The surface-enhanced Raman scattering (SERS) technique, that uses magnetic plasmonic particles (MPPs), is an advanced SERS detection platform owing to the synergetic effects of the particles’ magnetic and plasmonic properties. As well as being an ultrasensitive and reliable SERS material, MPPs perform various functions, such as aiding in separation, drug delivery, and acting as a therapeutic material.
- Surface-Enhanced Raman Scattering (SERS)
- magnetic nanoparticles
- plasmonic nanoparticles
- detection
- drug delivery
- cancer therapy
- biological application
1. Introduction
2. Types of MPPs
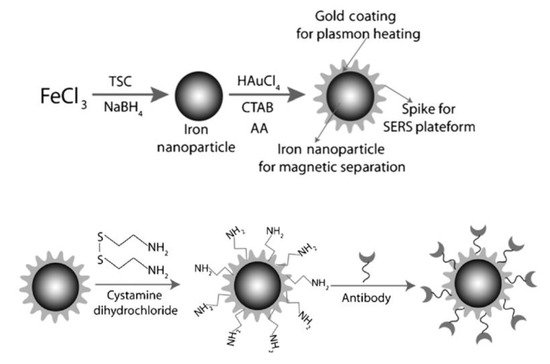
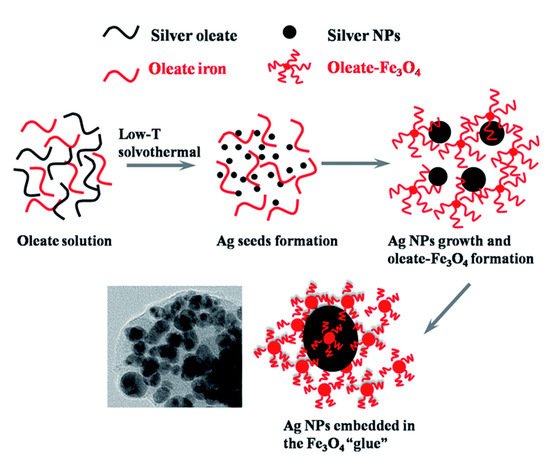
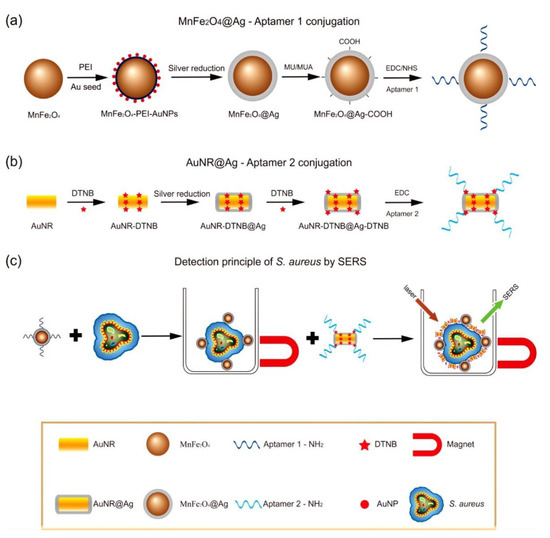
3. Bioapplication of MPPs
3.1. Detection and Separation
As SERS substrates, MPPs can detect targets with high sensitivity[22][76][77][78][79]. Additionally, as magnetic NPs, MPPs can be manipulated by an external magnetic field so that MPPs can be used to separate and enrich the target before SERS signal detection. Therefore, MPPs are often used to enhance the separation and detection of low concentration targets. These MPPs can also help isolate nucleic acids, proteins, and other small molecules in a complex biological matrix [59][80][81].
3.1.1. Nucleic Acids
Nucleic acid diagnostics, with its high sensitivity and specificity, plays a vital role in many fields, such as biology, medicine, and environmental science. Nucleic acid fragments and nucleotides can be targeted by magnetic SERS based detection. SERS enables high sensitivity and specificity, demonstrated by its ability to separate and collect targets with concentrations as low as fM[81][82][83], such as: typical Fe3O4 and Au based MPPs for adenine detection with detection limit as 0.7 µM[84]; dual MPPs including ultrabright Au- Ag core- gap-shell NPs with Raman reporter in the gap and magnetic NPs that were utilized for DNA detection with a detection limit as low as 100 aM[85]; and Fe3O4@Ag MPPs and duplex-specific nuclease signal amplification in microRNA detection with detection limit Nucleic acid diagnostics, with its high sensitivity and specificity.
Alula et al. developed MPPs containing iron and gold NPs for adenine detection [84]. A magnetic core is produced by coprecipitation and then coated by a polymer. Au NPs are then produced on the polymer-coated surface by a photochemical reduction method. The MPPs are stable in aqueous solution, disperse well in solution, and easily attach to the target adenine. Owing to their magnetic properties, the MPPs can concentrate the target to facilitate SERS imaging. Thereby, adenine can be detected at concentrations as low as 0.7 µM [84].
Ngo et al. reported an MPPs method to discriminate wild and mutant type malaria, via DNA detection and single nucleotide polymorphism[85]. Dual MPPs with magnetic NPs and Au/Ag NPs were labeled by DNA probes matching the target sequence. A hybridized sandwich of magnetic NPs-target sequence-Au/Ag NPs is formed via specific recognition of complementary DNAs, which are concentrated at the SERS detector. The limit of detection of the detection platform was approximately 100 aM.
MicroRNAs are short single stranded RNA molecules that are the primary regulatory factor in various biological pathways, such as mRNA degradation, translational inhibition, cell proliferation, differentiation, and apoptosis. They can also cause cancer[81][86][87][88][89][90][91]. Therefore, microRNAs are used as biomarkers for several diseases, including cancer. Pang et al. fabricated a Fe3O4@Ag MPPs designed for microRNA detection[81]. The Fe3O4@Ag MPPs are modified by Raman tags-DNA probes. Target microRNA are captured on the surface of Fe3O4@Ag MPPs through DNA/RNA hybridization. The SERS signal is significantly amplified by duplex specific nuclease, yielding a detection limit of 0.3 fM. The Fe3O4@Ag MPPs have been successfully applied in microRNAs’ capture, concentration, and direct quantification, overcoming disadvantages in previous microRNAs’ diagnostic tools, such as single sequence and low abundance[81]
In addition, MPPs have been employed in determining anti-cancer drug interaction with DNA [32], and for DNA methyltransferase activity[92]measurements, which uses SERS to detect DNA fragments via interaction between the fragments and plasmonic metals. These studies showed the MPPs also supported functionalization and recovery for recycling [32], and had excellent separation ability[92].
3.1.2. Protein
Like nucleic acids, proteins are key biological molecules that have a multitude of roles in organisms. There are numerous examples of proteins dispersed through an organism, including hormones, antibodies, cell-surfaced molecules, enzymes, and structural proteins. As abnormal protein quantities can lead to disease, sensitive and specific diagnosis techniques are necessary. Therefore, magnetic SERS has been applied in disease-based protein detection. Target proteins include human immunoglobulin G[20][36], prostate specific antigen[26][35][93], urinary erythropoietin[94], sepsis-specific biomarkers[95] carcinoembryonic antigen[96], thrombin, platelet derived growth factor BB, immunoglobulin E[62], and glycated hemoglobin[97]. These MPPs are designed with very specific components, such as antibodies, aptamers or chemical molecules[62][95][97]. The MPPs enable easy extraction, isolation, collection, and purification of the specific target from biological samples[62][93][94][95][96][97] and facilitate measurement at low concentrations.
3.1.3. Small Molecules
As an ultrasensitive sensor, SERS not only detects macromolecules like nucleic acids and proteins, but also single, small molecules[22]. Therefore, magnetic SERS have been applied in single molecule level diagnosis. The surfaces of the MPPs are labeled with a specific target antibody[30][98][99], aptamer[27], or chemical molecule [63]. The limit of target detection concentration can be as low as fg. mL−1 [30]. The magnetic parts act as a high-speed extractor[98] and simple isolator[99]. Additionally, the magnetic parts concentrate[63] and aggregate particles to generate hot spots, which increase the SERS intensity and thus aid sensitivity[100]. The process is used in a wide selection of applications, such as: determination of microcystin-LR in blood plasma[98]; anthrax biomarker poly-γ-D-glutamic acid in serum[99]; cotinine and benzoylecg onine in saliva[100]; thiocyanate and tetracycline in food products[27][41]; nitrite ions in environmental, biological, and food samples[63]; and N-terminal pro-brain natriuretic peptide as a biomarker for heart failure[30].
