Testosterone derivatives and related compounds (such as anabolic-androgenic steroids—AAS) are frequently misused by athletes (both professional and amateur) wishing to promote muscle development and strength or to cover AAS misuse.
- androgen receptor
- antiandrogens
- aging
- longevity
- medicinal natural products
1. Introduction
T and related compounds primarily act as androgens, promoting the development and maintenance of male sex characteristics such as maturation of the sex organs, voice deepening, and growth of facial and body hair. They also have an anabolic activity that promotes the storage of protein and stimulates the growth of bone and muscles, and these functions are especially important from a medicinal standpoint [1]. Indeed, tremendous efforts have been put into developing agents with increased anabolic activity such as the recently discovered selective androgen receptors modulators (SARMs). However, there is still no single anabolic molecule from which the androgenic activity has been fully eliminated. T and other AAS still find their use in the treatment of a wide range of human diseases, including hypogonadism, male sexual impotence, and some types of breast cancer in women. They are also of value in various types of wasting syndromes, for example in patients suffering from acquired immune deficiency syndrome (AIDS) anorexia, or alcoholism, and for those with severe burns, muscle, tendon or bone injury, osteoporosis, certain types of anemias, and hereditary angioedema [2]. T has also recently been discussed in connection with longevity. As a person ages, their physiological levels of T decrease. The T decline has been associated with aging symptoms such as hypertension, obesity, diabetes, overall fatigue, depression, and cognitive decline [2][3]. The current trend in some countries is to use T with its pleiotropic effects to combat several age-related changes, rather than a combination of drugs each treating one symptom. Supplementation with T or related compounds, however, may cause serious adverse effects, including skin disorders, hepatotoxicity (especially true for the orally-active T derivatives), altered blood lipid profiles, hypertension, cardiovascular conditions, kidney disorders, behavioral changes, and reproduction disorders [4]. Regardless of their safety and side-effect profile, T and its analogues, at the correct formulation and dose for the appropriate condition, may still offer several beneficial pharmacological responses and may be considered very valuable pharmaceutical agents.
One of the biggest current problems associated with AAS is that there has been an increasing number of recent reports of AAS abuse by non-professional athletes, mostly young people seeking to improve performance, build muscle and stamina, and have a great looking body [5][6]. Apart from the aforementioned side effects, AAS use may lead to withdrawal symptoms after these drugs have been discontinued. These symptoms are very similar to those observed in subjects with age-related T decline, including increased fat storage, loss of muscle mass and bone strength, mood swings, irritability, extreme fatigue, restlessness, and depression. Thus, for many users, the only way to overcome these symptoms is to start taking AAS again, and ultimately, they become addicted to these drugs (however, it is a relatively special type of addiction that is different from other drugs). As there are some indications that the abuse of AAS by amateur athletes is increasing, this may pose a challenge to the health care system and addiction centres.
2. Available Testosterone Analogues
Table 1. Medicinal use, usual routes of administration, and available forms of compounds discussed in this entry.
| Compound | Main Areas of Medicinal Application | Usual Route of Administration/Available Forms |
|---|---|---|
| AAS | ||
| Testosterone | Male hypogonadism, oestrogen-dependent breast cancer in women, adjunct to hormone replacement therapy in menopausal women (to improve libido), testosterone replacement therapy (TRT) | Transdermal (patch, gel, cream), oral tablets (undecanoate ester), buccal, sublingual, and intranasal formulations, subcutaneous implants, and various esters for intramuscular injection (caproate, cypionate, decanoate, enanthate, isobutyrate, phenylpropionate, propionate, undecanoate) |
