Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 2 by Catherine Yang and Version 1 by Artur Y. Prilepskii.
Nanomaterials are proven to affect the biological activity of mammalian and microbial cells profoundly. It has been revealed that the shape of the nanomaterial plays a crucial role. This aentrticley reviews the influence of nanomaterial shape on various biological activities of mammalian and microbial cells, such as proliferation, differentiation, and metabolism.
- nanoparticle
- shape
- microbial cell
- mammalian cell
1. Introduction
Nanomaterials are widely used in medicine as platforms for developing advanced drug delivery systems with controllable drug loading efficacy, biodistribution and cell/tissue targeting, therapeutic actions, cytotoxicity, selectivity, imaging ability, blood circulation time, half-life, and excretion. It is widely thought that all these properties are connected only with nanomaterial surface chemistry, total surface area, hydrodynamic size, the loaded drug, etc. The phenomenon of nanomaterial shape is usually considered in the context of systemic toxicity, biodistribution, and blood circulation time. However, its influence on the biological activities of mammalian and microbial cells has been reported in many articles, such as shape-induced directed differentiation [1], cellular death via apoptosis [2], necrosis [3], gene transfection and transfer [4], metabolism alteration [5], and other processes. These effects arise from different surface areas, uptake level, protein corona, physical disruption of the cell membrane, and particles’ wettability and surface curvature [6]. Moreover, the shape control of particle morphology demonstrates the importance of the ratio of the different crystal facet surface areas, which have a different dissolution rate in solution [7].
The shape control of nanomaterials represents a major field in materials science, and a large number of approaches exist towards the production of sphere- [8], ellipsoid- [9], dumbbell- [10], cube- [11], polyhedra- [12], rod- [13], urchin- [14], star- [15], chain- [16], ribbon- [17], hollow [18], prism- [19], and hexagon-shaped [20] nanomaterials. Nanomaterial shape control stems from effects such as selective adsorption growth of reactive facets; spontaneous aggregation and agglomeration; seeded growth on particularly-shaped templates; controllable crystal fusion via orientation attachment; self-assembly via selective strong interactions, e.g., chemical and hydrogen bonds; and Ostwald ripening directed at free surface energy minimization. Several synthetic approaches exist toward synthesizing nanomaterials with controlled morphologies, e.g., using polymeric additives, surface-capping surfactants, solvo/hydrothermal approaches, etc.
We focus on the mechanisms involved in nanomaterial shape control, synthetic approaches toward its production, and the influence of nanomaterial shape on various biological activities of mammalian and microbial cells, e.g., proliferation, differentiation, and metabolism, as well as reviewing the prospects of this emerging field.
2. Biological Effect and Application
2.1. Mammalian Cells
2.1.1. Cell Death
Apoptosis
Using nanoparticles to kill cells is one of the most common tasks. Nanoparticles are usually considered for use as drug carriers and heat or light conductors. However, even in the case of drug delivery and retention, the shape is of great importance [75][21]. Nevertheless, nanoparticles themselves can cause cell death, and not because of their toxicity. The following are studies showing the difference in the cellular response due to the various nanoparticle shapes.
Gold nanoparticles were among the first to be used in medicine. Widely developed methods for controlling the shape of gold nanoparticles have made it possible to obtain various shapes and sizes. However, when considering gold nanoparticles, it is challenging to separate shape effects from the size or surface functionalization effects. Thus, one of the most common methods of shape control is using surfactants. In [2], gold spheres, rods, and stars were compared. Anisotropic rod and star nanoparticles were obtained using CTAB, which is known for its cytotoxicity. The authors recorded a significant increase in the level of pro-apoptotic protein (Bax) and a decrease in that of anti-apoptotic protein (Bcl-2) for star and rod shapes. The authors also recorded the induction of autophagy in tested cells. In general, osteosarcoma MG-63 and 143B cells were more susceptible to nanoparticles than normal osteoblast hFOB 1.19 cells.
In most cases, increased toxicity (and subsequently, induction of cell death) is associated with “sharp” shapes [76][22]. However, some authors report opposite results. For instance, in a work by Favi et al., commercially purchased citric-stabilized gold nanospheres were found to be more toxic than gold nanostars. In that work, a star-looking shape was obtained using HEPES buffer as a reducing agent. On the other hand, Enea et al. reporting that nanospheres and nanostars capped with 11-mercaptoundecanoic acid (MUA) had similar cytotoxic properties [77][23]. In this case, the aspect of the shape fades into the background. However, in a work by Sultana et al. [78][24], flower-like gold nanoparticles and gold nanospheres with the same PEG shell and size were found to have different uptake and cytotoxic profiles. Authors associate the increased toxicity of gold nanoflowers with high surface roughness, mediating uptake and disturbance of the cytoskeleton.
Mesoporous silica nanoparticles (MSNs) have long been used for drug delivery and theranostics applications [79][25]. Little attention is usually paid to their shape since widespread protocols allow one to obtain mostly spherical particles. It was shown that silica nanorods with a length of about 450 nm (NLR450) are absorbed by cells much faster than rods with a length of 240 nm (NSR240) or spherical 100-nm particles (NS100) [80][26]. Consequently, NLR450 caused a greater cytotoxic effect, disturbances in the cytoskeleton structure and an increase in the number of apoptotic cells by 10% compared to other tested shapes. The authors also noted the influence of nanoparticles with various shapes on the migration and adhesion of cells, which may be associated with a cytoskeleton disruption.
Another widespread material, titanium dioxide, was also found to cause cytoskeleton damage in a shape-dependent manner [81][27]. Spherical TiO2 particles were discovered to penetrate the blood–brain barrier, probably through the disturbance of F-actin fibers. However, in addition to the shape, these particles also had a different crystalline phase.
When mentioning the crystalline phase, it should be noted that some authors equate the effect of the shape of particles and their crystalline phase [21][28]. It can be sometimes true; however, that the crystalline phase has more to do with surface morphology. For instance, TiO2 is known to have two crystalline phases—anatase and rutile—in addition to an amorphous form. Anatase was shown to have pronounced cytotoxic effects that were associated with the density of defect sites [82][29]. The anatase phase was also more toxic than rutile in a similar work, even though rutile particles were rod-like and sharp [83][30]. This observation suggests the necessity of the careful consideration of shape-dependent effects, especially in the context of inorganic particles.
Another example, in which a dependence of one kind can be mistaken for a dependence of another kind, is given in the work of Xu et al. [84][31]. Hydroxyapatite particles (HAP) with four different shapes were tested for general cytotoxicity manifestations (MTT assay), ALP activity, apoptosis assay (p53 and cytochrome C expression), and ROS generation. Almost all effects were in close correlation with particle-specific surface area but not with shape.
Usually, the shape of nanoparticles is discussed in the context of inorganic particles since a large number of approaches have been developed for them to create spatially anisotropic shapes. However, organic particles also have variations in shapes other than spherical. Biocompatible PLGA and PEG are some of the most common polymers used to create particles. However, it was shown that changing the shape of a particle from spherical to elongated leads to a significant increase in the particles’ cytotoxicity and induction of apoptosis [85][32]. The primary mechanism is assumed to be the rupture of lysosomes, followed by the launch of a cascade through caspase-3 and DNA damage.
Another example of organic particles with shape-dependent cytotoxicity is poly(3,4-ethylenedioxythiophene) (PEDT) polymer [86][33]. Oh et al. compared three types of PEDT nanomaterials with an average diameter of 55 nm. PEDT-1 was sphere-like, PEDT-2–rod-like, and PEDT-3 took the form of 1350 nm-long tubes. It was found that all samples caused a dose-dependent increase in LDH release in normal cells (IMR90 and J774A.1) by 5–50%. The ATP amount decreased in the same manner by 5–70%. The highest effect was observed for the smallest sphere-like particles. Furthermore, they caused a significant increase in the number of apoptotic cells (by 30%). However, the longest particles lead to considerable elevation of IL-1, IL-6, and TNF-α levels after 24 h of incubation.
Caspase-3 levels were elevated upon incubation of PC12 cells with graphene nanostructures of different dimensions [87][34]. The authors compared graphene layers (G) and carbon nanotubes (SWCNT), showing that SWNCT leads to LDH leakage, generation of ROS, and subsequent induction of apoptosis. The sharp shape of SWCNT was listed as the main reason for increased cytotoxicity.
Aluminum salts have long been widely used as an adjuvant for vaccines, whereas aluminum oxide (alumina) was used as an adsorbent. At the same time, methods for the synthesis of alumina usually include hydrothermal and ultrasonic-assisted approaches, in which precise control of the shape is not always possible [88][35]. Dong et al. managed to produce two types of alumina nanoparticles in the shapes of flakes and rods and studied them in detail using a metabolomics approach [89][36]. The main focus of the work was the study of brain cells, including primary cultures, which is especially important in the context of the medical use of alumina. Rod-like particles have been shown to have greater uptake ability, connected with increased cytotoxicity in astrocytes. Apoptotic markers and pro-inflammatory cytokine levels were increased in a dose-dependent and shape-dependent manner (similarly to the cytotoxic results). A careful analysis of 66 metabolites showed that nanorods cause more significant metabolic changes (21 unique differential metabolites (DMs) vs. 15 for nanorods and nanoflakes, respectively). These DMs mainly included amino acids, lipids, and carbohydrates.
Necrosis
In some cases, the process of cell death upon interaction with nanomaterials can follow the necrosis pathway [90][37]. From the therapeutic point of view, such an outcome is unfavorable, leading to inflammation [91][38]. However, in the context of screening potential nanomaterials for biomedical applications, it is imperative to conduct additional research for signs of necrotic cell death.
One of the most frequently used approaches to distinguish apoptotic and necrotic pathways is staining with FITC-labeled annexin. Using this technique, Huang et al. made a distinction between four shapes of hydroxyapatite nanoparticles (HAP) [3]. The authors found that cytotoxicity (and, similarly, the percentage of necrotic cells) rose in the order of plate > sphere > needle > rod. The difference between HAP plates and rods in the number of necrotic cells was almost twofold (17.13% vs. 9.67%, respectively). The impact of HAP on mitochondrial membrane potential and lysosome integrity followed the same shape-dependent trend. The authors concluded that the primary influence on cytotoxicity was exerted by the surface area, conductivity, and zeta potential of nanoparticles. However, cellular uptake of HAP was higher for the most toxic shapes—a fact that should be kept in mind.
