Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 2 by Vicky Zhou and Version 1 by Francisco Tustumi.
Achalasia is an esophageal motor disease characterized by the lack of relaxation of the lower esophageal sphincter (LES) and aperistalsis. The first time this condition was described was in 1672 by Sir Thomas Willis, but the "achalasia" term was only created in 1929 by Hurst and Rake, meaning "no relaxation".
- achalasia
- esophagus
- squamous cell carcinoma
- carcinoma
- neoplasm
1. Introduction
The main mechanism for which achalasia appears is due to neuronal degeneration of the myenteric plexus, although the reason for it to happen is still uncertain [1]. Reduced density of Cajal cells in the gastroesophageal junction, the gastrointestinal pacemakers, is another mechanism that may be related in some patients [2].
Achalasia is more often seen in South American countries due to its association with Chagas disease [3]. Patients with achalasia can initially show dysphagia, recurrent chest pain, regurgitation, and weight loss [4]. Usually, symptoms are nonspecific, and there is a significant delay in diagnosis, so the majority of the patients begin their treatment in advanced stages of the disease, already presenting malnutrition, underweight, and recurrent pneumonia [4]. Manometry is the gold-standard diagnostic test for achalasia. The typical findings are incomplete relaxation of LES and uncoordinated esophageal body contractions. Frequently, LES is hypertensive [5]. Barium swallow tests reveal a dilated esophagus, the reason for which achalasia is often called megaesophagus. Besides, barium swallow tests may reveal uncoordinated esophageal contractions and a narrowing at the gastroesophageal junction [6]. Patients with achalasia have an increased risk for developing esophageal cancer [7][8].
Fagge was probably the first author to point out a possible association between achalasia and cancer in 1872 [9]. He described a case of a benign esophageal condition associated with a tumor. Sato et al. [10], analyzing a sample size of 2714 achalasia patients, evidenced that the risk of developing cancer was 16.7 times higher for men with achalasia and 8.8 times higher for women with achalasia.
