Postprandial hyperglycemia (PPHG) is strongly linked with the future development of cardiovascular complications in type 2 diabetes (T2D). . Hence, reducing postprandial glycemic excursions is essential in T2D treatment to slow progressive deficiency of β-cell function and prevent cardiovascular complications. Most of the metabolic processes involved in PPHG, i.e., β-cell secretory function, GLP-1 secretion, insulin sensitivity, muscular glucose uptake, and hepatic glucose production, are controlled by the circadian clock and display daily oscillation. Consequently, postprandial glycemia displays diurnal variation with a higher glycemic response after meals with the same carbohydrate content, consumed at dusk compared to the morning. T2D and meal timing schedule not synchronized with the circadian clock (i.e., skipping breakfast) are associated with disrupted clock gene expression and are linked to PPHG. In contrast, greater intake in the morning (i.e., high energy breakfast) than in the evening has a resetting effect on clock gene oscillations and beneficial effects on weight loss, appetite, and reduction of PPHG, independently of total energy intake. Therefore, resetting clock gene expression through a diet intervention consisting of meal timing aligned to the circadian clock, i.e., shifting most calories and carbohydrates to the early hours of the day, is a promising therapeutic approach to improve PPHG in T2D. This review will focus on recent studies, showing how a high-energy breakfast diet (Bdiet) has resetting and synchronizing actions on circadian clock genes expression, improving glucose metabolism, postprandial glycemic excursions along weight loss in T2D.
- clock genes 2
- big breakfast 3
- PPHG 4
- T2D 5
- circadian rhythms
Role of High Energy Breakfast “Big Breakfast Diet” in Clock Gene Regulation of Postprandial Hyperglycemia and Weight Loss in Type 2 Diabetes
1. Introduction
1. Introduction
Postprandial hyperglycemia (PPHG) in type 2 diabetes (T2D) strongly contributes to glycated hemoglobin (HbA1c) values
Postprandial hyperglycemia (PPHG) in type 2 diabetes (T2D) strongly contributes to glycated hemoglobin (HbA1c) values
[1]
. It is linked to increased risk for the development of cardiovascular complications, even when glycemic control is restored
. Further, PPHG leads to a progressive decline of β-cell function and deficient and delayed early postprandial insulin response
. Hence, the reduction of glycemic peaks is an essential “target” in the treatment of T2D to mitigate the decline of β-cell secretion and prevent cardiovascular complications
.
The circadian clock temporally coordinates the metabolism over a 24-h period to anticipate daily recurring feeding-fasting cycles and optimize the metabolic efficiency at an appropriate time of the day, thereby preventing metabolic dysregulation
[8][9][10][11][12][13][14][15][16]
. Most of the hormonal and enzymatic functions controlling PPHG, i.e., secretion of insulin
, glucagon-like peptide-1 (GLP-1)
[20]
, GLUT-4 expression in skeletal muscle
, and hepatic glucose production
; are regulated by the circadian clock and display diurnal variations.
The circadian clock is controlled by light/dark signals and other external inputs such as meal timing or food availability
. The insulin sensitivity, β-cell responsiveness, GLUT-4 activity, and muscular glucose uptake are all enhanced in the early hours of the day compared to dusk or evening
[8][16][17][18][19][20][21][22][23][25][26][27][28][29][30][31][32][33]
. Therefore, the metabolism is optimized for food intake in breakfast, while the evening and nighttime are optimal for fasting and sleep
. Indeed, postprandial glycemia displays a clear circadian pattern, with the higher glycemic response after meals with the same carbohydrate content, consumed at dusk compared to the morning, both in healthy
, and T2D individuals
.
Meal timing exerts a critical influence on peripheral clocks involved in postprandial glycemia
.
Circadian misalignment, with day/night cycle, often imposed in modern society, like shift workers, skipping breakfast, snacking all day, including in the evening hours, are associated with disrupted clock gene expression and linked with aberrant metabolic responses, weight gain, PPHG, increased risk for T2D
[12][27][34][37][42][45][46][47][48][49][50][51][52]
, and cardiovascular complications
[53]
. Breakfast skipping is also linked to a significant increase in HbA1c even without overeating in the evening
[47]
.
