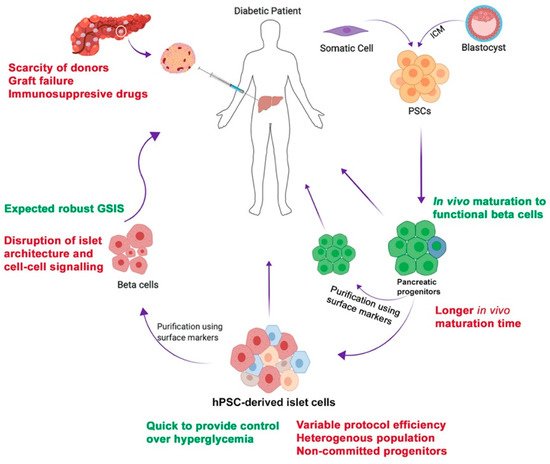
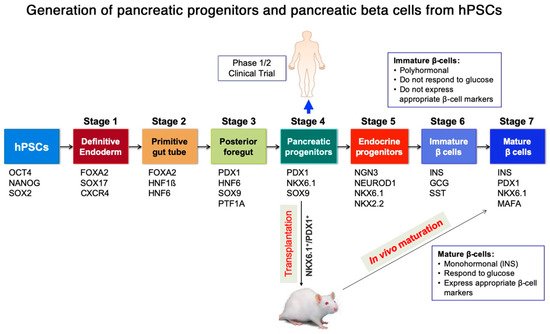
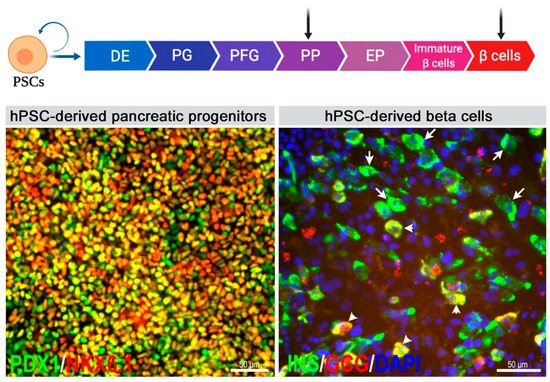
4. Encapsulation of hPSC-Derived Pancreatic Progeny for Cell Therapy
The in vivo maturation of hPSC-derived pancreatic progenitors or pancreatic beta cells indispensably requires a suitable transplantation site, as well as an appropriate encapsulation material or device. The pancreas provides an appropriate microenvironment for the maturation of islets. However, a surgical method for delivery and retreivability has limited its consideration as a candidate for transplantation site. One study transplanted islets in rats and found that normoglycemia is achieved with fewer islets, compared to the extra-pancreatic sites, such as the liver and the kidney
[78][92]. However, it is worth noting that pancreatic progenitors transplanted subcutaneously or under kidney capsules have, nevertheless, resulted in their differentiation into functional beta cells
[15][30][15,30], despite not being exposed to a ‘pancreatic’ microenvironment. For example, a previous study investigated the effect of the transplantation site on the generation of monohormonal insulin-secreting cells and found that the transplantation of pancreatic progenitors under mammary fat pads or kidney capsules do not affect their maturation into beta cells
[15]. It is plausible that the crucial vascular system supplying nutrients and oxygen to these transplanted progenitors carries cues sufficient to facilitate their maturation into beta cells.
An encapsulation device is, therefore, a crucial factor affecting the performance of the transplanted cells and it can control teratoma formation if the transplanted cells are contaminated with undifferentiated cells. Encapsulation of the transplanted pancreatic lineage is crucial in case of T1D to prevent an autoimmune reaction against the transplanted cells. Additionally, if the cell therapy product is from an allogenic source, for example, commercialized off-the-shelf hPSC-derived beta cells, then encapsulation is still a requirement. However, in the case of T2D or monogenic diabetes, there is a possibility of transplanting pancreatic progeny derived from the patient’s own cells, that circumvents the need for encapsulation
[5].
