Antibiotic resistance has increased markedly in Gram-negative bacteria, causing severe infections intractable with traditional drugs and amplifying mortality and healthcare costs. Consequently, to find novel antimicrobial compounds, active on multidrug resistant bacteria, is mandatory. In this regard, cationic antimicrobial peptides (CAMPs)—able to kill pathogens on contact—could represent an appealing solution. However, low selectivity, hemolytic toxicity and cost of manufacturing, hamper their massive clinical application. In the recent years—starting from CAMPs as template molecules—less toxic and lower-cost synthetic mimics of CAMPs, including cationic peptides, polymers and dendrimers, have been developed. Although the pending issue of hemolytic toxicity and biodegradability is still left not completely solved, cationic antimicrobial polymers (CAPs), compared to small drug molecules, thanks to their high molecular weight, own appreciable selectivity, reduced toxicity toward eukaryotic cells, more long-term activity, stability and non-volatility. With this background, an updated overview concerning the state of the art of the main manufactured types of CAPs, active on Gram-negative bacteria, is herein reported, including synthetic procedure and action’s mechanism. Information about the antibacterial activity, advantages and drawbacks of the most appealing compounds was also provided.
Introduction
Polymers have gained increasing attention by the scientific community as promising materials to prepare antimicrobial agents because of several advantages. An antibacterial polymeric device, differently from small drug molecules, could be endowed with more long-term activity, limited residual toxicity, chemical stability, non-volatility and incapacity to permeate through the skin thanks to its macromolecular structure and high molecular weight (MW)
[1,2][1][2].
The developed antimicrobial polymers can be divided into three wide families as reported in .
Table 1. Categories of antimicrobial polymer systems.
Since it is reasonable to think that mechanism of action of synthetic CAPs would be similar to that of CAMPs previously described and considered “membrane active agents”, the main strategies premeditated to design them, depended on the structural features of the outer envelope of the different bacterial cells. Since the scope of this work is to review CAPs active on Gram-negative bacteria, a list of the some representatives of this class was reported in , while a brief description of the cell wall of Gram-negative bacteria was provided in the subsequent Section.
Table 2. Examples of Gram-negative bacteria.
| Family |
Genus |
Species |
| biocidal polymer |
necessarily
cationic |
quaternary phosphonium |
| Campylobacteraceae |
Campylobacter | unspecific electrostatic/disruptive interaction with negatively charged bacteria membranes |
no presence of toxic biocide |
no release of harmful biocides for environmental
minor trend to develop resistance |
hemolytic toxicity
|
Campylobacter coli
Campylobacter concisus
| fast clearance from circulation
high uptake in the reticuloendothelial system |
| Campylobacter jejuni |
| Campylobacter rectus |
guanidinium |
| Arcobacter |
tertiary sulfonium |
| primary, secondary, tertiary, quaternary ammonium |
| Arcobacter butzleri |
Arcobacter cryaerophilus |
| Enterobacteriaceae |
Citrobacter |
Citrobacter amalonaticus
Citrobacter braakii
Citrobacter farmeri
Citrobacter freundii
Citrobacter gillenii
Citrobacter koseri |
| Enterobacter |
Enterobacter aerogenes
Enterobacter agglomerans
Enterobacter cloacae
|
biocidal polymers embodied
by the entire macromolecule
not requiring biocidal monomers |
| Enterobacter cowanii |
Enterobacter gergoviae |
polymeric biocide |
from polymerization of antimicrobial monomers
unnecessary cationic
presence of repeated antimicrobial functionalities |
same action of the attached biocide moieties |
lower systemic toxicity
lower hemolytic toxicity
lower clearance |
less active than free biocide drugs for steric hindrance cause by polymer |
| biocide-releasing polymer |
unnecessary cationic
not intrinsic activity of polymer
presence of loaded cleavable antimicrobial drugs covalently linked or by physically entrapped |
by releasing the entrapped or bond antimicrobial drugs |
target release of biocide
higher concentration of biocide at the target site
excellent efficacy |
significant reduction of activity in time
toxicity of free biocide |
In the biocidal polymers the antimicrobial site of biocidal polymers is embodied by the entire macromolecule, they do not require bioactive repeating units and are necessarily cationic.