3.1.4. Cancer Diagnostic
Cancer is a group of diseases caused by abnormal growth of cells[101]. It can occur anywhere in the human body and often spreads. From the early stages of cancer, biomarkers are released[102]. If these biomarkers can be detected, then early diagnosis is possible, which aids in effective treatment. Magnetic SERS can accurately detect very low concentrations of biomarkers. In these MPPs, the plasmonic property targets the biomarkers and the magnetic property contributes to the agglomeration and the amplification of the signal. Many studies have demonstrated magnetic SERS’ usefulness in cancer marker detection with high sensitivity. MPPs are synthesized with specific biological components, including antibodies or aptamers, in order to target cancer markers or cancer cells, such as carcinoembryonic antigens[101], circulating tumor cells[102], RNA extract from cancer cells[81], breast-cancer cells, floating leukemia cells[57], and Bronchioalveolar stem cells[103]. Even at extremely low concentrations (~fM), the MPPs can separate and collect the target using an external magnetic field. The SERS signal is also amplified by the magnetic gathering[81]. Figure 4 shows an example of SERS detection of a cancer biomarker. In this case, the MPPs are formed from magnetic NiFe@Au and plasmonic Au NPs, with Raman labeling and suitable antibodies. The biomarker/MPPs combinations are separated, gathered by an external magnet, and detected by SERS amplification[101].
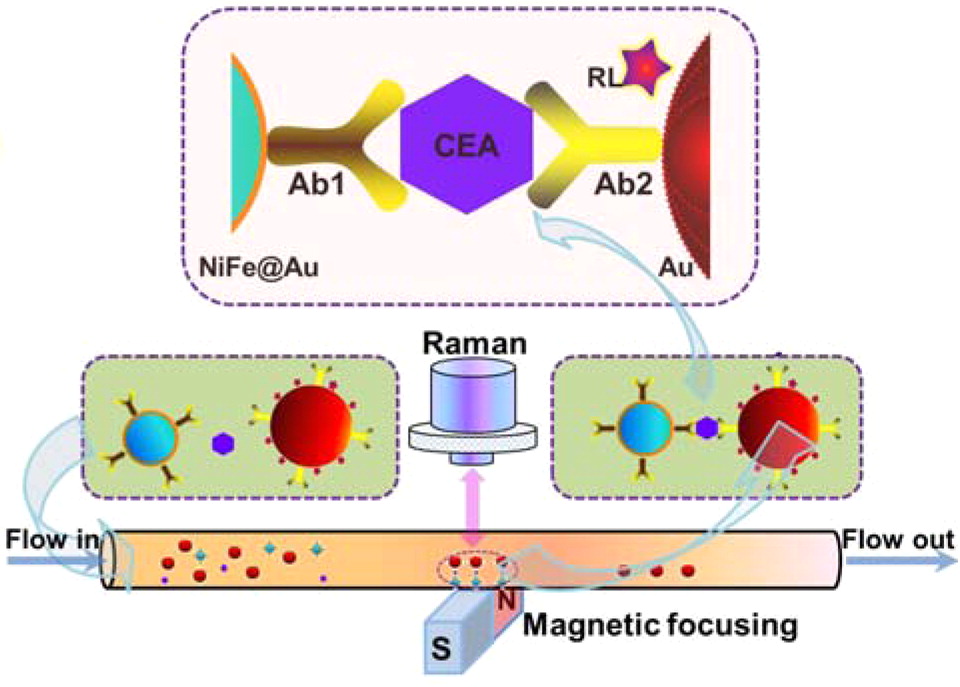
Figure 4. Illustration of SERS detection of cancer biomarker CEA (carcinoembryonic antigen). Using functional nanoprobes consisting of Au coated NiFe magnetic nanoparticles (NiFe@Au), capture antibodies (Ab1), detection antibodies (Ab2), and Raman labels (RL). Reprinted with permission from ref[101]. Copyright © 2015 American Chemical Society.
3.1.5. Detection of Pathogens
The magnetic SERS technique is one method for the diagnosis of infectious diseases caused by pathogens, such as bacteria or viruses[25][34][104][105]. Compared with other methods—including polymerase chain reaction, enzyme-linked immunosorbent assay, and culture and colony counting of bacteria—magnetic SERS has advantages, such as simpler sample preparation requirements, improved sensitivity, and rapid and multiplexed detection[104]. Additionally, magnetic properties assist in the concentration of the target to improve separation[15], accuracy, and sensitivity[106]. Typically, pathogens can be determined directly[34][106], or indirectly via specific capture with antibodies[24][104][107][108][109] or aptamers[25][110]. The MPPs used can be magnetic plasmonic core-shell mono particles[24] or dual particles including magnetic and plasmonic particles[25]. Additionally, the MPPs can be immobilized on a surface by specific capture[107], or without labeling[34]. Once these MPPs interact with targets, the targets are gathered by the magnetic force. The pathogens have a distinct, identifiable SER signal. Magnetic SERS techniques have ultrasensitive capabilities, down to 10 pathogen cells in 1 mL [25].
3.2. Drug Delivery and Therapy
The needs of drug delivery and therapy often coexist, in order to carry a drug to the affected location and provide medical treatment (Figure 5). Magnetic NPs have the potential to be next-generation drug carriers because of their unique physical-chemical properties[53][56]. Magnetic NPs can move easily and rapidly to the target position under a magnetic field. The toxicity and biocompatibility of magnetic NPs can be adjusted by surface modifications, such as coatings of polyethyleneimine (PEI)-g-polyethylene glycol (PEG), chitosan, dextran, N-(2-hydroxypropyl)methacrylamide (HPMA), polymeric micelles, starches, proteins, and polyvinyl alcohol (PVA)[52][53][55]. After being immobilized with the targeting ligands (enzymes, peptides, antibodies, aptamers etc.), the magnetic NPs accurately reach the target position and release drug molecules[54][55]. For cancer and tumor treatment, magnetic NPs have been utilized as a beneficial mechanism for thermotherapy[111][112][113][114][115]. In thermotherapy (high temperature treatment), magnetic particles act as heat transferrers, causing apoptosis of the tumor cells. In a study on mice by Xie et al., effective thermotherapy was achieved with four injections, each of dose 28 mg Fe. kg−1 body weight, using an alternating current magnetic field of 390 kHz and 12 A, with 30 min exposure at 43 °C [114]. Additionally, plasmonic NPs, such as Au and Ag, have been investigated for application in smart drug delivery and cancer therapy[54][55][111][116][117][118][119]. Several studies have also demonstrated that Au and Ag NPs possess antitumor properties and have the potential to enhance cancer therapy[120][121][122][123][124][125][126][127][128]. Briefly, endocytosis particles are carried by vesicles to a cell’s cytoplasm and nucleus; there they produce a toxic effect and cause apoptosis or programmed cell death through reactive oxygen species, tumor necrosis factor, or interleukin-6[117]. Meyers et al.[129] reported that peptide-targeted Au NPs successfully carried and delivered Phthalocyanine 4, an photodynamic agent, to cancer cells and then killed the cancer cells (concentration 1 µM). Ag NPs and their composite with other metals have also been investigated for antibacterial characteristics[127][130][131][132][133][134][135]. Ag NPs have been shown to effectively inhibit various pathogenic bacteria, fungi and viruses, including Staphylococcus aureus, Escherichia coli, Pseudomonas aeruginosa, dermatophyte, and HIV-1[127]. The antibacterial characteristics depend on the size, shape, concentration, and charge of the particles[111][127]. The optimum parameters for antibacterial Ag NPs were found to be spherical and less than 10 nm in size, rather than triangular, linear, or cubic in shape, and larger in size[111][127]. Additionally, accumulation over time and positive surface charge can help the Ag NPs increase their antibacterial effect[127]. An Ag-Co-Cr alloy has been investigated as an antibacterial medical implant[135].
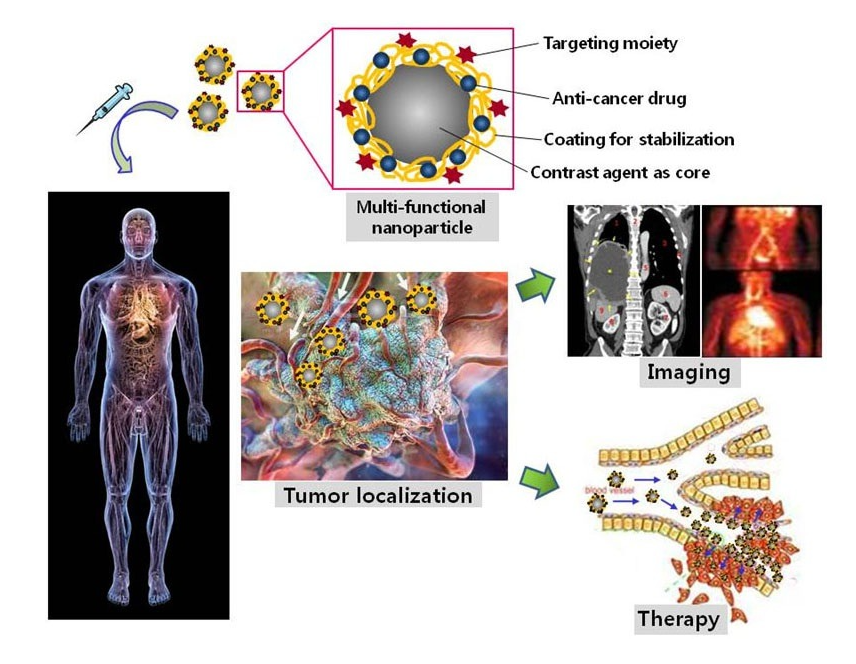
MPPs that combine magnetic and plasmonic nanoparticles have shown the same properties as the constituent magnetic NPs and plasmonic NPs. In a study by Tomitaka, magnetic plasmonic core-shell nanostar NPs demonstrated image-guided drug delivery and NIR-triggered drug release applications[51]. Under an alternating magnetic field, MPPs act as imaging contrast agents, assisting in the production of clear magnetic particle images. The Au parts of the MPPs bind the drug, which is then released at the desired location under NIR stimulation. Another study by Lal et al. demonstrated that iron oxide gold nanobowls can be guided magnetically, and that they possess distinct SERS capabilities. These characteristics show MPPs’ potential as a therapeutic drug storing and imaging contrast material[136]. Moreover, iron gold alloy NPs, in a study by Li et al., demonstrated their potential in thermotherapy-mediated controlled drug release for cancer therapy. According to this report, the thermotherapeutic properties of MPPs trigger drug release from the drug-MPPs conjugate. Furthermore, the magnetic properties in MPPs also aid in the drug unbinding under an external magnetic field[137].
In addition, the MPPs have exhibited photothermal ability that enables thermotherapeutic destruction of bacteria[24][108]. Popcorn shaped Fe core-Au shell MPPs have shown their antibacterial properties via a study on the multidrug resistant Salmonella DT104 bacterial strain[24]. The experiment achieved approximately 100% bacteria cell death using 2 mL of bacteria cells, at 1.2 × 105 CFU mL−1, incubated with 100 µL of particles and exposed to light (670 nm at 1.5 W cm−2) for 10 min. In another study, a mixture containing 150 µL of 1.3 × 104 CFU mL−1 E. coli cells, with 50 µL of MPPs solution, was exposed to light (670 nm, 2.5 W cm−2) for 12 min, and showed approximately 90% cell death[108]. In addition, the magnetic properties of MPPs aid the rapid separation of target pathogens in solution.
3.3. Imaging
Iron or gold-based NPs have been utilized as imaging contrast agents under alternating magnetic fields, such as in MRI and computed tomography; specifically, aimed at bio-imaging tumors and enabling their associated cancer diagnosis[111][138][139][140][141][142][143][144]. Iron and gold-based MPPs’ properties are used to upgrade the quality of cancer therapy by facilitating real-time diagnosis or image-guided drug delivery and therapy, and provides clear magnetic particle imaging. The high-quality images enable the control of the MPPs’ position under a magnetic environment. Therefore, once the MPPs reach the target position (tumor or cancer cell), the local environment’s structure is imaged and can help in diagnosis. Moreover, in drug delivery, observable drug release improves control of the drug dose and thereby ensures more effective treatment. Therefore, by imaging, a drug can be monitored at the tumor’s location and the drug’s concentration controlled[21][28][51][56][104] (Figure 5). For example, in Tomitaka et al.‘s research, MPPs demonstrated image-guided, near-infrared (NIR) responsive, and triggered drug release capabilities[51]. The MPPs produced clear magnetic particle imaging that enabled the MPPs to be guided to the correct position, and thereby deliver the drug to the required location. The Au parts in MPPs act as drug binders that release the drug under NIR stimulation. Thus, MPPs are candidates for applications in image-guided drug delivery therapy. These novel imaging properties could improve human health care in the future.
4. Conclusion and Future Perspectives
The above discussions demonstrate that MPPs are multifunctional nanomaterials that combine the synergistic effects of their magnetic and plasmonic parts. MPPs have improved the quality of existing imaging techniques and have the potential to monitor exceptionally low concentration targets and provide real-time delivery and release of therapeutic drugs. The latter has already been successfully applied at the in vitro and in vivo levels in rats. MPPs can be efficiently used in various systems to provide improved disease diagnosis, monitoring, and treatment. With real-time control, drug dose can be easily monitored, and release rates appropriately adjusted for each individual situation.
Application MPPs in thermotherapy is further widely performed due to not only high effectiveness but also less side effects. For example, in the case of intracranial thermotherapy of glioblastoma multiforme, magnetic NPs combined with external beam radiotherapy did not produce the side effects commonly seen with traditional treatments, such as headaches, nausea, vomitus, and allergic reactions. Additionally, no neurological deficits or infections were evident in the treated regions[112]. This study has shown the potential of next-generation MPPs for cancer treatments that decrease side effects and aid in healing.
MPPs also exhibit potential antibacterial properties. Bacterial inhibition without the use of antibiotics will help prevent the evolution of bacteria capable of defeating antibiotics. Overall, the potential and prospective applications of MPPs could bring significant benefits in the future.
Beyond this, there are many reports that have discussed the application of machine and artificial intelligence in the SERS technique[145][146][147][148]. It has provided a novel idea that it is possible for artificial intelligence which learns using MPPs in bioapplication to raise a new approach in future chemistry.
References
- Pilot, R.; Signorini, R.; Durante, C.; Orian, L.; Bhamidipati, M.; Fabris, L. A review on surface-enhanced raman scattering. Biosensors 2019, 9, 57.
- Schlucker, S. Surface-enhanced raman spectroscopy: Concepts and chemical applications. Angew. Chem. Int. Ed. 2014, 53, 4756–4795.
- Etchegoin, P.G.; Le Ru, E.C. A perspective on single molecule sers: Current status and future challenges. Phys. Chem. Chem. Phys. 2008, 10, 6079–6089.
- Kneipp, K.; Wang, Y.; Kneipp, H.; Perelman, L.T.; Itzkan, I.; Dasari, R.R.; Feld, M.S. Single molecule detection using surface-enhanced raman scattering (sers). Phys. Rev. Lett. 1997, 78, 1667.
- Lee, H.M.; Jin, S.M.; Kim, H.M.; Suh, Y.D. Single-molecule surface-enhanced raman spectroscopy: A perspective on the current status. Phys. Chem. Chem. Phys. 2013, 15, 5276–5287.
- Figueroa, M.; Schraer, S.; Pourrezaei, K.; Tyagi, S. Surface-Enhanced Raman Scattering and Microwave Absorption in Silver Nanoparticle Inks; SPIE BiOS: San Francisco, CA, USA, 2012; Volume 8234.
- Figueroa, M.; Pourrezaei, K.; Tyagi, S. Detection of Hyaluronic Acid on a Functionalized Surface Enhanced Raman Scattering Substrate. In Proceedings of the 2011 IEEE 37th Annual Northeast Bioengineering Conference (NEBEC), Troy, NY, USA, 1–3 April 2011; pp. 1–2.
- Contarino, M.; Kamat, V.; Keough, E.; Babu, N.; Sergi, M.; Ishino, T.; Papazoglou, E.; Chaiken, I.; Tyagi, S.; Pourrezaei, K. Bio-nano-Optics for Cellular Investigations; SPIE: Philadelphia, PA, USA, 2004; Volume 5588.
- Tyagi, S.; Figueroa, M. Flexible Sers Substrates with Filtering Capabilities. United States Patents US9278855B2, 8 March 2016.
- Lawandy, N.M. Chemical and Biological Sensing Using Metallic Particles in Amplifying and Absorbing Media. United States Patents US7355704B2, 8 April 2008.
- Tyagi, S.; Pourrezaei, K. Method for the Formation of Sers Substrates. United States Patents US8559002B2, 15 October 2013.
- Lu, A.H.; Salabas, E.L.; Schuth, F. Magnetic nanoparticles: Synthesis, protection, functionalization, and application. Angew. Chem. Int. Ed. 2007, 46, 1222–1244.
- Xie, W.; Schlucker, S. Rationally designed multifunctional plasmonic nanostructures for surface-enhanced raman spectroscopy: A review. Rep. Prog. Phys. 2014, 77, 116502.
- Park, S.-M.; Aalipour, A.; Vermesh, O.; Yu, J.H.; Gambhir, S.S. Towards clinically translatable in vivo nanodiagnostics. Nat. Rev. Mater. 2017, 2, 17014.
- Park, S.-M.; Wong, D.J.; Ooi, C.C.; Kurtz, D.M.; Vermesh, O.; Aalipour, A.; Suh, S.; Pian, K.L.; Chabon, J.J.; Lee, S.H.; et al. Molecular profiling of single circulating tumor cells from lung cancer patients. Proc. Natl. Acad. Sci. USA 2016, 113, E8379.
- Vermesh, O.; Aalipour, A.; Ge, T.J.; Saenz, Y.; Guo, Y.; Alam, I.S.; Park, S.-M.; Adelson, C.N.; Mitsutake, Y.; Vilches-Moure, J.; et al. An intravascular magnetic wire for the high-throughput retrieval of circulating tumour cells in vivo. Nat. Biomed. Eng. 2018, 2, 696–705.
- Lai, H.S.; Xu, F.G.; Wang, L. A review of the preparation and application of magnetic nanoparticles for surface-enhanced raman scattering. J. Mater. Sci. 2018, 53, 8677–8698.
- Wang, C.G.; Meloni, M.M.; Wu, X.Z.; Zhuo, M.; He, T.G.; Wang, J.F.; Wang, C.W.; Dong, P.T. Magnetic plasmonic particles for sers-based bacteria sensing: A review. Aip. Adv. 2019, 9.
- Rhouati, A.; Catanante, G.; Nunes, G.; Hayat, A.; Marty, J.-L. Label-free aptasensors for the detection of mycotoxins. Sensors 2016, 16, 2178.
- Balzerova, A.; Fargasova, A.; Markova, Z.; Ranc, V.; Zboril, R. Magnetically-assisted surface enhanced raman spectroscopy (ma-sers) for label-free determination of human immunoglobulin g (igg) in blood using nanocomposite. Anal. Chem. 2014, 86, 11107–11114.
- Han, Y.; Lei, S.-L.; Lu, J.-H.; He, Y.; Chen, Z.-W.; Ren, L.; Zhou, X. Potential use of sers-assisted theranostic strategy based on fe3o4/au cluster/shell nanocomposites for bio-detection, mri, and magnetic hyperthermia. Mater. Sci. Eng. C 2016, 64, 199–207.
- Xiao, R.; Wang, C.W.; Zhu, A.N.; Long, F. Single functional magnetic-bead as universal biosensing platform for trace analyte detection using sers-nanobioprobe. Biosens. Bioelectron. 2016, 79, 661–668.
- Agoston, R.; Izake, E.L.; Sivanesan, A.; Lott, W.B.; Sillence, M.; Steel, R. Rapid isolation and detection of erythropoietin in blood plasma by magnetic core gold nanoparticles and portable raman spectroscopy. Nanomed. Nanotechnol. Biol. Med. 2016, 12, 633–641.
- Fan, Z.; Senapati, D.; Khan, S.A.; Singh, A.K.; Hamme, A.; Yust, B.; Sardar, D.; Ray, P.C. Popcorn-shaped magnetic core–plasmonic shell multifunctional nanoparticles for the targeted magnetic separation and enrichment, label-free sers imaging, and photothermal destruction of multidrug-resistant bacteria. Chem. A Eur. J. 2013, 19, 2839–2847.
- Wang, J.; Wu, X.; Wang, C.; Shao, N.; Dong, P.; Xiao, R.; Wang, S. Magnetically assisted surface-enhanced raman spectroscopy for the detection of staphylococcus aureus based on aptamer recognition. ACS Appl. Mater. Interfaces 2015, 7, 20919–20929.
- Yang, K.; Hu, Y.; Dong, N.; Zhu, G.; Zhu, T.; Jiang, N. A novel sers-based magnetic aptasensor for prostate specific antigen assay with high sensitivity. Biosens. Bioelectron. 2017, 94, 286–291.
- Li, H.; Chen, Q.; Mehedi Hassan, M.; Chen, X.; Ouyang, Q.; Guo, Z.; Zhao, J. A magnetite/pmaa nanospheres-targeting sers aptasensor for tetracycline sensing using mercapto molecules embedded core/shell nanoparticles for signal amplification. Biosens. Bioelectron. 2017, 92, 192–199.
- Carrouée, A.; Allard-Vannier, E.; Même, S.; Szeremeta, F.; Beloeil, J.-C.; Chourpa, I. Sensitive trimodal magnetic resonance imaging-surface-enhanced resonance raman scattering-fluorescence detection of cancer cells with stable magneto-plasmonic nanoprobes. Anal. Chem. 2015, 87, 11233–11241.
- Yang, X.; He, Y.; Wang, X.; Yuan, R. A sers biosensor with magnetic substrate for sensitive detection of hg2+. Appl. Surf. Sci. 2017, 416, 581–586.
- He, Y.; Wang, Y.; Yang, X.; Xie, S.; Yuan, R.; Chai, Y. Metal organic frameworks combining cofe2o4 magnetic nanoparticles as highly efficient sers sensing platform for ultrasensitive detection of n-terminal pro-brain natriuretic peptide. ACS Appl. Mater. Interfaces 2016, 8, 7683–7690.
- Shen, J.; Zhou, Y.; Huang, J.; Zhu, Y.; Zhu, J.; Yang, X.; Chen, W.; Yao, Y.; Qian, S.; Jiang, H.; et al. In-situ sers monitoring of reaction catalyzed by multifunctional @ag-au microspheres. Appl. Catal. B Environ. 2017, 205, 11–18.
- Ilkhani, H.; Hughes, T.; Li, J.; Zhong, C.J.; Hepel, M. Nanostructured sers-electrochemical biosensors for testing of anticancer drug interactions with DNA. Biosens. Bioelectron. 2016, 80, 257–264.
- Srinoi, P.; Chen, Y.T.; Vittur, V.; Marquez, M.D.; Lee, T.R. Bimetallic nanoparticles: Enhanced magnetic and optical properties for emerging biological applications. Appl. Sci. 2018, 8, 1106.
- Wang, C.; Wang, J.; Li, M.; Qu, X.; Zhang, K.; Rong, Z.; Xiao, R.; Wang, S. A rapid sers method for label-free bacteria detection using polyethylenimine-modified au-coated magnetic microspheres and nanoparticles. Analyst 2016, 141, 6226–6238.
- Zhou, X.; Xu, W.; Wang, Y.; Kuang, Q.; Shi, Y.; Zhong, L.; Zhang, Q. Fabrication of cluster/shell fe3o4/au nanoparticles and application in protein detection via a sers method. J. Phys. Chem. C 2010, 114, 19607–19613.
- Wang, C.; Xu, J.; Wang, J.; Rong, Z.; Li, P.; Xiao, R.; Wang, S. Polyethylenimine-interlayered silver-shell magnetic-core microspheres as multifunctional sers substrates. J. Mater. Chem. C 2015, 3, 8684–8693.
- Wang, C.; Li, P.; Wang, J.; Rong, Z.; Pang, Y.; Xu, J.; Dong, P.; Xiao, R.; Wang, S. Polyethylenimine-interlayered core–shell–satellite 3d magnetic microspheres as versatile sers substrates. Nanoscale 2015, 7, 18694–18707.
- He, Q.; Zhao, A.; Li, L.; Sun, H.; Wang, D.; Guo, H.; Sun, M.; Chen, P. Fabrication of @ag magnetic–plasmonic nanospindles as highly efficient sers active substrates for label-free detection of pesticides. New J. Chem. 2017, 41, 1582–1590.
- Sun, Z.; Du, J.; Yan, L.; Chen, S.; Yang, Z.; Jing, C. Multifunctional –au satellite structured sers probe for charge selective detection of food dyes. ACS Appl. Mater. Inter. 2016, 8, 3056–3062.
- Jeong, C.; Kim, H.-M.; Park, S.Y.; Cha, M.G.; Park, S.-J.; Kyeong, S.; Pham, X.-H.; Hahm, E.; Ha, Y.; Jeong, D.H.; et al. Highly sensitive magnetic-sers dual-function silica nanoprobes for effective on-site organic chemical detection. Nanomaterials 2017, 7, 146.
- Hou, T.; Liu, Y.; Xu, L.; Wu, Y.; Ying, Y.; Wen, Y.; Guo, X.; Yang, H. Au dotted magnetic graphene sheets for sensitive detection of thiocyanate. Sens. Actuators B Chem. 2017, 241, 376–382.
- Ding, G.; Xie, S.; Zhu, Y.; Liu, Y.; Wang, L.; Xu, F. Graphene oxide wrapped nanohybrid as sers substrate for aromatic dye detection. Sens. Actuators B Chem. 2015, 221, 1084–1093.
- Liu, Z.; Wang, Y.; Deng, R.; Yang, L.; Yu, S.; Xu, S.; Xu, W. particles for surface magnet solid-phase extraction surface-enhanced raman scattering (smspe-sers): From sample pretreatment to detection all-in-one. ACS Appl. Mater. Inter. 2016, 8, 14160–14168.
- Contreras-Cáceres, R.; Abalde-Cela, S.; Guardia-Girós, P.; Fernández-Barbero, A.; Pérez-Juste, J.; Alvarez-Puebla, R.A.; Liz-Marzán, L.M. Multifunctional microgel magnetic/optical traps for sers ultradetection. Langmuir 2011, 27, 4520–4525.
- Zhao, M.; Guo, H.; Liu, W.; Tang, J.; Wang, L.; Zhang, B.; Xue, C.; Liu, J.; Zhang, W. Silica cladding of ag nanoparticles for high stability and surface-enhanced raman spectroscopy performance. Nanoscale Res. Lett. 2016, 11, 403.