Asynchrony of the circadian clock is central in the pathophysiology of T2D
. Lower transcripts of clock gene expression in T2D are linked to insulin resistance, delayed β-cell secretion and reduced β-cell proliferation
, PPHG, and increased HbA1c
. Moreover, breakfast’s omission in T2D patients causes further disruption in clock gene expression, and it is linked to PPHG and delayed and deficient early insulin and GLP-1 responses after subsequent meals
. In contrast, meal-timing pattern, aligned with the circadian clock, consuming high in energy breakfast, exerts a powerful effect on the clock network temporal synchronization, thereby improving the postprandial glycemic responses across the day in healthy and T2D patients
.
Therefore, breakfast consumption might be critical in T2D for the achievement of metabolic homeostasis and improvement of PPHG
[14][15][23][24][25][26][27][28][29][30][31][32][33][34][35][36][37][38][39][40][43][44][45][46][47][48][49][50][51][52][53][54][55][56]
. Hence, resetting clock gene expression through a diet intervention consisting of meal timing aligned to the circadian clock is a promising therapeutic intervention approach to improve PPHG
.
2. Circadian Clock Regulation of Metabolism
2.1. Central and Peripheral Clocks
The diurnal variation of the hormonal and enzymatic functions related to glucose metabolism and postprandial glycemia is synchronized by the circadian clock
.
The central or “Master Clock” is found in the hypothalamus in the suprachiasmatic nucleus (SCN). The clock genes located in SCN are synchronized by light signals and generate the body endogenous ~24-h rhythm. Other clock genes are disseminated in almost all the peripheral tissues, i.e., muscle, liver, intestinal L-cells, and in β-cells
. The clock genes located in peripheral tissues are coordinated by signals coming from the SCN
. However, they are mostly entrained to the time of food intake and food availability
. It allows the peripheral clocks to anticipate the secretion of metabolic hormones and enzymes just before the food consumption at a specific hour of the day
, leading to synchronized, hormonal, digestive, absorptive, and metabolic functions
. The synchronization of peripheral clock genes to the time of food intake is achieved in a tissue-specific fashion through multiple postprandial signals, i.e., absorbed nutrients, post-meal glucose excursions, β-cell, and GLP-1 secretion
.
Noteworthy is that the first meal of the day, i.e., breakfast, exerts a more powerful resetting effect on the clock network than other meals, underscoring the damage caused by the absence or delayed breakfast on the clock regulation of glucose metabolism and PPHG
(
).
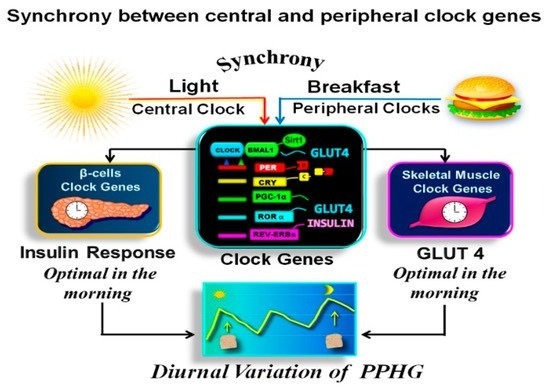
Synchronization between central and peripheral clock genes. In the above illustration, we observe that the breakfast in synchrony with downlight “turns on” the clock, activating the
complex. It drives the transcription of
,
,
,
genes,
coactivator 1α (
),
, and other transcriptional elements promoting downstream expression of several proteins encoded by tissue-specific “clock-controlled genes,” relaying the clock information to cellular processes. As a result, β-cells insulin response and muscular GLUT-4 activity are optimal in the early hours of day. Hence the glucose response after isocaloric meals is significantly higher in the evening than in the morning.