The aggregation state of nanomaterials plays an essential role in cytotoxicity [92][39]. Lee et al. [93][40] showed that iron oxide rods that remain stable under physiological condition were more toxic than aggregated nanospheres. The number of necrotic cells was increased after incubation with rod-shaped particles by about 20% at a 200 μg/mL concentration. However, considering important observations connected with ROS generation and aggregation state, the straight correlation between shape and cytotoxicity is not clear.
Ferroptosis
In discussing the possible effect of nanoparticles on cell viability, the phenomenon of ferroptosis cannot be ignored. This form of cell death exists in many cancer cells and therefore attracts particular interest in cancer treatment [94][41]. The main object of studies in nanotechnology has become iron-containing nanoparticles, since it is iron that participates in the Fenton reaction, leading to ferroptosis. The primary mechanism involves the release of iron ions Fe2+ or Fe3+ after the nanoparticles enter the lysosomes or an acidic environment. The ion release process is directly related to the surface area of the nanoparticles, which is sometimes associated with the shape. Smaller particle sizes are preferable since they tend to generate more iron ions. Ferumoxytol is used as a contrast for MRI [95][42]. It is a stabilized magnetite (Fe3O4) nanoparticles with a size of about 50 nm [96][43]. This size range is greatly suited for cellular uptake. It has been shown that ferumoxytol can cause the polarization of macrophages in cancerous tumors, reducing their growth [97][44]. Tumor size was halved on day 21 in the ferumoxytol-treated group. To note, the polarization of macrophages plays an essential role in the treatment and diagnosis of cancer, which was the subject of our recent review [97][44]. Amorphous forms of iron also cause Fe2+ release and can be used to trigger the Fenton reaction [98][45]. The advantage of the amorphous form over the crystalline one was that, under the action of an acidic environment, the release of ions from the amorphous form was significantly higher. In the first 6 h, the release from the amorphous form was 100%, whereas for the crystalline form, it was only 20%.
Combined strategies, such as drug loading, are successfully implemented along with iron-oxide nanoparticles [99][46]. The delivery of hydrogen peroxide can be considered one of the relevant strategies [100][47]. A complex structure, consisting of a PLGA shell incorporated with magnetite nanoparticles and loaded with H2O2, was developed. The release of H2O2 was carried out using ultrasound. Tumor size was reduced by eight times compared with the control group on the 22nd day of the experiment.
Non-iron-containing engineered nanomaterials have also been reported to cause ferroptosis. A relatively large amount of papers have been published recently on manganese oxide (MnO2) particles [101,102,103][48][49][50]. Though no shape-dependent activity has been reported, the investigated particles possessed developed surfaces (nanoflowers or nanobubbles). Another example is two-dimensional transition metal dichalcogenides. WS2 and MoS2 nanosheets were shown to cause ferroptosis in epithelial (BEAS-2B) and macrophage (THP-1) cells [104][51]. However, cell death was connected with surface vacancy, not the 2D nature of the nanomaterials.
2.1.2. Disturbance of Cell Function
Alterations of cell proliferation are not always accompanied by cell death and should be considered separately. In this section, we describe and discuss the shape-assisted alteration of cellular metabolism, genotoxicity, mammalian cell differentiation, as well as the influence of external stimuli on these processes (Figure 31).
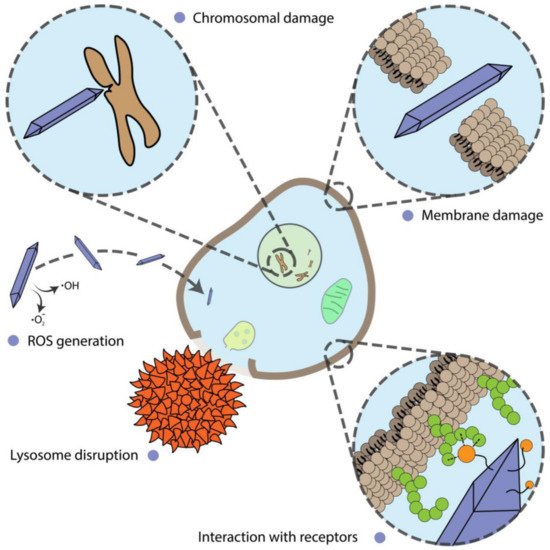

Figure 31.
Different mechanisms of nanoparticle action on cells.
Metabolic Alterations
In a typical investigation, various assays are used to assess cell proliferation by measuring cell metabolic activity. In most cases, they do not answer the question of what the exact cause of metabolic alterations is. Nevertheless, there are clear connections between nanoparticle shape and metabolic changes.
Six types of gold nanoparticle shapes were tested using the WST-1 assay on normal and cancer cell lines [5]. Aside from shape differences, spheres were the smallest ones (10 nm). Rods were about 40 nm in length; the rest of the particles exceeded 100 nm. Moreover, rods, prisms, and stars were synthesized in the presence of CTAB. A size-dependent effect was definitely found for rods and spheres, which were the most toxic ones. Shape-dependent behavior was obviously inherent to spiky nanoprisms and nanostars but not nanoflowers, the edges of which were smoother. Additionally, HeLa cells were much more resistant to the toxic effects of nanoparticles than HEK293T cells.
In the study by Arnida et al., several spherical- and rod-like nanoparticles were tested on prostate cancer cell line PC-3 [105][52]. It was found that nanospheres with 30–50 nm size have the highest cell uptake. PEGylation of particles drastically reduces cell uptake. No specific effects were associated with nanoparticle shape.
Research has shown that multiwalled carbon nanotubes (MWCNTs) have a similar structure to asbestos [106][53]. The latter is known for its ability to cause multiple pleural diseases [107][54]. Xu et al. [108][55] proved a hypothesis that the shape of MWCNT plays an important role in lung-associated diseases. Large (rod-like) and short (cotton candy-like) MWCNTs were administered in the rat lungs for 24 weeks. The authors found a shape-dependent formation of foci of mesothelial cell proliferation. Rod-like tubes also contributed to increased proliferating cell nuclear antigen (CD68) and pro-inflammatory cytokine levels.
Rod-shaped cerium oxide nanoparticles caused the same effect on pro-inflammatory cytokine levels [109][56]. The authors compared three sizes of rod-shaped particles (5.5–8.1 nm in diameter and 44–70 nm in length) and two sizes of octahedral-like particles (11.3–16.2 nm). All particles induced concentration-dependent cytotoxicity. All rod samples generate almost twice-higher TNF-α levels compared to octahedral particles. Additionally, the smaller the rods were, the more toxic they were. A similar dependence was found for LDH release. The smallest rod-like particles have the biggest surface area, which may be the reason for higher toxicity due to the release of ions. However, there was no ROS generation detected for all types of particles.
Genotoxicity
Nanoparticles are known to generate reactive oxygen species upon lysosomal or cytoplasmic degradation [110,111,112][57][58][59]. ROS is one of the primary causes of DNA damage [113][60]. However, ROS production can depend not only on chemistry, surface area, and size, but also on nanoparticle shape.
Rod-shaped zinc oxide nanoparticles were found to be significantly more genotoxic than sphere-like nanoparticles, mostly due to elevated ROS generation [114][61]. The authors assumed that increased ROS generation may be associated with a bigger surface area or higher uptake of rod-shaped particles, although no proof was provided.
Mesoporous silica nanoparticles cause genotoxicity in the DT40 cell line in a similar shape-dependent manner [115][62]. Rod-shaped particles were synthesized in the presence of CTAB. However, they were further calcined at 500 °C. In the test for chromosomal aberrations, rod-like particles showed a ~20% increase.
Cell Differentiation
Stem cells are of particular interest in many areas of biomedical research. Getting stem cells to turn into the required cells is challenging. It was found that some nanoparticles can force stem cells to differentiate, although the detailed mechanisms for this are not always known [1]. From the nanotechnology point of view, different nanoparticle shapes can lead to different effects.
Human mesenchymal stem cells (HMSCs) are multipotent cells that can replicate as undifferentiated cells. They have the potential to differentiate into mesenchymal tissues such as bones, muscle, marrow stroma, etc. [116][63]. Differentiation into bones and cartilage is of particular interest because of the prevalence of bone fractures, implanting of metal grafts, and other musculoskeletal system-associated disorders.
The effect of gold nanoparticles on osteogenic differentiation has been known for more than ten years [117,118,119][64][65][66]. However, the shape-dependent effect has not been studied well in detail. Li et al. studied full spectra of gold nanoparticles with three types of shapes (spheres, rods, and stars) and three different sizes [120][67]. The careful selection of particle parameters allowed the authors to distinguish shape effects clearly. Spheres and rods with dimensions close to 50 nm (but not stars) caused the most pronounced effect on the expression of osteogenic markers. Only nanorods with a length of 40 nm were found to be significantly cytotoxic, and therefore downregulated osteogenic markers.
Single- and multi-walled carbon nanotubes were also tested for their ability to alter HMSCs’ differentiation [121][68]. Both types inhibited cell proliferation, differentiation rate, and mineralization. The effect of single-walled CNTs was slightly more pronounced (within a 10% difference).
Ion Channel Disturbance
Though some researchers are not exploring the difference between particle shapes, shape dependence can still arise from external stimuli. For instance, when using an external magnetic field, the shape of the particles must be strictly defined. One of the uses of magnetic particles to control cell biological functions is related to the control of the activity of ion channels. Gregurec et al. [122][69] showed that magnetite nanodiscs with sizes of 98–226 nm could act as transducers of magnetic torque into cell stimuli [123][70]. Under the application of an external magnetic field with low flux and frequency, it could trigger Ca2+ influx.
A similar shape-based approach was described in the work of Lee et al. [124][71]. The authors assembled a complex structure, defined as an m-Torquer, from octahedron magnetite nanoparticles with predefined volume magnetization distributions. The m-Torquer was able to affect mechanosensitive ion channels both in vitro and in vivo.
Membrane Integrity
Although some particles can damage the cell membrane by themselves, sometimes their action can be controlled. In the case of plasmon resonant particles, their behaviour can be regulated by light excitation. The shape of some particles can serve as antennas for converting optical energy into heat [125][72]. Such a structure can allow the penetration of cell membranes in a controlled manner, which can be used for cell transfection. However, even slight alterations in nanoparticle morphology can lead to different responses to light irradiation [126][73].
