Porous tantalum (Ta) is a promising biomaterial and has been applied in orthopedics and dentistry for nearly two decades. The high porosity and interconnected pore structure of porous Ta promise fine bone ingrowth and new bone formation within the inner space, which further guarantee rapid osteointegration and bone–implant stability in the long term. Porous Ta has high wettability and surface energy that can facilitate adherence, proliferation and mineralization of osteoblasts. Meanwhile, the low elastic modulus and high friction coefficient of porous Ta allow it to effectively avoid the stress shield effect, minimize marginal bone loss and ensure primary stability. Accordingly, the satisfactory clinical application of porous Ta-based implants or prostheses is mainly derived from its excellent biological and mechanical properties.
- porous tantalum
- clinical application
- additive manufacturing
- surface modification
- bone tissue engineering
1. Introduction
| Pore Size (μm) | Strut Size (μm) | Elastic Modulus (GPa) | Compressive Strength (MPa) | Yield Strength (MPa) | 0.2% Proof Strength (MPa) | Ref | |||
|---|---|---|---|---|---|---|---|---|---|
| Cortical bone | 3–5 | 7–30 | 100–230 | [47] | |||||
| Trabecular bone | 50–90 | 0.01–3.0 | 2–12 | ||||||
| CVD (porous carbon scaffold) |
75–85 | 400–600 | 40–60 | 2.5–3.9 | 42–78 | [33] | |||
| CVD (porous SiC scaffold) |
70–85 |
2.3. Knee Arthroplasty
2.3. Knee Arthroplasty
2.4. Ankle Arthrodesis and Arthroplasty
2.4. Ankle Arthrodesis and Arthroplasty
2.5. Dental Implants
2.5. Dental Implants
3. New Development of Porous Ta for Bone Tissue Engineering
3.1. Additive Manufactured Porous Ta
3.1. Additive Manufactured Porous Ta
| Porosity%/Samples | In Vitro Tests Results | In Vivo Tests Results | Ref. |
|---|---|---|---|
| 80% Ta | Cytotoxicity test (L929 mammalian cells)
|
Histological evaluation (rat femur defect model)
|
[52] |
| 70% Ta vs. 70% Ti |
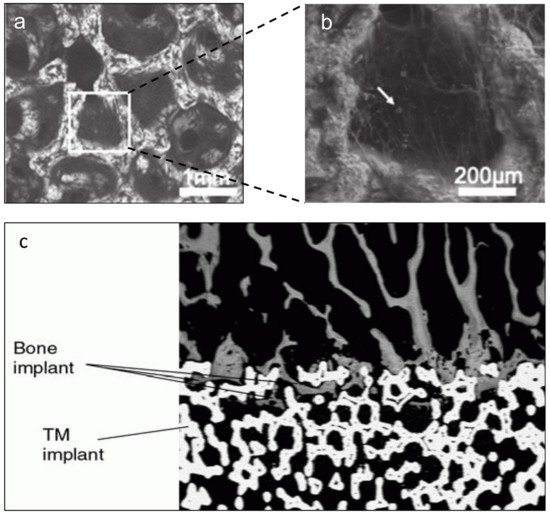
3.2. Surface Modification
3.2. Surface Modification
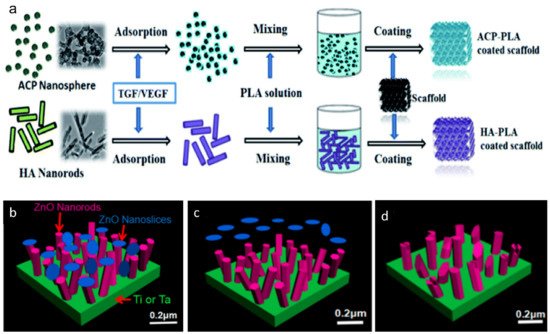
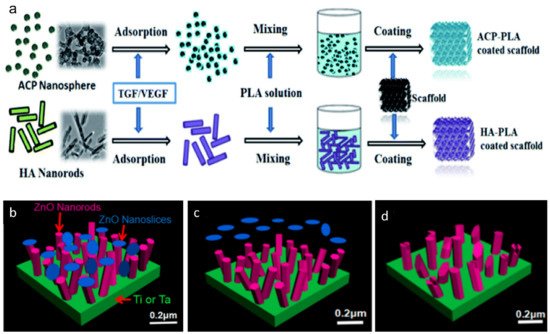
| Surface Modification | In Vitro Test Results | In Vivo Test Results | Ref. | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| ACP nanospheres–PLA coating HA nanorods–PLA coating |
Mineralization in SBF
|
Subchondral bone defect repair
|
[166] | Cell morphologies (hBMSCs)
| ||||||
| CaP nanospheres–PLA coating | Mineralization in SBF
|
|
|
Subchondral bone defect repair
|
||||||
| [ | 167 | ] | 80% Ta vs. 80% Ti | Cell morphologies (hBMSCs)
|
Histological evaluationsand fluorescence labeling (rabbit distal femoral defect model)
|
[55] | ||||
| 30% Ta vs. 30% Ti modified with TiO2 nanotubes, 30% Ti and solid Ti | ||||||||||
| BMP-7 coating | Not mentioned | Cartilage defect restoration (rabbit model)
|
[168] | Not mentioned | ||||||
| Ta2O5 nanotubes films | Anticorrosion test
| 150–400 | 40–60 | 10–30 | 35–100 |
|
Not mentioned | [51] |
| |||||||||||||||
|
| [ | 48 | ] | |||||||||||
| Not mentioned | [169] | Foam impregnation | 65–80 | 400–600 | 2.0–4.6 | 100–170 | 27% Ta and 45% Ta vs. 27% Ti | Cell morphologies (hFOB CRL-11372)
|
| [49 | |||||
| Nanoporous Ta oxide layers | Cell proliferation and morphology (L929 mouse fibroblasts)
|
Not mentioned | ] | ||||||||||||
| [170] | Powder metallurgy | ||||||||||||||
| MAO combined with NaOH treatment | Mineralization in SBF
| 100–400 |
|
Bone ingrowth (rabbit cranial defect model)
| 2.0 ± 0.3 |
| 50.3 ± 0.5 |
| [50] | ||||||
| [171] | LENS | 55 | |||||||||||||
| PHAs (PHB, PHBV and PHB4HB)–Genta coating | Cytotoxicity and cell adhesion (SaOS-2 cells)
| Antibacterial properties | 1.5 ± 0.3 | (S. aureus and E. coli) | 100 ± 10 | [51] | |||||||||
| Cell proliferation |
|
Not mentioned | [172] | 45 | |||||||||||
| ZnO nanorods−nanoslices hierarchical structure coating | Antibacterial Properties (S. aureus and E. coli)
| 7 ± 0.6 |
|
In vivo Infected Studies (KM mice subcutaneous implantation)
| 192 ± 7 | ||||||||||
|
[ | 173] | 27 | 20 ± 1.9 | 746 ± 27 | ||||||||||
| SLM | 80 | 500 | 150 | 1.22 ± 0.07 | 28.3 ± 1.2 | 12.7 ± 0.6 | [52] | ||||||||
| SEBM | 75 | 540 | 23.98 ± 1.72 | [53] | |||||||||||
| 80 | 392 | 19.48 ± 1.45 | |||||||||||||
| 85 | 386 | 6.78 ± 0.85 | |||||||||||||
| SLM | 70 | 500 | 400 | 3.10 ± 0.03 | [54] | ||||||||||
| SLM | 80 | 300–400 | 2.34 ± 0.2 | 78.54 ± 9.1 | [55] | ||||||||||
| Osseous Tissues | Manufacturing Technique | Porosity (%) | Pore Size (μm) | Strut Size (μm) | Elastic Modulus (GPa) | Compressive Strength (MPa) | Yield Strength (MPa) | 0.2% Proof Strength (MPa) | Ref | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Porosity%/Samples | In Vitro Tests Results | In Vivo Tests Results | Ref. | ||||||||||||
| Cortical bone | 3–5 | ||||||||||||||
| 80% Ta | Cytotoxicity test (L929 mammalian cells)
|
| Histological evaluation | 7–30 | (rat femur defect model) | 100–230 |
| [47] | |||||||
| [ | 52 | ] | Trabecular bone | 50–90 | |||||||||||
| 70% Ta vs. 70% Ti | Cell morphologies (hBMSCs)
| 0.01–3.0 |
Cell differentiation
| 2–12 |
| ||||||||||
Histological evaluations (rabbit distal femoral defect model)
|
[54] | CVD (porous carbon scaffold) |
75–85 | 400–600 | |||||||||||
| 80% Ta vs. 80% Ti | 40–60 | Cell morphologies (hBMSCs) | 2.5–3.9 | 42–78 |
| Cell differentiation
| [33] | ||||||||
Histological evaluationsand fluorescence labeling (rabbit distal femoral defect model)
|
[55] | CVD (porous SiC scaffold) |
|||||||||||||
| 30% Ta vs. 30% Ti modified with TiO2 | 70–85 | nanotubes, 30% Ti and solid Ti | Not mentioned | Histological analysis (rats distal femur model)
| 150–400 |
| 40–60 |
| 10–30 | 35–100 | FESEM micrographs
| [48] | |||
| Foam impregnation | 65–80 | 400–600 | 2.0–4.6 | 100–170 | [49] | ||||||||||