2.2. Molecular Mechanism of the Circadian Clock-Driven Metabolism
The molecular clock mechanism is identical in central and peripheral clocks. It consists of self-sustained transcriptional-translational feedback loops
. The transcriptional activators
(circadian locomotor output cycles protein kaput) and
(brain and muscle ARNT-like 1) act as positive elements in the feedback loop.
heterodimer drive the transcription of periods (
) and cryptochromes (
) genes. The resulting PER and CRY proteins dimerize in the cytoplasm. After ~24 h, they are translocated back into the nucleus to interact with the
complex, directly suppressing their own transcription, thus generating a cycle that recurs every ~24 h
. In a secondary regulatory loop,
mediates the transcription of the repressor nuclear receptor
and one promoter gene, the retinoic acid receptor-related orphan receptor (
), maintaining further the circadian (24 h) oscillation of the clock
.
The
heterodimer also mediates the transcription of tissue-specific output genes,
,
,
, and
clock genes, along with PPARγ coactivator 1α (
), and
and other transcriptional elements, which promote downstream expression of several tissue-specific proteins, hormones, and enzymes, relaying the clock information to cellular processes, like β-cell secretion, GLUT-4 activity, hepatic glycogenolysis, and gluconeogenesis
.
2.3. Circadian Clock Regulation of Glucose Metabolism and Postprandial Glycemia
complex plays a critical role in the transcription of tissue-specific elements, which regulate the circadian processes involved in glucose homeostasis and postprandial glycemia
.
,
, and
positively regulate the circadian β-cells secretion
, insulin sensitivity
, muscular GLUT-4 activity, and glucose uptake
, and β-cell replicative capacity and survival
.
and
integrity are also necessary for circadian GLP-1 secretion in the intestinal L-cells
. In the liver, the expression of
and
and
, modulates the gluconeogenic enzymes, and the rhythms of the hepatic glycolysis pathway
.
,
,
, and
, through posttranslational regulation of cAMP signaling, reduces the glucagon-stimulated hepatic glucose production
, and coordinate the nocturnal oscillation of hepatic glucose output; glycogenolysis in the first part of night and gluconeogenesis in the second part, before waking up
.
Adenosine monophosphate-activated protein kinase (
), plays a crucial role in the clock regulation of glucose metabolism
. Upregulation of
expression significantly enhances GLUT-4 translocation and muscular glucose uptake, ensuring metabolic efficiency and improving postprandial glucose and insulin responses
.
exerts a positive effect on
, associated with beneficial effects on insulin sensitivity and β-cells viability
.
2.4. Disrupted Clock Genes Expression in Type 2 Diabetes
Asynchrony of clock gene expression is essential in the pathophysiology of obesity, metabolic syndrome, and T2D
. It is also associated with circadian misalignment of meal timing or sleeping hours like in shift-workers
. Disrupted clock gene expression is associated with reduced and delayed β-cell response, insulin resistance, and a low rate of β-cell replication
. Deficient
and
expression in T2D is associated with PPHG and higher HbA1c levels
.
2.5. Synchronization between Central and Peripheral Clocks
For the functionality of the circadian clock, the individual clocks must be synchronized one to another and with the external environment
. This coordination is achieved when the feeding/fasting cycle is aligned with the day/night cycle
. Therefore, both stimuli, “light” and “food”, should occur simultaneously “in synchrony” (
). As breakfast consumption has a powerful resetting effect on the clock network, the temporal synchronization between breakfast and downlight is critical for achieving metabolic homeostasis
.
In
is shown how the breakfast in synchrony with the downlight “turns on” the clock gene machinery in the early morning. This further regulates the clock-controlled output genes relaying the clock information downstream to the tissue-specific proteins and the rhythms of cellular processes
.
2.6. Asynchrony between Central and Peripheral Clocks
Eating and sleeping out of synchrony, delaying the first meal or increasing the frequency of the meals, with calories and CH uniformly spread across the day, including evening hours assigned to rest, promote the uncoupling or desynchronization between the peripheral and the central clock genes, and disrupted regulation of metabolic processes. This misalignment may result in altered thermogenesis, weight gain, increased lipids, insulin resistance, fatty liver, and worsening of postprandial glycemia, as it was shown in preclinical studies
, clinical studies in non-diabetic
and in T2D individuals
(
).