In contrast to light-induced membrane penetration, Sun et al. used suspended magnetic hedgehog-like pollen grain particle for intracellular drug deliverys based on controlled membrane perforation under the action of a magnetic field [127][74]. Unlike the previous example, Qian et al. studied the interaction of the nickel urchin-like nanoparticle with tumor cells under the action of a rotating magnetic field [128][75]. Their results showed that the urchin-like particles induce a much higher necrosis rate than spherical nickel nanoparticles under the same rotating magnetic field. These examples revealed the potential of magnetic particles with branched structures for intracellular delivery or cell death action. However, these biological effects are closely interconnected and strictly depend on the size and shape of spikes on the particle’s surface and the magnetic field amplitude and frequency.
2.2. Microbial Cells
Microbial cells are one of the most important organisms on Earth. Their great diversity in genetic mechanisms, metabolic pathways, habitats in extreme environments, and survival strategies make them ideal organisms for biotechnology [129][76], molecular biology [130][77] and even in materials science [131][78]. Despite their laboratory and industrial applications [132][79], many diseases in the human body are connected with microbial cells. A comprehensive review describes not only the disease-mediated interaction of microbes and humans [133,134][80][81] but also emphasizes their crucial role in health. Hence, the control of microbial cells for humanity purposes may be helpful from several points: (i) the treatment of bacteria-mediated disease and (ii) microbial cells as factories for the production of valuable substances.
The treatment of bacterial infection and disease for almost one hundred years has been interconnected with antibiotics. Even though antibiotics are common and even traditionally used in therapy, researchers are now pushing back against an antimicrobial resistance wall [135][82]. The broad use and abuse of antibiotics has led to the formation of multidrug-resistant bacteria. The number of species resistant to traditional treatment is increasing rapidly and has become a major global healthcare problem. Over the last few years, the application of nanotechnology in biomedicine has become an alternative strategy for the treatment of disease, especially of microbial cell-mediated infection [136][83]. The nanoparticle platform has been used in drug delivery systems [10,137][10][84] and as part of implants coating against biofilms [138,139][85][86]. These examples expand the potential of synthetic and natural strategies to overcome antibiotic resistance and, consequently, serve as a universal approach for treating microbe-mediated disease.
The microbial cell factory is an alternative way to produce highly beneficial products such as pharmaceutics [140][87], chemicals [141][88], and biofuels [142][89]. The commonly used strategy to advance such systems is metabolic pathway design [141][88] and microbial cell engineering [143][90]. In contrast to treatment, the use of nanoparticles in microbial engineering is less widespread in the literature. Examples include protective nanoshells to overcome conditions in harsh environments [144[91][92],145], the integration of synthetic solar-to-chemical energy transformation pathways in non-photosynthetic organisms [146[93][94],147], and magnetic and light control of metabolic reactions [14,148][14][95]. The synergy of synthetic biology and materials science creates a unique possibility for the novel generation of microbes-based factories with remote control and the on-demand production of valuable products.
Despite the great potential of nanoparticles for biofilm therapy and improving microbial factories, most papers demonstrating the use of spherical nanoparticles focus on surface chemistry rather than the particle shape. To date, materials science allows particles to be designed for particular process-of-interest and creates a myriad of novel applications of nanoparticles in regulating microbes’ biochemistry, arising from nanoparticle shape. In this section, we outline the state of the art in the nanoparticle shape-dependent effect on microbial cells.
2.2.1. Antimicrobial Shape-Dependent Effect
Nanoparticles of different shapes are widely used for the treatment of biofilms, as well as the induction of microbial cell death. Silver nanoparticles represent the majority of nanoparticle-mediated therapy. Ag NPs have received considerable attention due to their wide use as antimicrobial agents, which were shown to be very effective [149][96]. The toxicity of silver nanoparticles is usually associated with ROS generation, membrane damage, and ion release, which induce the dysregulation of DNA synthesis [148][95]. The pioneering work on the shape-dependent toxicity of silver nanoparticles was carried out by Pal et al. [150][97]. Triangular silver nanoplates with a {111} lattice plane have a stronger biocidal action than spherical and rod-shaped nanoparticles. The authors suggested the contribution of facets with distinct activity ({100} < {111}) for different shapes. Cheon et al. studied the shape-dependent toxicity of Ag nanoparticles with spherical-, disk- and plate-like shapes [151][98]. The antimicrobial effect was associated with different ion release kinetics displayed by nanoparticles with different shapes. The most pronounced antimicrobial activity was for spherical Ag NPs; the disk Ag NPs were more toxic than triangular plate Ag NPs. Acharya et al. studied the toxicity of spherical and rod-shaped silver particles [152][99]. They also provided data about the larger bactericidal effects of spheres due to their larger surface area, damaging the cell wall of both Gram-negative and Gram-positive bacteria. Thus, the shape of silver nanoparticles plays a crucial role in bactericidal activity with the dominance of the spherical shape above others.
The use of Au NPs has been documented for diverse applications [153][100] in biological systems due to their unique physio-chemical properties, which can be easily tuned by tuning the chemical routes [125][72]. Au NPs possess the ability to undergo localized surface plasmon resonance (SPR), which depends on the particle’s size, shape, and surface morphology. To date, the bactericidal action of gold nanoparticles as an antimicrobial drug carrier and as a photothermally-activated actuator has been shown [154][101]. Although size plays a crucial role [155][102] in triggering biological effects, it is necessary to consider the shape of the gold nanoparticles. Penders et al. studied the bactericidal performance of spheres, stars, and flowers with similar dimensions [156][103]. It is worth noting that particles’ antimicrobial activity was studied along with their biocompatibility to mammalian cells. The authors found that gold nanoflowers possessed the most promising non-cytotoxic mammalian cell behavior, with the greatest shape-dependent antibacterial activity-promising properties. Meanwhile, the nanospheres had no significant impact on S. aureus viability.
A comprehensive study of gold particle-mediated antimicrobial activity was carried out by Chmielewska and others [157][104]. The effect of Au rods, peanuts, and stars was assessed on a large set of Gram-positive and Gram-negative pathogenic bacteria. The study presented the well-pronounced bactericidal activity of a whole set of particles, primarily caused by ROS generation and related to the destruction of bacterial membranes. The gold peanuts have a prevailing bactericidal effect over other shapes. Hameed et al. demonstrated that gold nanocubes showed the most significant bactericidal properties, even at lower concentrations, against E. coli, P. aeruginosa, and S. aureus, followed by Au NSps (nanospheres) and Au NSts (nanostars) [158][105].
A special role in the literature is allocated for magnetically powered particles commonly based on Fe3O4 [159,160][106][107]. The magnetic field is a promising physical tool to control particle dynamic media due to the absence of energy dissipation in biological media, and the ability to generate heat by magnetic hyperthermia and mechanical force via magnetically induced particle oscillation and rotation under the action of an alternating magnetic field [161,162][108][109]. However, most papers describe the application of isotropic nanosized particles [163][110], as well as the mutual interaction of magnetic nanoparticle results in terms of vortex formation, which is efficiently operated by a magnetic field, and have been focused on accurate therapy [164,165,166][111][112][113] and cargo delivery [167,168][114][115]. Dong et al. designed a magnetic vortex (microswarm) consisting of porous magnetite particles, which, under the action of the rotating magnetic field, could disrupt the biofilm and generate ROS [169][116]. Moreover, the presented microswarm can be transported to confinement and narrow spaces for the effective disruption of biofilms. A similar concept was demonstrated by Hwang et al. [170][117]. Isotropic magnetic nanoparticles with catalytic activity were implemented as the building blocks of a catalytic antimicrobial robot designed as a result of the aggregation of nanoparticle and 3D molds. The robot could completely remove the biofilm without regrowing and be driven to the difficult-to-reach surfaces and even into human teeth.
Another exciting application of magnetic particles is inducing the shape-transformation of liquid metals. Gallium-based alloys remain liquid at room temperature and have a unique set of characteristics and behaviors, which originate from their simultaneous metallic and liquid natures. This has prompted the emergence of applications in various fields in catalysis, physical and synthetic chemistry [18], and biomedicine [171][118]. Elbourne et al. studied the shape transformation of liquid metal nanoparticles under the action of the magnetic field and the application of the designed shapes for the elimination of biofilms [172][119]. The bactericidal action of synthesized magnetic liquid particles was achieved only under the action of rotating magnetic fields, resulting in completely removed biofilms after 90 min of magnetic field exposition. The authors continued their work and used particles to efficiently eliminate multi-species biofilms, characterized as more complex structures due to their pathogenicity and resistance to the treatment and environmental factors [173][120].
Another bactericidal strategy involves the use of nanostructured surfaces inspired by nature. Insects commonly fabricate surfaces with high-aspect-ratio elements to damage the bacterial cell membrane [174][121]. The bactericidal effect depends on various parameters like height, width, spacing, nature, and geometry of the nanostructured surface. The influence of these parameters is the main focus of experimental [175,176][122][123] and theoretical work [177,178,179][124][125][126] and review papers [4[4][127][128],180,181], in which scientist have been looking for the mechanism of bacteria destruction and what exactly causes of cell death. Moreover, these surfaces are used for bactericidal effects and as a surface for improved growth and differentiation of mammalian cells [176,182][123][129]. Precisely, the mechanism of bacteria cell death involves the stretching and rupture of the cell membrane between the pillars, and the pillars are not expected to substantially deflect upon contact with bacteria. These facts have inspired scientist to look towards novel bioinspired methods for fabricating such nanostructured materials and using them in biomedicine. Valiei et al. studied the interaction of zinc oxide nanopillars with bacteria, which caused cell death [183][130]. This is interesting, but the interaction itself does not provide a bactericidal effect. The primary cause of cell death is the moving of the air–liquid interface between the media and the nanopillars. Linklater et al. fabricated vertically aligned carbon nanotubes [184][131] and demonstrated their significant antibacterial properties due to the storage and release of mechanical energy sufficient for bacteria membrane rupture. Taken together, the presented strategy would be helpful for the design of the next generation of implants to avoid the common problems with biofilms and induce the proliferation of mammalian cells.
