Therapeutic monoclonal antibodies (mAbs) have been the subject of widespread investigation focusing on two target-based groups, i.e., non-SARS-CoV-2 specific mAbs, that target immune system responses, and SARS-CoV-2 specific mAbs, designed to neutralize the virus protein structure.
- COVID-19 treatment
- clinical trials
- monoclonal antibodies
- non-SARS-CoV-2 specific
- SARS-CoV-2 specific
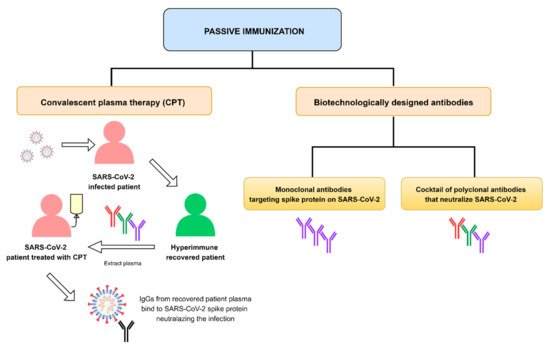
1. Introduction
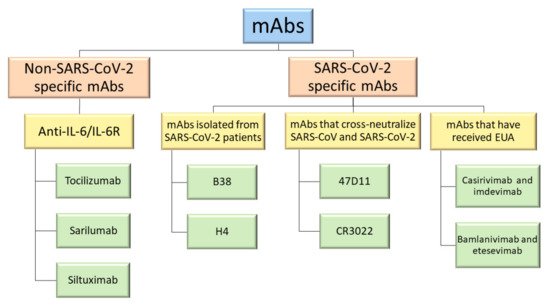
2. Non-SARS-CoV-2 Specific Monoclonal Antibodies
3. SARS-CoV-2 Specific Monoclonal Antibodies
Phylogenetic analysis has shown that SARS-CoV and SARS-CoV-2 are very similar in genetic and structural terms. This helped scientists understand the pathogenesis of COVID-19 and provided a basis for initial research. Indeed, the glycosylated spike (S) proteins from both coronaviruses have a primary amino acid sequence homology of 77.5% [7,8][7][8]. The S proteins are located on the surface of these coronaviruses and are a key component in infection. They mediate viral host cell entry by binding to the host cell receptor angiotensin-converting enzyme 2 (ACE2). The SARS-CoV-2 S protein is composed of 1273 amino acids (aa) and consists of three segments: an extracellular N-terminus, a transmembrane (TM), and a short intracellular C-terminal segment. A signal peptide is located at the N-terminus domain which consists of a few aa (1–13 residues). The rest of the protein is divided into two large regions: S1 and S2 subunits. The S1 subunit (14–685 aa) is responsible for receptor binding and consists of an N-terminal domain (NTD) and a receptor-binding domain (RBD). The S2 subunit (686–1273 aa) comprises the fusion peptide (FP), heptapeptide repeat sequence 1 (HR1), HR2, TM domain, and cytoplasm domain, and is involved in the fusion of the viral and host cell membranes and the consequent release of the viral genome into the host cell (Figure 3a). The RBD region is considered a critical target for neutralizing antibodies (nAbs) since it allows the spike protein to bind to the cell receptor ACE2. A deeper knowledge about the structure and configuration of the SARS-CoV-2 S protein can be found in references [45,46][16][17].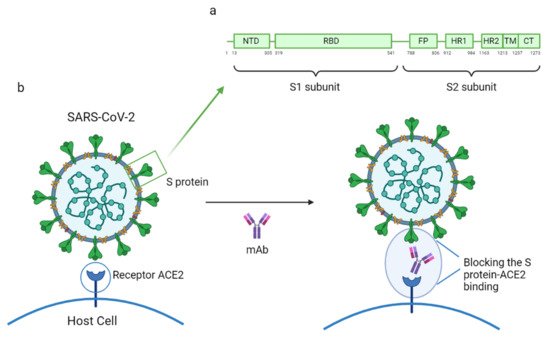
Groups of Specific mAbs | Name | Binding Site and Mechanism of Action | ||||||
|---|---|---|---|---|---|---|---|---|
MAbs isolated from SARS-CoV-2 patients | B5 | SARS-CoV-2 RBD; partial competition with ACE2 | ||||||
B38 | SARS-CoV-2 RBD; complete competition with ACE2 | |||||||
H2 | SARS-CoV-2 RBD; no competition with ACE2 | |||||||
H4 | SARS-CoV-2 RBD; complete competition with ACE2 | |||||||
EY6A | SARS-CoV-2 RBD and SARS-CoV RBD with lower affinity; site spatially separate from that of ACE2 | |||||||
MAbs that cross-neutralize SARS-CoV and SARS-CoV-2 | 47D11 | SARS-CoV-2 and SARS-CoV RBD; conserved epitope in the RBD | ||||||
CR3022 | SARS-CoV RBD and SARS-CoV-2 RBD with lower affinity; conserved epitope in the RBD. Do not neutralize SARS-CoV-2 | |||||||
MAbs that have received Emergency Use Authorization (EUA) | Bamlanivimab (LY-CoV555) | SARS-CoV-2 RBD; EUA revoked | ||||||
Casirivimab (REGN10933) and imdevimab (REGN10987) in a combined therapy | Non-overlapping epitopes of the SARS-CoV-2 RBD | |||||||
Bamlanivimab (LY-CoV555) and etesevimab (LY-CoV016) in a combined therapy | Different, but overlapping, epitopes of the SARS-CoV-2 RBD |
References
- Renn, A.; Fu, Y.; Hu, X.; Hall, M.D.; Simeonov, A. Fruitful Neutralizing Antibody Pipeline Brings Hope To Defeat SARS-Cov-2. Trends Pharmacol. Sci. 2020, 41, 815–829.