Achieving the right design of an encapsulation device requires putting together variables, such as the biocompatibility properties of the membrane, exposure to the blood stream, and availability of nutrition and oxygen for the encapsulated cells amongst others
[79][93]. Studies are being done on modification of the available materials to improve these properties of the biomaterial, and have mainly been developed for islet transplantation, both in macro- and micro-encapsulation systems. The assessment of islet function and viability following their coating with alginate derivatives is being widely investigated for improving islet transplantation outcomes. Purified alginate improved the survival of encapsulated islets and had a moderate effect on necrosis compared to non-purified alginate capsules
[80][81][94,95]. Furthermore, certain alginate modifications are particularly interesting to study as they could circumvent immune response following transplantation of allogenic islets. Modification of alginate capsules using triazole-derivatives showed positive results in preventing immune cell activation at capsule surfaces in mice and non-human primates
[82][96]. The incorporation of the chemokine CXCL12 in the alginate capsule protected the islets and improved their function by serving as an immune-isolating material without the need for immune-suppression
[83][97]. Likewise, such alginate-based microencapsulation methods are now being applied for stem cell therapy, such as for hPSC-derived beta cells. CXCL12 coating was recently shown to prolong the viability of hPSC-derived beta cells in immune-competent mice without requiring immunosuppression by preventing fibrotic overgrowth
[84][98]. In addition, the CXCL12 coating enhanced beta cell function by improving their glucose responsiveness, thereby making it an important biomaterial to study further for beta cell encapsulation.
While alginate-based microencapsulation is a promising option for beta cell replacement therapy, complete retrieval of the implanted microencapsulated islets in the portal vein, under kidney capsule or in the peritoneal space is challenging, often being invasive and incomplete
[3][85][86][3,99,100]. Therefore, their use as an encapsulant for hPSC-derived beta cells, without a protective retrievable device could raise concerns as hPSC-derived beta cells have not yet been proven to be completely identical to pancreatic adult islets
[27] and require their retrieval periodically to assess their viability, function, and to detect any teratomas formed. While studies on the formation of teratoma by unsorted terminally differentiated hPSC cultures containing target cells and uncommitted progenitors have revealed their expected frequency
[87][101], it is unclear how the variability in differentiation efficiencies by different cell lines for the generation of beta cells would affect teratoma formation in vivo. However, the purification of fully mature hPSC-derived beta cells using surface markers prior to their microencapsulation could nullify some of these concerns. Therefore, further studies are required for the identification of these markers and their application.
On the other hand, minimally invasive macro-encapsulation devices have also shown progress in delivering cell therapy products. The bilaminar TheraCyte macro-encapsulation device, which has an outer surface that promotes tissue engraftment or infiltration of blood vessels thus providing a close proximity of vasculature to the implanted cells and an inner membrane that prevents immune cell diffusion, has been shown to protect against immune-rejection of the transplanted pancreatic tissues in rodents. In addition, the device has allowed maturation of the hPSC-derived pancreatic lineages into pancreatic beta cells in animal models and proven competent at controlling hyperglycemia
[9][29][31][88][89][90][91][92][9,29,31,102,103,104,105,106]. ViaCyte, leading the only clinical trial using hPSC-derived product for T1D treatment has reviewed the performance of its macro-encapsulation device, PEC-Encap, a TheraCyte adaptation, in the first phase of their study. The PEC-Encap is a combination of the hPSC-derived pancreatic progenitors enclosed in a semi-permeable, Encaptra drug delivery system, which was transplanted under the skin of T1D patients (
https://viacyte.com/products/pec-encap-vc-01). The preliminary results indicated that the device is not effective in allowing its engraftment into the host tissue, which may impair supply of oxygen and nutrients to the therapeutic cells within the device (
https://viacyte.com/products/pec-encap-vc-01). However, it was shown to protect the cells from immune invasion. To overcome these limitations, another ViaCyte system called the PEC-Direct, with a modified encapsulation membrane, is being currently employed in another clinical trial to assess its safety T1D hypoglycemic patients, however it requires immune-suppression (
https://viacyte.com/products/pec-direct). Nonetheless, novel cell encapsulation systems are being developed, such as the Cell Pouch by Sernova (
https://www.sernova.com/technology/) that facilitates formation of a pre-vascularized scaffold at the target site before cell delivery, that can advance cell therapy
[93][107]. Another innovative device that aims to enhance oxygen availability to the encapsulated cells is the Beta-O
2 device that consists of a gas chamber next to the encapsulated cells that allows the diffusion of oxygen to the cells (
https://beta-o2.com/living-with-sair/). This chamber can be refilled occasionally to maintain a continuous supply of oxygen.