The biocidal polymers can be obtained either by the polymerization of cationic monomers not necessarily active and frequently without antibacterial activity or by functionalizing an inactive polymeric scaffold with inactive cationic groups to form a cationic macromolecule with antimicrobial activity due to its high density of positive charge. Biocidal polymers are active because they are positively charged macromolecules that, miming CAMPs, are able to kill bacteria on contact, by a disruptive action on their anionic cell membranes
[3][3].
Differently, polymeric biocide derives from the polymerization of antibacterial monomers, which can be cationic, anionic or uncharged. Therefore, polymeric biocides are not necessarily cationic, they can also be anionic or neutral, are less active than monomers and act with the same mechanism of action as monomers. The antibacterial activity resides in the monomeric units and not in the polymer itself and in some cases, the polymer architecture can even nullify the antibacterial effects of the monomers. The mechanisms of action of polymeric biocides can therefore be of various types and do not necessarily interfere with the integrity of the bacterial membranes by destroying them. Lastly, biocide-releasing polymers consist of non-active polymers loaded with biocide moieties, covalently linked or physically entrapped, which can be released also in a targeted and/or protracted modality. In this work, it was reviewed the first class of antimicrobial polymers, i.e., the per se antimicrobial cationic polymers and in particular, those ones active on Gram-negative bacteria.
The cationic moieties possibly present in cationic antimicrobial polymers (CAPs) are in general guanidinium, tertiary sulfonium, primary, secondary, tertiary and quaternary ammonium, including also compounds containing heterocycles such as pyridine, imidazole, etc. with quaternized nitrogen atoms and quaternary phosphonium groups
[4,5][4][5].
In order to obtain good materials, the architecture of the designed polymer should be stable in long-term applications, stable during the required storage time and at the conditions of its targeted application and should have a low degree of toxicity. Among the developed CAPs, few natural and semi-synthetic macromolecules and a large variety of synthetic cationic polymers were manufactured.
| Escherichia |
| Escherichia coli |
| Klebsiella |
Klebsiella pneumoniae |
| Morganella |
Morganella morganii |
| Proteus |
Proteus vulgaris
Proteus mirabilis |
| Shigella |
Shigella dissenteriae |
| Salmonella |
Salmonella tiphy |
| Yersinia |
Yersinia pestis (responsible for the plague)
Yersinia pseudotuberculosis
Yersinia enterocolitica |
| Serratia |
Serratia marcescens |
| Aerobacter |
Aerobacter aerogenes |
| Enterobacter |
Enterobacter sakazakii |
| Moraxellaceae |
Acinetobacter |
Acinetobacter baumannii
Acinetobacter beijerinckii
Acinetobacter bereziniae
Acinetobacter boissieri |
| Moraxella |
Moraxella catarrhalis (Branhamella catarrhalis) |
| Neisseriaceae |
Neisseria |
Neisseria meningitidis |
| Hemophilus |
Hemophilus influenzae |
| Pasteurellaceae |
Pasteurella |
Pasteurella multocida |
| Pseudomonadaceae |
Pseudomonas |
Pseudomonas aeruginosa |
| Vibrionaceae |
Vibrio |
Vibrio cholerae (responsible for cholera)
Vibrio fischeri
Stenotrophomonas maltophilia |
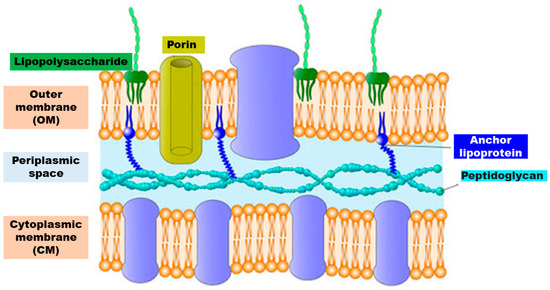
Structure of Gram-negative Cells Wall
Gram-negative outer envelope is composed of a thin peptidoglycan layer sandwiched between an inner cytoplasmic cell
membrane (CM) and a bacterial outer membrane (OM) ().