| Powder metallurgy | 100–400 | 2.0 ± 0.3 | 50.3 ± 0.5 | [50] | |||||||||||
| LENS | 55 | 1.5 ± 0.3 | 100 ± 10 | [51] | |||||||||||
| 45 | 7 ± 0.6 | 192 ± 7 | |||||||||||||
| 27 | 20 ± 1.9 | 746 ± 27 | |||||||||||||
| SLM | 80 | 500 | 150 | 1.22 ± 0.07 | 28.3 ± 1.2 | 12.7 ± 0.6 | [52] | ||||||||
| SEBM | 75 | 540 | 23.98 ± 1.72 | [53] | |||||||||||
| 80 | 392 | 19.48 ± 1.45 | |||||||||||||
| 85 | 386 | 6.78 ± 0.85 | |||||||||||||
| SLM | 70 | 500 | 400 | 3.10 ± 0.03 | [54] | ||||||||||
| SLM | 80 | 300–400 | 2.34 ± 0.2 | 78.54 ± 9.1 | [55] |
| Osseous Tissues | Manufacturing Technique | Porosity (%) |
|---|
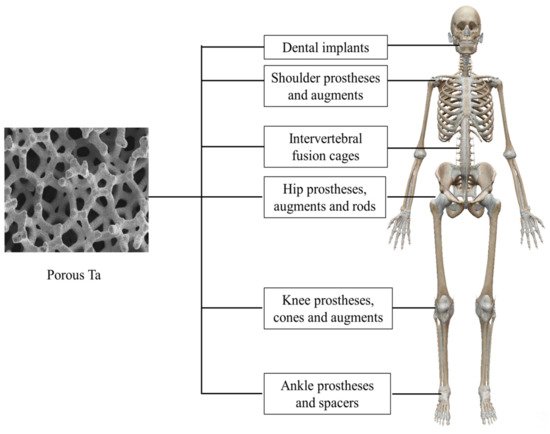

2. Clinical Application of Porous Ta in Orthopedics and Dentistry
2.1. Femoral Head Osteonecrosis
2.1. Femoral Head Osteonecrosis
2.2. Hip Arthroplasty
2.2. Hip Arthroplasty
| Surface Modification | In Vitro Test Results | In Vivo Test Results | Ref. |
|---|---|---|---|
| ACP nanospheres–PLA coating HA nanorods–PLA coating |
Mineralization in SBF
|
Subchondral bone defect repair
|
[163] |
| CaP nanospheres–PLA coating | Mineralization in SBF
|
Subchondral bone defect repair
|
[165] |
| BMP-7 coating | Not mentioned | Cartilage defect restoration (rabbit model)
|
[166] |
| Ta2O5 nanotubes films | Anticorrosion test
|
||
| 27% Ta and 45% Ta vs. 27% Ti | Cell morphologies (hFOB CRL-11372)
|
Not mentioned | [51] |
|
| |||
| Not mentioned | [167] | ||
| Nanoporous Ta oxide layers | Cell proliferation and morphology (L929 mouse fibroblasts)
|
Not mentioned | [168] |
| MAO combined with NaOH treatment | Mineralization in SBF
|
Bone ingrowth (rabbit cranial defect model)
|
[169] |
| PHAs (PHB, PHBV and PHB4HB)–Genta coating | Cytotoxicity and cell adhesion (SaOS-2 cells)
|
Not mentioned | [170] |
| ZnO nanorods−nanoslices hierarchical structure coating | Antibacterial Properties (S. aureus and E. coli)
|
In vivo Infected Studies (KM mice subcutaneous implantation)
|
[164] |
References
- Wauthle, R.; van der Stok, J.; Amin Yavari, S.; Van Humbeeck, J.; Kruth, J.P.; Zadpoor, A.A.; Weinans, H.; Mulier, M.; Schrooten, J. Additively manufactured porous tantalum implants. Acta Biomater. 2015, 14, 217–225. Weeks, M.E. The discovery of the elements. VII. Columbium, tantalum, and vanadium. J. Chem. Educ. 1932, 9, 863.
- Wang, H.; Su, K.; Su, L.; Liang, P.; Ji, P.; Wang, C. Comparison of 3D-printed porous tantalum and titanium scaffolds on osteointegration and osteogenesis. Mater. Sci. Eng. C Mater. Biol. Appl. 2019, 104, 109908. Fuerst, J.; Medlin, D.; Carter, M.; Sears, J.; Vander Voort, G. LASER Additive Manufacturing of Titanium-Tantalum Alloy Structured Interfaces for Modular Orthopedic Devices. JOM 2015, 67, 775–780.
- Guo, Y.; Xie, K.; Jiang, W.; Wang, L.; Li, G.; Zhao, S.; Wu, W.; Hao, Y. In Vitro and in Vivo Study of 3D-Printed Porous Tantalum Scaffolds for Repairing Bone Defects. ACS Biomater. Sci. Eng. 2018, 5, 1123–1133. Thijs, L.; Montero Sistiaga, M.L.; Wauthle, R.; Xie, Q.; Kruth, J.-P.; Van Humbeeck, J. Strong morphological and crystallographic texture and resulting yield strength anisotropy in selective laser melted tantalum. Acta Mater. 2013, 61, 4657–4668.
- Bandyopadhyay, A.; Mitra, I.; Shivaram, A.; Dasgupta, N.; Bose, S. Direct comparison of additively manufactured porous titanium and tantalum implants towards in vivo osseointegration. Addit. Manuf. 2019, 28, 259–266. Zhou, L.; Yuan, T.; Li, R.; Tang, J.; Wang, G.; Guo, K. Selective laser melting of pure tantalum: Densification, microstructure and mechanical behaviors. Mater. Sci. Eng. A 2017, 707, 443–451.
- Balla, V.K.; Bodhak, S.; Bose, S.; Bandyopadhyay, A. Porous tantalum structures for bone implants: Fabrication, mechanical and in vitro biological properties. Acta Biomater. 2010, 6, 3349–3359. Black, J. Biological performance of tantalum. Clin. Mater. 1994, 16, 167–173.
- Zhou, R.; Ni, H.-J.; Peng, J.-H.; Liu, N.; Chen, S.; Shao, J.-H.; Fu, Q.-W.; Liu, J.-J.; Chen, F.; Qian, Q.-R. The mineralization, drug release and in vivo bone defect repair properties of calcium phosphates/PLA modified tantalum scaffolds. RSC Adv. 2020, 10, 7708–7717. Wang, F.; Li, C.; Zhang, S.; Liu, H. Tantalum coated on titanium dioxide nanotubes by plasma spraying enhances cytocompatibility for dental implants. Surf. Coat. Technol. 2020, 382, 125161.
- Zhou, R.; Xu, W.; Chen, F.; Qi, C.; Lu, B.Q.; Zhang, H.; Wu, J.; Qian, Q.R.; Zhu, Y.J. Amorphous calcium phosphate nanospheres/polylactide composite coated tantalum scaffold: Facile preparation, fast biomineralization and subchondral bone defect repair application. Colloids Surf. B Biointerfaces 2014, 123, 236–245. Zhang, X.M.; Li, Y.; Gu, Y.X.; Zhang, C.N.; Lai, H.C.; Shi, J.Y. Ta-Coated Titanium Surface with Superior Bacteriostasis and Osseointegration. Int. J. Nanomedicine 2019, 14, 8693–8706.
- Wang, Q.; Zhang, H.; Gan, H.; Wang, H.; Li, Q.; Wang, Z. Application of combined porous tantalum scaffolds loaded with bone morphogenetic protein 7 to repair of osteochondral defect in rabbits. Int. Orthop. 2018, 42, 1437–1448. Lu, R.J.; Wang, X.; He, H.X.; E, L.L.; Li, Y.; Zhang, G.L.; Li, C.J.; Ning, C.Y.; Liu, H.C. Tantalum-incorporated hydroxyapatite coating on titanium implants: Its mechanical and in vitro osteogenic properties. J. Mater. Sci. Mater. Med. 2019, 30, 111.
- Wang, N.; Li, H.; Wang, J.; Chen, S.; Ma, Y.; Zhang, Z. Study on the anticorrosion, biocompatibility, and osteoinductivity of tantalum decorated with tantalum oxide nanotube array films. ACS Appl. Mater. Interfaces 2012, 4, 4516–4523. Rahmati, B.; Sarhan, A.A.D.; Basirun, W.J.; Abas, W.A.B.W. Ceramic tantalum oxide thin film coating to enhance the corrosion and wear characteristics of Ti 6Al 4V alloy. J. Alloys Compd. 2016, 676, 369–376.