2.2.2. Shape-Dependent Metabolic Alterations
Microalgae represent one of the most promising organisms for the production of valuable metabolites such as biofuel and novel plant-based drugs [185,186][132][133]. However, large-scale cultivation remains the main obstacle in the development of economically beneficial techniques [185][132]. To date, nanomaterials represent an alternative way to increase the efficiency of algae cultivation. Eroglu et al. synthesized plasmonic nanostructures to enhance the accumulation of microalgal pigment [187][134]. They found that gold nanorods with maximum surface plasmon resonance at 710 nm had the most significant effect on combined pigment content up to 150 μmol m−2 s−1. SPR tuning by nanoparticle shape allows their introduction as optical filters, which could alter the production yield.
Cruces et al. studied the toxicity mechanism of the graphene oxide and multi-walled carbon nanotubes in Microcystic aeruginosa [188][135]. Despite the apparent mismatch in nanomaterial physicochemical properties, the author revealed similar dependencies in the inhibition of photosynthesis esterase activity and growth rate. The effects of metabolism alternation after nanomaterial exposition are mainly associated with interaction-induced alteration of photosynthetic electron transfer. Samei et al. studied the shape- and size-dependent effects of zinc oxide nanoparticles on freshwater microalgae [189][136]. Spherical ZnO NPs acted more destructively (50% cell death) to microalgal cells than nanorods (30% cell death) with the same concentration.
Methane is considered one of the most widely used and important biofuels [190][137]. Metallic and metal-oxide nanoparticles possess great potential to improve biomethane production [191][138]. Ambuchi et al. studied the combination of magnetic nanoparticles with multi-walled carbon nanotubes on biogas and methane generation from sludge with microorganisms, revealing improved biomethanogenesis upon nanomaterial treatment [192][139]. Some authors claimed that a similar effect of nanomaterials on methanogenesis enhancement was achieved by direct interspecies electron transfer due to transition metal oxide catalyst [191,192,193,194,195][138][139][140][141][142]. From this point, the shape tunability allows the control of methane generation in sludge or a microorganism consortium.
Respiration is a crucial metabolic activity, from which almost all energy is derived. Instead of eukaryotic cells, in prokaryotes this is carried out in cell membranes. Thus, the strategy to modulate the bacterial respiration chain does not involve the uptake of nanomaterials. The interaction between nanomaterials and bacteria, which has a high aspect ratio (tubular and rod). requiring extensive contact with bacterial membranes, has been shown to regulate metabolic processes in bacteria, even on the gene expression level [196,197][143][144].
Mortimer et al. studied the carbon nanomaterials and nanoceria influences on Bradyrhizobium diazoefficiens, the essential microorganism for nitrogen fixation in agriculturally beneficial plants [196][143]. They showed that nanomaterials with similar physical properties significantly altered the expression of genes associated with the nodulation competitiveness of B. diazoefficien. Moreover, they also suppressed root exudate-induced gene expression in bacteria, suggesting that nanomaterials may interfere with plant-microbe signaling. The same group published a paper about the influence of multi-walled carbon nanotubes, inducing differential regulation of 111 genes in P. aeruginosa. Interestingly, graphene, boron nitride, and carbon black caused differential regulation only of 44, 26, and 25 genes, respectively, despite reports of the common interaction mechanism of these materials with bacteria [197][144]. The upregulation of genes by carbon nanotubes involves a list of metabolic pathways, such as nitrogen metabolism, sulfur metabolism, membrane proteins, and a two-component regulatory system associated with antibiotic resistance.
Li et al. [198][145] found that spherical and nanosheet carbon nanomaterials positively affect the upregulation of genes involved in amino acid and carbohydrate metabolism. Zhang et al. studied the effect of chronic exposure to low concentrations of graphene oxide on Escherichia coli [199][146]. The interaction between graphene oxide and bacteria causes the significant activation of the Cpx envelope stress response, resulting in a more than two-fold increase of extracellular protease release and biofilm formation. Moreover, bacteria after graphene oxide exposition exhibited higher pathogenicity than control cells.
3. Conclusions and Future Prospects
The growing field of study regarding anisotropic nanoparticle shape originally arose from the tuning of particles’ optical, electrical, and magnetic properties. The further assessment of the interactions of these nanoparticles with biological objects opened up an exciting field of biomedical research. This review presents an overview of the most relevant reports for this broad field of biomedical and biotechnological topics. Nevertheless, the nanoparticles shape-dependent effect on cells has a huge potential in the control and up- and downregulation of biochemical processes, as well as whole-organism behavior.
The study of nanoparticle shape within the microbial context is usually focused on antimicrobial action with common mechanisms, such as ROS generation, ion release, and physical disruption of the membrane. The great challenge in the generation of new bioapplications is the material-guided control of metabolic activity of the microbes, horizontal gene transfer, and modulation of quorum sensing without a significant effect on cell viability. Within this field, future research might include the intracellular delivery of the reaction substrate, gene circuit, signaling molecules to achieve increases in metabolic performance and on-demand control of the biocatalysis, with the separate growth of microbes and synthesis of the valuable product phases. The manipulation of biofilm growth through the precise delivery of the gene construction and quorum sensing moieties opens an avenue of application in the biomedical treatment of pathogenic bacteria via biological routes and advanced bioreactors for high-yield production. Mechanobiology and intracellular delivery represent a tremendous achievement for mammalian cells. Therefore, the transfer of these approaches to microbial cells could ultimately prove successful in next-generation therapy and biotechnology applications. Since shape and crystal engineering of inorganic particles has demonstrated sufficient flexibility for biological applications, novel applications could arise from shape-dependent modulation of the processes of interest. For example, photosynthesis and nitrogen fixation control in microalgae via anisotropic particles could enable the modulation of plant growth and development for agricultural purposes. In addition to shape-control itself, the unique physicochemical properties of these anisotropic particles allow them to bring physical stimuli, such as light and magnetic and electric fields, potentially enabling the remote modulation of biochemical processes via biophysical cues. Finally, the potential for nanoparticle shape-mediated increases in antimicrobial resistance, taking care of possible negative consequences using developed methods, is also worth mentioning. Interdisciplinary research is essential in order to take the next step toward the advanced future of biomedicine and biotechnology.
The study of the effects of nanoparticle shape within the mammalian cell context could be focused on such areas as cell reprogramming, cell differentiation, and more precise control over biological functions. It should be kept in mind that nanoparticles can be used in the form of surface coatings, not just as stand-alone nanoparticles. Surface structuring with nanoparticles can provide great opportunities in areas of cell differentiation and reprogramming. An outstanding review on high-aspect-ratio nanostructures showed the crucial importance of interactions between cells and nanomaterials [180][127]. Surface structuring can be implemented for graft coatings. Doctors have long used various implants for vascular surgery as substitutes for vessels, bone prostheses, etc. The issue of the survival rate of these artificial objects is extremely important. The development of surface treatments may enable significant progress in the creation of highly biocompatible implants. Currently, work in this direction is actively promoted only in the field of bone surgery. However, for instance, proper graft surface functionalization for vessel substitution applications could prevent thrombocyte aggregation or the overgrowth of endothelial cells.
Cell reprogramming is the newest technology that can be used in regenerative medicine or in the treatment of genetic diseases. It is known that many physical parameters such as surface topology, stiffness, and charge can influence the reprogramming process [200][147]. It has already been shown that a specific surface microstructure is extremely important for cell reprogramming [201][148]. At the same time, there have been no works devoted to the detailed study or, most importantly, the comparison of various shapes of nanoparticles for cell reprogramming.
Ferroptosis is another example of an area where shape dependence has not been investigated. The anisotropic surface of nanoparticles can positively influence both the release of ions and lead to other effects, providing a synergistic effect on cancer cells.
Another common problem for nanobiomedicine-related sciences is the weak link between chemists and biologists. It is necessary to distinguish clearly the effects of particle shape from the functionalization of the surface, charge, size, and other properties. Occasionally, non-obvious properties such as charge density [202][149], crystallinity [203][150], aggregation state [99][46], and the structure of local inhomogeneities [204][151], are essential. Thus, it is necessary to optimize the synthesis procedures and create particles that differ only in their shape.
References
- Dayem, A.A.; Choi, H.Y.; Yang, G.M.; Kim, K.; Saha, S.K.; Kim, J.H.; Cho, S.G. The potential of nanoparticles in stem cell differentiation and further therapeutic applications. Biotechnol. J. 2016, 11, 1550–1560.
- Steckiewicz, K.P.; Barcinska, E.; Malankowska, A.; Zauszkiewicz–Pawlak, A.; Nowaczyk, G.; Zaleska-Medynska, A.; Inkielewicz-Stepniak, I. Impact of gold nanoparticles shape on their cytotoxicity against human osteoblast and osteosarcoma in in vitro model. Evaluation of the safety of use and anti-cancer potential. J. Mater. Sci. Mater. Med. 2019, 30, 22.
- Huang, L.H.; Sun, X.Y.; Ouyang, J.M. Shape-dependent toxicity and mineralization of hydroxyapatite nanoparticles in A7R5 aortic smooth muscle cells. Sci. Rep. 2019, 9, 18979.
- Stewart, M.P.; Langer, R.; Jensen, K.F. Intracellular delivery by membrane disruption: Mechanisms, strategies, and concepts. Chem. Rev. 2018, 118, 7409–7531.
- Woźniak, A.; Malankowska, A.; Nowaczyk, G.; Grześkowiak, B.F.; Tuśnio, K.; Słomski, R.; Zaleska-Medynska, A.; Jurga, S. Size and shape-dependent cytotoxicity profile of gold nanoparticles for biomedical applications. J. Mater. Sci. Mater. Med. 2017, 28, 92.
- Villanueva-Flores, F.; Castro-Lugo, A.; Ramírez, O.T.; Palomares, L.A. Understanding cellular interactions with nanomaterials: Towards a rational design of medical nanodevices. Nanotechnology 2020, 31, 132002.
- Ivask, A.; Kurvet, I.; Kasemets, K.; Blinova, I.; Aruoja, V.; Suppi, S.; Vija, H.; Käkinen, A.; Titma, T.; Heinlaan, M.; et al. Size-dependent toxicity of silver nanoparticles to bacteria, yeast, algae, crustaceans and mammalian cells in vitro. PLoS ONE 2014, 9, e102108.