Figure 4. Schematic representation of the structure of the cell wall of Gram-negative bacteria.
The most important characteristic of the outer envelope of the Gram-negative bacteria cells is a net negative charge, frequently stabilized by the presence of divalent cations such as Mg
2+ and Ca
2+. The anionic character is due to LPS present in the OM, to the phospholipids of the OM and to those of CM, which in turn is composed of a phospholipid bilayer with embedded essential functional proteins, such as enzymes. CM is semi-permeable and controls the passage of solutes and metabolites in and out of the cell cytoplasm
[6][6].
The presence in the OM of porin channels, which slow down molecular diffusion and limit the antibacterial substances diffusion, is considered the reason of the high resistance of Gram-negative bacteria towards common antiseptics and disinfectants in comparison to Gram-positive
[7][7].
OM and CM represent the primary target for the antibacterial agents, whose main mechanism of action mimics that of CAMPs and since they are anionic hydrophilic–hydrophobic compartments, in order to promote their absorption on bacteria, the synthetic antimicrobial polymers were mainly designed as cationic hydrophilic–hydrophobic macromolecular systems.
Antimicrobial Cationic Polymers (CAPs) and their Antibacterial “Brute-force Action”
Several combinations of hydrophilic and hydrophobic polymeric constructions were explored in order to realize the ideal CAP.
As examples, polymers with controlled MW, precise structure and composition were synthetized by advanced polymerization techniques, as reversible deactivation radical polymerization (RDRP) or reversible addition-fragmentation chain transfer polymerization (RAFT).
Block polymers, owing bi-block links, made of a hydrocarbon nonpolar hydrophobic block and of a cationic one, were developed. Furthermore, random copolymers were achieved by polymerizing a hydrophobic monomer and a hydrophilic comonomer with a functional group. Hydrophobic polynorborane-based oligomers and polymers (see Table 5 and
Section 7.2) endowed with high antimicrobial effects against Gram-negative
E. coli rather than against
S. aureus, were synthetized.
In general, but not always, the developed CAPs are amphiphilic macromolecules and possess surface-activity properties, the adsorption/absorption ability of surfactants, high binding affinity for bacterial cells membrane and a proper lipophilicity, that allows them to cause effective damage to the structural organization and integrity of cell membranes and to lead to cell lysis
[1,8,9][1][8][9].
In particular, CAPs like CAMPs, commonly inhibit or kill bacteria immediately on contact by causing the bacterial cell to burst, through a series of steps
[1][1].
In particular, considering the Gram-negative bacteria of interest of this review, in the first step, the polymer adsorbs onto the OM of bacterial cell wall, in virtue of an electrostatic interaction and causes impairments, which translate in an improvement of OM permeability and pores formation. Second, the polymeric antimicrobial agent diffuses through the cell wall, adsorbs onto the CM and finally causes CM disruption. The subsequent leakage of cytoplasmic constituents including crucial cations as K
+ leads to the death of the bacteria cell. It is evident that, while small molecule antimicrobial agents are endowed with a weak adsorption capacity and a good diffusion ability, thanks to their low MW, CAPs excel at the adsorption steps, that are crucial for disrupting CM and kill the cells
[1][1].
Consequently, while the small drugs need to diffuse and enter into bacteria cells to affect vital processes, CAPs neutralize bacteria on contact without the need of interfering with more fine metabolic pathways.