When patients start to have symptoms of cancer, the tumor is usually already advanced. The symptoms of achalasia and esophagus cancer are similar, and the patient may undervalue the symptoms. Additionally, esophageal-related symptoms depend on the esophagus distension and longitudinal contraction of esophageal smooth muscle, and in achalasia, the esophagus is naturally enlarged [11]. In this way, achalasia patients may present only mild esophageal symptoms, even with a severe condition within it [12]. For the carcinoma to overlap the symptoms of achalasia, it must be large to obstruct a dilated esophagus [13]. Due to the delay in the differential diagnosis, many patients end up having a late cancer diagnosis, and the locally advanced disease leads to poor prognosis of these patients, often making the curative treatment unfeasible [8].
2. The Mechanisms for the Association of Cancer and Esophageal Dysmotility Disorders
Achalasia is a risk factor for developing cancer, including squamous cell carcinoma and adenocarcinoma. Figure 1 schemes the mechanism for DNA damage and mutations for cancer development in achalasia. Additionally, other esophageal dysmotility disorders mimicking achalasia symptoms may be associated with other malignant-related conditions, such as paraneoplastic manifestations and familial KIT-gene mutation.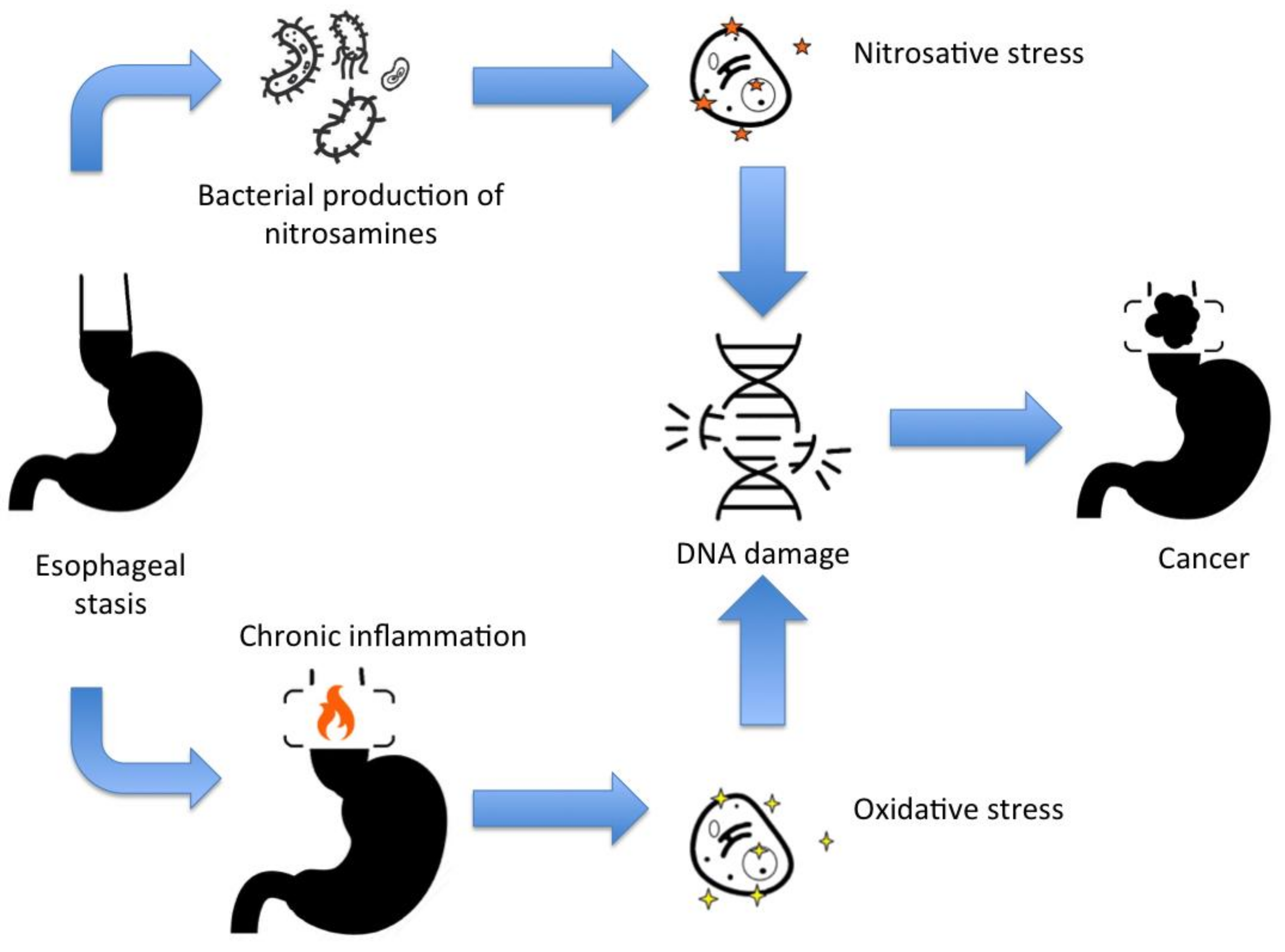 Figure 1. The main mechanisms for DNA damage and cancer development in achalasia.
Figure 1. The main mechanisms for DNA damage and cancer development in achalasia.References
- Lichtenstein, G.R. Achalasia. Gastroenterol. Hepatol. 2017, 13, 388.
- Kilic, A.; Luketich, J.D.; Landreneau, R.J.; Owens, S.R.; Krasinskas, A.M.; Schuchert, M.J. Alterations in the density of interstitial cells of Cajal in achalasia. Dig. Dis. Sci. 2008, 53, 1488–1492.
- Herbella, F.A.; Aquino, J.L.; Stefani-Nakano, S.; Artifon, E.L.; Sakai, P.; Crema, E.; Andreollo, N.A.; Lopes, L.R.; de Castro Pochini, C.; Corsi, P.R.; et al. Treatment of achalasia: Lessons learned with Chagas’ disease. Dis. Esophagus 2008, 21, 461–467.
- Blam, M.E.; Delfyett, W.; Levine, M.S.; Metz, D.C.; Katzka, D.A. Achalasia: A disease of varied and subtle symptoms that do not correlate with radiographic findings. Am. J. Gastroenterol. 2002, 97, 1916–1923.
- Müller, M. Impact of high-resolution manometry on achalasia diagnosis and treatment. Ann. Gastroenterol. Q. Publ. Hell. Soc. Gastroenterol. 2015, 28, 3.
- Ott, D.J.; Richter, J.E.; Chen, Y.M.; Wu, W.C.; Gelfand, D.W.; Castell, D.O. Esophageal radiography and manometry: Correlation in 172 patients with dysphagia. Am. J. Roentgenol. 1987, 149, 307–311.