- Kim, T.W.; Chung, P.W.; Lin, V.S.Y. Facile synthesis of monodisperse spherical MCM-48 mesoporous silica nanoparticles with controlled particle size. Chem. Mater. 2010, 22, 5093–5104.
- Tan, W.F.; Yu, Y.T.; Wang, M.X.; Liu, F.; Koopal, L.K. Shape evolution synthesis of monodisperse spherical, ellipsoidal, and elongated hematite (α-Fe2O3) nanoparticles using ascorbic acid. Cryst. Growth Des. 2014, 14, 157–164.
- Serov, N.; Darmoroz, D.; Lokteva, A.; Chernyshov, I.; Koshel, E.; Vinogradov, V. One-pot synthesis of template-free hollow anisotropic CaCO3structures: Towards inorganic shape-mimicking drug delivery systems. Chem. Commun. 2020, 56, 11969–11972.
- Gao, G.; Liu, X.; Shi, R.; Zhou, K.; Shi, Y.; Ma, R.; Takayama-Muromachi, E.; Qiu, G. Shape-controlled synthesis and magnetic properties of monodisperse Fe 3O4 nanocubes. Cryst. Growth Des. 2010, 10, 2888–2894.
- Eguchi, M.; Mitsui, D.; Wu, H.L.; Sato, R.; Teranishi, T. Simple reductant concentration-dependent shape control of polyhedral gold nanoparticles and their plasmonic properties. Langmuir 2012, 28, 9021–9026.
- Khalavka, Y.; Becker, J.; Sönnichsen, C. Synthesis of rod-shaped gold nanorattles with improved plasmon sensitivity and catalytic activity. J. Am. Chem. Soc. 2009, 131, 1871–1875.
- Kladko, D.V.; Zakharzhevskii, M.A.; Vinogradov, V.V. Magnetic Field-Mediated Control of Whole-Cell Biocatalysis. J. Phys. Chem. Lett. 2020.
- Liao, H.G.; Jiang, Y.X.; Zhou, Z.Y.; Chen, S.P.; Sun, S.G. Shape-controlled synthesis of gold nanoparticles in deep eutectic solvents for studies of structure-functionality relationships in electrocatalysis. Angew. Chem. Int. Ed. 2008, 47, 9100–9103.
- Hu, Y.; He, L.; Yin, Y. Magnetically Responsive Photonic Nanochains. Angew. Chem. 2011, 123, 3831–3834.
- Jiao, L.; Wang, X.; Diankov, G.; Wang, H.; Dai, H. Facile synthesis of high-quality graphene nanoribbons. Nat. Nanotechnol. 2010, 5, 321–325.
- Falchevskaya, A.S.; Prilepskii, A.Y.; Tsvetikova, S.A.; Koshel, E.I.; Vinogradov, V.V. Facile Synthesis of a Library of Hollow Metallic Particles through the Galvanic Replacement of Liquid Gallium. Chem. Mater. 2021, 33, 1571–1580.
- Lizandara-Pueyo, C.; Siroky, S.; Wagner, M.R.; Hoffmann, A.; Reparaz, J.S.; Lehmann, M.; Polarz, S. Shape anisotropy influencing functional properties: Trigonal prismatic ZnO nanoparticles as an example. Adv. Funct. Mater. 2011, 21, 295–304.
- Liu, L.; Song, P.; Wei, Q.; Zhong, X.; Yang, Z.; Wang, Q. Synthesis of porous SnO2 hexagon nanosheets loaded with Au nanoparticles for high performance gas sensors. Mater. Lett. 2017, 201, 211–215.
- Jurney, P.; Agarwal, R.; Singh, V.; Choi, D.; Roy, K.; Sreenivasan, S.V.; Shi, L. Unique size and shape-dependent uptake behaviors of non-spherical nanoparticles by endothelial cells due to a shearing flow. J. Control. Release 2017, 245, 170–176.
- Chu, Z.; Zhang, S.; Zhang, B.; Zhang, C.; Fang, C.Y.; Rehor, I.; Cigler, P.; Chang, H.C.; Lin, G.; Liu, R.; et al. Unambiguous observation of shape effects on cellular fate of nanoparticles. Sci. Rep. 2014, 4, 4495.
- Enea, M.; Pereira, E.; de Almeida, M.P.; Araújo, A.M.; de Lourdes Bastos, M.; Carmo, H. Gold nanoparticles induce oxidative stress and apoptosis in human kidney cells. Nanomaterials 2020, 10, 995.
- Sultana, S.; Djaker, N.; Boca-Farcau, S.; Salerno, M.; Charnaux, N.; Astilean, S.; Hlawaty, H.; De La Chapelle, M.L. Comparative toxicity evaluation of flower-shaped and spherical gold nanoparticles on human endothelial cells. Nanotechnology 2015, 26, 055101.
- Chen, L.; Liu, M.; Zhou, Q.; Li, X. Recent developments of mesoporous silica nanoparticles in biomedicine. Emergent Mater. 2020, 3, 381–405.
- Huang, X.; Teng, X.; Chen, D.; Tang, F.; He, J. The effect of the shape of mesoporous silica nanoparticles on cellular uptake and cell function. Biomaterials 2010, 31, 438–448.
- Liu, X.; Sui, B.; Sun, J. Size- and shape-dependent effects of titanium dioxide nanoparticles on the permeabilization of the blood-brain barrier. J. Mater. Chem. B 2017, 5, 9558–9570.
- Huang, Y.W.; Cambre, M.; Lee, H.J. The Toxicity of Nanoparticles Depends on Multiple Molecular and Physicochemical Mechanisms. Int. J. Mol. Sci. 2017, 18, 2707.
- Jiang, J.; Oberdörster, G.; Elder, A.; Gelein, R.; Mercer, P.; Biswas, P. Does nanoparticle activity depend upon size and crystal phase? Nanotoxicology 2008, 2, 33–42.
- Wu, J.; Sun, J.; Xue, Y. Involvement of JNK and P53 activation in G2/M cell cycle arrest and apoptosis induced by titanium dioxide nanoparticles in neuron cells. Toxicol. Lett. 2010, 199, 269–276.
- Xu, Z.; Liu, C.; Wei, J.; Sun, J. Effects of four types of hydroxyapatite nanoparticles with different nanocrystal morphologies and sizes on apoptosis in rat osteoblasts. J. Appl. Toxicol. 2012, 32, 429–435.
- Zhang, B.; Sai Lung, P.; Zhao, S.; Chu, Z.; Chrzanowski, W.; Li, Q. Shape dependent cytotoxicity of PLGA-PEG nanoparticles on human cells. Sci. Rep. 2017, 7, 7315.
- Oh, W.K.; Kim, S.; Yoon, H.; Jang, J. Shape-dependent cytotoxicity and proinflammatory response of poly(3,4-ethylenedioxythiophene) nanomaterials. Small 2010, 6, 872–879.
- Zhang, Y.; Ali, S.F.; Dervishi, E.; Xu, Y.; Li, Z.; Casciano, D.; Biris, A.S. Cytotoxicity effects of graphene and single-wall carbon nanotubes in neural phaeochromocytoma-derived pc12 cells. ACS Nano 2010, 4, 3181–3186.
- Chapurina, Y.E.; Drozdov, A.S.; Popov, I.; Vinogradov, V.V.; Dudanov, I.P.; Vinogradov, V.V. alumina nanoparticles as a promising thrombolytic colloid with prolonged action. J. Mater. Chem. B 2016, 4, 5921–5928.
- Dong, L.; Tang, S.; Deng, F.; Gong, Y.; Zhao, K.; Zhou, J.; Liang, D.; Fang, J.; Hecker, M.; Giesy, J.P.; et al. Shape-dependent toxicity of alumina nanoparticles in rat astrocytes. Sci. Total Environ. 2019, 690, 158–166.
- Galluzzi, L.; Vitale, I.; Aaronson, S.A.; Abrams, J.M.; Adam, D.; Agostinis, P.; Alnemri, E.S.; Altucci, L.; Amelio, I.; Andrews, D.W.; et al. Molecular mechanisms of cell death: Recommendations of the Nomenclature Committee on Cell Death 2018. Cell Death Differ. 2018, 25, 486–541.
- Vakkila, J.; Lotze, M.T. Inflammation and necrosis promote tumour growth. Nat. Rev. Immunol. 2004, 4, 641–648.
- Albanese, A.; Chan, W.C.W. Effect of gold nanoparticle aggregation on cell uptake and toxicity. ACS Nano 2011, 5, 5478–5489.
- Lee, J.H.; Ju, J.E.; Kim, B.I.; Pak, P.J.; Choi, E.K.; Lee, H.S.; Chung, N. Rod-shaped iron oxide nanoparticles are more toxic than sphere-shaped nanoparticles to murine macrophage cells. Environ. Toxicol. Chem. 2014, 33, 2759–2766.
- Shen, Z.; Song, J.; Yung, B.C.; Zhou, Z.; Wu, A.; Chen, X. Emerging Strategies of Cancer Therapy Based on Ferroptosis. Adv. Mater. 2018, 30, 1704007.
- Zanganeh, S.; Hutter, G.; Spitler, R.; Lenkov, O.; Mahmoudi, M.; Shaw, A.; Pajarinen, J.S.; Nejadnik, H.; Goodman, S.; Moseley, M.; et al. Iron oxide nanoparticles inhibit tumour growth by inducing pro-inflammatory macrophage polarization in tumour tissues. Nat. Nanotechnol. 2016, 11, 986–994.
- Provenzano, R.; Schiller, B.; Rao, M.; Coyne, D.; Brenner, L.; Pereira, B.J.G. Ferumoxytol as an intravenous iron replacement therapy in hemodialysis patients. Clin. J. Am. Soc. Nephrol. 2009, 4, 386–393.
- Dukhinova, M.S.; Prilepskii, A.Y.; Vinogradov, V.V.; Shtil, A.A. Metal oxide nanoparticles in therapeutic regulation of macrophage functions. Nanomaterials 2019, 9, 1631.
- Zhang, C.; Bu, W.; Ni, D.; Zhang, S.; Li, Q.; Yao, Z.; Zhang, J.; Yao, H.; Wang, Z.; Shi, J. Synthesis of iron nanometallic glasses and their application in cancer therapy by a localized fenton reaction. Angew. Chem. Int. Ed. 2016, 55, 2101–2106.