Chitosan-based cationic polysaccharides
[10[10][11],
11], polyvinyl-based phosphonium, quaternized ammonium salts and not quaternized amine polymers
[12][12], insoluble pyridinium-based polymers
[13,14][13][14], polymers peripheral functionalized with poly(vinyl-
N-pyridinium) salts
[15[15][16],
16], ε-poly
L-lysine (
ε-PL)
[17][17], cationic amphiphilic polyacrylates
[18] [18] and branched polyethyleneimine (
b-PEI)
[19,20,21] [19][20][21] are among others, some examples of the developed classes of natural and synthetic positively charged polymers endowed with antimicrobial properties. Details concerning the synthetic strategies and the mechanisms of action are reported in the text of
Section 6 and
Section 7. A brief description of changes caused by CAPs action in Gram-negative bacteria at molecular level and of the polymer’s structural factors that could influence their activity and toxicity are reported in
Section 8 and
Section 9.
Natural Positively Charged Antimicrobial Polymers
Among cationic polymers, up today, chitosan and poly(
ε-lysine) are the only natural polymers recognized to possess antimicrobial properties
[22][22].
Chitosan
Chitosan is a natural cationic polysaccharide deriving from chitin by deacetylation in basic solution and encompassing in its structure units of
β(1 → 4)-2-amido-2-deoxy-D-glucan (D-glucosamine) and
β(1 → 4)-acetoamido-2-deoxy-D-glucan (
N-acetyl glucosamine) joined by glycosidic bonds
[23,24,25][23][24][25].
Usually, commercially available contain > 75%–85% deacetylated units and have MW between 50 and 1000 kDa. The degree of deacetylation strongly influence chitosan solubility, its capacity to interact with polyanions and consequently its antimicrobial effects
[26][26].
In addition, also MW, concentration, physical state (e.g., in solution or in solid state, as fibers, particles or films) and type of microorganism may influence chitosan activity.
Chitosan can act in two modalities, passively by provoking a reduction of protein adsorption on bacteria surface that leads to impairing the adhesion capacity of pathogens (in this case, bacteria are not killed, but only repelled) or actively, killing bacteria on contact.
However, the dominant argument is that chitosan acts principally as an external membrane disruptor, by interacting electrostatically with Gram-negative bacteria LPSs and by causing the formation of pores. Once membrane permeability is hopelessly compromised, chitosan can also behave as penetration material and, if its MW is sufficiently low (MW < 5 kD), can enter into bacteria cells, bind with microbial DNA and/or mRNA, thus interfering with transcription and translation processes.
A third supposed mechanism is based on the property of chitosan to bind metals as bivalent cations that stabilized the OM, thus favoring OM destabilization and loss of integrity.
The chitosan capacity of interacting with the negative charges from the bacterial cell surface is more effective at low pH, that allows the amine groups to be protonated, while the chelation ability is more efficient at high pH, when the positive metal ions can bound to chitosan, non-protonated amino groups and the electron pair on the amine nitrogen is available for donation to metal ions
[26][26].
By synthetic quaternization of the nitrogen atoms of the amino groups of chitosan, semisynthetic chitosan derivatives permanently charged at any pH value were prepared
[27,28][27][28].
Many methods were proposed for realizing the N-quaternization of the chitosan nitrogen atoms and several positively charged derivatives were prepared with different amounts of quaternary ammonium salts moieties.
They are soluble in water and have proved high antimicrobial and antibiofilm activity depending on the number of cationic groups
[29,30,31] [29][30][31] and can be eligible for application in pharmaceutic and biomedical fields as agents against infection by implantation of medical devices.
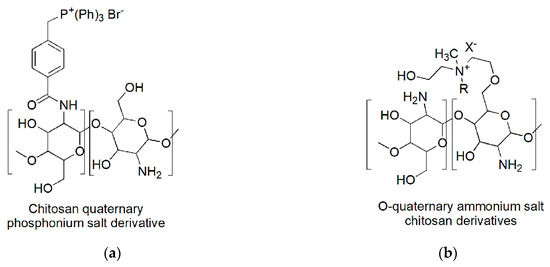
Chitosan derivatives permanently protonated were prepared by exploiting either the amine groups or the hydroxyls to insert phosphonium or ammonium salt functions. It is the case of the chitosan derivatives used by Zhu et al. (2016)
[32] [32] (a) and by Wang et al. (2016) (b)
[33][33].