- Uslu, E.; Oztatli, H.; Garipcan, B.; Ercan, B. Fabrication and cellular interactions of nanoporous tantalum oxide. J. Biomed. Mater. Res. B Appl. Biomater. 2020, 108, 2743–2753. Zhu, H.; Ji, X.; Guan, H.; Zhao, L.; Zhao, L.; Liu, C.; Cai, C.; Li, W.; Tao, T.; Reseland, J.E.; et al. Tantalum nanoparticles reinforced polyetheretherketone shows enhanced bone formation. Mater. Sci. Eng. C Mater. Biol. Appl. 2019, 101, 232–242.
- Gao, H.; Jie, Y.F.; Wang, Z.Q.; Wan, H.; Gong, L.; Lu, R.C.; Xue, Y.K.; Li, D.; Wang, H.Y.; Hao, L.N.; et al. Bioactive tantalum metal prepared by micro-arc oxidation and NaOH treatment. J. Mater. Chem. B 2014, 2, 1216–1224. Singh, B.; Singh, G.; Sidhu, B.S. In vitro investigation of Nb Ta alloy coating deposited on CoCr alloy for biomedical implants. Surf. Coat. Technol. 2019, 377.
- Rodriguez-Contreras, A.; Guillem-Marti, J.; Lopez, O.; Manero, J.M.; Ruperez, E. Antimicrobial PHAs coatings for solid and porous tantalum implants. Colloids Surf. B Biointerfaces 2019, 182, 110317. Skoog, S.A.; Kumar, G.; Goering, P.L.; Williams, B.; Stiglich, J.; Narayan, R.J. Biological Response of Human Bone Marrow-Derived Mesenchymal Stem Cells to Commercial Tantalum Coatings with Microscale and Nanoscale Surface Topographies. JOM 2016, 68, 1672–1678.
- Liao, H.; Miao, X.; Ye, J.; Wu, T.; Deng, Z.; Li, C.; Jia, J.; Cheng, X.; Wang, X. Falling Leaves Inspired ZnO Nanorods-Nanoslices Hierarchical Structure for Implant Surface Modification with Two Stage Releasing Features. ACS Appl. Mater. Interfaces 2017, 9, 13009–13015. Jin, W.; Wang, G.; Peng, X.; Li, W.; Qasim, A.M.; Chu, P.K. Tantalum nitride films for corrosion protection of biomedical Mg-Y-RE alloy. J. Alloys Compd. 2018, 764, 947–958.
- Wang, Q.; Zhang, H.; Gan, H.; Wang, H.; Li, Q.; Wang, Z. Application of combined porous tantalum scaffolds loaded with bone morphogenetic protein 7 to repair of osteochondral defect in rabbits. Int. Orthop. 2018, 42, 1437–1448. Wang, H.; Zheng, Y.; Jiang, C.; Li, Y.; Fu, Y. In vitro corrosion behavior and cytocompatibility of pure Fe implanted with Ta. Surf. Coat. Technol. 2017, 320, 201–205.
- Wang, N.; Li, H.; Wang, J.; Chen, S.; Ma, Y.; Zhang, Z. Study on the anticorrosion, biocompatibility, and osteoinductivity of tantalum decorated with tantalum oxide nanotube array films. ACS Appl. Mater. Interfaces 2012, 4, 4516–4523. Moreira, H.; Costa-Barbosa, A.; Marques, S.M.; Sampaio, P.; Carvalho, S. Evaluation of cell activation promoted by tantalum and tantalum oxide coatings deposited by reactive DC magnetron sputtering. Surf. Coat. Technol. 2017, 330, 260–269.
- Uslu, E.; Oztatli, H.; Garipcan, B.; Ercan, B. Fabrication and cellular interactions of nanoporous tantalum oxide. J. Biomed. Mater. Res. B Appl. Biomater. 2020, 108, 2743–2753. Harrison, P.L.; Harrison, T.; Stockley, I.; Smith, T.J. Does tantalum exhibit any intrinsic antimicrobial or antibiofilm properties? Bone Joint J. 2017, 99B, 1153–1156.
- Gao, H.; Jie, Y.F.; Wang, Z.Q.; Wan, H.; Gong, L.; Lu, R.C.; Xue, Y.K.; Li, D.; Wang, H.Y.; Hao, L.N.; et al. Bioactive tantalum metal prepared by micro-arc oxidation and NaOH treatment. J. Mater. Chem. B 2014, 2, 1216–1224. Schildhauer, T.A.; Robie, B.; Muhr, G.; Koller, M. Bacterial adherence to tantalum versus commonly used orthopedic metallic implant materials. J. Orthop. Trauma 2006, 20, 476–484.
- Rodriguez-Contreras, A.; Guillem-Marti, J.; Lopez, O.; Manero, J.M.; Ruperez, E. Antimicrobial PHAs coatings for solid and porous tantalum implants. Colloids Surf. B Biointerfaces 2019, 182, 110317. Shimabukuro, M.; Ito, H.; Tsutsumi, Y.; Nozaki, K.; Chen, P.; Yamada, R.; Ashida, M.; Nagai, A.; Hanawa, T. The Effects of Various Metallic Surfaces on Cellular and Bacterial Adhesion. Metals 2019, 9, 1145.
- Liao, H.; Miao, X.; Ye, J.; Wu, T.; Deng, Z.; Li, C.; Jia, J.; Cheng, X.; Wang, X. Falling Leaves Inspired ZnO Nanorods-Nanoslices Hierarchical Structure for Implant Surface Modification with Two Stage Releasing Features. ACS Appl. Mater. Interfaces 2017, 9, 13009–13015. Miyazaki, T.; Kim, H.-M.; Kokubo, T.; Ohtsuki, C.; Kato, H.; Nakamura, T. Mechanism of bonelike apatite formation on bioactive tantalum metal in a simulated body fluid. Biomaterials 2002, 23, 827–832.
- Levine, B.R.; Sporer, S.; Poggie, R.A.; Della Valle, C.J.; Jacobs, J.J. Experimental and clinical performance of porous tantalum in orthopedic surgery. Biomaterials 2006, 27, 4671–4681.
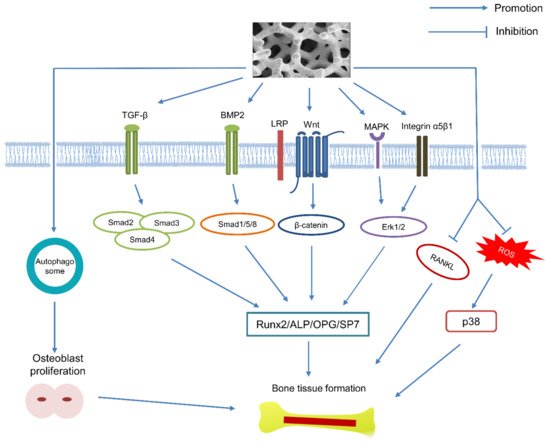
- Kang, C.; Wei, L.; Song, B.; Chen, L.; Liu, J.; Deng, B.; Pan, X.; Shao, L. Involvement of autophagy in tantalum nanoparticle-induced osteoblast proliferation. Int. J. Nanomedicine 2017, 12, 4323–4333.
- Shi, L.Y.; Wang, A.; Zang, F.Z.; Wang, J.X.; Pan, X.W.; Chen, H.J. Tantalum-coated pedicle screws enhance implant integration. Colloids Surf. B Biointerfaces 2017, 160, 22–32.
- Lu, M.M.; Wu, P.S.; Guo, X.J.; Yin, L.L.; Cao, H.L.; Zou, D. Osteoinductive effects of tantalum and titanium on bone mesenchymal stromal cells and bone formation in ovariectomized rats. Eur. Rev. Med. Pharmacol. Sci. 2018, 22, 7087–7104.
- Duan, P.; Bonewald, L.F. The role of the wnt/beta-catenin signaling pathway in formation and maintenance of bone and teeth. Int. J. Biochem. Cell Biol. 2016, 77, 23–29.
- Lu, M.; Zhuang, X.; Tang, K.; Wu, P.; Guo, X.; Yin, L.; Cao, H.; Zou, D. Intrinsic Surface Effects of Tantalum and Titanium on Integrin alpha5beta1/ ERK1/2 Pathway-Mediated Osteogenic Differentiation in Rat Bone Mesenchymal Stromal Cells. Cell. Physiol. Biochem. 2018, 51, 589–609.
- Qian, H.; Lei, T.; Ye, Z.; Hu, Y.; Lei, P. From the Performance to the Essence: The Biological Mechanisms of How Tantalum Contributes to Osteogenesis. Biomed. Res. Int. 2020, 2020, 5162524.
