme.