Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 2 by Dean Liu and Version 1 by Francesco Ardito.
KRAS mutational heterogeneity between primary colorectal cancer and liver metastases may present a challenge in assessing prognostic information prior to the multimodal treatment.
- KRAS mutation status
- KRAS discordance
- precision medicine
- colorectal tumor
- colorectal liver metastases
- overall survival
1. Introduction
Hepatic resection is currently the only treatment option that can offer, together with perioperative chemotherapy, a chance of long-term survival in patients with colorectal liver metastases (CRLM), resulting in 5-year survival rates of 40% [1,2][1][2], and exceeding 50% in selected patients [3,4,5][3][4][5]. Several clinico-pathologic factors have been used to assess prognosis following liver resection for CRLM [4,6][4][6]. However, it has been showed that these factors, often do not represent the biological heterogeneity of CRLM and are not adequate for predicting long-term outcome.
In the current era of targeted therapies and personalized medicine, molecular biomarkers have been studied as fundamental prognostic predictors that guide the type of chemotherapy and define a better selection of patients for surgery [7]. The KRAS oncogene is currently the most used molecular biomarker in patients with CRLM [8]. This mutation occurs in about 38% of colorectal tumors and in 15–35% of patients with resectable CRLM, involving mainly codons 12 and 13 in more than 95% of cases [9]. A mutation in KRAS is associated with resistance to treatment with epidermal growth factor receptor (EGFR) antibodies, showing a lower response rate to therapy. For this reason, KRAS mutation is associated with lower overall and disease-free survival following liver resection and with higher risk of relapse in the lungs [10,11][10][11].
However, it has been showed that resistance to anti-EGFR therapy may also occur among patients without KRAS mutation in colorectal tumors (KRAS wild-type). The discordance of KRAS mutation status between the primary colorectal tumor and the corresponding CRLM may represent a possible explanation for the resistance to monoclonal antibodies targeted therapies.
Generally, metastases have similar mutations to those of the corresponding colorectal primary tumor. For these reasons, in the clinical practice, the KRAS mutation status information is generally obtained from primary colorectal tumor (surgical resection or biopsy). However, if KRAS mutation status of primary tumor may be representative of the corresponding CRLM mutational pattern, is debatable. Indeed, additional mutations may occur, determining the heterogeneity between primary tumor and CRLM. It has been hypothesized that primary tumor and the corresponding CRLM may show a discrepancy in mutational pattern. This event may represent the predominant cause of resistance to therapy. In this situation, it is clear that the only evaluation of KRAS mutation status in primary colorectal tumor may be inadequate to predict response to anti-EGFR therapy of the corresponding CRLM and may provide limited information prior to multimodal treatment. Several studies have been published focusing on the incidence of discordance between primary colorectal tumor and metastases, with controversial results. Some studies reported 100% of concordance [12,13][12][13]. On the other hand, several studies have reported different rates of discordance, ranging from 4 to 32% [14,15,16,17,18,19][14][15][16][17][18][19]. This heterogeneity of results may be due to the bias of inclusion criteria in several studies, where the KRAS mutation status of primary tumor is compared with a wide variety of metastatic sites.
2. Incidence of KRAS Mutation
Among the 107 patients, 33 (30.8%) had a KRAS mutation in the primary tumor and 36 (33.6%) had a KRAS mutation in the corresponding CRLM (Table 21). The most frequent mutations were found in codon 12. Type of KRAS mutations were not significantly different between primary tumors and CRLM (Table 21).Table 21.
Distribution of KRAS mutation type in primary colorectal tumor and in CRLM.
The incidence of KRAS discordance varied according to the time interval between the KRAS mutation analysis of the primary colorectal tumor and the mutation analysis of the corresponding CRLM (Figure 2). This incidence showed a statistical significant negative linear correlation with the time interval between the two analyses (p = 0.025; Pearson correlation = 0.2).
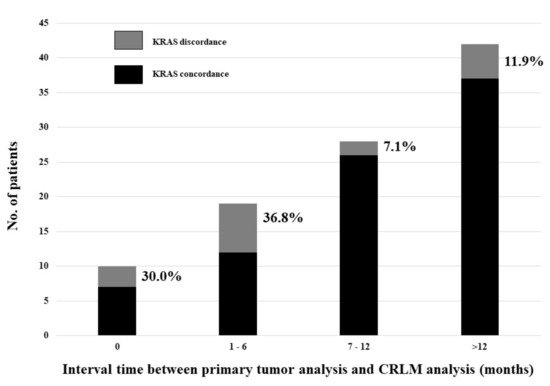

Figure 2. Incidence of KRAS discordance according to the time interval between the KRAS mutation analysis of the primary colorectal tumor and the mutation analysis of the corresponding CRLM.
5. Predictors of KRAS Discordance
A multivariable logistic regression analysis for the risk of KRAS discordance associated with the worst prognosis (wild-type in primary tumor, mutation in CRLM) was performed. The number of CRLM >3 was an independent risk factor for this type of KRAS discordance (OR = 4.600; 95%CI = 1.020–20.734; p = 0.047). Preoperative Bevacizumab administration showed a protective effect (OR = 0.072; 95%CI = 0.007–0.716; p = 0.025) (Table 42).
Table 42. Univariable and multivariable logistic regression analysis for the risk of KRAS discordance associated with the worst prognosis (wild-type in primary tumor, mutation in CRLM).
| Variable | Univariable Analysis p |
Multivariable Analysis OR (95% CI) |
p |
|---|---|---|---|
| Age (years) ≥70 | 0.018 | ||
| Male sex | 0.391 | ||
| Primary tumor location | |||
| Right-sided | 0.744 | ||
| Left-sided | 0.780 | ||
| Rectum | 0.469 | ||
| Positive lymph nodes in primary tumor | 0.775 | ||
| Synchronous CRLM | 0.798 | ||
| CRLM size ≥5 cm | 0.998 | ||
| >3 CRLM | 0.155 | 4.600 (1.020–20.734) | 0.047 |
| Bilobar CRLM | 0.206 | ||
| Initially unresectable CRLM | 0.746 | ||
| Administration of preoperative chemotherapy | 0.268 | ||
| Oxaliplatin-based chemotherapy | 0.655 | ||
| Irinotecan-based chemotherapy | 0.167 | ||
| Association of targeting agents | 0.033 | ||
| Association of Bevacizumab | 0.167 | 0.072 (0.007–0.716) | 0.025 |
| Association of Cetuximab | 0.291 | ||
| >6 cycles of chemotherapy | 0.280 | ||
| Progression after chemotherapy | 0.133 | ||
| Major hepatectomy | 0.598 |
| KRAS Mutation Analysis | Primary Tumor, No. (%) | CRLM, No. (%) | p |
|---|---|---|---|
| KRAS mutation | 33 (30.8) | 36 (33.6) | |
| Codon 12 | 16/33 (48.5) | 21/36 (58.3%) | 0.412 |
| p.(Gly12Val) | 6 | 7 | |
| p.(Gly12Asp) | 4 | 7 | |
| p.(Gly12Ser) | 2 | 2 | |
| p.(Gly12Cys) | 2 | 2 | |
| p.(Gly12Ala) | 2 | 2 | |
| p.(G12R/S/C) | - | 1 | |
| Codon 13 | 7/33 (21.2) | 7/36 (19.4) | 0.855 |
| p.(Gly13Asp) | 7 | 7 | |
| p.(Ala146Thr) | 6/33 (18.1) | 5/36 (13.9) | 0.626 |
| p.(Gln22Lys) | 2/33 (6.1) | 2/36 (5.6) | 0.928 |
| p.(Ala59Xaa) | - | 1/36 (2.8) | |
| p.(Gln61Xaa) | 2/33 (6.1) |
3. Incidence of KRAS Discordance
Sixty-four patients (59.8%) were KRAS wild-type in both primary tumor and CRLM and 26 (24.3%) were KRAS mutated in both primary tumor and CRLM (Figure 1). Discordance was documented in 17 patients (15.9%): in 10 patients (9.4%) KRAS was wild-type in primary tumor and mutated in CRLM and in 7 patients (6.5%) KRAS was mutated in primary tumor and wild-type in CRLM (Figure 1).
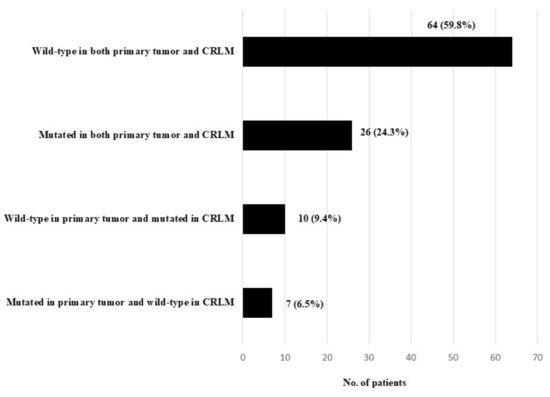

Figure 1.
KRAS mutation status in primary colorectal tumor and corresponding CRLM.
4. KRAS Discordance and Preoperative Chemotherapy
Twenty-seven patients (25.2%) did not undergo preoperative chemotherapy. Among these patients, the incidence of discordance was 18.5% (5 patients), not significantly different than that observed in patients who underwent preoperative chemotherapy (15.0%, 12/80 patients; p = 0.665).
Among the 74 patients with wild-type colorectal primary tumor, 33.9% (25 patients) underwent preoperative administration of anti-EGFR agents: 2 patients before simultaneous colorectal and liver resection and the other 23 patients prior to liver surgery. Among these 74 patients, the incidence of discordance was 13.5% (10 patients), not significantly different than that observed among the 33 patients with KRAS-mutated colorectal tumor (21.2%, 7 patients; p = 0.314). Forty-nine patients with wild-type colorectal primary tumor did not undergo preoperative administration of anti-EGFR agents. In such patients the incidence of discordance was 18.4% (9/49).
References
- House, M.G.; Ito, H.; Gonen, M.; Fong, Y.; Allen, P.J.; DeMatteo, R.P.; Brennan, M.F.; Blumgart, L.H.; Jarnagin, W.R.; D’Angelica, M.I. Survival after hepatic resection for metastatic colorectal cancer: Trends in outcomes for 1600 patients during two decades at a single institution. J. Am. Coll. Surg. 2010, 210, 744–752.
- Giuliante, F.; Ardito, F.; Vellone, M.; Ranucci, G.; Federico, B.; Giovannini, I.; Nuzzo, G. Role of the surgeon as a variable in long-term survival after liver resection for colorectal metastases. J. Surg. Oncol. 2009, 100, 538–545.
- Simmonds, P.C.; Primrose, J.N.; Colquitt, J.L.; Garden, O.J.; Poston, G.J.; Rees, M. Surgical resection of hepatic metastases from colorectal cancer: A systematic review of published studies. Br. J. Cancer 2006, 94, 982–999.
- Rees, M.; Tekkis, P.P.; Welsh, F.K.; O’Rourke, T.; John, T.G. Evaluation of long-term survival after hepatic resection for metastatic colorectal cancer: A multifactorial model of 929 patients. Ann. Surg. 2008, 247, 125–135.
- de Haas, R.J.; Wicherts, D.A.; Andreani, P.; Pascal, G.; Saliba, F.; Ichai, P.; Adam, R.; Castaing, D.; Azoulay, D. Impact of expanding criteria for resectability of colorectal metastases on short- and long-term outcomes after hepatic resection. Ann. Surg. 2011, 253, 1069–1079.
- Fong, Y.; Fortner, J.; Sun, R.L.; Brennan, M.F.; Blumgart, L.H. Clinical score for predicting recurrence after hepatic resection for metastatic colorectal cancer: Analysis of 1001 consecutive cases. Ann. Surg. 1999, 230, 309–318.
- Yamashita, S.; Chun, Y.S.; Kopetz, S.E.; Vauthey, J.N. Biomarkers in colorectal liver metastases. Br. J. Surg. 2018, 105, 618–627.
- Brudvik, K.W.; Jones, R.P.; Giuliante, F.; Shindoh, J.; Passot, G.; Chung, M.H.; Song, J.; Li, L.; Dagenborg, V.J.; Fretland, Å.A.; et al. RAS Mutation Clinical Risk Score to Predict Survival After Resection of Colorectal Liver Metastases. Ann. Surg. 2019, 269, 120–126.