Figure 5. Quaternized chitosan derivatives permanently cationic: (a) Chitosan phosphonium salt; (b) o-quaternary chitosan ammonium salts. R: –CH2Ph (BNQAS–CS); –C12H25 (C12QAS–CS); – C14H29 (C14QAS–CS); – C16H33 (C16QAS–CS); – C18H37 (C18QAS–CS); X: Cl, Br.
N-quaternary phosphonium chitosan derivatives (N–QPCSxy) were prepared by partial amidation of NH2 moieties of chitosan with 4-(2,5-dioxo-pyrrolidin-1-yloxycarbonyl)-benzyl)-triphenyl-phosphonium bromides (NHS–QPS).
The best compounds achieved proved water solubility over the pH range of 3 to 12 and antibacterial activities significantly improved if compared to chitosan and low cytotoxicity. In particular, the minimum bactericidal concentration (MBC) against
E. coli was observed at a dosage of 500 µg/mL, which allowed a red blood cell viability of 74.1%
[32][32].
On the contrary, the chitosan chemical modification proposed by Wang et al., that led to obtain water-soluble
O-quaternary chitosan ammonium salt (QAS–CS) bearing
N-methyl-
N-R-
N-bis(2-hydroxyethyl)ammonium bromides [R =–benzyl (chloride, BNQAS–CS),–dodecyl (C
12QAS–CS),–tetradecyl(C
14QAS–CS),–hexadecyl(C
16QAS–CS),–octadecyl(C
18QAS–CS)], allowed to achieve molecules with good antibacterial abilities against Gram-positive bacteria, but bad against Gram-negative bacteria
[33][33].
ε-Polylysine (ε-PL)
ε-PL is a cationic polyamide consisting of
L-lysine units (n = 25–30), linked together by the
ε-amino and the
α-carboxyl groups.
ε-PL toxicity is significantly lower than that of CAMPs
[16] [16] and
ε-PL has received increasing attention in food industry as preservative additive, thanks to its strong antimicrobial effects and established safety [16]. In vivo investigations for evaluating acute oral toxicity, proved that
ε-PL is non-toxic at the high dosage of 5 g/kg in rats [16].
Concerning its mechanism of action, Ye et al. (2013)
[34] [34] carried out studies to explain
ε-PL antibacterial mechanism of action against
E. coli O157:H7 at the molecular level.
The results asserted that, initially the main approach is an electrostatic interaction with OM, then quickly
ε-PL strips of the membrane, causing alteration of cytoplasm distribution, formation of pores and the onset of structural defects. The increased permeability of membrane favors bacteria penetration and DNA binding. The disruption of membrane integrity jointed to the detrimental interaction of
ε-PL with genetic material induce oxidative stress by radical oxygen species (ROS) production and influences various gene expressions leading to bacteria death
[35,36,37,38][35][36][37][38].
Synthetic Cationic Antimicrobial Polymers (CAPs)
Polymers Containing Quaternary Phosphonium and/or Ammonium and/or Guanidinium Groups
Polymeric quaternary ammonium salts (PQASs), quaternary phosphonium salts (PQPSs), polymeric guanidine (PGSs) and biguanidine salts (PBGSs) are classes of cationic polymer materials with high potential as antimicrobial agents, due to the high and permanent cationic character of their quaternary groups
[39,40,41,42,43,44,45][39][40][41][42][43][44][45].
Polymers Containing Quaternary Phosphonium and/or Ammonium Groups
PQASs and PQPSs can be obtained either through direct polymerization of monomers already containing quaternary groups, by incorporating the quaternary moieties into the previously synthesized polymers or including them by electrostatic interactions with previously synthetized sulfonate polymers.