- Son, H.Y.; Kim, K.R.; Lee, J.B.; Le Kim, T.H.; Jang, J.; Kim, S.J.; Yoon, M.S.; Kim, J.W.; Nam, Y.S. Bioinspired synthesis of mesoporous gold-silica hybrid microspheres as recyclable colloidal sers substrates. Sci. Rep. 2017, 7, 14728.
- Zou, Y.; Chen, H.; Li, Y.; Yuan, X.; Zhao, X.; Chen, W.; Cao, F.; Cai, N.; Huang, X.; Yang, F.; et al. Synthesis of mesoporous-silica coated multi-branched gold nanoparticles for surface enhanced raman scattering evaluation of 4-bromomethcathinone. J. Saudi Chem. Soc. 2019, 23, 378–383.
- Li, M.; Qiu, Y.; Fan, C.; Cui, K.; Zhang, Y.; Xiao, Z. Design of sers nanoprobes for raman imaging: Materials, critical factors and architectures. Acta Pharm. Sin. B 2018, 8, 381–389.
- Li, F.; Huang, Y.; Huang, K.; Lin, J.; Huang, P. Functional magnetic graphene composites for biosensing. Int. J. Mol. Sci. 2020, 21, 390.
- Shan, Y.; Yang, Y.; Cao, Y.; Huang, Z. Facile solvothermal synthesis of ag/fe3o4 nanocomposites and their sers applications in on-line monitoring of pesticide contaminated water. RSC Adv. 2015, 5, 102610–102618.
- Tomitaka, A.; Arami, H.; Ahmadivand, A.; Pala, N.; McGoron, A.J.; Takemura, Y.; Febo, M.; Nair, M. Magneto-plasmonic nanostars for image-guided and nir-triggered drug delivery. Sci. Rep. 2020, 10, 10115.
- McBain, S.C.; Yiu, H.H.P.; Dobson, J. Magnetic nanoparticles for gene and drug delivery. Int. J. Nanomed. 2008, 3, 169–180.
- Mou, X.; Ali, Z.; Li, S.; He, N. Applications of magnetic nanoparticles in targeted drug delivery system. J. Nanosci. Nanotechnol. 2015, 15, 54–62.
- Lombardo, D.; Kiselev, M.A.; Caccamo, M.T. Smart nanoparticles for drug delivery application: Development of versatile nanocarrier platforms in biotechnology and nanomedicine. J. Nanomater. 2019, 2019, 3702518.
- Ulbrich, K.; Holá, K.; Šubr, V.; Bakandritsos, A.; Tuček, J.; Zbořil, R. Targeted drug delivery with polymers and magnetic nanoparticles: Covalent and noncovalent approaches, release control, and clinical studies. Chem. Rev. 2016, 116, 5338–5431.
- Bae, K.H.; Chung, H.J.; Park, T.G. Nanomaterials for cancer therapy and imaging. Mol. Cells 2011, 31, 295–302.
- Jun, B.H.; Noh, M.S.; Kim, J.; Kim, G.; Kang, H.; Kim, M.S.; Seo, Y.T.; Baek, J.; Kim, J.H.; Park, J.; et al. Multifunctional silver-embedded magnetic nanoparticles as sers nanoprobes and their applications. Small 2010, 6, 119–125.
- Doering, W.E.; Piotti, M.E.; Natan, M.J.; Freeman, R.G. Sers as a foundation for nanoscale, optically detected biological labels. Adv. Mater. 2007, 19, 3100–3108.
- Jun, B.H.; Noh, M.S.; Kim, G.; Kang, H.; Kim, J.H.; Chung, W.J.; Kim, M.S.; Kim, Y.K.; Cho, M.H.; Jeong, D.H.; et al. Protein separation and identification using magnetic beads encoded with surface-enhanced raman spectroscopy. Anal. Biochem. 2009, 391, 24–30.
- Padilla-Cruz, A.L.; Garza-Cervantes, J.A.; Vasto-Anzaldo, X.G.; García-Rivas, G.; León-Buitimea, A.; Morones-Ramírez, J.R. Synthesis and design of ag–fe bimetallic nanoparticles as antimicrobial synergistic combination therapies against clinically relevant pathogens. Sci. Rep. 2021, 11, 5351.
- Al-Asfar, A.; Zaheer, Z.; Aazam, E.S. Eco-friendly green synthesis of bimetallic nanoparticles: Antioxidant, antimicrobial and photocatalytic degradation of bromothymol blue. J. Photochem. Photobiol. B Biol. 2018, 185, 143–152.
- He, J.; Li, G.; Hu, Y. Aptamer recognition induced target-bridged strategy for proteins detection based on magnetic chitosan and silver/chitosan nanoparticles using surface-enhanced raman spectroscopy. Anal. Chem. 2015, 87, 11039–11047.
- Chen, J.; Pang, S.; He, L.; Nugen, S.R. Highly sensitive and selective detection of nitrite ions using /au magnetic nanoparticles by surface-enhanced raman spectroscopy. Biosens. Bioelectron. 2016, 85, 726–733.
- Pour Yazdankhah, S.; Hellemann, A.-L.; Rønningen, K.; Olsen, E. Rapid and sensitive detection of staphylococcus species in milk by elisa based on monodisperse magnetic particles. Vet. Microbiol. 1998, 62, 17–26.
- Alarcón, B.; Vicedo, B.; Aznar, R. Pcr-based procedures for detection and quantification of staphylococcus aureus and their application in food. J. Appl. Microbiol. 2006, 100, 352–364.
- Zelada-Guillén, G.A.; Sebastián-Avila, J.L.; Blondeau, P.; Riu, J.; Rius, F.X. Label-free detection of staphylococcus aureus in skin using real-time potentiometric biosensors based on carbon nanotubes and aptamers. Biosens. Bioelectron. 2012, 31, 226–232.
- Wang, X.-Y.; Yang, J.-Y.; Wang, Y.-T.; Zhang, H.-C.; Chen, M.-L.; Yang, T.; Wang, J.-H. M13 phage-based nanoprobe for sers detection and inactivation of staphylococcus aureus. Talanta 2021, 221, 121668.
- Zhu, A.; Ali, S.; Xu, Y.; Ouyang, Q.; Chen, Q. A sers aptasensor based on aunps functionalized pdms film for selective and sensitive detection of staphylococcus aureus. Biosens. Bioelectron. 2021, 172, 112806.
- Strickland, A.D.; Batt, C.A. Detection of carbendazim by surface-enhanced raman scattering using cyclodextrin inclusion complexes on gold nanorods. Anal. Chem. 2009, 81, 2895–2903.
- Guerrini, L.; Garcia-Ramos, J.V.; Domingo, C.; Sanchez-Cortes, S. Nanosensors based on viologen functionalized silver nanoparticles: Few molecules surface-enhanced raman spectroscopy detection of polycyclic aromatic hydrocarbons in interparticle hot spots. Anal. Chem. 2009, 81, 1418–1425.
- Guerrini, L.; Garcia-Ramos, J.V.; Domingo, C.; Sanchez-Cortes, S. Sensing polycyclic aromatic hydrocarbons with dithiocarbamate-functionalized ag nanoparticles by surface-enhanced raman scattering. Anal. Chem. 2009, 81, 953–960.
- Zhang, H.; Li, F.; Chen, H.; Ma, Y.; Qi, S.; Chen, X.; Zhou, L. Aunps colorimetric sensor for detecting platelet-derived growth factor-bb based on isothermal target-triggering strand displacement amplification. Sens. Actuators B Chem. 2015, 207, 748–755.
- Zhu, D.; Zhou, X.; Xing, D. A new kind of aptamer-based immunomagnetic electrochemiluminescence assay for quantitative detection of protein. Biosens. Bioelectron. 2010, 26, 285–288.
- Jin, X.; Zhao, J.; Zhang, L.; Huang, Y.; Zhao, S. An enhanced fluorescence polarization strategy based on multiple protein–DNA–protein structures for sensitive detection of pdgf-bb. RSC Adv. 2014, 4, 6850–6853.
- Wang, C.-W.; Chang, H.-T. Sensitive detection of platelet-derived growth factor through surface-enhanced raman scattering. Anal. Chem. 2014, 86, 7606–7611.
- Niu, C.; Zou, B.; Wang, Y.; Cheng, L.; Zheng, H.; Zhou, S. Highly sensitive and reproducible sers performance from uniform film assembled by magnetic noble metal composite microspheres; Langmuir: America, 2016; pp. 32.
- Shuangmei Zhu; Chunzhen Fan; Junqiao Wang; Fan Chunzhen; Erjun Liang; Mingju Chao; Realization of high sensitive SERS substrates with one-pot fabrication of Ag–Fe 3 O 4 nanocomposites. Journal of Colloid and Interface Science 2015, 438, 116-121, 10.1016/j.jcis.2014.09.015.