- Ma, P.; Xiao, H.; Yu, C.; Liu, J.; Cheng, Z.; Song, H.; Zhang, X.; Li, C.; Wang, J.; Gu, Z.; et al. Enhanced Cisplatin Chemotherapy by Iron Oxide Nanocarrier-Mediated Generation of Highly Toxic Reactive Oxygen Species. Nano Lett. 2017, 17, 928–937.
- Li, W.P.; Su, C.H.; Chang, Y.C.; Lin, Y.J.; Yeh, C.S. Ultrasound-Induced Reactive Oxygen Species Mediated Therapy and Imaging Using a Fenton Reaction Activable Polymersome. ACS Nano 2016, 10, 2017–2027.
- Liu, X.; Sui, B.; Camargo, P.H.C.; Wang, J.; Sun, J. Tuning band gap of MnO2 nanoflowers by Alkali metal doping for enhanced Ferroptosis/phototherapy synergism in Cancer. Appl. Mater. Today 2021, 23, 101027.
- Wang, S.; Li, F.; Qiao, R.; Hu, X.; Liao, H.; Chen, L.; Wu, J.; Wu, H.; Zhao, M.; Liu, J.; et al. Arginine-Rich Manganese Silicate Nanobubbles as a Ferroptosis-Inducing Agent for Tumor-Targeted Theranostics. ACS Nano 2018, 12, 12380–12392.
- An, P.; Gao, Z.; Sun, K.; Gu, D.; Wu, H.; You, C.; Li, Y.; Cheng, K.; Zhang, Y.; Wang, Z.; et al. Photothermal-Enhanced Inactivation of Glutathione Peroxidase for Ferroptosis Sensitized by an Autophagy Promotor. ACS Appl. Mater. Interfaces 2019, 11, 42988–42997.
- Xu, S.; Zheng, H.; Ma, R.; Wu, D.; Pan, Y.; Yin, C.; Gao, M.; Wang, W.; Li, W.; Liu, S.; et al. Vacancies on 2D transition metal dichalcogenides elicit ferroptotic cell death. Nat. Commun. 2020, 11, 3483.
- Arnida; Malugin, A.; Ghandehari, H. Cellular uptake and toxicity of gold nanoparticles in prostate cancer cells: A comparative study of rods and spheres. J. Appl. Toxicol. 2010, 30, 212–217.
- Nagai, H.; Toyokuni, S. Biopersistent fiber-induced inflammation and carcinogenesis: Lessons learned from asbestos toward safety of fibrous nanomaterials. Arch. Biochem. Biophys. 2010, 502, 1–7.
- Myers, R. Asbestos-related pleural disease. Curr. Opin. Pulm. Med. 2012, 18, 377–381.
- Xu, J.; Alexander, D.B.; Futakuchi, M.; Numano, T.; Fukamachi, K.; Suzui, M.; Omori, T.; Kanno, J.; Hirose, A.; Tsuda, H. Size- and shape-dependent pleural translocation, Deposition, Fibrogenesis, And mesothelial proliferation by multiwalled carbon nanotubes. Cancer Sci. 2014, 105, 763–769.
- Forest, V.; Leclerc, L.; Hochepied, J.F.; Trouvé, A.; Sarry, G.; Pourchez, J. Impact of cerium oxide nanoparticles shape on their in vitro cellular toxicity. Toxicol. Vitro 2017, 38, 136–141.
- Dayem, A.A.; Hossain, M.K.; Lee, S.B.; Kim, K.; Saha, S.K.; Yang, G.M.; Choi, H.Y.; Cho, S.G. The role of reactive oxygen species (ROS) in the biological activities of metallic nanoparticles. Int. J. Mol. Sci. 2017, 18, 120.
- Prilepskii, A.Y.; Serov, N.S.; Kladko, D.V.; Vinogradov, V.V. Nanoparticle-Based Approaches towards the Treatment of Atherosclerosis. Pharmaceutics 2020, 12, 1056.
- Nuzhina, J.V.; Shtil, A.A.; Prilepskii, A.Y.; Vinogradov, V.V. Preclinical evaluation and clinical translation of magnetite-based nanomedicines. J. Drug Deliv. Sci. Technol. 2019, 54, 101282.
- Chompoosor, A.; Saha, K.; Ghosh, P.S.; MacArthy, D.J.; Miranda, O.R.; Zhu, Z.J.; Arcaro, K.F.; Rotello, V.M. The role of surface functionality on acute cytotoxicity, ROS generation and DNA damage by cationic gold nanoparticles. Small 2010, 6, 2246–2249.
- Bhattacharya, D.; Santra, C.R.; Ghosh, A.N.; Karmakar, P. Differential toxicity of rod and spherical zinc oxide nanoparticles on human peripheral blood mononuclear cells. J. Biomed. Nanotechnol. 2014, 10, 707–716.
- Niu, M.; Zhong, H.; Shao, H.; Hong, D.; Ma, T.; Xu, K.; Chen, X.; Han, J.; Sun, J. Shape-dependent genotoxicity of mesoporous silica nanoparticles and cellular mechanisms. J. Nanosci. Nanotechnol. 2016, 16, 2313–2318.
- Pittenger, M.F.; Mackay, A.M.; Beck, S.C.; Jaiswal, R.K.; Douglas, R.; Mosca, J.D.; Moorman, M.A.; Simonetti, D.W.; Craig, S.; Marshak, D.R. Multilineage potential of adult human mesenchymal stem cells. Science 1999.
- Fan, J.H.; Li, W.T.; Hung, W.I.; Chen, C.P.; Yeh, J.M. Cytotoxicity and differentiation effects of gold nanoparticles to human bone marrow mesenchymal stem cells. Biomed. Eng. Appl. Basis Commun. 2011, 23, 141–152.
- Wei, M.; Li, S.; Yang, Z.; Zheng, W.; Le, W. Gold nanoparticles enhance the differentiation of embryonic stem cells into dopaminergic neurons via mTOR/p70S6K pathway. Nanomedicine 2017, 12, 1305–1317.
- Yi, C.; Liu, D.; Fong, C.C.; Zhang, J.; Yang, M. Gold nanoparticles promote osteogenic differentiation of mesenchymal stem cells through p38 MAPK pathway. ACS Nano 2010, 4, 6439–6448.
- Li, J.; Li, J.J.; Zhang, J.; Wang, X.; Kawazoe, N.; Chen, G. Gold nanoparticle size and shape influence on osteogenesis of mesenchymal stem cells. Nanoscale 2016, 8, 7992–8002.
- Liu, D.; Yi, C.; Zhang, D.; Zhang, J.; Yang, M. Inhibition of proliferation and differentiation of mesenchymal stem cells by carboxylated carbon nanotubes. ACS Nano 2010, 4, 2185–2195.
- Dobson, J. Remote control of cellular behaviour with magnetic nanoparticles. Nat. Nanotechnol. 2008, 3, 139–143.
- Gregurec, D.; Senko, A.W.; Chuvilin, A.; Reddy, P.D.; Sankararaman, A.; Rosenfeld, D.; Chiang, P.H.; Garcia, F.; Tafel, I.; Varnavides, G.; et al. Magnetic Vortex Nanodiscs Enable Remote Magnetomechanical Neural Stimulation. ACS Nano 2020, 14, 8036–8045.
- Lee, J.U.; Shin, W.; Lim, Y.; Kim, J.; Kim, W.R.; Kim, H.; Lee, J.H.; Cheon, J. Non-contact long-range magnetic stimulation of mechanosensitive ion channels in freely moving animals. Nat. Mater. 2021.
- Vanzha, E.; Pylaev, T.; Prilepskii, A.; Golubev, A.; Khlebtsov, B.; Bogatyrev, V.; Khlebtsov, N. Cell culture surfaces with immobilized gold nanostars: A new approach for laser-induced plasmonic cell optoporation. In Proceedings of the Saratov Fall Meeting 2016: Optical Technologies in Biophysics and Medicine XVIII, Saratov, Russia, 27–30 September 2016; Volume 10336, p. 103360L.
- Bibikova, O.; Popov, A.; Skovorodkin, I.; Prilepskyi, A.; Pylaev, T.; Bykov, A.; Staroverov, S.; Bogatyrev, V.; Tuchin, V.; Kinnunen, M.; et al. Plasmon-resonant gold nanoparticles with variable morphology as optical labels and drug carriers for cytological research. In Proceedings of the Optics InfoBase Conference Papers, Society of Photo-Optical Instrumentation Engineers (SPIE), Munich, Germany, 12–16 May 2013.
- Sun, M.; Liu, Q.; Fan, X.; Wang, Y.; Chen, W.; Tian, C.; Sun, L.; Xie, H. Autonomous Biohybrid Urchin-Like Microperforator for Intracellular Payload Delivery. Small 2020, 16, e1906701.
- Qian, Y.; Wang, D.; Tian, X.; Liu, H.; Wang, X.; Li, H.; Chen, Q.; Zhang, X.; Wang, H. Synthesis of urchin-like nickel nanoparticles with enhanced rotating magnetic field-induced cell necrosis and tumor inhibition. Chem. Eng. J. 2020, 400.
- Nielsen, J.; Keasling, J.D. Engineering Cellular Metabolism. Cell 2016, 164, 1185–1197.
- Sharma, D.; Saharan, B.S. (Eds.) Microbial Cell Factories; CRC Press: Boca Raton, FL, USA, 2018; ISBN 9780367657307.
- Tang, T.C.; An, B.; Huang, Y.; Vasikaran, S.; Wang, Y.; Jiang, X.; Lu, T.K.; Zhong, C. Materials design by synthetic biology. Nat. Rev. Mater. 2020, 6, 332–350.
- Wu, S.; Snajdrova, R.; Moore, J.C.; Baldenius, K.; Bornscheuer, U.T. Biocatalysis: Enzymatic Synthesis for Industrial Applications. Angew. Chem. Int. Ed. 2021, 60, 88–119.
- Chatterjee, A.; Modarai, M.; Naylor, N.R.; Boyd, S.E.; Atun, R.; Barlow, J.; Holmes, A.H.; Johnson, A.; Robotham, J.V. Quantifying drivers of antibiotic resistance in humans: A systematic review. Lancet Infect. Dis. 2018, 18, e368–e378.