Polymers with quaternary ammonium or phosphonium salts are materials widely explored as antimicrobial devices and have proved potent activity
[46,47,48,49] [46][47][48][49] and effectiveness even against bacteria that are resistant to other cationic antibacterial agents
[50][50].
Both of them proved to be more active of the corresponding starting small molecules monomers mainly against Gram-negative bacteria with an effectiveness that resulted enhanced by the increase of polymeric chain length and by the hydrophobicity of the macromolecules
[44, 51,52,53][44][51][52][53].
In general, low MW antimicrobial agents including cationic monomers present several issues counting environmental toxicity and short-term antimicrobial ability. Their introduction into polymer molecules achieving biocide polymers as PQASs and PQPSs allows enhancing their efficacy and selectivity, prolonging their lifetime, minimizing the environmental problems and the residual toxicity
[1][1].
PQASs and PQPSs, when inserted on surfaces to achieve antimicrobial surfaces able to kill airborne as well as waterborne microbes, significantly limit bacteria colonization without release of antimicrobials into the environment.
In addition, they proved higher activity of non-polymeric small antimicrobial drugs because, while their action consists in impairing the adhesion of bacteria by reducing their contact ability to the surface (without killing them), PQASs and PQPSs with quaternary ammonium or phosphonium units, commonly kill bacteria on contact
[1,53,54,55,56,57][1][53][54][55][56][57].
Based on the mode of incorporation of quaternary ammonium or quaternary phosphonium monomers in the polymers, PQASs and PQPSs are classified in two categories: ionically bound or covalently attached.
Generally, the polymeric materials with active cations electrostatically bound exhibited strong antibacterial action, thanks to the release in the aqueous environment of the active cationic groups through an ion exchange mechanism. Differently, in order to exert an antimicrobial action, the class of compounds where the active cations are covalently linked, requires the contact of the polymer with the microorganisms
[39][39].
In addition, studies of comparison between PQASs and PQPSs showed that the latter possess antimicrobial activity higher than that of polymeric quaternary ammonium salts, because of a difference of electronegativity between nitrogen and carbon atoms and phosphorous and carbon atoms
[52][52].
The reason is attributable to the mechanism of action of the ammonium and phosphonium polymers that involves as usual, a destructive electrostatic interaction with the bacteria cell wall
[39,58][39][58].
Concerning Gram-negative bacteria, the results of investigations carried with different experimental methods in the last decades, supported the hypothesis that antimicrobial polymers bearing cationic charges on the quaternary ammonium/phosphonium groups, kill bacteria by electrostatic interaction with the outer membrane (OM) and cytoplasmic membrane (CM), followed by their damage, cell lysis with release of crucial ions such as potassium
[59][59].
Due to the difference of electronegativity existing between nitrogen and phosphorous atoms and the adjacent carbons, in ammonium cation, nitrogen exhibits a negative charge, while in phosphonium, phosphorous owns a positive charge
[60][60].
Consequently, the stronger polarization and positive charge of phosphorous atoms in PQPSs favor easier interactions with bacteria wall thus resulting in higher effectiveness
[61][61]. A positively charged antimicrobial random co-polymer encompassing both kinds of cationic groups was synthetized, via free radical polymerization (FRP) of acrylamide (AM), diallyl dimethyl ammonium chloride (DADMAC) and (4-penten-1-yl) triphenylphosphonium bromide (PTBT).
The obtained poly(PTPB-r-AM-r-DADMAC) copolymers, different for the content of PTBT, in addition to possess antiviral activity, proved to be effective against
E. coli but only when the content in phosphonium monomer was higher than 49%, confirming the higher effectiveness of cation phosphonium
[62][62].
Later, with a similar procedure, tri-blocks copolymers containing both phosphonium and ammonium groups were synthetized by AM, tributyl(4-vinylbenzyl)phosphonium (QPM) and [2-(acryloyloxy)ethyltrimethylammonium chloride (ATC) and were tested for antimicrobial and antiviral activities.