- Urach, D.R.; Tomlinson, G.A.; Harnish, J.L.; Martino, R.; Diamant, N.E. A measure of disease-specific health-related quality of life for achalasia. Am. J. Gastroenterol. 2005, 100, 1668–1676.
- Tustumi, F.; Bernardo, W.M.; da Rocha, J.R.; Szachnowicz, S.; Seguro, F.C.; Bianchi, E.T.; Sallum, R.A.; Cecconello, I. Esophageal achalasia: A risk factor for carcinoma. A systematic review and meta-analysis. Dis. Esophagus 2017, 30, 1–8.
- Fagge, C.H. A case of simple stenosis of the oesophagus followed by epithelioma. Guy’s Hosp. Rep. 1872, 171, 413.
- Sato, H.; Terai, S.; Shimamura, Y.; Tanaka, S.; Shiwaku, H.; Minami, H.; Sato, C.; Ogawa, R.; Yokomichi, H.; Inoue, H. Achalasia and esophageal cancer: A large database analysis in Japan. J. Gastroenterol. 2021, 56, 360–370.
- Miwa, H.; Kondo, T.; Oshima, T.; Fukui, H.; Tomita, T.; Watari, J. Esophageal sensation and esophageal hypersensitivity-overview from bench to bedside. J. Neurogastroenterol. Motil. 2010, 16, 353.
- Tustumi, F.; Morrell, A.L.; Szor, D.J.; Dias, A.R. Achalasia: A mechanical and sensitivity disorder. United Eur. Gastroenterol. J. 2020, 8, 1126–1127.
- Meijssen, M.A.; Tilanus, H.W.; Van Blankenstein, M.; Hop, W.C.; Ong, G.L. Achalasia complicated by oesophageal squamous cell carcinoma: A prospective study in 195 patients. Gut 1992, 33, 155–158.
- Gillies, C.L.; Farrukh, A.; Abrams, K.R.; Mayberry, J.F. Risk of esophageal cancer in achalasia cardia: A meta-analysis. JGH Open 2019, 3, 196–200.
- Sterne, J.A.; Gavaghan, D.; Egger, M. Publication and related bias in meta-analysis: Power of statistical tests and prevalence in the literature. J. Clin. Epidemiol. 2000, 53, 1119–1129.
- Torres-Aguilera, M.; Troche, J.M. Achalasia and esophageal cancer: Risks and links. Clin. Exp. Gastroenterol. 2018, 11, 309.
- Boller, D.A.; Borovicka, J.; Studer, R.; Zuercher, U.; Kradolfer, D.; Neuweiler, J.; Spieler, P.; Schoenegg, R.; Grossenbacher, R.; Meyenberger, C. Lugol chromoendoscopy combined with image cytometry and p53 LOH in patients at risk for esophageal squamous cell carcinoma. Gastrointest. Endosc. 2006, 63, AB130.
- Gockel, I.; Exner, C.; Junginger, T. Morbidity and mortality after esophagectomy for esophageal carcinoma: A risk analysis. World J. Surg. Oncol. 2005, 3, 1–8.
- Kennovin, G.D.; Hirst, D.G.; Stratford, M.R.; Flitney, F.W. Inducible nitric oxide synthase is expressed in tumor-associated vasculature: Inhibition retards tumor growth in vivo. Biol. Nitric Oxide Part 1994, 4, 473–479.
- De Wilt, J.H.; Manusama, E.R.; Van Etten, B.; Van Tiel, S.T.; Jorna, A.S.; Seynhaeve, A.L.; ten Hagen, T.L. Nitric oxide synthase inhibition results in synergistic anti-tumour activity with melphalan and tumour necrosis factor alpha-based isolated limb perfusions. Br. J. Cancer 2000, 83, 1176–1182.
- Orucevic, A.; Lala, P.K. Effects ofNG-Nitro-L-arginine Methyl Ester, an Inhibitor of Nitric Oxide Synthesis, on IL-2-Induced LAK Cell Generationin Vivoandin Vitroin Healthy and Tumor-Bearing Mice. Cell. Immunol. 1996, 169, 125–132.
- Surh, Y.J.; Kundu, J.K.; Na, H.K.; Lee, J.S. Redox-sensitive transcription factors as prime targets for chemoprevention with anti-inflammatory and antioxidative phytochemicals. J. Nutr. 2005, 135, 2993S–3001S.
More