- Vickery, K.; Hu, H.; Jacombs, A.S.; Bradshaw, D.A.; Deva, A.K. A review of bacterial biofilms and their role in device-associated infection. Healthc. Infect. 2013, 18, 61–66.
- Kälicke, T.; Schierholz, J.; Schlegel, U.; Frangen, T.M.; Köller, M.; Printzen, G.; Seybold, D.; Klöckner, S.; Muhr, G.; Arens, S. Effect on infection resistance of a local antiseptic and antibiotic coating on osteosynthesis implants: An in vitro and in vivo study. J. Orthop. Res. 2006, 24, 1622–1640.
- Liu, Y.; Shi, L.; Su, L.; Van der Mei, H.C.; Jutte, P.C.; Ren, Y.; Busscher, H.J. Nanotechnology-based antimicrobials and delivery systems for biofilm-infection control. Chem. Soc. Rev. 2019, 48, 428–446.
- Sims, K.R.; Liu, Y.; Hwang, G.; Jung, H.I.; Koo, H.; Benoit, D.S.W. Enhanced design and formulation of nanoparticles for anti-biofilm drug delivery. Nanoscale 2019, 11, 219–236.
- Agarwalla, S.V.; Ellepola, K.; Silikas, N.; Castro Neto, A.H.; Seneviratne, C.J.; Rosa, V. Persistent inhibition of Candida albicans biofilm and hyphae growth on titanium by graphene nanocoating. Dent. Mater. 2021, 37, 370–377.
- Yuan, Z.; Tao, B.; He, Y.; Mu, C.; Liu, G.; Zhang, J.; Liao, Q.; Liu, P.; Cai, K. Remote eradication of biofilm on titanium implant via near-infrared light triggered photothermal/photodynamic therapy strategy. Biomaterials 2019, 223, 119479.
- Ferrer-Miralles, N.; Domingo-Espín, J.; Corchero, J.; Vázquez, E.; Villaverde, A. Microbial factories for recombinant pharmaceuticals. Microb. Cell Fact. 2009, 8, 1–8.
- Ko, Y.S.; Kim, J.W.; Lee, J.A.; Han, T.; Kim, G.B.; Park, J.E.; Lee, S.Y. Tools and strategies of systems metabolic engineering for the development of microbial cell factories for chemical production. Chem. Soc. Rev. 2020, 49, 4615–4636.
- Peralta-Yahya, P.P.; Zhang, F.; Del Cardayre, S.B.; Keasling, J.D. Microbial engineering for the production of advanced biofuels. Nature 2012, 488, 320–328.
- Ding, Q.; Diao, W.; Gao, C.; Chen, X.; Liu, L. Microbial cell engineering to improve cellular synthetic capacity. Biotechnol. Adv. 2020, 45, 107649.
- Li, W.; Liu, Z.; Liu, C.; Guan, Y.; Ren, J.; Qu, X. Manganese Dioxide Nanozymes as Responsive Cytoprotective Shells for Individual Living Cell Encapsulation. Angew. Chem. Int. Ed. 2017, 56, 13661–13665.
- Wang, L.; Hu, Z.Y.; Yang, X.Y.; Zhang, B.B.; Geng, W.; Van Tendeloo, G.; Su, B.L. Polydopamine nanocoated whole-cell asymmetric biocatalysts. Chem. Commun. 2017, 53, 6617–6620.
- Zhang, H.; Liu, H.; Tian, Z.; Lu, D.; Yu, Y.; Cestellos-Blanco, S.; Sakimoto, K.K.; Yang, P. Bacteria photosensitized by intracellular gold nanoclusters for solar fuel production. Nat. Nanotechnol. 2018, 13, 900–905.
- Guo, J.; Suástegui, M.; Sakimoto, K.K.; Moody, V.M.; Xiao, G.; Nocera, D.G.; Joshi, N.S. Light-driven fine chemical production in yeast biohybrids. Science 2018, 362, 813–816.
- Ding, Q.; Ma, D.; Liu, G.Q.; Li, Y.; Guo, L.; Gao, C.; Hu, G.; Ye, C.; Liu, J.; Liu, L.; et al. Light-powered Escherichia coli cell division for chemical production. Nat. Commun. 2020, 11, 2262.
- Chernousova, S.; Epple, M. Silver as antibacterial agent: Ion, nanoparticle, and metal. Angew. Chem. Int. Ed. 2013, 52, 1636–1653.
- Pal, S.; Tak, Y.K.; Song, J.M. Does the antibacterial activity of silver nanoparticles depend on the shape of the nanoparticle? A study of the gram-negative bacterium Escherichia coli. Appl. Environ. Microbiol. 2007, 73, 1712–1720.
- Cheon, J.Y.; Kim, S.J.; Rhee, Y.H.; Kwon, O.H.; Park, W.H. Shape-dependent antimicrobial activities of silver nanoparticles. Int. J. Nanomed. 2019, 14, 2773–2780.
- Acharya, D.; Singha, K.M.; Pandey, P.; Mohanta, B.; Rajkumari, J.; Singha, L.P. Shape dependent physical mutilation and lethal effects of silver nanoparticles on bacteria. Sci. Rep. 2018, 8, 201.
- Yeh, Y.C.; Creran, B.; Rotello, V.M. Gold nanoparticles: Preparation, properties, and applications in bionanotechnology. Nanoscale 2012, 4, 1871–1880.
- Canaparo, R.; Foglietta, F.; Giuntini, F.; Della Pepa, C.; Dosio, F.; Serpe, L. Recent developments in antibacterial therapy: Focus on stimuli-responsive drug-delivery systems and therapeutic nanoparticles. Molecules 2019, 24, 1991.
- Dreaden, E.C.; Alkilany, A.M.; Huang, X.; Murphy, C.J.; El-Sayed, M.A. The golden age: Gold nanoparticles for biomedicine. Chem. Soc. Rev. 2012, 41, 2740–2779.
- Penders, J.; Stolzoff, M.; Hickey, D.J.; Andersson, M.; Webster, T.J. Shape-dependent antibacterial effects of non-cytotoxic gold nanoparticles. Int. J. Nanomed. 2017, 12, 2457–2468.
- Chmielewska, S.J.; Skłodowski, K.; Depciuch, J.; Deptuła, P.; Piktel, E.; Fiedoruk, K.; Kot, P.; Paprocka, P.; Fortunka, K.; Wollny, T.; et al. Bactericidal properties of rod-, peanut-, and star-shaped gold nanoparticles coated with ceragenin CSA-131 against multidrug-resistant bacterial strains. Pharmaceutics 2021, 13, 425.
- Hameed, S.; Wang, Y.; Zhao, L.; Xie, L.; Ying, Y. Shape-dependent significant physical mutilation and antibacterial mechanisms of gold nanoparticles against foodborne bacterial pathogens (Escherichia coli, Pseudomonas aeruginosa and Staphylococcus aureus) at lower concentrations. Mater. Sci. Eng. C 2020, 108, 110338.
- Shabanova, E.M.; Drozdov, A.S.; Ivanovski, V.; Suvorova, I.I.; Vinogradov, V.V. magnetite: Proteolytic composite for magnetically targeted minimally invasive surgery. RSC Adv. 2016, 6, 84354–84362.
- Shabanova, E.M.; Drozdov, A.S.; Fakhardo, A.F.; Dudanov, I.P.; Kovalchuk, M.S.; Vinogradov, V.V. Fe 3 O 4 nanoparticles for use as a hemostatic agent in internal bleeding. Sci. Rep. 2018, 8, 233.
- Soto, F.; Wang, J.; Ahmed, R.; Demirci, U. Medical Micro/Nanorobots in Precision Medicine. Adv. Sci. 2020, 7.
- Yang, Z.; Zhang, L. Magnetic Actuation Systems for Miniature Robots: A Review. Adv. Intell. Syst. 2020, 2, 2000082.
- Wang, J.; Wang, L.; Pan, J.; Zhao, J.; Tang, J.; Jiang, D.; Hu, P.; Jia, W.; Shi, J. Magneto-Based Synergetic Therapy for Implant-Associated Infections via Biofilm Disruption and Innate Immunity Regulation. Adv. Sci. 2021, 8, 2004010.
- Yan, X.; Zhou, Q.; Vincent, M.; Deng, Y.; Yu, J.; Xu, J.; Xu, T.; Tang, T.; Bian, L.; Wang, Y.X.J.; et al. Multifunctional biohybrid magnetite microrobots for imaging-guided therapy. Sci. Robot. 2017, 2, eaaq1155.
- Prilepskii, A.Y.; Fakhardo, A.F.; Drozdov, A.S.; Vinogradov, V.V.; Dudanov, I.P.; Shtil, A.A.; Bel’tyukov, P.P.; Shibeko, A.M.; Koltsova, E.M.; Nechipurenko, D.Y.; et al. Urokinase-conjugated magnetite nanoparticles as a promising drug delivery system for targeted thrombolysis: Synthesis and preclinical evaluation. ACS Appl. Mat. Int. 2018, 10(43), 36764–36775.
- Drozdov, A.S.; Volodina, K.V.; Vinogradov, V.V.; Vinogradov, V.V. Biocomposites for wound-healing based on sol–gel magnetite. RSC Adv. 2015, 5, 82992–82997.
- Yan, X.; Zhou, Q.; Yu, J.; Xu, T.; Deng, Y.; Tang, T.; Feng, Q.; Bian, L.; Zhang, Y.; Ferreira, A.; et al. Magnetite Nanostructured Porous Hollow Helical Microswimmers for Targeted Delivery. Adv. Funct. Mater. 2015, 25, 5333–5342.
- Vinogradov, V.V.; Drozdov, A.S.; Mingabudinova, L.R.; Shabanova, E.M.; Kolchina, N.O.; Anastasova, E.I.; Markova, A.A.; Shtil, A.A.; Milichko, V.A.; Starova, G.L.; et al. Composites based on heparin and MIL-101 (Fe): The drug releasing depot for anticoagulant therapy and advanced medical nanofabrication. J. Mater. Chem. B 2018, 6(16), 2450–2459.
- Dong, Y.; Wang, L.; Yuan, K.; Ji, F.; Gao, J.; Zhang, Z.; Du, X.; Tian, Y.; Wang, Q.; Zhang, L. Magnetic Microswarm Composed of Porous Nanocatalysts for Targeted Elimination of Biofilm Occlusion. ACS Nano 2021, 15, 5056–5067.