| Dihydrotestosterone (DHT; androstanolone) | Hypogonadism, gynecomastia, breast cancer (discontinued in some countries) | Transdermal gel, buccal and sublingual formulations, enanthate, propionate, and valerate esters (intramuscular injection) |
| Methyltestosterone | Delayed puberty, hypogonadism, cryptorchidism, erectile dysfunction, menopausal symptoms (osteoporosis, hot flashes, to improve libido), postpartum breast pain and engorgement, breast cancer in women | Oral tablets, buccal and sublingual formulations |
| Methandriol | Breast cancer in women (now discontinued in most countries) | Oral tablets or propionate and bisenanthoyl acetate esters (intramuscular injection) |
| Boldenone | Wasting syndrome, osteoporosis (now discontinued in most countries) | Undecylenate ester (intramuscular injection) |
| Fluoxymesterone (halotestin) | Hypogonadism, delayed puberty in males, breast cancer in women, some types of anemia | Oral tablets |
| Metandienone (dianabol®) | Hypogonadism (now discontinued in most countries) | |
| Drostanolone | Breast cancer (now discontinued in most countries) | Propionate ester (intramuscular injection) |
| Methenolone | Bone marrow failure-associated anemia, wasting syndromes, osteoporosis, sarcopenia | Acetate (orally active) and enanthate esters (intramuscular injection) |
| Oxandrolone | Osteoporosis-derived pain, weight loss, protein catabolism, AIDS-induced wasting, alcoholic hepatitis, severe burns, anemia, hereditary angioedema, Turner syndrome, hypogonadism, idiopathic short stature | Oral tablets |
| Oxymetholone | Anemia, osteoporosis (largely discontinued for these conditions), muscle wasting, AIDS wasting syndrome | |
| Stanozolol | Anemia, osteoporosis, burns, skeletal muscle injury (largely discontinued for these conditions), hereditary angioedema | Aqueous suspensions (intramuscular injection) or oral tablets |
| Turinabol | Osteoporosis (now discontinued in most countries) | Acetate ester (intramuscular injection) or oral tablets |
| Nandrolone | Burns, breast cancer, anemia, osteoporosis, HIV-induced wasting (now discontinued in most countries) | Decanoate and phenylpropionate esters (intramuscular or subcutaneous injections) or sulfate (eye drop formulation) |
| Ethylestrenol | Wasting syndromes, osteoporosis-associated pain, burns, severe injuries, various types of anemias, conditions of veins and arteries, arthritis, short stature (now discontinued in most countries) | Oral tablets |
| Norethandrolone | Wasting syndromes, burns, severe injuries, various types of anemias (now discontinued in most countries) | |
| Trenbolone | Wasting syndromes (now discontinued in most countries) | Acetate and hexahydrobenzylcarbonate esters (intramuscular injection) |
| T and Nandrolone Prodrugs | ||
| Androstenediol | To raise T levels (never marketed for medicinal use) | Oral tablets |
| Androstenedione | ||
| 19-nor-5-androstenediol | ||
| 19-nor-4-androstenediol | ||
| 19-nor-5-androstenedione | ||
| 19-nor-4-androstenedione | ||
| Dehydroepiandrosterone (DHEA) | To raise T levels (never marketed for medicinal use; though it is available as a dietary supplement) | |
| Experimental AAS | ||
| Trestolone | Under development as a male contraceptive, for TRT, and hypogonadism | Subcutaneous implants, intramuscular injection (acetate ester) |
| Dimethandrolone | Under development as a male contraceptive and for TRT | Oral tablets |
| Designer Steroids | ||
| 1-testosterone | Never marketed for medicinal use | Intramuscular injections |
| Methasterone | Oral tablets | |
| Desoxymethyltestosterone | ||
| Tetrahydrogestrinone | Oral tablets, intramuscular injection | |
| Norboletone | ||
| Metribolone | ||
| Methylstenbolone | ||
| SARMs | ||
| Enobosarm | Under development for cancer-related wasting, sarcopenia, breast cancer, osteoporosis, and stress urinary incontinence in menopausal women | Oral tablets |
| Ligandrol | Under development for wasting syndrome and osteoporosis | |
| BMS 564929 | Under development for wasting syndrome, osteoporosis, diabetes, hypertension, reduced libido, depression | |
| AC-262356 | In pre-clinical development | |
| LGD-2226 | ||
| LGD-3303 | ||
| S-40503 | ||
| S-23 | ||
| S-1 |
2.1. Analogues of Testosterone with Agonistic Activity
19
4
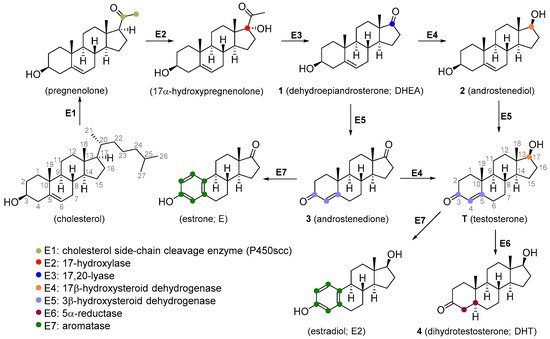
Figure 1.