The results from antibacterial evaluations on
S. aureus and
E. coli and from viricidal investigations on influenza virus and adenovirus, demonstrated an excellent antibacterial activity
versus both Gram-positive and Gram-negative bacteria and antiviral activity
versus both enveloped and non-enveloped viruses
[63][63].
In a study by Kougia et al. (2015), a library of homopolymers and copolymers with quaternary cationic groups either electrostatically bound or covalently linked were prepared. Furthermore, copolymers, obtained by copolymerizing the cationic monomer vinylbenzyl dimethylhexadecylammonium chloride (VBCHAM) and either hydrophilic or hydrophobic comonomers, were synthetized. Even if phosphonium co-polymers were also investigated, the study mainly focused on quaternary ammonium polymers. The antimicrobial activity, determined as a function of the contact time at 4 °C and 22 °C was evaluated against
P. aeruginosa,
E. coli,
S. aureus and
Enterococcus fecalis and eventual relationship between polymer chemical structure and antimicrobial activity was investigated and discussed
[39][39].
VBCHAM-based copolymers in which acrylic acid (AA) was used as the comonomer and copolymers presenting both covalently attached and electrostatically bound quaternary ammonium groups showed the highest antimicrobial activity
[39][39].
In particular, seven cationic macromolecules from this study named poly(cetyltrimethylammonium-4-styrene)sulfonate (PSSAmC
16), poly(cetyltrimethylphosphonium-4-styrene)sulfonate (PSSPhC
16), poly(VBCHAM), poly(methymetacrylate-co-VBCHAM) [P(MMA-co-VBCHAM)], poly(cetyltrimethylammonium-4-styrene) sodium sulfonate-co-VBCHAM) [P(SSNa-co-VBCHAM)], poly(acrylamide-co-VBCHAM) [P(AA-co-VBCHAM)] and poly(cetyltrimethylammonium-4-styrene)sulfonate-co-VBCHAM) [P(SSAmC
16-co-VBCHAM)] proved antimicrobial activity against Gram-negative bacteria.
In order to evaluate practical applications of developed CAPs, it was investigated if their antimicrobial activity could be maintained when they are embodied in polysulfone (PSF), a polymer typically used in medical devices. For this experiment, the copolymers P(AA-co-VBCHAM) and P(SSAmC
16-co-VBCHAM) and the homopolymer PSSAmC
16, which were the most active, were entrapped in PSF and the CAPs-enriched PSF-based membrane were essayed. The results confirmed that the developed antimicrobial materials remain remarkably efficient even when they are incorporated in PSF membranes
[39][39].
Polymers Containing Quaternary Guanidinium Groups
Among biguanidinium polymers, poly(hexamethylene biguanide chloride) (PHMB) was the first antimicrobial polymer whose mechanism of interaction with phospholipid membranes was studied by Broxton and coworkers on
E. coli [64,65][64][65].
In particular, it was observed, that the sequence of events during PHMB interaction with the cell envelope of
E. coli involves first, a rapid attraction of PHMB toward the negatively charged bacterial cell surface, thanks to strong and specific adsorption to phosphate groups of compounds present in OM. As a consequence, the integrity of the OM is impaired and PHMB is allowed to proceed and to be attracted to the inner CM, where an additional binding of PHMB to phospholipids occurs. Consequently, also the integrity of inner membrane begin to impair and if PHMB concentrations are low, the increment of membrane permeability causes only the loss of potassium ions (K
+) and provokes bacterial stasis. Progressively, higher concentrations of PHMB, increases the extent of the damage and the size of pores, which allow the loss of larger inorganic species as Cs
+, Na
+, Li
+ and inorganic phosphate. This event leads to a complete loss of membrane functionalities with leak of other essential cellular components, precipitation of intracellular constituents and bacterial death
[64][64].
The damage to the CM by PHMB is non-specific, immediate and irreversible. Practically, a scenario similar to that observed for polyvinyl benzyl dimethyl butyl ammonium chloride
[59,40][40][59].