- Hwang, G.; Paula, A.J.; Hunter, E.E.; Liu, Y.; Babeer, A.; Karabucak, B.; Stebe, K.; Kumar, V.; Steager, E.; Koo, H. Catalytic antimicrobial robots for biofilm eradication. Sci. Robot. 2019, 4, eaaw2388.
- Xie, W.; Allioux, F.M.; Ou, J.Z.; Miyako, E.; Tang, S.Y.; Kalantar-Zadeh, K. Gallium-Based Liquid Metal Particles for Therapeutics. Trends Biotechnol. 2020.
- Elbourne, A.; Cheeseman, S.; Atkin, P.; Truong, N.P.; Syed, N.; Zavabeti, A.; Mohiuddin, M.; Esrafilzadeh, D.; Cozzolino, D.; McConville, C.F.; et al. Antibacterial Liquid Metals: Biofilm Treatment via Magnetic Activation. ACS Nano 2020, 14, 802–817.
- Cheeseman, S.; Elbourne, A.; Kariuki, R.; Ramarao, A.V.; Zavabeti, A.; Syed, N.; Christofferson, A.J.; Kwon, K.Y.; Jung, W.; Dickey, M.D.; et al. Broad-spectrum treatment of bacterial biofilms using magneto-responsive liquid metal particles. J. Mater. Chem. B 2020, 8, 10776–10787.
- Watson, G.S.; Watson, J.A.; Cribb, B.W. Diversity of Cuticular Micro- and Nanostructures on Insects: Properties, Functions, and Potential Applications. Annu. Rev. Entomol. 2017, 62, 185–205.
- Dixit, H.G.; Starr, R.; Dundon, M.L.; Pairs, P.I.; Yang, X.; Zhang, Y.; Nampe, D.; Ballas, C.B.; Tsutsui, H.; Forman, S.J.; et al. Massively-Parallelized, Deterministic Mechanoporation for Intracellular Delivery. Nano Lett. 2019, 20, 860–867.
- Ganjian, M.; Modaresifar, K.; Ligeon, M.R.O.; Kunkels, L.B.; Tümer, N.; Angeloni, L.; Hagen, C.W.; Otten, L.G.; Hagedoorn, P.L.; Apachitei, I.; et al. Nature Helps: Toward Bioinspired Bactericidal Nanopatterns. Adv. Mater. Interfaces 2019, 6.
- Pogodin, S.; Hasan, J.; Baulin, V.A.; Webb, H.K.; Truong, V.K.; Phong Nguyen, T.H.; Boshkovikj, V.; Fluke, C.J.; Watson, G.S.; Watson, J.A.; et al. Biophysical model of bacterial cell interactions with nanopatterned cicada wing surfaces. Biophys. J. 2013, 104, 835–840.
- Li, X. Bactericidal mechanism of nanopatterned surfaces. Phys. Chem. Chem. Phys. 2015, 18, 1311–1316.
- Xie, X.; Xu, A.M.; Angle, M.R.; Tayebi, N.; Verma, P.; Melosh, N.A. Mechanical model of vertical nanowire cell penetration. Nano Lett. 2013, 13, 6002–6008.
- Higgins, S.G.; Becce, M.; Belessiotis-Richards, A.; Seong, H.; Sero, J.E.; Stevens, M.M. High-Aspect-Ratio Nanostructured Surfaces as Biological Metamaterials. Adv. Mater. 2020, 32, 1–44.
- Linklater, D.P.; Juodkazis, S.; Ivanova, E.P. Nanofabrication of mechano-bactericidal surfaces. Nanoscale 2017, 9, 16564–16585.
- Harris, A.R.; Jreij, P.; Fletcher, D.A. Mechanotransduction by the Actin Cytoskeleton: Converting Mechanical Stimuli into Biochemical Signals. Annu. Rev. Biophys. 2018, 47, 617–631.
- Valiei, A.; Lin, N.; Bryche, J.F.; McKay, G.; Canva, M.; Charette, P.G.; Nguyen, D.; Moraes, C.; Tufenkji, N. Hydrophilic mechano-bactericidal nanopillars require external forces to rapidly kill bacteria. Nano Lett. 2020, 20, 5720–5727.
- Linklater, D.P.; De Volder, M.; Baulin, V.A.; Werner, M.; Jessl, S.; Golozar, M.; Maggini, L.; Rubanov, S.; Hanssen, E.; Juodkazis, S.; et al. High Aspect Ratio Nanostructures Kill Bacteria via Storage and Release of Mechanical Energy. ACS Nano 2018, 12, 6657–6667.
- Khan, M.I.; Shin, J.H.; Kim, J.D. The promising future of microalgae: Current status, challenges, and optimization of a sustainable and renewable industry for biofuels, feed, and other products. Microb. Cell Fact. 2018, 17, 36.
- Vargas-Estrada, L.; Torres-Arellano, S.; Longoria, A.; Arias, D.M.; Okoye, P.U.; Sebastian, P.J. Role of nanoparticles on microalgal cultivation: A review. Fuel 2020, 280.
- Eroglu, E.; Eggers, P.K.; Winslade, M.; Smith, S.M.; Raston, C.L. Enhanced accumulation of microalgal pigments using metal nanoparticle solutions as light filtering devices. Green Chem. 2013, 15, 3155–3159.
- Cruces, E.; Barrios, A.C.; Cahue, Y.P.; Januszewski, B.; Gilbertson, L.M.; Perreault, F. Similar toxicity mechanisms between graphene oxide and oxidized multi-walled carbon nanotubes in Microcystis aeruginosa. Chemosphere 2021, 265, 129137.
- Samei, M.; Sarrafzadeh, M.H.; Faramarzi, M.A. The impact of morphology and size of zinc oxide nanoparticles on its toxicity to the freshwater microalga, Raphidocelis subcapitata. Environ. Sci. Pollut. Res. 2019, 26, 2409–2420.
- Lyu, Z.; Shao, N.; Akinyemi, T.; Whitman, W.B. Methanogenesis. Curr. Biol. 2018, 28, R727–R732.
- Zhu, X.; Blanco, E.; Bhatti, M.; Borrion, A. Impact of metallic nanoparticles on anaerobic digestion: A systematic review. Sci. Total Environ. 2021, 757, 143747.
- Ambuchi, J.J.; Zhang, Z.; Shan, L.; Liang, D.; Zhang, P.; Feng, Y. Response of anaerobic granular sludge to iron oxide nanoparticles and multi-wall carbon nanotubes during beet sugar industrial wastewater treatment. Water Res. 2017, 117, 87–94.
- Yun, S.; Xing, T.; Han, F.; Shi, J.; Wang, Z.; Fan, Q.; Xu, H. Enhanced direct interspecies electron transfer with transition metal oxide accelerants in anaerobic digestion. Bioresour. Technol. 2021, 320, 124294.
- Zhou, W.; Wang, Y.; Wang, M.; Qian, B.; Li, L.; Gao, B. The co-effect of ampicillin and multi-walled carbon nanotubes on activated sludge in sequencing batch reactors: Microbial status, microbial community structure and ARGs propagation. Environ. Sci. Nano 2021, 8, 204–216.
- Yang, X.; He, Q.; Guo, F.; Sun, X.; Zhang, J.; Chen, Y. Impacts of carbon-based nanomaterials on nutrient removal in constructed wetlands: Microbial community structure, enzyme activities, and metabolism process. J. Hazard. Mater. 2021, 401, 123270.
- Mortimer, M.; Li, D.; Wang, Y.; Holden, P.A. Physical Properties of Carbon Nanomaterials and Nanoceria Affect Pathways Important to the Nodulation Competitiveness of the Symbiotic N2-Fixing Bacterium Bradyrhizobium diazoefficiens. Small 2020, 16, e1906055.
- Mortimer, M.; Devarajan, N.; Li, D.; Holden, P.A. Multiwall Carbon Nanotubes Induce More Pronounced Transcriptomic Responses in Pseudomonas aeruginosa PG201 than Graphene, Exfoliated Boron Nitride, or Carbon Black. ACS Nano 2018, 12, 2728–2740.
- Li, X.; Ban, Z.; Yu, F.; Hao, W.; Hu, X. Untargeted Metabolic Pathway Analysis as an Effective Strategy to Connect Various Nanoparticle Properties to Nanoparticle-Induced Ecotoxicity. Environ. Sci. Technol. 2020, 54, 3395–3406.
- Zhang, Q.; Zhang, C. Chronic Exposure to Low Concentration of Graphene Oxide Increases Bacterial Pathogenicity via the Envelope Stress Response. Environ. Sci. Technol. 2020, 54, 12412–12422.
- Fang, J.; Hsueh, Y.Y.; Soto, J.; Sun, W.; Wang, J.; Gu, Z.; Khademhosseini, A.; Li, S. Engineering Biomaterials with Micro/Nanotechnologies for Cell Reprogramming. ACS Nano 2020, 14, 1296–1318.
- Carlson, A.L.; Bennett, N.K.; Francis, N.L.; Halikere, A.; Clarke, S.; Moore, J.C.; Hart, R.P.; Paradiso, K.; Wernig, M.; Kohn, J.; et al. Generation and transplantation of reprogrammed human neurons in the brain using 3D microtopographic scaffolds. Nat. Commun. 2016, 7, 10862.
- Prilepskii, A.Y.; Kalnin, A.Y.; Fakhardo, A.F.; Anastasova, E.I.; Nedorezova, D.D.; Antonov, G.A.; Vinogradov, V.V. Cationic magnetite nanoparticles for increasing siRNA hybridization rates. Nanomaterials 2020, 10, 1018.
- Iswarya, V.; Bhuvaneshwari, M.; Alex, S.A.; Iyer, S.; Chaudhuri, G.; Chandrasekaran, P.T.; Bhalerao, G.M.; Chakravarty, S.; Raichur, A.M.; Chandrasekaran, N.; et al. Combined toxicity of two crystalline phases (anatase and rutile) of Titania nanoparticles towards freshwater microalgae: Chlorella sp. Aquat. Toxicol. 2015, 161, 154–169.
- Fratoddi, I.; Venditti, I.; Cametti, C.; Russo, M.V. How toxic are gold nanoparticles? The state-of-the-art. Nano Res. 2015, 8, 1771–1799.
More