- Oliveira, C.; Westra, J.L.; Arango, D.; Ollikainen, M.; Domingo, E.; Ferreira, A.; Velho, S.; Niessen, R.; Lagerstedt, K.; Alhopuro, P.; et al. Distinct patterns of KRAS mutations in colorectal carcinomas according to germline mismatch repair defects and hMLH1 methylation status. Hum. Mol. Genet. 2004, 13, 2303–2311.
- Vauthey, J.N.; Zimmitti, G.; Kopetz, S.E.; Shindoh, J.; Chen, S.S.; Andreou, A.; Curley, S.A.; Aloia, T.A.; Maru, D.M. RAS mutation status predicts survival and patterns of recurrence in patients undergoing hepatectomy for colorectal liver metastases. Ann. Surg. 2013, 258, 619–627.
- Kemeny, N.E.; Chou, J.F.; Capanu, M.; Gewirtz, A.N.; Cercek, A.; Kingham, T.P.; Jarnagin, W.R.; Fong, Y.C.; DeMatteo, R.P.; Allen, P.J.; et al. KRAS mutation influences recurrence patterns in patients undergoing hepatic resection of colorectal metastases. Cancer 2014, 120, 3965–3971.
- Zauber, P.; Sabbath-Solitare, M.; Marotta, S.P.; Bishop, D.T. Molecular changes in the Ki-ras and APC genes in primary colorectal carcinoma and synchronous metastases compared with the findings in accompanying adenomas. Mol. Pathol. 2003, 56, 137–140.
- Etienne-Grimaldi, M.C.; Formento, J.L.; Francoual, M.; François, E.; Formento, P.; Renée, N.; Laurent-Puig, P.; Chazal, M.; Benchimol, D.; Delpero, J.R.; et al. K-Ras mutations and treatment outcome in colorectal cancer patients receiving exclusive fluoropyrimidine therapy. Clin. Cancer Res. 2008, 14, 4830–4835.
- Artale, S.; Sartore-Bianchi, A.; Veronese, S.M.; Gambi, V.; Sarnataro, C.S.; Gambacorta, M.; Lauricella, C.; Siena, S. Mutations of KRAS and BRAF in primary and matched metastatic sites of colorectal cancer. J. Clin. Oncol. 2008, 26, 4217–4219.
- Santini, D.; Loupakis, F.; Vincenzi, B.; Floriani, I.; Stasi, I.; Canestrari, E.; Rulli, E.; Maltese, P.E.; Andreoni, F.; Masi, G.; et al. High concordance of KRAS status between primary colorectal tumors and related metastatic sites: Implications for clinical practice. Oncologist 2008, 13, 1270–1275.
- Molinari, F.; Martin, V.; Saletti, P.; De Dosso, S.; Spitale, A.; Camponovo, A.; Bordoni, A.; Crippa, S.; Mazzucchelli, L.; Frattini, M. Differing deregulation of EGFR and downstream proteins in primary colorectal cancer and related metastatic sites may be clinically relevant. Br. J. Cancer 2009, 100, 1087–1094.
- Knijn, N.; Mekenkamp, L.J.; Klomp, M.; Vink-Börger, M.E.; Tol, J.; Teerenstra, S.; Meijer, J.W.; Tebar, M.; Riemersma, S.; van Krieken, J.H.; et al. KRAS mutation analysis: A comparison between primary tumours and matched liver metastases in 305 colorectal cancer patients. Br. J. Cancer 2011, 104, 1020–1026.
- Li, Z.Z.; Bai, L.; Wang, F.; Zhang, Z.C.; Wang, F.; Zeng, Z.L.; Zeng, J.B.; Zhang, D.S.; Wang, F.H.; Wang, Z.Q.; et al. Comparison of KRAS mutation status between primary tumor and metastasis in Chinese colorectal cancer patients. Med. Oncol. 2016, 33, 71.
- Bhullar, D.S.; Barriuso, J.; Mullamitha, S.; Saunders, M.P.; O’Dwyer, S.T.; Aziz, O. Biomarker concordance between primary colorectal cancer and its metastases. EBioMedicine 2019, 40, 363–374.
More