- Dou, X.; Wei, X.; Liu, G.; Wang, S.; Lv, Y.; Li, J.; Ma, Z.; Zheng, G.; Wang, Y.; Hu, M.; et al. Effect of porous tantalum on promoting the osteogenic differentiation of bone marrow mesenchymal stem cells in vitro through the MAPK/ERK signal pathway. J. Orthop. Translat. 2019, 19, 81–93.
- Wang, L.; Hu, X.; Ma, X.; Ma, Z.; Zhang, Y.; Lu, Y.; Li, X.; Lei, W.; Feng, Y. Promotion of osteointegration under diabetic conditions by tantalum coating-based surface modification on 3-dimensional printed porous titanium implants. Colloids Surf. B Biointerfaces 2016, 148, 440–452.
- Van Bael, S.; Chai, Y.C.; Truscello, S.; Moesen, M.; Kerckhofs, G.; Van Oosterwyck, H.; Kruth, J.P.; Schrooten, J. The effect of pore geometry on the in vitro biological behavior of human periosteum-derived cells seeded on selective laser-melted Ti6Al4V bone scaffolds. Acta Biomater. 2012, 8, 2824–2834.
- Van der Stok, J.; Van der Jagt, O.P.; Amin Yavari, S.; De Haas, M.F.; Waarsing, J.H.; Jahr, H.; Van Lieshout, E.M.; Patka, P.; Verhaar, J.A.; Zadpoor, A.A.; et al. Selective laser melting-produced porous titanium scaffolds regenerate bone in critical size cortical bone defects. J. Orthop. Res. 2013, 31, 792–799.
- Bobbert, F.S.L.; Zadpoor, A.A. Effects of bone substitute architecture and surface properties on cell response, angiogenesis, and structure of new bone. J. Mater. Chem. B 2017, 5, 6175–6192.
- Arabnejad, S.; Burnett Johnston, R.; Pura, J.A.; Singh, B.; Tanzer, M.; Pasini, D. High-strength porous biomaterials for bone replacement: A strategy to assess the interplay between cell morphology, mechanical properties, bone ingrowth and manufacturing constraints. Acta Biomater. 2016, 30, 345–356.
- Zardiackas, L.D.; Parsell, D.E.; Dillon, L.D.; Mitchell, D.W.; Nunnery, L.A.; Poggie, R. Structure, metallurgy, and mechanical properties of a porous tantalum foam. J. Biomed. Mater. Res. 2001, 58, 180–187.
- Sinclair, S.K.; Konz, G.J.; Dawson, J.M.; Epperson, R.T.; Bloebaum, R.D. Host bone response to polyetheretherketone versus porous tantalum implants for cervical spinal fusion in a goat model. Spine 2012, 37, 571–580.
- Gruen, T.A.; Poggie, R.A.; Lewallen, D.G.; Hanssen, A.D.; Lewis, R.J.; O’Keefe, T.J.; Stulberg, S.D.; Sutherland, C.J. Radiographic evaluation of a monoblock acetabular component: A multicenter study with 2- to 5-year results. J. Arthroplasty 2005, 20, 369–378.
- Piglionico, S.; Bousquet, J.; Fatima, N.; Renaud, M.; Collart-Dutilleul, P.Y.; Bousquet, P. Porous Tantalum VS. Titanium Implants: Enhanced Mineralized Matrix Formation after Stem Cells Proliferation and Differentiation. J. Clin. Med. 2020, 9, 3657.
- Bobyn, J.D.; Toh, K.K.; Hacking, S.A.; Tanzer, M.; Krygier, J.J. Tissue response to porous tantalum acetabular cups: A canine model. J. Arthroplasty 1999, 14, 347–354.
- Bobyn, J.D.; Stackpool, G.J.; Hacking, S.A.; Tanzer, M.; Krygier, J.J. Characteristics of bone ingrowth and interface mechanics of a new porous tantalum biomaterial. J. Bone Joint Surg. Br. 1999, 81, 907–914.
- Gordon, W.J.; Conzemius, M.G.; Birdsall, E.; Wannemuehler, Y.; Mallapragada, S.; Lewallen, D.G.; Yaszemski, M.J.; O’Driscoll, S.W. Chondroconductive potential of tantalum trabecular metal. J. Biomed. Mater. Res. B Appl. Biomater. 2005, 75, 229–233.
- Hacking, S.A.; Bobyn, J.D.; Toh, K.; Tanzer, M.; Krygier, J.J. Fibrous tissue ingrowth and attachment to porous tantalum. J. Biomed. Mater. Res. 2000, 52, 631–638.
- Reach, J.S., Jr.; Dickey, I.D.; Zobitz, M.E.; Adams, J.E.; Scully, S.P.; Lewallen, D.G. Direct tendon attachment and healing to porous tantalum: An experimental animal study. J. Bone Joint Surg. Am. 2007, 89, 1000–1009.
- Zhang, Y.; Ahn, P.B.; Fitzpatrick, D.C.; Heiner, A.D.; Poggie, R.A.; Brown, T.D. Interfacial Frictional Behavior: Cancellous Bone, Cortical Bone, and a Novel Porous Tantalum Biomaterial. J. Musculoskelet. Res. 2011, 3, 245–251.
- Mandal, B.B.; Kundu, S.C. Cell proliferation and migration in silk fibroin 3D scaffolds. Biomaterials 2009, 30, 2956–2965.
- Sagomonyants, K.B.; Hakim-Zargar, M.; Jhaveri, A.; Aronow, M.S.; Gronowicz, G. Porous tantalum stimulates the proliferation and osteogenesis of osteoblasts from elderly female patients. J. Orthop. Res. 2011, 29, 609–616.
- Levine, B.; Della Valle, C.J.; Jacobs, J.J. Applications of porous tantalum in total hip arthroplasty. J. Am. Acad. Orthop. Surg. 2006, 14, 646–655.
- Harrison, A.K.; Gioe, T.J.; Simonelli, C.; Tatman, P.J.; Schoeller, M.C. Do porous tantalum implants help preserve bone?: Evaluation of tibial bone density surrounding tantalum tibial implants in TKA. Clin. Orthop. Relat. Res. 2010, 468, 2739–2745.
- Wang, X.; Xu, S.; Zhou, S.; Xu, W.; Leary, M.; Choong, P.; Qian, M.; Brandt, M.; Xie, Y.M. Topological design and additive manufacturing of porous metals for bone scaffolds and orthopaedic implants: A review. Biomaterials 2016, 83, 127–141.
- Wei, X.; Zhao, D.; Wang, B.; Wang, W.; Kang, K.; Xie, H.; Liu, B.; Zhang, X.; Zhang, J.; Yang, Z. Tantalum coating of porous carbon scaffold supplemented with autologous bone marrow stromal stem cells for bone regeneration in vitro and in vivo. Exp. Biol. Med. 2016, 241, 592–602.
- Wang, Q.; Zhang, H.; Li, Q.; Ye, L.; Gan, H.; Liu, Y.; Wang, H.; Wang, Z. Biocompatibility and osteogenic properties of porous tantalum. Exp. Ther Med. 2015, 9, 780–786.
- Zhou, Y.; Zhu, Y. Three-dimensional Ta foams produced by replication of NaCl space-holders. Mater. Lett. 2013, 99, 8–10.
- Balla, V.K.; Bodhak, S.; Bose, S.; Bandyopadhyay, A. Porous tantalum structures for bone implants: Fabrication, mechanical and in vitro biological properties. Acta Biomater. 2010, 6, 3349–3359.
- Wauthle, R.; van der Stok, J.; Amin Yavari, S.; Van Humbeeck, J.; Kruth, J.P.; Zadpoor, A.A.; Weinans, H.; Mulier, M.; Schrooten, J. Additively manufactured porous tantalum implants. Acta Biomater. 2015, 14, 217–225.
- Tang, H.P.; Yang, K.; Jia, L.; He, W.W.; Yang, L.; Zhang, X.Z. Tantalum Bone Implants Printed by Selective Electron Beam Manufacturing (SEBM) and Their Clinical Applications. JOM 2020, 72, 1016–1021.
- Wang, H.; Su, K.; Su, L.; Liang, P.; Ji, P.; Wang, C. Comparison of 3D-printed porous tantalum and titanium scaffolds on osteointegration and osteogenesis. Mater. Sci. Eng. C Mater. Biol. Appl. 2019, 104, 109908.
- Guo, Y.; Xie, K.; Jiang, W.; Wang, L.; Li, G.; Zhao, S.; Wu, W.; Hao, Y. In Vitro and in Vivo Study of 3D-Printed Porous Tantalum Scaffolds for Repairing Bone Defects. ACS Biomater. Sci. Eng. 2018, 5, 1123–1133.
- Qian, H.; Lei, T.; Lei, P.; Hu, Y. Additively Manufactured Tantalum Implants for Repairing Bone Defects: A Systematic Review. Tissue Eng. Part. B. Rev. 2021, 27, 166–180.