- Sergio H. Toma; Jonnatan Santos; Koiti Araki; Henrique E. Toma; Pushing the surface-enhanced Raman scattering analyses sensitivity by magnetic concentration: A simple non core–shell approach. Analytica Chimica Acta 2015, 855, 70-75, 10.1016/j.aca.2014.12.005.
- Jianhua Shen; Yihua Zhu; Xiaoling Yang; Jie Zong; Chunzhong Li; Multifunctional Fe3O4@Ag/SiO2/Au Core–Shell Microspheres as a Novel SERS-Activity Label via Long-Range Plasmon Coupling. Langmuir 2012, 29, 690-695, 10.1021/la304048v.
- Yuling Wang; Sandeep Ravindranath; Joseph Irudayaraj; Separation and detection of multiple pathogens in a food matrix by magnetic SERS nanoprobes. Analytical and Bioanalytical Chemistry 2010, 399, 1271-1278, 10.1007/s00216-010-4453-6.
- Yuanfeng Pang; Chongwen Wang; Jing Wang; Zhiwei Sun; Rui Xiao; Shengqi Wang; Fe3O4@Ag magnetic nanoparticles for microRNA capture and duplex-specific nuclease signal amplification based SERS detection in cancer cells. Biosensors and Bioelectronics 2016, 79, 574-580, 10.1016/j.bios.2015.12.052.
- Jinnan Zhang; Padmanabh Joshi; Yan Zhou; Rui Ding; Quantitative SERS-based DNA detection assisted by magnetic microspheres. Chemical Communications 2015, 51, 15284-15286, 10.1039/C5CC06571B.
- Long Wu; Xiaoyan Xiao; Kun Chen; Wenmin Yin; Qin Li; Pan Wang; Zhicheng Lu; Jing Ma; Heyou Han; Ultrasensitive SERS detection of Bacillus thuringiensis special gene based on Au@Ag NRs and magnetic beads. Biosensors and Bioelectronics 2017, 92, 321-327, 10.1016/j.bios.2016.11.005.
- Melisew Tadele Alula; Jyisy Yang; Photochemical decoration of gold nanoparticles on polymer stabilized magnetic microspheres for determination of adenine by surface-enhanced Raman spectroscopy. Microchimica Acta 2014, 182, 1017-1024, 10.1007/s00604-014-1429-9.
- Hoan T. Ngo; Naveen Gandra; Andrew M. Fales; Steve M. Taylor; Tuan Vo-Dinh; Sensitive DNA detection and SNP discrimination using ultrabright SERS nanorattles and magnetic beads for malaria diagnostics. Biosensors and Bioelectronics 2016, 81, 8-14, 10.1016/j.bios.2016.01.073.
- Lin He; Gregory J. Hannon; MicroRNAs: small RNAs with a big role in gene regulation. Nature Reviews Genetics 2004, 5, 522-531, 10.1038/nrg1379.
- Carl D. Novina; Phillip A. Sharp; The RNAi revolution. Nature 2004, 430, 161-164, 10.1038/430161a.
- Thalia A. Farazi; Jessica I. Spitzer; Pavel Morozov; Thomas Tuschl; miRNAs in human cancer. The Journal of Pathology 2010, 223, 102-115, 10.1002/path.2806.
- Wei Kang; Joanna H. M. Tong; Anthony W. H. Chan; Raymond W. M. Lung; Shuk Ling Chau; Queenie W. L. Wong; Nathalie Wong; Jun Yu; Alfred S. L. Cheng; Ka Fai To; et al. Stathmin1 Plays Oncogenic Role and Is a Target of MicroRNA-223 in Gastric Cancer. PLOS ONE 2012, 7, e33919, 10.1371/journal.pone.0033919.
- Nozomu Yanaihara; Natasha Caplen; Elise Bowman; Masahiro Seike; Kensuke Kumamoto; Ming Yi; Robert M. Stephens; Aikou Okamoto; Jun Yokota; Tadao Tanaka; et al.George Adrian CalinChang-Gong LiuCarlo M. CroceCurtis C. Harris Unique microRNA molecular profiles in lung cancer diagnosis and prognosis. Cancer Cell 2006, 9, 189-198, 10.1016/j.ccr.2006.01.025.
- Tugba Kilic; Seda Nur Topkaya; Dilsat Ozkan Ariksoysal; Mehmet Ozsoz; Petek Ballar; Yasemin Erac; Oguz Gozen; Electrochemical based detection of microRNA, mir21 in breast cancer cells. Biosensors and Bioelectronics 2012, 38, 195-201, 10.1016/j.bios.2012.05.031.
- Ping Ping Hu; Hui Liu; Shu Jun Zhen; Chun Mei Li; Cheng Zhi Huang; Nanosilver-based surface-enhanced Raman spectroscopic determination of DNA methyltransferase activity through real-time hybridization chain reaction. Biosensors and Bioelectronics 2015, 73, 228-233, 10.1016/j.bios.2015.06.009.
- Rongke Gao; Ziyi Cheng; Andrew J. Demello; Jaebum Choo; Wash-free magnetic immunoassay of the PSA cancer marker using SERS and droplet microfluidics. Lab on a Chip 2016, 16, 1022-1029, 10.1039/c5lc01249j.
- Yesim Somay Selbes; Mehmet Gokhan Caglayan; Merve Eryilmaz; Ismail Hakki Boyaci; Necdet Saglam; Arif Ahmet Basaran; Ugur Tamer; Surface-enhanced Raman probe for rapid nanoextraction and detection of erythropoietin in urine. Analytical and Bioanalytical Chemistry 2016, 408, 8447-8456, 10.1007/s00216-016-9966-1.
- Anh H. Nguyen; Yesol Shin; Sang Jun Sim; Development of SERS substrate using phage-based magnetic template for triplex assay in sepsis diagnosis. Biosensors and Bioelectronics 2016, 85, 522-528, 10.1016/j.bios.2016.05.043.
- Yan Lin; Guanhong Xu; Fangdi Wei; Aixia Zhang; Jing Yang; Qin Hu; Detection of CEA in human serum using surface-enhanced Raman spectroscopy coupled with antibody-modified Au and γ-Fe 2 O 3 @Au nanoparticles. Journal of Pharmaceutical and Biomedical Analysis 2016, 121, 135-140, 10.1016/j.jpba.2016.01.027.
- Duygu Deniz Usta; Kouroush Salimi; Asli Pinar; Ilknur Coban; Turgay Tekinay; Ali Tuncel; A Boronate Affinity-Assisted SERS Tag Equipped with a Sandwich System for Detection of Glycated Hemoglobin in the Hemolysate of Human Erythrocytes. ACS Applied Materials & Interfaces 2016, 8, 11934-11944, 10.1021/acsami.6b00138.
- Waleed A. Hassanain; Emad L. Izake; Michael S. Schmidt; Godwin Ayoko; Gold nanomaterials for the selective capturing and SERS diagnosis of toxins in aqueous and biological fluids. Biosensors and Bioelectronics 2017, 91, 664-672, 10.1016/j.bios.2017.01.032.
- Rongke Gao; Juhui Ko; Kiweon Cha; Jun Ho Jeon; Gi-Eun Rhie; Jonghoon Choi; Andrew J. Demello; Jaebum Choo; Fast and sensitive detection of an anthrax biomarker using SERS-based solenoid microfluidic sensor. Biosensors and Bioelectronics 2015, 72, 230-236, 10.1016/j.bios.2015.05.005.
- Tianxi Yang; Xiaoyu Guo; Hui Wang; Shuyue Fu; Ying Wen; Haifeng Yang; Magnetically optimized SERS assay for rapid detection of trace drug-related biomarkers in saliva and fingerprints. Biosensors and Bioelectronics 2015, 68, 350-357, 10.1016/j.bios.2015.01.021.
- Holubovska Polina; Zakiya Skeete; Shiyao Shan; Shan Yan; Katarzyna Kurzatkowska; Wei Zhao; Quang Minh Ngo; Polina Holubovska; Jin Luo; Maria Hepel; et al.Chuan-Jian Zhong Surface Enhanced Raman Scattering Detection of Cancer Biomarkers with Bifunctional Nanocomposite Probes. Analytical Chemistry 2015, 87, 10698-10702, 10.1021/acs.analchem.5b03456.
- Changlong Sun; Ren Zhang; Mingxia Gao; Xiangmin Zhang; A rapid and simple method for efficient capture and accurate discrimination of circulating tumor cells using aptamer conjugated magnetic beads and surface-enhanced Raman scattering imaging. Analytical and Bioanalytical Chemistry 2015, 407, 8883-8892, 10.1007/s00216-015-9049-8.
- Mi Suk Noh; Bong-Hyun Jun; Seongyong Kim; Homan Kang; Min-Ah Woo; Arash Minai-Tehrani; Ji-Eun Kim; Jaeyun Kim; Jooyoung Park; Hwang-Tae Lim; et al.Se-Chang ParkTaeghwan HyeonYong-Kweon KimDae Hong JeongYoon-Sik LeeMyung-Haing Cho Magnetic surface-enhanced Raman spectroscopic (M-SERS) dots for the identification of bronchioalveolar stem cells in normal and lung cancer mice. Biomaterials 2009, 30, 3915-3925, 10.1016/j.biomaterials.2009.03.059.