- Cantini, F.; Goletti, D.; Petrone, L.; Najafi Fard, S.; Niccoli, L.; Foti, R. Immune Therapy, or Antiviral Therapy, or Both for COVID-19: A Systematic Review. Drugs 2020, 80, 1929–1946.
- Gavriatopoulou, M.; Ntanasis-Stathopoulos, I.; Korompoki, E.; Fotiou, D.; Migkou, M.; Tzanninis, I.-G.; Psaltopoulou, T.; Kastritis, E.; Terpos, E.; Dimopoulos, M.A. Emerging treatment strategies for COVID-19 infection. Clin. Exp. Med. 2021, 21, 167–179.
- RECOVERY Collaborative Group. Effect of Hydroxychloroquine in Hospitalized Patients with Covid-19. N. Engl. J. Med. 2020, 383, 2030–2040.
- WHO Solidarity Trial Consortium. Repurposed Antiviral Drugs for Covid-19—Interim WHO Solidarity Trial Results. N. Engl. J. Med. 2021, 384, 497–511.
- Horby, P.W.; Mafham, M.; Bell, J.L.; Linsell, L.; Staplin, N.; Emberson, J.; Palfreeman, A.; Raw, J.; Elmahi, E.; Prudon, B.; et al. Lopinavir–ritonavir in patients admitted to hospital with COVID-19 (RECOVERY): A randomised, controlled, open-label, platform trial. Lancet 2020, 396, 1345–1352.
- Owji, H.; Negahdaripour, M.; Hajighahramani, N. Immunotherapeutic approaches to curtail COVID-19. Int. Immunopharmacol. 2020, 88, 106924.
- Jahanshahlu, L.; Rezaei, N. Monoclonal antibody as a potential anti-COVID-19. Biomed. Pharmacother. 2020, 129, 110337.
- An, Z. Therapeutic Monoclonal Antibodies; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2009; ISBN 9780470485408.
- Zarkali, A.; Karageorgopoulos, D.E.; Rafailidis, P.I.; Falagas, M.E. Frequency of the off-label use of monoclonal antibodies in clinical practice: A systematic review of the literature. Curr. Med. Res. Opin. 2014, 30, 471–480.
- Brechner, R.J.; Rosenfeld, P.J.; Babish, J.D.; Caplan, S. Pharmacotherapy for Neovascular Age-Related Macular Degeneration: An Analysis of the 100% 2008 Medicare Fee-For-Service Part B Claims File. Am. J. Ophthalmol. 2011, 151, 887–895.
- DeFrancesco, L. COVID-19 antibodies on trial. Nat. Biotechnol. 2020, 38, 1242–1252.
- Fajgenbaum, D.C.; June, C.H. Cytokine Storm. N. Engl. J. Med. 2020, 383, 2255–2273.
- European Medicines Agency. Treatments and Vaccines for COVID-19—European Medicines Agency. Available online: (accessed on 16 March 2021).
- Khan, F.A.; Stewart, I.; Fabbri, L.; Moss, S.; Robinson, K.; Smyth, A.R.; Jenkins, G. Systematic review and meta-analysis of anakinra, sarilumab, siltuximab and tocilizumab for COVID-19. Thorax 2021, 1–13.
- Huang, Y.; Yang, C.; Xu, X.; Xu, W.; Liu, S. Structural and functional properties of SARS-CoV-2 spike protein: Potential antivirus drug development for COVID-19. Acta Pharmacol. Sin. 2020, 41, 1141–1149.
- Valdez-Cruz, N.A.; García-Hernández, E.; Espitia, C.; Cobos-Marín, L.; Altamirano, C.; Bando-Campos, C.G.; Cofas-Vargas, L.F.; Coronado-Aceves, E.W.; González-Hernández, R.A.; Hernández-Peralta, P.; et al. Integrative overview of antibodies against SARS-CoV-2 and their possible applications in COVID-19 prophylaxis and treatment. Microb. Cell Fact. 2021, 20, 88.
- Marovich, M.; Mascola, J.R.; Cohen, M.S. Monoclonal Antibodies for Prevention and Treatment of COVID-19. JAMA 2020, 324, 131.
- Wu, Y.; Wang, F.; Shen, C.; Peng, W.; Li, D.; Zhao, C.; Li, Z.; Li, S.; Bi, Y.; Yang, Y.; et al. A noncompeting pair of human neutralizing antibodies block COVID-19 virus binding to its receptor ACE2. Science 2020, 368, 1274–1278.