- Butler, J.S.; Lui, D.F.; Malhotra, K.; Suarez-Huerta, M.L.; Yu, H.; Selvadurai, S.; Agu, O.; Molloy, S. 360-Degree Complex Primary Reconstruction Using Porous Tantalum Cages for Adult Degenerative Spinal Deformity. Global Spine J. 2019, 9, 613–618.
- Pakos, E.E.; Megas, P.; Paschos, N.K.; Syggelos, S.A.; Kouzelis, A.; Georgiadis, G.; Xenakis, T.A. Modified porous tantalum rod technique for the treatment of femoral head osteonecrosis. World J. Orthop. 2015, 6, 829–837.
- Witek, L.; Alifarag, A.M.; Tovar, N.; Lopez, C.D.; Gil, L.F.; Gorbonosov, M.; Hannan, K.; Neiva, R.; Coelho, P.G. Osteogenic parameters surrounding trabecular tantalum metal implants in osteotomies prepared via osseodensification drilling. Medicina Oral, Patologia Oral y Cirugia Bucal 2019, 24, e764–e769.
- Simon, S.; Frank, B.J.H.; Aichmair, A.; Dominkus, M.; Hofstaetter, J.G. Reconstruction of Proximal Metaphyseal Femoral Defects Using Trabecular Metal Augments in Revision Total Hip Arthroplasty. Arthroplast. Today 2021, 8, 216–221.
- Liu, F.; Wang, W.; Yang, L.; Wang, B.; Wang, J.; Chai, W.; Zhao, D. An epidemiological study of etiology and clinical characteristics in patients with nontraumatic osteonecrosis of the femoral head. J. Res. Med. Sci 2017, 22, 15.
- Ikeuchi, K.; Hasegawa, Y.; Seki, T.; Takegami, Y.; Amano, T.; Ishiguro, N. Epidemiology of nontraumatic osteonecrosis of the femoral head in Japan. Mod. Rheumatol. 2015, 25, 278–281.
- Scully, S.P.; Aaron, R.K.; Urbaniak, J.R. Survival analysis of hips treated with core decompression or vascularized fibular grafting because of avascular necrosis. J. Bone Joint Surg. Am. 1998, 80, 1270–1275.
- Gonzalez del Pino, J.; Knapp, K.; Gomez Castresana, F.; Benito, M. Revascularization of femoral head ischemic necrosis with vascularized bone graft: A CT scan experimental study. Skeletal Radiol. 1990, 19, 197–202.
- Tsao, A.K.; Roberson, J.R.; Christie, M.J.; Dore, D.D.; Heck, D.A.; Robertson, D.D.; Poggie, R.A. Biomechanical and clinical evaluations of a porous tantalum implant for the treatment of early-stage osteonecrosis. J. Bone Joint Surg. Am. 2005, 87 (Suppl. 2), 22–27.
- Veillette, C.J.; Mehdian, H.; Schemitsch, E.H.; McKee, M.D. Survivorship analysis and radiographic outcome following tantalum rod insertion for osteonecrosis of the femoral head. J. Bone Joint Surg. Am. 2006, 88 (Suppl. 3), 48–55.
- Shuler, M.S.; Rooks, M.D.; Roberson, J.R. Porous tantalum implant in early osteonecrosis of the hip: Preliminary report on operative, survival, and outcomes results. J. Arthroplasty 2007, 22, 26–31.
- Liu, G.; Wang, J.; Yang, S.; Xu, W.; Ye, S.; Xia, T. Effect of a porous tantalum rod on early and intermediate stages of necrosis of the femoral head. Biomed. Mater. 2010, 5, 065003.
- Zhang, X.; Wang, J.; Xiao, J.; Shi, Z. Early failures of porous tantalum osteonecrosis implants: A case series with retrieval analysis. Int. Orthop. 2016, 40, 1827–1834.
- Papapietro, N.; Di Martino, A.; Niccoli, G.; Palumbo, A.; Salvatore, G.; Forriol, F.; Denaro, V. Trabecular metal screw implanted for avascular necrosis of the femoral head may complicate subsequent arthroplasty surgery. Eur. J. Orthop. Surg. Traumatol. 2014, 24, 931–938.
- Liu, Y.; Su, X.; Zhou, S.; Wang, L.; Wang, C.; Liu, S. A modified porous tantalum implant technique for osteonecrosis of the femoral head: Survivorship analysis and prognostic factors for radiographic progression and conversion to total hip arthroplasty. Int. J. Clin. Exp. Med. 2015, 8, 1918–1930.
- Liu, Z.H.; Guo, W.S.; Li, Z.R.; Cheng, L.M.; Zhang, Q.D.; Yue, D.B.; Shi, Z.C.; Wang, B.L.; Sun, W.; Zhang, N.F. Porous tantalum rods for treating osteonecrosis of the femoral head. Genet. Mol. Res. 2014, 13, 8342–8352.
- Ma, J.; Sun, W.; Gao, F.; Guo, W.; Wang, Y.; Li, Z. Porous Tantalum Implant in Treating Osteonecrosis of the Femoral Head: Still a Viable Option? Sci Rep. 2016, 6, 28227.
- Tanzer, M.; Bobyn, J.D.; Krygier, J.J.; Karabasz, D. Histopathologic retrieval analysis of clinically failed porous tantalum osteonecrosis implants. J. Bone Joint Surg. Am. 2008, 90, 1282–1289.
- Varitimidis, S.E.; Dimitroulias, A.P.; Karachalios, T.S.; Dailiana, Z.H.; Malizos, K.N. Outcome after tantalum rod implantation for treatment of femoral head osteonecrosis: 26 hips followed for an average of 3 years. Acta Orthop. 2009, 80, 20–25.
- Zhao, D.; Zhang, Y.; Wang, W.; Liu, Y.; Li, Z.; Wang, B.; Yu, X. Tantalum rod implantation and vascularized iliac grafting for osteonecrosis of the femoral head. Orthopedics 2013, 36, 789–795.
- Zhao, D.; Liu, B.; Wang, B.; Yang, L.; Xie, H.; Huang, S.; Zhang, Y.; Wei, X. Autologous bone marrow mesenchymal stem cells associated with tantalum rod implantation and vascularized iliac grafting for the treatment of end-stage osteonecrosis of the femoral head. Biomed. Res. Int. 2015, 2015, 240506.
- Sculco, T.P. The acetabular component: An elliptical monoblock alternative. J. Arthroplasty 2002, 17, 118–120.
- Meneghini, R.M.; Ford, K.S.; McCollough, C.H.; Hanssen, A.D.; Lewallen, D.G. Bone remodeling around porous metal cementless acetabular components. J. Arthroplasty 2010, 25, 741–747.
- Jasty, M.; Bragdon, C.R.; Haire, T.; Mulroy, R.D., Jr.; Harris, W.H. Comparison of bone ingrowth into cobalt chrome sphere and titanium fiber mesh porous coated cementless canine acetabular components. J. Biomed. Mater. Res. 1993, 27, 639–644.
- Macheras, G.; Kateros, K.; Kostakos, A.; Koutsostathis, S.; Danomaras, D.; Papagelopoulos, P.J. Eight- to ten-year clinical and radiographic outcome of a porous tantalum monoblock acetabular component. J. Arthroplasty 2009, 24, 705–709.
- Garbuz, D.S. Revision total hip: A novel modular cementless acetabular system for reconstruction of severe acetabular bone loss. Oper. Tech. Orthop. 2004, 14, 117–120.
- Van Kleunen, J.P.; Lee, G.C.; Lementowski, P.W.; Nelson, C.L.; Garino, J.P. Acetabular revisions using trabecular metal cups and augments. J. Arthroplasty 2009, 24, 64–68.
- Sporer, S.M.; Paprosky, W.G. The use of a trabecular metal acetabular component and trabecular metal augment for severe acetabular defects. J. Arthroplasty 2006, 21, 83–86.
- Sporer, S.M.; Paprosky, W.G. Acetabular revision using a trabecular metal acetabular component for severe acetabular bone loss associated with a pelvic discontinuity. J. Arthroplasty 2006, 21, 87–90.
- Siegmeth, A.; Duncan, C.P.; Masri, B.A.; Kim, W.Y.; Garbuz, D.S. Modular tantalum augments for acetabular defects in revision hip arthroplasty. Clin. Orthop. Relat. Res. 2009, 467, 199–205.
- Malkani, A.L.; Price, M.R.; Crawford, C.H., 3rd; Baker, D.L. Acetabular component revision using a porous tantalum biomaterial: A case series. J. Arthroplasty 2009, 24, 1068–1073.
- Issack, P.S. Use of porous tantalum for acetabular reconstruction in revision hip arthroplasty. J. Bone Joint Surg. Am. 2013, 95, 1981–1987.
- Nehme, A.; Lewallen, D.G.; Hanssen, A.D. Modular porous metal augments for treatment of severe acetabular bone loss during revision hip arthroplasty. Clin. Orthop. Relat. Res. 2004, 429, 201–208.