- Hongdeok Jang; Eun Young Hwang; Yongshin Kim; Jaebum Choo; Jihoon Jeong; Dong Woo Lim; Surface-Enhanced Raman Scattering and Fluorescence-Based Dual Nanoprobes for Multiplexed Detection of Bacterial Pathogens. Journal of Biomedical Nanotechnology 2016, 12, 1938-1951, 10.1166/jbn.2016.2309.
- Xingang Zhang; Xiaolei Zhang; Changliang Luo; Zhengqi Liu; Yiyun Chen; Shilian Dong; Changzhong Jiang; Shikuan Yang; Fubing Wang; Xiangheng Xiao; et al. Volume‐Enhanced Raman Scattering Detection of Viruses. Small 2019, 15, e1805516, 10.1002/smll.201805516.
- Lei Zhang; Jiajie Xu; Luo Mi; Heng Gong; Shaoyi Jiang; Qiuming Yu; Multifunctional magnetic–plasmonic nanoparticles for fast concentration and sensitive detection of bacteria using SERS. Biosensors and Bioelectronics 2012, 31, 130-136, 10.1016/j.bios.2011.10.006.
- Junfeng Wang; Xuezhong Wu; Chongwen Wang; Zhen Rong; Hongmei Ding; Hui Li; Shaohua Li; Ningsheng Shao; Peitao Dong; Rui Xiao; et al.Sheng-Qi Wang Facile Synthesis of Au-Coated Magnetic Nanoparticles and Their Application in Bacteria Detection via a SERS Method. ACS Applied Materials & Interfaces 2016, 8, 19958-19967, 10.1021/acsami.6b07528.
- Thomas J. Ondera; Ashton T. Hamme Ii; Magnetic-optical nanohybrids for targeted detection, separation, and photothermal ablation of drug-resistant pathogens. The Analyst 2015, 140, 7902-7911, 10.1039/c5an00497g.
- Yang Sun; Li Xu; Fengdi Zhang; Zhigang Song; Yunwen Hu; Yongjia Ji; Jiayin Shen; Ben Li; Hongzhou Lu; Haifeng Yang; et al. A promising magnetic SERS immunosensor for sensitive detection of avian influenza virus. Biosensors and Bioelectronics 2017, 89, 906-912, 10.1016/j.bios.2016.09.100.
- Yi Liang; Ji-Lai Gong; Yong Huang; Yue Zheng; Jian-Hui Jiang; Guo-Li Shen; Ru-Qin Yu; Biocompatible core-shell nanoparticle-based surface-enhanced Raman scattering probes for detection of DNA related to HIV gene using silica-coated magnetic nanoparticles as separation tools. Talanta 2007, 72, 443-449, 10.1016/j.talanta.2006.11.002.
- Kim-Hung Huynh; Xuan-Hung Pham; Jaehi Kim; Sang Hun Lee; Hyejin Chang; Won-Yeop Rho; Bong-Hyun Jun; Synthesis, Properties, and Biological Applications of Metallic Alloy Nanoparticles. International Journal of Molecular Sciences 2020, 21, 5174, 10.3390/ijms21145174.
- Klaus Maier-Hauff; Ronny Rothe; Regina Scholz; Uwe Gneveckow; Peter Wust; Burghard Thiesen; Annelie Feussner; Andreas Von Deimling; Norbert Waldoefner; Roland Felix; et al.Andreas Jordan Intracranial Thermotherapy using Magnetic Nanoparticles Combined with External Beam Radiotherapy: Results of a Feasibility Study on Patients with Glioblastoma Multiforme. Journal of Neuro-Oncology 2006, 81, 53-60, 10.1007/s11060-006-9195-0.
- Sally J. DeNardo; Gerald L. DeNardo; Laird A. Miers; Arutselvan Natarajan; Alan R. Foreman; Cordula Gruettner; Grete N. Adamson; Robert Ivkov; Development of Tumor Targeting Bioprobes (111In-Chimeric L6 Monoclonal Antibody Nanoparticles) for Alternating Magnetic Field Cancer Therapy. Clinical Cancer Research 2005, 11, 7087s-7092s, 10.1158/1078-0432.ccr-1004-0022.
- Jun Xie; Yu Zhang; Caiyun Yan; Lina Song; Song Wen; Fengchao Zang; Gong Chen; Qi Ding; Changzhi Yan; Ning Gu; et al. High-performance PEGylated Mn–Zn ferrite nanocrystals as a passive-targeted agent for magnetically induced cancer theranostics. Biomaterials 2014, 35, 9126-9136, 10.1016/j.biomaterials.2014.07.019.
- Jun Xie; Caiyun Yan; Yu Yan; Ling Chen; Lina Song; Fengchao Zang; Yanli An; Gaojun Teng; Ning Gu; Yu Zhang; et al. Multi-modal Mn–Zn ferrite nanocrystals for magnetically-induced cancer targeted hyperthermia: a comparison of passive and active targeting effects. Nanoscale 2016, 8, 16902-16915, 10.1039/c6nr03916b.
- Tarryn L. Botha; Elias E. Elemike; Suranie Horn; Damian C. Onwudiwe; John P. Giesy; Victor Wepener; Cytotoxicity of Ag, Au and Ag-Au bimetallic nanoparticles prepared using golden rod (Solidago canadensis) plant extract. Scientific Reports 2019, 9, 1-8, 10.1038/s41598-019-40816-y.
- Heerak Chugh; Damini Sood; Ishita Chandra; Vartika Tomar; Gagan Dhawan; Ramesh Chandra; Role of gold and silver nanoparticles in cancer nano-medicine. Artificial Cells, Nanomedicine, and Biotechnology 2018, 46, 1210-1220, 10.1080/21691401.2018.1449118.
- Miko Yamada; Matthew Foote; Tarl W. Prow; Therapeutic gold, silver, and platinum nanoparticles. WIREs Nanomedicine and Nanobiotechnology 2014, 7, 428-445, 10.1002/wnan.1322.
- João Conde; Gonçalo Doria; Pedro Baptista; Noble Metal Nanoparticles Applications in Cancer. Journal of Drug Delivery 2011, 2012, 1-12, 10.1155/2012/751075.
- 120. Shanmugasundaram, T.; Radhakrishnan, M.; Gopikrishnan, V.; Kadirvelu, K.; Balagurunathan, R; . Biocompatible silver, gold and silver/gold alloy nanoparticles for enhanced cancer therapy: In vitro and in vivo perspectives. Nanoscale 2017, 9, 16773–16790.
- Hector Katifelis; Anna Lyberopoulou; Iuliia Mukha; Nadiia Vityuk; Gallina Grodzyuk; George E. Theodoropoulos; Efstathios P. Efstathopoulos; Maria Gazouli; Ag/Au bimetallic nanoparticles induce apoptosis in human cancer cell lines via P53, CASPASE-3 and BAX/BCL-2 pathways. Artificial Cells, Nanomedicine, and Biotechnology 2018, 46, S389-S398, 10.1080/21691401.2018.1495645.
- Igor Shmarakov; Iuliia Mukha; Nadiia Vityuk; Vira Borschovetska; Nelya Zhyshchynska; Galyna Grodzyuk; Anna Eremenko; Antitumor Activity of Alloy and Core-Shell-Type Bimetallic AgAu Nanoparticles. Nanoscale Research Letters 2017, 12, 333, 10.1186/s11671-017-2112-y.
- Miao Hao; Chenfei Kong; Chengwei Jiang; Ruizhi Hou; XiaoMing Zhao; Jing Li; Yuqian Wang; Yiyao Gao; Hao Zhang; Bai Yang; et al.Jinlan Jiang Polydopamine-coated Au-Ag nanoparticle-guided photothermal colorectal cancer therapy through multiple cell death pathways. Acta Biomaterialia 2019, 83, 414-424, 10.1016/j.actbio.2018.10.032.
- Sathishkumar Gnanasekar; Dhivyabharathi Balakrishnan; Prabukumar Seetharaman; Pugazhendhi Arivalagan; Rajkuberan Chandrasekaran; Sivaramakrishnan Sivaperumal; Chrysin-Anchored Silver and Gold Nanoparticle-Reduced Graphene Oxide Composites for Breast Cancer Therapy. ACS Applied Nano Materials 2020, 3, 4574-4585, 10.1021/acsanm.0c00630.
- Wafa I Abdel-Fattah; Ghareib W Ali; On the anti-cancer activities of silver nanoparticles. Journal of Applied Biotechnology & Bioengineering 2018, 5, 43-46, 10.15406/jabb.2018.05.00116.
