Bone sarcomas are rare cancers which often present with metastatic disease and are still associated with poor survival rates. Studies in the last decade have identified that exosomes, a type of extracellular vesicle released by cells, play an important role in tumour progression and dissemination. Through the transfer of their cargo (RNAs, proteins, and lipids) across cells, they are involved in cellular cross-talk and can induce changes in cellular behaviour. Exosomes have been shown to be important in metastasis organotropism, induction of angiogenesis and vascular permeability, the education of cells towards a pro-metastatic phenotype or the interaction between stromal and tumour cells. Due to the importance exosomes have in disease progression and the high incidence of metastasis in bone sarcomas, recent studies have evaluated the implications of these extracellular vesicles in bone sarcomas.
- bone sarcoma
- exosomes
- metastasis
- osteosarcoma
- Ewing sarcoma
- chondrosarcoma
1. Exosomes
1.1. Exosome Biogenesis, Release, and Uptake
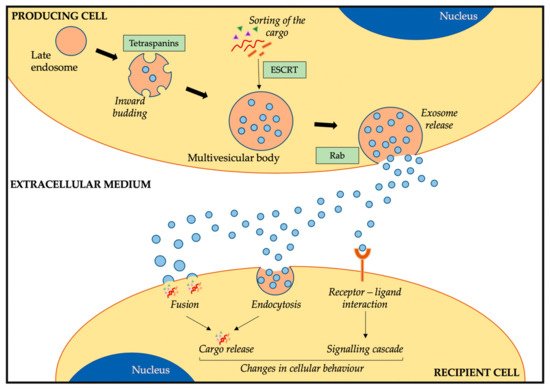
1.2. Exosome Cargo and Composition
1.3. Function of Exosomes
2. The Role of Exosomes in Bone Sarcoma Metastasis
Bone sarcoma metastasis represents the most adverse clinical factor and is associated with poor survival rates [1][35]. This is partly due to the lack of understanding on the molecular mechanisms behind tumour dissemination and metastatic disease [1,74,75][35][36][37]. In both OS and ES, the most common metastatic site is the lung, followed by bone and bone marrow. Interestingly, lung metastasis is associated with better prognosis than non-lung metastasis [1,76][35][38]. Due to the importance of metastasis in these bone sarcomas, many studies have focused on deciphering the pathways behind disease progression. Amongst the different genes identified in OS, we find overexpression of CD155 [77][39], loss of TP53, RB1, and PTEN [78][40], and upregulation of Notch genes [79][41] to be important in metastatic OS. Moreover, different novel treatment strategies are evaluating their efficacy in OS metastatic patients through clinical trials [75][37], with the aim of finding novel treatment strategies for these patients. In ES, similar studies have resulted in several genes linked to metastasis, such as ROR1 [80][42], MSH2, MSH6, RPA2, and RFC2 genes from the mismatch repair pathway [81][43], PPP1R1A [82][44] and TWIST1 proteins [83][45], the Cad11 adhesion molecule [84][46], or ERBB4 via activation of the PI3K-Akt-FAK cascade [85][47]. Moreover, similar to other cancers, hypoxia has been associated with induction of metastasis in OS and ES via regulation of HIF1α through HIF1α [86][48] or overexpression of CXCR4 in ES [87][49], amongst others. Similar to OS, different clinical trials are evaluating the response of metastatic ES patients to different treatment approaches in order to improve survival rates [76][38]. In contrast to OS and ES, CS is usually a non-metastatic disease with locally aggressive tumours [88][50]. However, some evidence suggests integrins are involved in the metastatic organotropism of CS to the lungs [89,90][51][52].2.1. Exosomes in OS
| Origin Cell | Recipient Cell | Cargo | Change | Ref. | |||||
|---|---|---|---|---|---|---|---|---|---|
| OS cells and conditioned media | - | Profiling of proteome and secretome | Exosome proteins involved in migration, adhesion, and angiogenesis | [98][60] | |||||
| Metastatic and non-metastatic OS cell lines | - | Profiling of miRNAs and target genes | miRNA of metastatic OS exosomes target metastasis-associated genes, cell adhesion, and apoptosis | [99][61] | |||||
| Metastatic and non-metastatic OS cell lines | Osteoblasts | ES cell linesmiR-675 (miRNA profiling) |