- Unger, A.S.; Lewis, R.J.; Gruen, T. Evaluation of a porous tantalum uncemented acetabular cup in revision total hip arthroplasty: Clinical and radiological results of 60 hips. J. Arthroplasty 2005, 20, 1002–1009.
- Weeden, S.H.; Schmidt, R.H. The use of tantalum porous metal implants for Paprosky 3A and 3B defects. J. Arthroplasty 2007, 22, 151–155.
- Del Gaizo, D.J.; Kancherla, V.; Sporer, S.M.; Paprosky, W.G. Tantalum augments for Paprosky IIIA defects remain stable at midterm followup. Clin. Orthop. Relat. Res. 2012, 470, 395–401.
- Flecher, X.; Appy, B.; Parratte, S.; Ollivier, M.; Argenson, J.N. Use of porous tantalum components in Paprosky two and three acetabular revision. A minimum five-year follow-up of fifty one hips. Int. Orthop. 2017, 41, 911–916.
- Jenkins, D.R.; Odland, A.N.; Sierra, R.J.; Hanssen, A.D.; Lewallen, D.G. Minimum Five-Year Outcomes with Porous Tantalum Acetabular Cup and Augment Construct in Complex Revision Total Hip Arthroplasty. J. Bone Joint Surg. Am. 2017, 99, e49.
- Lochel, J.; Janz, V.; Hipfl, C.; Perka, C.; Wassilew, G.I. Reconstruction of acetabular defects with porous tantalum shells and augments in revision total hip arthroplasty at ten-year follow-up. Bone Joint J. 2019, 101-B, 311–316.
- Brown, T.S.; Salib, C.G.; Rose, P.S.; Sim, F.H.; Lewallen, D.G.; Abdel, M.P. Reconstruction of the hip after resection of periacetabular oncological lesions: A systematic review. Bone Joint J. 2018, 100-B, 22–30.
- Rose, P.S.; Halasy, M.; Trousdale, R.T.; Hanssen, A.D.; Sim, F.H.; Berry, D.J.; Lewallen, D.G. Preliminary results of tantalum acetabular components for THA after pelvic radiation. Clin. Orthop. Relat. Res. 2006, 453, 195–198.
- Joglekar, S.B.; Rose, P.S.; Lewallen, D.G.; Sim, F.H. Tantalum acetabular cups provide secure fixation in THA after pelvic irradiation at minimum 5-year followup. Clin. Orthop. Relat. Res. 2012, 470, 3041–3047.
- De Paolis, M.; Zucchini, R.; Romagnoli, C.; Romantini, M.; Mariotti, F.; Donati, D.M. Middle term results of tantalum acetabular cups in total hip arthroplasty following pelvic irradiation. Acta Orthop Traumatol. Turc. 2019, 53, 165–169.
- Mahoney, C.R.; Garvin, K.L. Periprosthetic acetabular stress fracture causing pelvic discontinuity. Orthopedics 2002, 25, 83–85.
- Springer, B.D.; Berry, D.J.; Cabanela, M.E.; Hanssen, A.D.; Lewallen, D.G. Early postoperative transverse pelvic fracture: A new complication related to revision arthroplasty with an uncemented cup. J. Bone Joint Surg. Am. 2005, 87, 2626–2631.
- Levine, B.; Sporer, S.; Della Valle, C.J.; Jacobs, J.J.; Paprosky, W. Porous tantalum in reconstructive surgery of the knee: A review. J. Knee Surg. 2007, 20, 185–194.
- Helm, A.T.; Kerin, C.; Ghalayini, S.R.; McLauchlan, G.J. Preliminary results of an uncemented trabecular metal tibial component in total knee arthroplasty. J. Arthroplasty 2009, 24, 941–944.
- O’Keefe, T.J.; Winter, S.; Lewallen, D.G.; Robertson, D.D.; Poggie, R.A. Clinical and radiographic evaluation of a monoblock tibial component. J. Arthroplasty 2010, 25, 785–792.
- Kamath, A.F.; Lee, G.C.; Sheth, N.P.; Nelson, C.L.; Garino, J.P.; Israelite, C.L. Prospective results of uncemented tantalum monoblock tibia in total knee arthroplasty: Minimum 5-year follow-up in patients younger than 55 years. J. Arthroplasty 2011, 26, 1390–1395.
- Unger, A.S.; Duggan, J.P. Midterm results of a porous tantalum monoblock tibia component clinical and radiographic results of 108 knees. J. Arthroplasty 2011, 26, 855–860.
- De Martino, I.; D’Apolito, R.; Sculco, P.K.; Poultsides, L.A.; Gasparini, G. Total Knee Arthroplasty Using Cementless Porous Tantalum Monoblock Tibial Component: A Minimum 10-Year Follow-Up. J. Arthroplasty 2016, 31, 2193–2198.
- DeFrancesco, C.J.; Canseco, J.A.; Nelson, C.L.; Israelite, C.L.; Kamath, A.F. Uncemented Tantalum Monoblock Tibial Fixation for Total Knee Arthroplasty in Patients Less Than 60 Years of Age: Mean 10-Year Follow-up. J. Bone Joint Surg. Am. 2018, 100, 865–870.
- Sambaziotis, C.; Lovy, A.J.; Koller, K.E.; Bloebaum, R.D.; Hirsh, D.M.; Kim, S.J. Histologic retrieval analysis of a porous tantalum metal implant in an infected primary total knee arthroplasty. J. Arthroplasty 2012, 27, 1413–1419.
- Meneghini, R.M.; de Beaubien, B.C. Early failure of cementless porous tantalum monoblock tibial components. J. Arthroplasty 2013, 28, 1505–1508.
- Long, W.J.; Scuderi, G.R. Porous tantalum cones for large metaphyseal tibial defects in revision total knee arthroplasty: A minimum 2-year follow-up. J. Arthroplasty 2009, 24, 1086–1092.
- Potter, G.D., 3rd; Abdel, M.P.; Lewallen, D.G.; Hanssen, A.D. Midterm Results of Porous Tantalum Femoral Cones in Revision Total Knee Arthroplasty. J. Bone Joint Surg. Am. 2016, 98, 1286–1291.
- Kamath, A.F.; Lewallen, D.G.; Hanssen, A.D. Porous tantalum metaphyseal cones for severe tibial bone loss in revision knee arthroplasty: A five to nine-year follow-up. J. Bone Joint Surg. Am. 2015, 97, 216–223.
- Bohl, D.D.; Brown, N.M.; McDowell, M.A.; Levine, B.R.; Sporer, S.M.; Paprosky, W.G.; Della Valle, C.J. Do Porous Tantalum Metaphyseal Cones Improve Outcomes in Revision Total Knee Arthroplasty? J. Arthroplasty 2018, 33, 171–177.
- Nasser, S.; Poggie, R.A. Revision and salvage patellar arthroplasty using a porous tantalum implant. J. Arthroplasty 2004, 19, 562–572.
- Ries, M.D.; Cabalo, A.; Bozic, K.J.; Anderson, M. Porous tantalum patellar augmentation: The importance of residual bone stock. Clin. Orthop. Relat. Res. 2006, 452, 166–170.
- Kamath, A.F.; Gee, A.O.; Nelson, C.L.; Garino, J.P.; Lotke, P.A.; Lee, G.C. Porous tantalum patellar components in revision total knee arthroplasty minimum 5-year follow-up. J. Arthroplasty 2012, 27, 82–87.
- Pantalone, A.; Guelfi, M.; Salini, V.; Guelfi, M.G.B. Ankle Arthritis: Etiology and Classifications. In Ankle Joint Arthroscopy; Allegra, F., Cortese, F., Lijoi, F., Eds.; Springer International Publishing: Cham, Switzerland, 2020; pp. 159–163.
- Adukia, V.; Mangwani, J.; Issac, R.; Hussain, S.; Parker, L. Current concepts in the management of ankle arthritis. J. Clin. Orthop Trauma 2020, 11, 388–398.
- Sagherian, B.H.; Claridge, R.J. The Use of Tantalum Metal in Foot and Ankle Surgery. Orthop. Clin. North. Am. 2019, 50, 119–129.
- Frigg, A.; Dougall, H.; Boyd, S.; Nigg, B. Can porous tantalum be used to achieve ankle and subtalar arthrodesis?: A pilot study. Clin. Orthop. Relat. Res. 2010, 468, 209–216.
- Sagherian, B.H.; Claridge, R.J. Porous tantalum as a structural graft in foot and ankle surgery. Foot Ankle Int. 2012, 33, 179–189.
- Bouchard, M.; Barker, L.G.; Claridge, R.J. Technique tip: Tantalum: A structural bone graft option for foot and ankle surgery. Foot Ankle Int. 2004, 25, 39–42.