- A.C. Gomathi; S.R. Xavier Rajarathinam; A. Mohammed Sadiq; S. Rajeshkumar; Anticancer activity of silver nanoparticles synthesized using aqueous fruit shell extract of Tamarindus indica on MCF-7 human breast cancer cell line. Journal of Drug Delivery Science and Technology 2020, 55, 101376, 10.1016/j.jddst.2019.101376.
- Li Xu; Yi-Yi Wang; Jie Huang; Chun-Yuan Chen; Zhen-Xing Wang; Hui Xie; Silver nanoparticles: Synthesis, medical applications and biosafety. Theranostics 2020, 10, 8996-9031, 10.7150/thno.45413.
- Mi Suk Noh; Somin Lee; Homan Kang; Jin-Kyoung Yang; Hyunmi Lee; Doyk Hwang; Jong Woo Lee; Sinyoung Jeong; Yoonjeong Jang; Bong-Hyun Jun; et al.Dae Hong JeongSeong Keun KimYoon-Sik LeeMyung-Haing Cho Corrigendum to “Target-specific near-IR induced drug release and photothermal therapy with accumulated Au/Ag hollow nanoshells on pulmonary cancer cell membranes” [Biomaterials 45 (2015) 81–92]. Biomaterials 2015, 45, 81-92, 10.1016/j.biomaterials.2015.06.047.
- Joseph D. Meyers; Yu Cheng; Ann-Marie Broome; Richard S. Agnes; Mark D. Schluchter; Seunghee Margevicius; Xinning Wang; Malcolm Kenney; Clemens Burda; James P. Basilion; et al. Peptide-Targeted Gold Nanoparticles for Photodynamic Therapy of Brain Cancer. Particle & Particle Systems Characterization 2014, 32, 448-457, 10.1002/ppsc.201400119.
- Zengchao Guo; Yun Chen; Yi-Han Wang; Hui Jiang; Xuemei Wang; Advances and challenges in metallic nanomaterial synthesis and antibacterial applications. Journal of Materials Chemistry B 2020, 8, 4764-4777, 10.1039/d0tb00099j.
- Samuel Cheeseman; Andrew J. Christofferson; Rashad Kariuki; Daniel Cozzolino; Torben Daeneke; Russell J. Crawford; Vi Khanh Truong; James Chapman; Aaron Elbourne; Antimicrobial Metal Nanomaterials: From Passive to Stimuli‐Activated Applications. Advanced Science 2020, 7, 1902913, 10.1002/advs.201902913.
- Gina V Vimbela; Sang M. Ngo; Carolyn Fraze; Lei Yang; David A. Stout; Antibacterial properties and toxicity from metallic nanomaterials. International Journal of Nanomedicine 2017, 12, 3941-3965, 10.2147/ijn.s134526.
- Mayur Valodkar; Shefaly Modi; Angshuman Pal; Sonal Thakore; Synthesis and anti-bacterial activity of Cu, Ag and Cu–Ag alloy nanoparticles: A green approach. Materials Research Bulletin 2011, 46, 384-389, 10.1016/j.materresbull.2010.12.001.
- Xiaona Hu; Yuyun Zhao; Zhijian Hu; Aditya Saran; Shuai Hou; Tao Wen; Wenqi Liu; Yinglu Ji; Xingyu Jiang; Xiaochun Wu; et al. Gold nanorods core/AgPt alloy nanodots shell: A novel potent antibacterial nanostructure. Nano Research 2013, 6, 822-835, 10.1007/s12274-013-0360-4.
- Feilong Jiang; Weiwei Zhu; Cancan Zhao; Yulei Li; Pengbo Wei; Tian Wan; Haixia Ye; Shuai Pan; Fuzeng Ren; A strong, wear- and corrosion-resistant, and antibacterial Co–30 at.% Cr–5 at.% Ag ternary alloy for medical implants. Materials & Design 2019, 184, 108190, 10.1016/j.matdes.2019.108190.
- Alexander H. Mo; Preston B. Landon; Karla Santacruz Gomez; Heemin Kang; Joon Lee; Chen Zhang; Woraphong Janetanakit; Vrinda Sant; Tianyu Lu; David Alexander Colburn; et al.Siddhartha AkkirajuSamuel DossouYue CaoKuo-Fen LeeShyni VargheseGennadi GlinskyRatnesh Lal Magnetically-responsive silica-gold nanobowls for targeted delivery and SERS-based sensing.. Nanoscale 2016, 8, 11840-50, 10.1039/c6nr02445a.
- Yun-Qian Li; Meng Xu; Udesh Dhawan; Wai Ching Liu; Kou-Ting Wu; Xin-Rui Liu; ChingPo Lin; Gang Zhao; Yu-Chuan Wu; Ren-Jei Chung; et al. Iron–gold alloy nanoparticles serve as a cornerstone in hyperthermia-mediated controlled drug release for cancer therapy. International Journal of Nanomedicine 2018, 13, 5499-5509, 10.2147/ijn.s163721.
- 138. Naha, P.C.; Lau, K.C.; Hsu, J.C.; Hajfathalian, M.; Mian, S.; Chhour, P.; Uppuluri, L.; McDonald, E.S.; Maidment, A.D.; Cormode, D.P; et al. Gold silver alloy nanoparticles (gsan): An imaging probe for breast cancer screening with dual-energy mammography or computed tomography. Nanoscale 2016, 8, 13740–13754.
- Shinya Maenosono; Toshimasa Suzuki; Soichiro Saita; Superparamagnetic FePt nanoparticles as excellent MRI contrast agents. Journal of Magnetism and Magnetic Materials 2008, 320, L79-L83, 10.1016/j.jmmm.2008.01.026.
- Hong Yang; Jingjing Zhang; Qiwei Tian; He Hu; Yong Fang; Huixia Wu; Shiping Yang; One-pot synthesis of amphiphilic superparamagnetic FePt nanoparticles and magnetic resonance imaging in vitro. Journal of Magnetism and Magnetic Materials 2010, 322, 973-977, 10.1016/j.jmmm.2009.11.039.
- Hong Yang; Xuejian Li; Hong Zhou; Yeming Zhuang; He Hu; Huixia Wu; Shiping Yang; Monodisperse water-soluble Fe–Ni nanoparticles for magnetic resonance imaging. Journal of Alloys and Compounds 2011, 509, 1217-1221, 10.1016/j.jallcom.2010.09.191.
- Shang-Wei Chou; Chien-Liang Liu; Tzu-Ming Liu; Yu-Fang Shen; Lun-Chang Kuo; Cheng-Ham Wu; Tsung-Yuan Hsieh; Pei-Chun Wu; Ming-Rung Tsai; Che-Chang Yang; et al.Kai-Yao ChangMeng-Hua LuPai-Chi LiShi-Ping ChenYu-Hsin WangChen-Wen LuYi-An ChenChih-Chia HuangChurng-Ren Chris WangJong-Kai HsiaoMeng-Lin LiPi-Tai Chou Infrared-active quadruple contrast FePt nanoparticles for multiple scale molecular imaging. Biomaterials 2016, 85, 54-64, 10.1016/j.biomaterials.2016.01.053.
- 143. Nune, S.K.; Gunda, P.; Thallapally, P.K.; Lin, Y.-Y.; Laird Forrest, M.; Berkland, C.J.; Nanoparticles for biomedical imaging. Expert Opin. Drug Del 2009, 6, 1175–1194.
- Hak Soo Choi; John V. Frangioni; Nanoparticles for Biomedical Imaging: Fundamentals of Clinical Translation. Molecular Imaging 2010, 9, 7290, 10.2310/7290.2010.00031.
- Félix Lussier; Vincent Thibault; Benjamin Charron; Gregory Q. Wallace; Jean-Francois Masson; Deep learning and artificial intelligence methods for Raman and surface-enhanced Raman scattering. TrAC Trends in Analytical Chemistry 2020, 124, 115796, 10.1016/j.trac.2019.115796.
- Wei Hu; Sheng Ye; Yujin Zhang; Tianduo Li; Guozhen Zhang; Yi Luo; Shaul Mukamel; Jun Jiang; Machine Learning Protocol for Surface-Enhanced Raman Spectroscopy. The Journal of Physical Chemistry Letters 2019, 10, 6026-6031, 10.1021/acs.jpclett.9b02517.
- Christopher T. Mandrell; Torrey E. Holland; James F. Wheeler; Sakineh M. A. Esmaeili; Kshitij Amar; Farhan Chowdhury; Poopalasingam Sivakumar; Machine Learning Approach to Raman Spectrum Analysis of MIA PaCa-2 Pancreatic Cancer Tumor Repopulating Cells for Classification and Feature Analysis. Life 2020, 10, 181, 10.3390/life10090181.
- Phuong H. L. Nguyen; Brandon Hong; Shimon Rubin; Yeshaiahu Fainman; Machine learning for composition analysis of ssDNA using chemical enhancement in SERS. Biomedical Optics Express 2020, 11, 5092-5121, 10.1364/boe.397616.