Osteoblasts, osteoclasts in 3D scaffoldMetastasis-associated exosomes induce migration and invasion in osteoblasts via miR-675/CALN1 axis | [100][62] | |||||
| EZH2 mRNA | (target of interest) | Transfer of EZH2 mRNA to MSC (increase expression), osteoblasts (no change), and osteoclasts (reduction expression) | [118][82] | Metastatic OS cell lines | Macrophages, osteoclasts, endothelial cells | miR-148a, miR-21-5p (RNA profiling) |
Induction of osteoclast-like gene expression (macrophage), increase in bone resorption (osteoclasts) and angiogenesis (endothelial cells) via miRNA transfer | [101][63] | |
| ESCD99neg cell line model | ES cell lines (normal CD99) | Increased miR-34a | Regulation of NFκB via miR-34a through reduction of Notch. Increase in neural differentiation (similar to direct CD99 silencing) | [119][83] | Metastatic and non-metastatic OS cell lines | ||||
| ESCD99neg cell line model | MSC | TGFβ (induction of IL6) |
Internalization of TGFβ induces IL6 production, cell growth, and lung metastasis in vivo | [102][64] | |||||
| Doxorubicin-resistant OS cell lines | Sensitive OS cell lines | MRP1, Pgp(multidrug resistant proteins) | Increase in doxorubicin resistance in recipient cells; increase in MRP1 and Pgp mRNA levels. | [103][65] | |||||
| Bone marrow (conditioned media) | Metastatic and non-metastatic OS cells | uPA (secreted, paracrine loop) | Increase in migration on recipient cells, induction of OS metastasis in vivo | [104][66] | |||||
| CAF | OS cell lines | miR-1228 (miRNA profiling) |
Increase in migration and invasion via miR-1228 transfer | [105][67] | |||||
| MSC | OS cell lines | miR-143 (synthetic introduction) | Reduction of migration via exosome transfer (better than transfection) | [106][68] | |||||
| OS cell lines | - | Profiling of miRNA as OS biomarkers | Better biomarker than ALP or patient stratification according to chemotherapy response | [107,108][69][70], |
2.2. Exosomes in ES
| Origin cell | Recipient cell | Cargo | Change | Ref. |
|---|---|---|---|---|
| ES cell lines | - | EWSR1-FLI1, EZH2, and 10 more mRNAs (mRNA profiling of known ES targets) |
Suitable as circulating biomarkers for ES, detectable in spike-in healthy blood samples | [116][80] |
| ES cell lines | ES cell lines | EWSR1-FLI1 mRNA | Labelled EWSR1-FLI1 transferred to other ES cells, not to OS | [117][81] |
| ES cell lines (normal CD99) | ||||
| miR-199a-3p | (miRNA profiling) miR-199a-3p |
Induction of different gene expression profiles, neural differentiation, and neurogenesis; reduction of cell growth and migration (similar to miR mimic) | [120][84] |
2.3. Exosomes in CS
2.4. Other Roles of Exosomes in Bone Sarcomas
References
- Pegtel, D.M.; Gould, S.J. Exosomes. Annu. Rev. Biochem. 2019, 88, 487–514.
- Johnstone, R.M.; Adam, M.; Hammond, J.R.; Orr, L.; Turbide, C. Vesicle formation during reticulocyte maturation. Association of plasma membrane activities with released vesicles (exosomes). J. Biol. Chem. 1987, 262, 9412–9420.
- Miller, I.V.; Grunewald, T.G.P. Tumour-derived exosomes: Tiny envelopes for big stories. Biol. Cell 2015, 107, 287–305.
- Théry, C.; Zitvogel, L.; Amigorena, S. Exosomes: Composition, biogenesis and function. Nat. Rev. Immunol. 2002, 2, 569.
- Abels, E.R.; Breakefield, X.O. Introduction to Extracellular Vesicles: Biogenesis, RNA Cargo Selection, Content, Release, and Uptake. Cell. Mol. Neurobiol. 2016, 36, 301–312.
- Kharaziha, P.; Ceder, S.; Li, Q.; Panaretakis, T. Tumor cell-derived exosomes: A message in a bottle. Biochim. Biophys. Acta Rev. Cancer 2012, 1826, 103–111.
- György, B.; Szabó, T.G.; Pásztói, M.; Pál, Z.; Misják, P.; Aradi, B.; László, V.; Pállinger, É.; Pap, E.; Kittel, Á.; et al. Membrane vesicles, current state-of-the-art: Emerging role of extracellular vesicles. Cell. Mol. Life Sci. 2011, 68, 2667–2688.