- Economopoulos, K.; Barker, L.; Beauchamp, C.; Claridge, R. Case report: Reconstruction of the distal tibia with porous tantalum spacer after resection for giant cell tumor. Clin. Orthop. Relat. Res. 2010, 468, 1697–1701.
- Kreulen, C.; Lian, E.; Giza, E. Technique for Use of Trabecular Metal Spacers in Tibiotalocalcaneal Arthrodesis With Large Bony Defects. Foot Ankle Int. 2017, 38, 96–106.
- Lebhar, J.; Kriegel, P.; Chatellier, P.; Breton, Y.; Ropars, M.; Huten, D. Tantalum implants for posterior lumbar interbody fusion: A safe method at medium-term follow-up? Orthop. Traumatol. Surg. Res. 2020, 106, 269–274.
- Boone, D.W. Complications of iliac crest graft and bone grafting alternatives in foot and ankle surgery. Foot Ankle Clin. 2003, 8, 1–14.
- Heary, R.F.; Schlenk, R.P.; Sacchieri, T.A.; Barone, D.; Brotea, C. Persistent iliac crest donor site pain: Independent outcome assessment. Neurosurgery 2002, 50, 510–516; discussion 516–517.
- Sagherian, B.H.; Claridge, R.J. Salvage of failed total ankle replacement using tantalum trabecular metal: Case series. Foot Ankle Int. 2015, 36, 318–324.
- Aubret, S.; Merlini, L.; Fessy, M.; Besse, J.L. Poor outcomes of fusion with Trabecular Metal implants after failed total ankle replacement: Early results in 11 patients. Orthop Traumatol Surg Res. 2018, 104, 231–237.
- Sundet, M.; Johnsen, E.; Eikvar, K.H.; Eriksen, M.L. Retrograde nailing, trabecular metal implant and use of bone marrow aspirate concentrate after failed ankle joint replacement. Foot Ankle Surg. 2021, 27, 123–128.
- Onggo, J.R.; Nambiar, M.; Phan, K.; Hickey, B.; Galvin, M.; Bedi, H. Outcome after total ankle arthroplasty with a minimum of five years follow-up: A systematic review and meta-analysis. Foot Ankle Surg. 2020, 26, 556–563.
- Daniels, T.R.; Younger, A.S.; Penner, M.; Wing, K.; Dryden, P.J.; Wong, H.; Glazebrook, M. Intermediate-term results of total ankle replacement and ankle arthrodesis: A COFAS multicenter study. J. Bone Joint Surg. Am. 2014, 96, 135–142.
- Popelka, S.; Sosna, A.; Vavrik, P.; Jahoda, D.; Bartak, V.; Landor, I. [Eleven-Year Experience with Total Ankle Arthroplasty]. Acta Chir. Orthop. Traumatol. Cech. 2016, 83, 74–83.
- Lamothe, J.; Deland, J.; Schon, L.; Saltzman, C.; Ellis, S. Total ankle replacement through a lateral approach. Tech. Foot Ankle Surg. 2015, 14, 69–78.
- DeVries, J.G.; Derksen, T.A.; Scharer, B.M.; Limoni, R. Perioperative Complications and Initial Alignment of Lateral Approach Total Ankle Arthroplasty. J. Foot Ankle Surg. 2017, 56, 996–1000.
- Bianchi, A.; Martinelli, N.; Hosseinzadeh, M.; Flore, J.; Minoli, C.; Malerba, F.; Galbusera, F. Early clinical and radiological evaluation in patients with total ankle replacement performed by lateral approach and peroneal osteotomy. BMC Musculoskelet Disord. 2019, 20, 132.
- Usuelli, F.G.; Indino, C.; Maccario, C.; Manzi, L.; Salini, V. Total ankle replacement through a lateral approach: Surgical tips. SICOT J. 2016, 2, 38.
- Tan, E.W.; Maccario, C.; Talusan, P.G.; Schon, L.C. Early Complications and Secondary Procedures in Transfibular Total Ankle Replacement. Foot Ankle Int. 2016, 37, 835–841.
- Tiusanen, H.; Kormi, S.; Kohonen, I.; Saltychev, M. Results of Trabecular-Metal Total Ankle Arthroplasties With Transfibular Approach. Foot Ankle Int. 2020, 41, 411–418.
- Barg, A.; Bettin, C.C.; Burstein, A.H.; Saltzman, C.L.; Gililland, J. Early Clinical and Radiographic Outcomes of Trabecular Metal Total Ankle Replacement Using a Transfibular Approach. J. Bone Joint Surg. Am. 2018, 100, 505–515.
- Brigido, S.A.; DiDomenico, L.A. Primary Zimmer Trabecular Metal Total Ankle Replacement. In Primary and Revision Total Ankle Replacement; Roukis, T.S., Berlet, G.C., Bibbo, C., Hyer, C.F., Penner, M.J., Wünschel, M., Prissel, M.A., Eds.; Springer International Publishing: Cham, Switzerland, 2016; pp. 131–149.
- Epperson, R.T.; Barg, A.; Williams, D.L.; Saltzman, C.L. Histological Analysis of a Retrieved Porous Tantalum Total Ankle Replacement: A Case Report. JBJS Case Connect. 2020, 10, e0379.
- Le Guehennec, L.; Soueidan, A.; Layrolle, P.; Amouriq, Y. Surface treatments of titanium dental implants for rapid osseointegration. Dent. Mater. 2007, 23, 844–854.
- Rupp, F.; Scheideler, L.; Olshanska, N.; de Wild, M.; Wieland, M.; Geis-Gerstorfer, J. Enhancing surface free energy and hydrophilicity through chemical modification of microstructured titanium implant surfaces. J. Biomed. Mater. Res. A 2006, 76, 323–334.
- Goldman, M.; Juodzbalys, G.; Vilkinis, V. Titanium surfaces with nanostructures influence on osteoblasts proliferation: A systematic review. J. Oral Maxillofac Res. 2014, 5.
- De Arriba, C.C.; Alobera Gracia, M.A.; Coelho, P.G.; Neiva, R.; Tarnow, D.P.; Del Canto Pingarron, M.; Aguado-Henche, S. Osseoincorporation of Porous Tantalum Trabecular-Structured Metal: A Histologic and Histomorphometric Study in Humans. Int. J. Periodontics Restorative Dent. 2018, 38, 879–885.
- Lee, J.W.; Wen, H.B.; Gubbi, P.; Romanos, G.E. New bone formation and trabecular bone microarchitecture of highly porous tantalum compared to titanium implant threads: A pilot canine study. Clin. Oral Implants Res. 2018, 29, 164–174.
- El Chaar, E.; Castano, A. A Retrospective Survival Study of Trabecular Tantalum Implants Immediately Placed in Posterior Extraction Sockets Using a Flapless Technique. J. Oral Implantol. 2017, 43, 114–124.
- Bencharit, S.; Byrd, W.C.; Hosseini, B. Immediate placement of a porous-tantalum, trabecular metal-enhanced titanium dental implant with demineralized bone matrix into a socket with deficient buccal bone: A clinical report. J. Prosthet. Dent. 2015, 113, 262–269.
- Brauner, E.; Guarino, G.; Jamshir, S.; Papi, P.; Valentini, V.; Pompa, V.; Pompa, G. Evaluation of Highly Porous Dental Implants in Postablative Oral and Maxillofacial Cancer Patients: A Prospective Pilot Clinical Case Series Report. Implant. Dent. 2015, 24, 631–637.
- Papi, P.; Jamshir, S.; Brauner, E.; Di Carlo, S.; Ceci, A.; Piccoli, L.; Pompa, G. Clinical evaluation with 18 months follow-up of new PTTM enhanced dental implants in maxillo-facial post-oncological patients. Ann. Stomatol. 2014, 5, 136–141.
- Schlee, M.; Pradies, G.; Mehmke, W.U.; Beneytout, A.; Stamm, M.; Meda, R.G.; Kamm, T.; Poiroux, F.; Weinlich, F.; del Canto Pingarron, M.; et al. Prospective, Multicenter Evaluation of Trabecular Metal-Enhanced Titanium Dental Implants Placed in Routine Dental Practices: 1-Year Interim Report From the Development Period (2010 to 2011). Clin. Implant. Dent. Relat. Res. 2015, 17, 1141–1153.
- Dimaira, M. Immediate Placement of Trabecular Implants in Sites of Failed Implants. Int. J. Oral Maxillofac. Implants 2019, 34, 77–83.
- Schlee, M.; van der Schoor, W.P.; van der Schoor, A.R. Immediate loading of trabecular metal-enhanced titanium dental implants: Interim results from an international proof-of-principle study. Clin. Implant. Dent. Relat. Res. 2015, 17 (Suppl. 1), 308–320.
- Edelmann, A.R.; Patel, D.; Allen, R.K.; Gibson, C.J.; Best, A.M.; Bencharit, S. Retrospective analysis of porous tantalum trabecular metal-enhanced titanium dental implants. J. Prosthet. Dent. 2019, 121, 404–410.