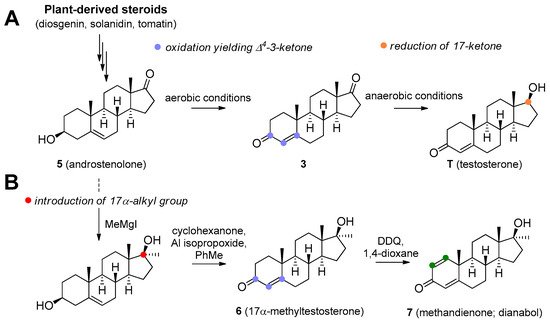
Figure 2.
A
17
B
1
2
3
4
5
Dioscorea
Trigonella foenum-graecum
Solanum
Lycopersicon
5
6
7
T is not orally active as it readily undergoes hepatic metabolism (though there are some oral forms, such as undecanoate ester; attachment of a very long-chain ester at 17β position increases oral activity). The usual mode of administration includes injections or subcutaneous implants of its ester forms. Dermal patches are available and this is the method of choice for the treatment of hypogonadism [1][7]. T is also available in other therapeutic modalities, including topical hydroalcoholic gels [8], buccal [9], sublingual [10], and intranasal formulations [11].
4
6
Figure 2) is an orally active agent that is used for hypogonadism, erectile dysfunction, suppression of menopausal symptoms (hot flashes, osteoporosis, low libido), and in the treatment of breast cancer [13][14]. Mesterolone (8,
4 double bond and is also orally active. Its androgenic activity is slightly higher than the anabolic effect, and it is of value for increasing low T levels, but it is hardly ever prescribed now [15][16]. Mesterolone has very low to no oestrogenic activity and shows only slight hepatotoxicity. The introduction of a methyl group in position 1α leads to an increased oral activity. Oral activity may also be achieved by the introduction of the 17α-alkyl group (as seen in methyltestosterone). This modification leads to reduced metabolism in the liver and increased bioavailability, but hepatotoxicity is also increased [17][18]. Methandriol (
9
Figure 3) is available in both oral and injectable forms as dipropionate, propionate, and bisenanthoyl acetate esters. It has almost exclusively been used in the treatment of breast cancer in women [19][20].
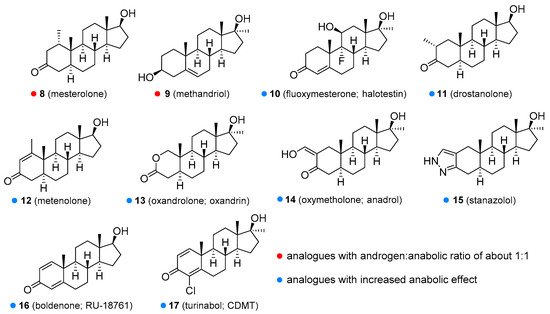
Figure 3.
1
2
9
10
11
12
4
2
2
19
®
7
10
11
12
13
2
14
15
Figure 3) has been used in the treatment of osteoporosis [36] and is currently being evaluated as a treatment for hereditary angioedema [37][38]. Unlike other anabolic steroids, it is not available in esterified form but as an aqueous solution or tablets. The use of this drug in humans was discontinued in many countries, but it is still widely used in veterinary medicine for the same conditions as in humans. Boldenone (
16
1-testosterone. It is available as a undecylenate ester and is exclusively used in veterinary medicine [16][39]. Among other activities, it increases appetite and also stimulates the release of erythropoietin. Boldenone has relatively low hepatotoxicity, androgenic potency and does not interact with receptors for progesterone. Turinabol (chlorodehydromethyltestosterone;
17
19
18
Figure 4) is available as decanoate and phenylpropionate esters, but these are not orally active and must be administered via subcutaneous or intramuscular injection. They are used in the treatment of anemia, severe burns, wasting syndrome in patients suffering from AIDS [41], osteoporosis [42], or breast cancer [39][43]. It is also available in the form of eye drops as nandrolone sulfate [44]. Ethylestrenol (
19
Figure 4) lacks the 3-keto group. It was used for muscle promotion and weight gain, in treatment of bone pain and osteoporosis, as an adjunct therapy for corticosteroid-induced wasting and severe injuries, arthritis, aplastic anemia and anemia of chronic kidney disease [45][46][47], conditions of arteries and veins (e.g., thrombosis, Behçet’s disease, Raynaud’s disease, Degos disease) [48][49][50], and short stature in youths [51]. It is no longer used medicinally but is still available for veterinary use. Norethandrolone (
20
21
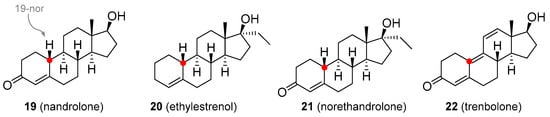
Figure 4.
22