- El Andaloussi, S.; Mäger, I.; Breakefield, X.O.; Wood, M.J.A. Extracellular vesicles: Biology and emerging therapeutic opportunities. Nat. Rev. Drug Discov. 2013, 12, 347.
- Baietti, M.F.; Zhang, Z.; Mortier, E.; Melchior, A.; Degeest, G.; Geeraerts, A.; Ivarsson, Y.; Depoortere, F.; Coomans, C.; Vermeiren, E.; et al. Syndecan-syntenin-ALIX regulates the biogenesis of exosomes. Nat. Cell Biol. 2012, 14, 677–685.
- Trajkovic, K.; Hsu, C.; Chiantia, S.; Rajendran, L.; Wenzel, D.; Wieland, F.; Schwille, P.; Brügger, B.; Simons, M. Ceramide Triggers Budding of Exosome Vesicles into Multivesicular Endosomes. Science 2008, 319, 1244–1247.
- Kowal, J.; Tkach, M.; Théry, C. Biogenesis and secretion of exosomes. Curr. Opin. Cell Dev. Biol. 2014, 29, 116–125.
- Villarroya-Beltri, C.; Baixauli, F.; Gutiérrez-Vázquez, C.; Sánchez-Madrid, F.; Mittelbrunn, M. Sorting it out: Regulation of exosome loading. Semin. Cancer Biol. 2014, 28, 3–13.
- Laulagnier, K.; Motta, C.; Hamdi, S.; Roy, S.; Fauvelle, F.; Pageaux, J.-F.; Kobayashi, T.; Salles, J.-P.; Perret, B.; Bonnerot, C.; et al. Mast cell- and dendritic cell-derived exosomes display a specific lipid composition and an unusual membrane organization. Biochem. J. 2004, 380, 161–171.
- Ostrowski, M.; Carmo, N.B.; Krumeich, S.; Fanget, I.; Raposo, G.; Savina, A.; Moita, C.F.; Schauer, K.; Hume, A.N.; Freitas, R.P.; et al. Rab27a and Rab27b control different steps of the exosome secretion pathway. Nat. Cell Biol. 2010, 12, 19.
- Parolini, I.; Federici, C.; Raggi, C.; Lugini, L.; Palleschi, S.; De Milito, A.; Coscia, C.; Iessi, E.; Logozzi, M.; Molinari, A.; et al. Microenvironmental pH is a key factor for exosome traffic in tumor cells. J. Biol. Chem. 2009, 284, 34211–34222.
- Horibe, S.; Tanahashi, T.; Kawauchi, S.; Murakami, Y.; Rikitake, Y. Mechanism of recipient cell-dependent differences in exosome uptake. BMC Cancer 2018, 18, 1–9.
- Tian, T.; Zhu, Y.L.; Zhou, Y.Y.; Liang, G.F.; Wang, Y.Y.; Hu, F.H.; Xiao, Z.D. Exosome uptake through clathrin-mediated endocytosis and macropinocytosis and mediating miR-21 delivery. J. Biol. Chem. 2014, 289, 22258–22267.
- Svensson, K.J.; Christianson, H.C.; Wittrup, A.; Bourseau-Guilmain, E.; Lindqvist, E.; Svensson, L.M.; Mörgelin, M.; Belting, M. Exosome uptake depends on ERK1/2-heat shock protein 27 signaling and lipid raft-mediated endocytosis negatively regulated by caveolin-1. J. Biol. Chem. 2013, 288, 17713–17724.
- Munich, S.; Sobo-Vujanovic, A.; Buchser, W.J.; Beer-Stolz, D.; Vujanovic, N.L. Dendritic cell exosomes directly kill tumor cells and activate natural killer cells via TNF superfamily ligands. Oncoimmunology 2012, 1, 1074–1083.
- Théry, C. Exosomes: Secreted vesicles and intercellular communications. F1000 Biol. Rep. 2011, 3, 1–8.
- Henne, W.M.; Buchkovich, N.J.; Zhao, Y.; Emr, S.D. The endosomal sorting complex ESCRT-II mediates the assembly and architecture of ESCRT-III helices. Cell 2012, 151, 356–371.
- Pols, M.S.; Klumperman, J. Trafficking and function of the tetraspanin CD63. Exp. Cell Res. 2009, 315, 1584–1592.
- Thery, C.; Boussac, M.; Veron, P.; Ricciardi-Castagnoli, P.; Raposo, G.; Garin, J.; Amigorena, S. Proteomic Analysis of Dendritic Cell-Derived Exosomes: A Secreted Subcellular Compartment Distinct from Apoptotic Vesicles. J. Immunol. 2001, 166, 7309–7318.
- Valadi, H.; Ekström, K.; Bossios, A.; Sjöstrand, M.; Lee, J.J.; Lötvall, J.O. Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat. Cell Biol. 2007, 9, 654–659.
- Thakur, B.K.; Zhang, H.; Becker, A.; Matei, I.; Huang, Y.; Costa-Silva, B.; Zheng, Y.; Hoshino, A.; Brazier, H.; Xiang, J.; et al. Double-stranded DNA in exosomes: A novel biomarker in cancer detection. Cell Res. 2014, 24, 766–769.
- Kahlert, C.; Melo, S.A.; Protopopov, A.; Tang, J.; Seth, S.; Koch, O.; Zhang, J.; Weitz, J.; Chin, L.; Futreal, A.; et al. Identification of doublestranded genomic dna spanning all chromosomes with mutated KRAS and P53 DNA in the serum exosomes of patients with pancreatic cancer. J. Biol. Chem. 2014, 289, 3869–3875.
- Mathivanan, S.; Ji, H.; Simpson, R.J. Exosomes: Extracellular organelles important in intercellular communication. J. Proteom. 2010, 73, 1907–1920.
- Llorente, A.; Skotland, T.; Sylvänne, T.; Kauhanen, D.; Róg, T.; Orłowski, A.; Vattulainen, I.; Ekroos, K.; Sandvig, K. Molecular lipidomics of exosomes released by PC-3 prostate cancer cells. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2013, 1831, 1302–1309.
- Skotland, T.; Sandvig, K.; Llorente, A. Lipids in exosomes: Current knowledge and the way forward. Prog. Lipid Res. 2017, 66, 30–41.
- Théry, C.; Ostrowski, M.; Segura, E. Membrane vesicles as conveyors of immune responses. Nat. Rev. Immunol. 2009, 9, 581–593.
- Lachenal, G.; Pernet-Gallay, K.; Chivet, M.; Hemming, F.J.; Belly, A.; Bodon, G.; Blot, B.; Haase, G.; Goldberg, Y.; Sadoul, R. Release of exosomes from differentiated neurons and its regulation by synaptic glutamatergic activity. Mol. Cell. Neurosci. 2011, 46, 409–418.
- Ratajczak, J.; Miekus, K.; Kucia, M.; Zhang, J.; Reca, R.; Dvorak, P.; Ratajczak, M.Z. Embryonic stem cell-derived microvesicles reprogram hematopoietic progenitors: Evidence for horizontal transfer of mRNA and protein delivery. Leukemia 2006, 20, 847–856.
- Dias, M.V.S.; Costa, C.S.; DaSilva, L.L.P. The Ambiguous Roles of Extracellular Vesicles in HIV Replication and Pathogenesis. Front. Microbiol. 2018, 9, 1–13.
- Tai, Y.L.; Chen, K.C.; Hsieh, J.T.; Shen, T.L. Exosomes in cancer development and clinical applications. Cancer Sci. 2018, 109, 2364–2374.
- Casali, P.G.; Bielack, S.; Abecassis, N.; Aro, H.T.; Bauer, S.; Biagini, R.; Bonvalot, S.; Boukovinas, I.; Bovee, J.V.M.G.; Brennan, B.; et al. Bone sarcomas: ESMO-PaedCan-EURACAN Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2018, 29, iv79–iv95.
- Grünewald, T.G.P.; Cidre-Aranaz, F.; Surdez, D.; Tomazou, E.M.; de Álava, E.; Kovar, H.; Sorensen, P.H.; Delattre, O.; Dirksen, U. Ewing sarcoma. Nat. Rev. Dis. Primers 2018, 4, 5.
- Kansara, M.; Teng, M.W.; Smyth, M.J.; Thomas, D.M. Translational biology of osteosarcoma. Nat. Rev. Cancer 2014, 14, 722–735.
- Gaspar, N.; Hawkins, D.S.; Dirksen, U.; Lewis, I.J.; Ferrari, S.; Le Deley, M.C.; Kovar, H.; Grimer, R.; Whelan, J.; Claude, L.; et al. Ewing sarcoma: Current management and future approaches through collaboration. J. Clin. Oncol. 2015, 33, 3036–3046.
- Zhuo, B.; Li, Y.; Gu, F.; Li, Z.; Sun, Q.; Shi, Y.; Shen, Y.; Zhang, F.; Wang, R.; Wang, X. Overexpression of CD155 relates to metastasis and invasion in osteosarcoma. Oncol. Lett. 2018, 15, 7312–7318.
- Xu, H.; Zhu, X.; Bao, H.; Wh Shek, T.; Huang, Z.; Wang, Y.; Wu, X.; Wu, Y.; Chang, Z.; Wu, S.; et al. Genetic and clonal dissection of osteosarcoma progression and lung metastasis. Int. J. Cancer 2018, 143, 1134–1142.
- Mu, X.; Isaac, C.; Greco, N.; Huard, J.; Weiss, K. Notch signaling is associated with ALDH activity and an aggressive metastatic phenotype in murine osteosarcoma cells. Front. Oncol. 2013, 3, 143.
- Potratz, J.; Tillmanns, A.; Berning, P.; Korsching, E.; Schaefer, C.; Lechtape, B.; Schleithoff, C.; Unland, R.; Schäfer, K.L.; Müller-Tidow, C.; et al. Receptor tyrosine kinase gene expression profiles of Ewing sarcomas reveal ROR1 as a potential therapeutic target in metastatic disease. Mol. Oncol. 2016, 10, 677–692.
- Li, G.; Zhang, P.; Zhang, W.; Lei, Z.; He, J.; Meng, J.; Di, T.; Yan, W. Identification of key genes and pathways in Ewing’s sarcoma patients associated with metastasis and poor prognosis. Onco Targets Ther. 2019, 12, 4153–4165.
- Luo, W.; Xu, C.; Ayello, J.; Dela Cruz, F.; Rosenblum, J.M.; Lessnick, S.L.; Cairo, M.S. Protein phosphatase 1 regulatory subunit 1A in ewing sarcoma tumorigenesis and metastasis. Oncogene 2018, 37, 798–809.
- Choo, S.; Wang, P.; Newbury, R.; Roberts, W.; Yang, J. Reactivation of TWIST1 contributes to Ewing sarcoma metastasis. Pediatr. Blood Cancer 2018, 65, 1–18.
- Hatano, M.; Matsumoto, Y.; Fukushi, J.I.; Matsunobu, T.; Endo, M.; Okada, S.; Iura, K.; Kamura, S.; Fujiwara, T.; Iida, K.; et al. Cadherin-11 regulates the metastasis of Ewing sarcoma cells to bone. Clin. Exp. Metastasis 2015, 32, 579–591.
- Mendoza-Naranjo, A.; El-Naggar, A.; Wai, D.H.; Mistry, P.; Lazic, N.; Ayala, F.R.R.; da Cunha, I.W.; Rodriguez-Viciana, P.; Cheng, H.; Tavares Guerreiro Fregnani, J.H.; et al. ERBB4 confers metastatic capacity in Ewing sarcoma. EMBO Mol. Med. 2013, 5, 1019–1034.
- El-Naggar, A.M.; Veinotte, C.J.; Cheng, H.; Grunewald, T.G.P.; Negri, G.L.; Somasekharan, S.P.; Corkery, D.P.; Tirode, F.; Mathers, J.; Khan, D.; et al. Translational Activation of HIF1α by YB-1 Promotes Sarcoma Metastasis. Cancer Cell 2015, 27, 682–697.
- Krook, M.A.; Nicholls, L.A.; Scannell, C.A.; Chugh, R.; Thomas, D.G.; Lawlor, E.R. Stress-induced CXCR4 promotes migration and invasion of Ewing Sarcoma. Mol. Cancer Res. 2014, 12, 953–964.
- Chow, W.A. Chondrosarcoma: Biology, genetics, and epigenetics. F1000Research 2018, 7, 1826.
- Wu, C.M.; Li, T.M.; Hsu, S.F.; Su, Y.C.; Kao, S.T.; Fong, Y.C.; Tang, C.H. IGF-I enhances α5β1 integrin expression and cell motility in human chondrosarcoma cells. J. Cell. Physiol. 2011, 226, 3270–3277.
- Lai, T.H.; Fong, Y.C.; Fu, W.M.; Yang, R.S.; Tang, C.H. Stromal cell-derived factor-1 increase αvβ3 integrin expression and invasion in human chondrosarcoma cells. J. Cell. Physiol. 2009, 218, 334–342.
- Valery, P.C.; Laversanne, M.; Bray, F. Bone cancer incidence by morphological subtype: A global assessment. Cancer Causes Control 2015, 26, 1127–1139.
- Mirabello, L.; Troisi, R.J.; Savage, S.A. Osteosarcoma incidence and survival improvement. Cancer 2009, 115, 1531–1543.
- Abarrategi, A.; Tornin, J.; Lucia, M.C.; Hamilton, A.; Enrique, M.C.; Rodrigo, J.P.; González, M.V.; Baldini, N.; Javier, G.C.; Rodriguez, R. Osteosarcoma: Cells-of-Origin, Cancer stem cells, and targeted therapies. Stem Cells Int. 2016, 2016, 3631764.
- Bayani, J.; Zielenska, M.; Pandita, A.; Al-Romaih, K.; Karaskova, J.; Harrison, K.; Bridge, J.A.; Sorensen, P.; Thorner, P.; Squire, J.A. Spectral karyotyping identifies recurrent complex rearrangements of chromosomes 8, 17, and 20 in osteosarcomas. Genes Chromosoms Cancer 2003, 36, 7–16.
- Stephens, P.J.; Greenman, C.D.; Fu, B.; Yang, F.; Bignell, G.R.; Mudie, L.J.; Pleasance, E.D.; Lau, K.W.; Beare, D.; Stebbings, L.A.; et al. Massive genomic rearrangement acquired in a single catastrophic event during cancer development. Cell 2011, 144, 27–40.
- Daw, N.C.; Chou, A.J.; Jaffe, N.; Rao, B.N.; Billups, C.A.; Rodriguez-Galindo, C.; Meyers, P.A.; Huh, W.W. Recurrent osteosarcoma with a single pulmonary metastasis: A multi-institutional review. Br. J. Cancer 2015, 112, 278–282.
- Isakoff, M.S.; Bielack, S.S.; Meltzer, P.; Gorlick, R. Osteosarcoma: Current treatment and a collaborative pathway to success. J. Clin. Oncol. 2015, 33, 3029–3035.
- Jerez, S.; Araya, H.; Thaler, R.; Charlesworth, M.C.; López-Solís, R.; Kalergis, A.M.; Céspedes, P.F.; Dudakovic, A.; Stein, G.S.; van Wijnen, A.J.; et al. Proteomic Analysis of Exosomes and Exosome-Free Conditioned Media From Human Osteosarcoma Cell Lines Reveals Secretion of Proteins Related to Tumor Progression. J. Cell. Biochem. 2017, 118, 351–360.
- Jerez, S.; Araya, H.; Hevia, D.; Irarrázaval, C.E.; Thaler, R.; van Wijnen, A.J.; Galindo, M. Extracellular vesicles from osteosarcoma cell lines contain miRNAs associated with cell adhesion and apoptosis. Gene 2019, 710, 246–257.
- Gong, L.; Bao, Q.; Hu, C.; Wang, J.; Zhou, Q.; Wei, L.; Tong, L.; Zhang, W.; Shen, Y. Exosomal miR-675 from metastatic osteosarcoma promotes cell migration and invasion by targeting CALN1. Biochem. Biophys. Res. Commun. 2018, 500, 170–176.
- Raimondi, L.; De Luca, A.; Gallo, A.; Costa, V.; Russelli, G.; Cuscino, N.; Manno, M.; Raccosta, S.; Carina, V.; Bellavia, D.; et al. Osteosarcoma cell-derived exosomes affect tumor microenvironment by specific packaging of microRNAs. Carcinogenesis 2019, 1–12.
- Baglio, S.R.; Lagerweij, T.; Pérez-Lanzón, M.; Ho, X.D.; Léveillé, N.; Melo, S.A.; Cleton-Jansen, A.M.; Jordanova, E.S.; Roncuzzi, L.; Greco, M.; et al. Blocking tumor-educated MSC paracrine activity halts osteosarcoma progression. Clin. Cancer Res. 2017, 23, 3721–3733.
- Torreggiani, E.; Roncuzzi, L.; Perut, F.; Zini, N.; Baldini, N. Multimodal transfer of MDR by exosomes in human osteosarcoma. Int. J. Oncol. 2016, 49, 189–196.
- Endo-Munoz, L.; Cai, N.; Cumming, A.; Macklin, R.; De Long, L.M.; Topkas, E.; Mukhopadhyay, P.; Hill, M.; Saunders, N.A. Progression of osteosarcoma from a non-metastatic to a metastatic phenotype is causally associated with activation of an autocrine and paracrine uPA axis. PLoS ONE 2015, 10, e0133592.
- Wang, J.W.; Wu, X.F.; Gu, X.J.; Jiang, X.H. Exosomal miR-1228 from cancer-associated fibroblasts promotes cell migration and invasion of osteosarcoma by directly targeting SCAI. Oncol. Res. 2019, 27, 979–986.
- Shimbo, K.; Miyaki, S.; Ishitobi, H.; Kato, Y.; Kubo, T.; Shimose, S.; Ochi, M. Exosome-formed synthetic microRNA-143 is transferred to osteosarcoma cells and inhibits their migration. Biochem. Biophys. Res. Commun. 2014, 445, 381–387.
- Fujiwara, T.; Uotani, K.; Yoshida, A.; Morita, T.; Nezu, Y.; Kobayashi, E.; Yoshida, A.; Uehara, T.; Omori, T.; Sugiu, K.; et al. Clinical significance of circulating miR-25-3p as a novel diagnostic and prognostic biomarker in osteosarcoma. Oncotarget 2017, 8, 33375–33392.
- Xu, J.-F.; Wang, Y.-P.; Zhang, S.-J.; Chen, Y.; Gu, H.-F.; Dou, X.-F.; Xia, B.; Bi, Q.; Fan, S.-W. Exosomes containing differential expression of microRNA and mRNA in osteosarcoma that can predict response to chemotherapy. Oncotarget 2017, 8, 75968–75978.
- Muz, B.; de la Puente, P.; Azab, F.; Azab, A.K. The role of hypoxia in cancer progression, angiogenesis, metastasis, and resistance to therapy. Hypoxia 2015, 3, 83–92.
- Stiller, C.A.; Trama, A.; Serraino, D.; Rossi, S.; Navarro, C.; Chirlaque, M.D.; Zielonk, N.; Van Eycken, E.; Sundseth, H.; Hedelin, G.; et al. Descriptive epidemiology of sarcomas in Europe: Report from the RARECARE project. Eur. J. Cancer 2013, 49, 684–695.
- Delattre, O.; Zucman, J.; Plougastel, B.; Desmaze, C.; Melot, T.; Peter, M.; Kovar, H.; Joubert, I.; de Jong, P.; Rouleau, G. Gene fusion with an ETS DNA-binding domain caused by chromosome translocation in human tumours. Nature 1992, 359, 162–165.
- Peter, M.; Gilbert, E.; Delattre, O. A multiplex Real-Time PCR assay for the detection of gene fusions observed in solid tumors. Lab. Investig. 2001, 81, 905–912.
- Pinto, A.; Dickman, P.; Parham, D. Pathobiologic markers of the Ewing sarcoma family of tumors: State of the art and prediction of behaviour. Sarcoma 2011, 2011, 856190.
- Kreppel, M.; Aryee, D.N.T.; Schaefern, K.L.; Amann, G.; Kofler, R.; Poremba, C.; Kovar, H. Suppression of KCMF1 by constitutive high CD99 expression is involved in the migratory ability of Ewing’s sarcoma cells. Oncogene 2006, 25, 2795–2800.
- Rocchi, A.; Manara, M. CD99 inhibits neural differentiation of human Ewing sarcoma cells and thereby contributes to oncogenesis. J. Clin. Investig. 2010, 120, 668–680.
- Balamuth, N.J.; Womer, R.B. Ewing’s sarcoma. Lancet Oncol. 2010, 11, 184–192.
- Rodriguez-Galindo, C.; Spunt, S.L.; Pappo, A.S. Treatment of Ewing sarcoma family of tumors: Current status and outlook for the future. Med. Pediatr. Oncol. 2003, 40, 276–287.
- Miller, I.V.; Raposo, G.; Welsch, U.; Prazeres da Costa, O.; Thiel, U.; Lebar, M.; Maurer, M.; Bender, H.U.; von Luettichau, I.; Richter, G.H.S.; et al. First identification of Ewing’s sarcoma-derived extracellular vesicles and exploration of their biological and potential diagnostic implications. Biol. Cell 2013, 105, 289–303.
- Tsugita, M.; Yamada, N.; Noguchi, S.; Yamada, K.; Moritake, H.; Shimizu, K.; Akao, Y.; Ohno, T. Ewing sarcoma cells secrete EWS/Fli-1 fusion mRNA via microvesicles. PLoS ONE 2013, 8, e77416.
- Villasante, A.; Marturano-Kruik, A.; Ambati, S.R.; Liu, Z.; Godier-Furnemont, A.; Parsa, H.; Lee, B.W.; Moore, M.A.S.; Vunjak-Novakovic, G. Recapitulating the size and cargo of tumor exosomes in a tissue-engineered model. Theranostics 2016, 6, 1119–1130.
- Ventura, S.; Aryee, D.N.T.; Felicetti, F.; De Feo, A.; Mancarella, C.; Manara, M.C.; Picci, P.; Colombo, M.P.; Kovar, H.; Care, A.; et al. CD99 regulates neural differentiation of Ewing sarcoma cells through miR-34a-Notch-mediated control of NF-κB signaling. Oncogene 2016, 35, 3944–3954.
- De Feo, A.; Sciandra, M.; Ferracin, M.; Felicetti, F.; Astolfi, A.; Pignochino, Y.; Picci, P.; Carè, A.; Scotlandi, K. Exosomes from CD99-deprived Ewing sarcoma cells reverse tumor malignancy by inhibiting cell migration and promoting neural differentiation. Cell Death Dis. 2019, 10, 1–15.
- Kim, M.J.; Cho, K.J.; Ayala, A.G.; Ro, J.Y. Chondrosarcoma: With updates on molecular genetics. Sarcoma 2011, 2011, 405437.
- Cintra, F.F.; Etchebehere, M.; Gonçalves, J.C.B.; Cassone, A.E.; Amstalden, E.M.I. Vascular pattern in enchondroma and chondrosarcoma: Clinical and immunohistologic study. Appl. Immunohistochem. Mol. Morphol. 2014, 22, 600–605.
- Liu, G.T.; Chen, H.T.; Tsou, H.K.; Tan, T.W.; Fong, Y.C.; Chen, P.C.; Yang, W.H.; Wang, S.W.; Chen, J.C.; Tang, C.H. CCL5 promotes VEGF-dependent angiogenesis by downregulating miR-200B through PI3K/Akt signaling pathway in human chondrosarcoma cells. Oncotarget 2014, 5, 10718–10731.
- Liu, G.T.; Huang, Y.L.; Tzeng, H.E.; Tsai, C.H.; Wang, S.W.; Tang, C.H. CCL5 promotes vascular endothelial growth factor expression and induces angiogenesis by down-regulating miR-199a in human chondrosarcoma cells. Cancer Lett. 2015, 357, 476–487.
- Sun, X.; Charbonneau, C.; Wei, L.; Chen, Q.; Terek, R.M. MiR-181a targets RGS16 to promote chondrosarcoma growth, angiogenesis, and metastasis. Mol. Cancer Res. 2015, 13, 1347–1357.
- Zhang, P.; Li, J.; Song, Y.; Wang, X. MiR-129-5p inhibits proliferation and invasion of chondrosarcoma cells by regulating SOX4/Wnt/β-catenin signaling pathway. Cell. Physiol. Biochem. 2017, 42, 242–253.
- Lu, N.; Lin, T.; Wang, L.; Qi, M.; Liu, Z.; Dong, H.; Zhang, X.; Zhai, C.; Wang, Y.; Liu, L.; et al. Association of SOX4 regulated by tumor suppressor miR-30a with poor prognosis in low-grade chondrosarcoma. Tumor Biol. 2015, 36, 3843–3852.
- Horng, C.T.; Shieh, P.C.; Tan, T.W.; Yang, W.H.; Tang, C.H. Paeonol suppresses chondrosarcoma metastasis through up-regulation of miR-141 by modulating PKCδ and c-Src signaling pathway. Int. J. Mol. Sci. 2014, 15, 11760–11772.
- Tsai, C.H.; Yang, D.Y.; Lin, C.Y.; Chen, T.M.; Tang, C.H.; Huang, Y.L. Sphingosine-1-phosphate suppresses chondrosarcoma metastasis by upregulation of tissue inhibitor of metalloproteinase 3 through suppressing miR-101 expression. Mol. Oncol. 2017, 11, 1380–1398.
- Aghebati-Maleki, A.; Nami, S.; Baghbanzadeh, A.; Karzar, B.H.; Noorolyai, S.; Fotouhi, A.; Aghebati-Maleki, L. Implications of exosomes as diagnostic and therapeutic strategies in cancer. J. Cell. Physiol. 2019, 234, 21694–21706.
- Gudbergsson, J.M.; Jønsson, K.; Simonsen, J.B.; Johnsen, K.B. Systematic review of targeted extracellular vesicles for drug delivery—Considerations on methodological and biological heterogeneity. J. Control. Release 2019, 306, 108–120.