- Bencharit, S.; Byrd, W.C.; Altarawneh, S.; Hosseini, B.; Leong, A.; Reside, G.; Morelli, T.; Offenbacher, S. Development and applications of porous tantalum trabecular metal-enhanced titanium dental implants. Clin. Implant. Dent. Relat. Res. 2014, 16, 817–826.
- Bandyopadhyay, A.; Mitra, I.; Shivaram, A.; Dasgupta, N.; Bose, S. Direct comparison of additively manufactured porous titanium and tantalum implants towards in vivo osseointegration. Addit. Manuf. 2019, 28, 259–266.
- Wang, F.; Chen, H.; Yang, P.; Muheremu, A.; He, P.; Fan, H.; Yang, L. Three-dimensional printed porous tantalum prosthesis for treating inflammation after total knee arthroplasty in one-stage surgery—A case report. J. Int. Med. Res. 2020, 48, 300060519891280.
- Engesaeter, I.O.; Lie, S.A.; Lehmann, T.G.; Furnes, O.; Vollset, S.E.; Engesaeter, L.B. Neonatal hip instability and risk of total hip replacement in young adulthood: Follow-up of 2,218,596 newborns from the Medical Birth Registry of Norway in the Norwegian Arthroplasty Register. Acta Orthop. 2008, 79, 321–326.
- Cheng, L.; Zhao, D.; Yang, L.; Li, J.; Ma, Z.; Wang, Z.; Tian, F.; Tian, S. The application of 3D printed customized porous tantalum acetabular patch for adult DDH hip reconstruction. Chin. J. Orthop 2018, 38, 650–657.
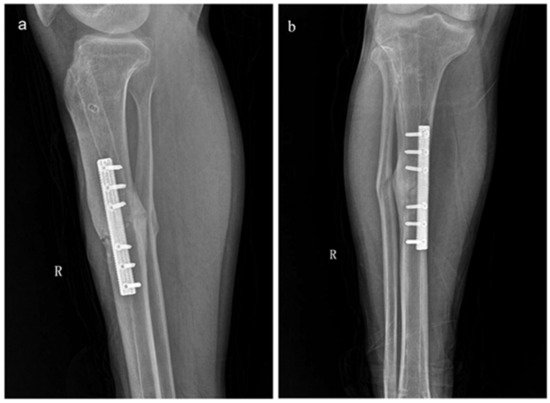
- Zhao, D.W.; Ma, Z.J.; Wang, T.N.; Liu, B.Y. Biocompatible Porous Tantalum Metal Plates in the Treatment of Tibial Fracture. Orthop. Surg. 2019, 11, 325–329.
- Zhou, R.; Ni, H.-J.; Peng, J.-H.; Liu, N.; Chen, S.; Shao, J.-H.; Fu, Q.-W.; Liu, J.-J.; Chen, F.; Qian, Q.-R. The mineralization, drug release and in vivo bone defect repair properties of calcium phosphates/PLA modified tantalum scaffolds. RSC Adv. 2020, 10, 7708–7717.
- Liao, H.; Miao, X.; Ye, J.; Wu, T.; Deng, Z.; Li, C.; Jia, J.; Cheng, X.; Wang, X. Falling Leaves Inspired ZnO Nanorods-Nanoslices Hierarchical Structure for Implant Surface Modification with Two Stage Releasing Features. ACS Appl. Mater. Interfaces 2017, 9, 13009–13015.
- Zhou, R.; Xu, W.; Chen, F.; Qi, C.; Lu, B.Q.; Zhang, H.; Wu, J.; Qian, Q.R.; Zhu, Y.J. Amorphous calcium phosphate nanospheres/polylactide composite coated tantalum scaffold: Facile preparation, fast biomineralization and subchondral bone defect repair application. Colloids Surf. B Biointerfaces 2014, 123, 236–245.
- Wang, Q.; Zhang, H.; Gan, H.; Wang, H.; Li, Q.; Wang, Z. Application of combined porous tantalum scaffolds loaded with bone morphogenetic protein 7 to repair of osteochondral defect in rabbits. Int. Orthop. 2018, 42, 1437–1448.
- Wang, N.; Li, H.; Wang, J.; Chen, S.; Ma, Y.; Zhang, Z. Study on the anticorrosion, biocompatibility, and osteoinductivity of tantalum decorated with tantalum oxide nanotube array films. ACS Appl. Mater. Interfaces 2012, 4, 4516–4523.
- Uslu, E.; Oztatli, H.; Garipcan, B.; Ercan, B. Fabrication and cellular interactions of nanoporous tantalum oxide. J. Biomed. Mater. Res. B Appl. Biomater. 2020, 108, 2743–2753.
- Gao, H.; Jie, Y.F.; Wang, Z.Q.; Wan, H.; Gong, L.; Lu, R.C.; Xue, Y.K.; Li, D.; Wang, H.Y.; Hao, L.N.; et al. Bioactive tantalum metal prepared by micro-arc oxidation and NaOH treatment. J. Mater. Chem. B 2014, 2, 1216–1224.
- Rodriguez-Contreras, A.; Guillem-Marti, J.; Lopez, O.; Manero, J.M.; Ruperez, E. Antimicrobial PHAs coatings for solid and porous tantalum implants. Colloids Surf. B Biointerfaces 2019, 182, 110317.
- Garbuz, D.S.; Hu, Y.; Kim, W.Y.; Duan, K.; Masri, B.A.; Oxland, T.R.; Burt, H.; Wang, R.; Duncan, C.P. Enhanced gap filling and osteoconduction associated with alendronate-calcium phosphate-coated porous tantalum. J. Bone Joint Surg. Am. 2008, 90, 1090–1100.
- Tanzer, M.; Karabasz, D.; Krygier, J.J.; Cohen, R.; Bobyn, J.D. The Otto Aufranc Award: Bone augmentation around and within porous implants by local bisphosphonate elution. Clin. Orthop. Relat. Res. 2005, 441, 30–39.
- Wen, H.B.; Dalmeijer, R.A.J.; Cui, F.Z.; Van Blitterswijk, C.A.; De Groot, K. Preparation of calcium phosphate coating on porous tantalum. J. Mater. Sci. Lett. 1998, 17, 925–930.
- Papanna, M.C.; Al-Hadithy, N.; Somanchi, B.V.; Sewell, M.D.; Robinson, P.M.; Khan, S.A.; Wilkes, R.A. The use of bone morphogenic protein-7 (OP-1) in the management of resistant non-unions in the upper and lower limb. Injury 2012, 43, 1135–1140.
- Dimitriou, R.; Dahabreh, Z.; Katsoulis, E.; Matthews, S.J.; Branfoot, T.; Giannoudis, P.V. Application of recombinant BMP-7 on persistent upper and lower limb non-unions. Injury 2005, 36 (Suppl. 4), S51–S59.
- Van Houwelingen, A.P.; McKee, M.D. Treatment of osteopenic humeral shaft nonunion with compression plating, humeral cortical allograft struts, and bone grafting. J. Orthop. Trauma 2005, 19, 36–42.
- Vinall, R.L.; Lo, S.H.; Reddi, A.H. Regulation of articular chondrocyte phenotype by bone morphogenetic protein 7, interleukin 1, and cellular context is dependent on the cytoskeleton. Exp. Cell Res. 2002, 272, 32–44.
- Zhao, Q.-M.; Li, G.-Z.; Yang, H.-L.; Gu, X.-F. Surface modification of biomedical tantalum by micro-arc oxidation. Mater. Technol. 2016, 32, 90–95.
- Bai, Y.; Park, I.S.; Lee, S.J.; Bae, T.S.; Duncan, W.; Swain, M.; Lee, M.H. One-step approach for hydroxyapatite-incorporated TiO2 coating on titanium via a combined technique of micro-arc oxidation and electrophoretic deposition. Appl. Surf. Sci. 2011, 257, 7010–7018.
- Hickok, N.J.; Shapiro, I.M.; Chen, A.F. The Impact of Incorporating Antimicrobials into Implant Surfaces. J. Dent. Res. 2018, 97, 14–22.
- Zeng, Q.; Zhu, Y.; Yu, B.; Sun, Y.; Ding, X.; Xu, C.; Wu, Y.W.; Tang, Z.; Xu, F.J. Antimicrobial and Antifouling Polymeric Agents for Surface Functionalization of Medical Implants. Biomacromolecules 2018, 19, 2805–2811.
- Raza, Z.A.; Abid, S.; Banat, I.M. Polyhydroxyalkanoates: Characteristics, production, recent developments and applications. Int. Biodeterior. Biodegrad. 2018, 126, 45–56.
- Rodríguez-Contreras, A.; García, Y.; Manero, J.M.; Rupérez, E. Antibacterial PHAs coating for titanium implants. Eur. Polym. J. 2017, 90, 66–78.