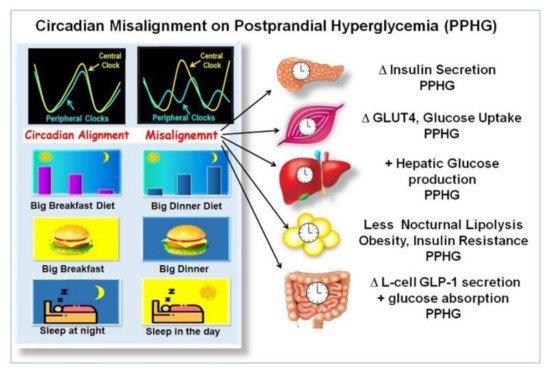
Effects of misalignment on postprandial hyperglycemia (PPHG). The illustration, shows, how eating and sleeping at hours not aligned with the circadian clock, i.e., small breakfast, big dinner, sleeping during the day, etc., produces a misalignment between central and peripheral clocks and disrupted clock gene expression. It is associated with deficient β-cell secretion, GLUT-4 activity, muscular glucose uptake, increased hepatic glucose output, adipogenesis, reduced lipolysis, insulin resistance, alteration of GLP-1 secretion, and increased intestinal glucose absorption. All of which may result in worsening of PPHG “Adapted with permission”
.
3. Effect of High Energy Breakfast “Big Breakfast Diet” on Resetting Clock Gene Expression and Reduction of PPHG in T2D
The circadian clock regulation of PPHG is influenced by the meal timing schedule
. Breakfast skipping and over-eating in the evening led to asynchrony of the circadian clock and is linked to weight gain, PPHG, and diabetes
.
Several recent reports suggest that eating in synchrony with the circadian clock by shifting more energy and CH to the morning hours (i.e., high energy and CH breakfast), and reducing energy and CH consumption in the evening hours, facilitate weight loss, improve postprandial glycemia, and reduce appetite and craving in metabolic syndrome and in T2D, compared to the inverse pattern, i.e., “high in energy and CH dinner” and reduced breakfast
[15][27][30][35][39][40][61][73][74][75][76][77]
. Clinical and epidemiological studies have shown that late meals are linked to obesity and T2D
. A diet intervention not aligned with the circadian clock by shifting calories and CH to later hours of the day is associated with less weight loss and higher postprandial and overall glycemia among obese
and in T2D individuals
.
3.1. Effect Skipping Versus Eating Breakfast, on Clock Gene Expression and PPHG
In two crossover studies in T2D patients, we explored whether skipping breakfast in a single day (NoB) versus another day consuming high energy and CH breakfast (YesB) influences the clock gene expression and the PPHG after subsequent isocaloric meals.
. Breakfast skipping (NoB) acutely disrupts clock gene expression after lunch
. The absence of breakfast (NoB) down-regulated the mRNA expression of
and
,
and
expression, and this clock gene disruption in NoB day was associated with significantly higher postprandial glycemic response and deficient and delayed insulin, and intact GLP-1 postprandial secretion after lunch
(
). In contrast, high energy and CH breakfast consumption in YesB day led to an overall increased expression of these key metabolic clock genes, i.e.,
,
,
, and
. This resetting effect on clock genes mRNA expression in YesB day was associated with a significant reduction of postprandial glycemic response and enhanced and faster insulin and GLP-1 response after subsequent lunch. It suggests that the upregulation of these pivotal clock genes in the YesB day led to the improvement of PPHG
(
).
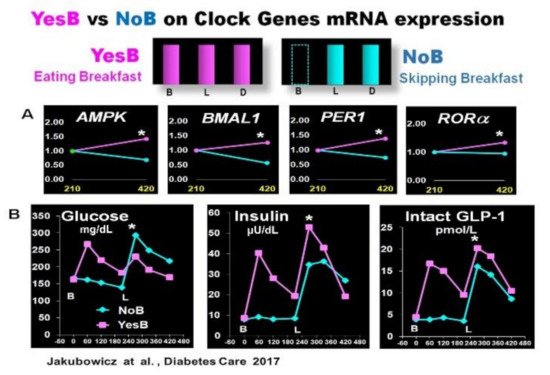
Effect of YesB vs. NoB on clock gene expression and postprandial glucose, insulin, intact GLP-1 response after breakfast and lunch in T2D. (
): Clock gene expression: Blood samples were collected 3.5 h after breakfast (YesB) or no breakfast (NoB) (time point 210 min) and 3.5 h after lunch (time point 420 min) Asterisks denote statistical differences (
< 0.05) between time point 210 min and time point 420 min. Data are means ± SE. (
): Line charts of glucose, insulin, and intact GLP-1 postprandial responses on YesB and NoB days: Breakfast (B) was given to the YesB group at time point 0. Lunch (L) was given to both groups at time point 210 min. Asterisks denote statistical differences between YesB and NoB at a specific time point. Data are means ± SE. “Reproduced and adapted with permission”
.
This research showed that just a single day of breakfast omission adversely influenced the clock gene expression and significantly increased glycemic response after lunch. It suggests a high relevance of breakfast consumption on the clock gene regulation of postprandial glycemia
.
In another crossover study in T2D patients, we reported that the omission of breakfast (NoB) versus breakfast consumption (YesB), was associated with significantly higher glycemic response, after lunch and also after subsequent dinner. Moreover, compared to the day when breakfast was consumed, the omission of breakfast led to reduced and delayed insulin, C-peptide, and iGLP-1 responses after lunch and dinner
(
).
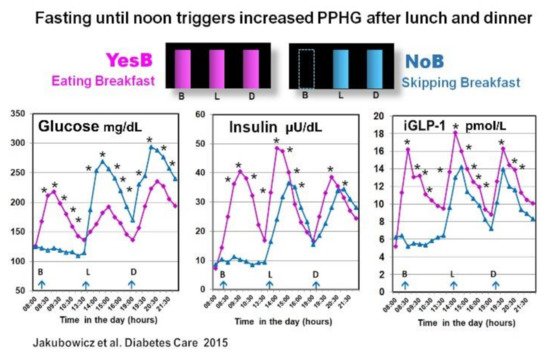
The reduction of postprandial glycemia and higher and faster insulin response after lunch with prior breakfast consumption in YesB day was previously reported in healthy and in T2D individuals
and was described as the second meal phenomenon
. It has been reported that previous breakfast consumption may enhance β-cell memory and β-cell responsiveness at the second meal (lunch)
.
However, in our study, this effect of breakfast was extended to dinner
. Indeed, the absence of breakfast led to higher postprandial glucose and decreased GLP-1 and insulin response after lunch and also after dinner
. It has been suggested that fasting until noon on NoB day may reduce the β-cell memory and β-cell responsiveness in an extended fashion, resulting in less and delayed postprandial insulin response after both lunch and dinner
.
The explanation is based on a recent report showing that nutrient depletion or starvation induces lysosomal degradation of nascent insulin secretory granules and to less β-cell secretory granule biogenesis
. It leads to deficient and delayed postprandial insulin response extended to lunch and dinner
. The increased postprandial GLP-1 on the YesB day is also associated with enhanced β-cell memory
. Further, it may explain the reduction of glycemic excursions after lunch and dinner on the day when breakfast was consumed
(
).
Moreover, the breakfast consumption triggers correct oscillatory clock gene expression, namely
, RORα, PER1, and
, and the clock gene regulation of glucose metabolism, thereby improving the postprandial GLP-1, insulin, and glucose responses after subsequent meals
.
The upregulation of
, in YesB day, significantly enhances GLUT-4 translocation, muscular glucose uptake, and postprandial insulin response further reducing post-meal glycemic excursions
.
is positively linked to
, and its beneficial effects on insulin sensitivity, β-cell proliferation, and viability
.
Breakfast consumption is essential when targeting glycemic control in T2D. The upregulation of the clock genes induced by breakfast consumption positively influences cardiovascular activity, heart rate, blood pressure, adipose tissue, and other metabolic organs
. Therefore, breakfast consumption may improve overall metabolism and reduce cardiometabolic complications of T2D.
3.2. High Energy Breakfast Diet “Breakfast Diet” (Bdiet) Reduces overall Postprandial Glycemia and Body Weight in Metabolic Syndrome
Studies in rodents and humans suggest that not only the amount but also the hour of food intake, especially the time of energy, protein, and CH intake, play an essential role in the circadian clock regulation of energy, and glucose homeostasis, thereby influencing the glycemic postprandial excursions
. Several reports showed that ingested calories are more efficiently used in the morning than at dusk
, and this is evidenced by less hyperglycemic excursions throughout the day and better weight loss, when most of the energy and CH are assigned to the early hours of the day, compared to iso-energetic calorie and CH intake mainly in the evening
.
We examined in participants with metabolic syndrome whether a diet with overall similar daily caloric intake but with different meal timing and distribution: either consuming a high-energy and CH breakfast (Bdiet) or high-energy and CH dinner (Ddiet) has a distinct influence on the glycemic postprandial response after breakfast, lunch, and dinner. We also explored the influence of Bdiet vs. Ddiet on overall glycemia, weight loss, and appetite scores. The energy distribution of Bdiet was: large breakfast (700 kcal, 50%), medium-sized lunch (600 kcal, 36%), and small dinner (200 kcal, 14%). In Ddiet, the plan was reversed; small breakfast and large dinner
(
).
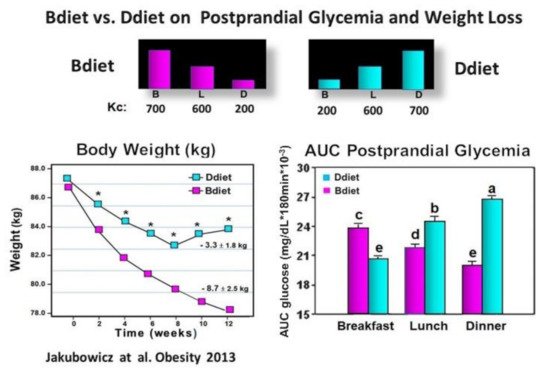
Effect of Bdiet versus Ddiet on postprandial glycemia and weight loss in metabolic syndrome. In the upper part is shown the caloric content for Breakfast (B), Lunch (L), and Dinner (D) in Bdiet and Ddiet. The line chart shows the changes in body weight, recorded every two weeks during the 12 weeks of the study in Bdiet and Diet groups. The bar graph illustrates AUC for postprandial glycemia calculated at 0–180 min after breakfast, lunch, and dinner meal challenges, of the assigned diet. The meal tests were performed on a single day during the second week of the study. Values are means ± SE; Bdiet–breakfast diet group; Ddiet—dinner diet group; Asterisks denotes
< 0.05; Bars with different letters, denote significant difference
< 0.05.“Reproduced and adapted with permission”
.
Over 12 wk. of the study, the body weight decreased significantly in both groups. However, the Bdiet group showed a 2.5-fold more significant weight loss (
). After the high-calorie dinner meal test in the Ddiet group, the postprandial glucose response was significantly higher compared to the postprandial glucose response to the isocaloric high-calorie breakfast meal in the Bdiet group (
). The overall postprandial response to breakfast, lunch, and dinner challenge meals, expressed as overall AUC for postprandial glycemia was significantly lower in the Bdiet group than the Ddiet group
. These results are in line with several recent reports suggesting metabolic disadvantages of high energy and CH consumption in evening hours, while high energy and CH meals consumed in the early hours of the day, may reduce the insulin resistance and glucose post-meal response in obese and prediabetics
.
A high-energy and CH breakfast (Bdiet) is more beneficial than a high-energy and CH dinner to reduce overall postprandial glycemia. Avoiding high energy and CH intake at dusk and in the evening may be advantageous, particularly for lowering postprandial glycemic excursions, and may reduce the risk of T2D and cardiovascular diseases.
3.3. High Energy Breakfast Diet “Breakfast Diet” (Bdiet) Versus High Energy Dinner Diet (Ddiet) Reduces overall PPHG in Type 2 Diabetes
Based on the previous study