In regard of growth inhibitory activity and bactericidal activity, PHMB with high MW, i.e., n ≥ 10, proved to be effective at very low value of MIC and MBC, PHMB with 2 > n < 10, proved good activity while the activity of low MW dimers was questionable. It failed to inhibit motility in actively growing cultures and did not totally inhibit growth
[64][64].
In a study by Ikeda et al. (1984), it was aroused a concern about the exact evaluation of antimicrobial activity of biguanidinium compounds in culture medium, because of interfering interactions between the polymeric biguanides and some culture medium constituents.
In this regard, acrylate monomers with pendant biguanide groups were successfully synthesized and their homopolymers and copolymers with acrylamide were prepared by radical polymerization using AIBN as initiator. Evaluated in a clean system, these CAPs showed to be higher effective against Gram-positive bacteria, rather than on Gram-negative strains, but to be much more active than the monomeric species
[66,59][66][59].
Synthetic guanidinium and biguanidinium antimicrobial polymers endowed with a proper amphiphilic balance, which allows high selectivity for bacteria and good antimicrobial activity, are considered the best mimics of CAMPs.
In this regard, polyhexamethylene guanidine hydrochloride (PHMG) and three its analogs, i.e., polybutamethylene guanidine hydrochloride, polyoctamethylene guanidine hydrochloride (POMG) and poly(m-xylylene) guanidine hydrochloride, were prepared by reacting guanidine hydrochloride with the proper di-alkyl amine. The antimicrobial properties of the obtained polymers were investigated on 370 clinical strains, often involved in nosocomial infections, 96 isolates of which were antibiotics-resistant
[67][67].
The best compound was POMG, that provided MIC values (0.5–16 µg/mL) even lower than those of chlorhexidine digluconate (2–64 µg/mL) against all the 370 antibiotics-susceptible and antibiotics-resistant clinical strains. Concerning the interest of the present review, POMG displayed excellent activity (2–16 µg/mL) against several representative of Gram-negative bacteria
[67].
The killing curves showed that POMG was bactericidal at 5 µg/mL, concentration that caused an approximate 6 log
10 reduction in the numbers of CFU for clinically isolated
P. aeruginosa at 4 h
[67][67].
The broad activity of POMG against antibiotic-resistant bacteria suggests that cationic guanidine-based polymers possess high potential for the development of novel potent antimicrobials for clinical applications.
Copolymers with functionalized guanidine pendant groups were prepared by RAFT polymerization and were evaluated for their antimicrobial activity against some Gram-positive strains,
C. albicans and
E. coli, as well as for the hemolytic toxicity
[68,69][68][69]. While antimicrobial activity
versus Gram-positive bacteria and antifungal effects resulted considerable, antibacterial activity on
E. coli was debatable.
Polynorborane-based Antimicrobial Polymers
Designed just to mimic CAMPs, polynorboranes (PNBs)-based antimicrobial polymers possess an amphiphilic structure characterized by having the cationic hydrophilic fragment segregated onto one region (or face) of the macromolecule and the hydrophobic portion, usually constituted by hydrocarbon chains, distinctly onto the opposite face.
In this regard, such polymers are called “facially amphiphilic” (FA) and were synthetized by polymerizing FA norbornene-based monomers, with different protonated groups, such as primary amine, guanidine or pyridine, located on a side alkyl chain, pending from the nitrogen atom of the bicyclic norbornane structure.
The mechanism of action of these polymers, involves as usually, an initial interaction with OM, the creation of pores, the insertion of the biocide into the bacteria cell wall, a second electrostatic interaction with CM, the impairment of its integrity, the progressive increase of its permeability up to its disruption, loss of cytoplasmic material and bacteria death. The type of counterions, the length of alkyl side chains and also the molecular charge density strongly influence the activity and the selectivity of PNBs polymers.
Thanks to their amphiphilic structure, antimicrobial PNBs possess particular ability in inserting and disrupting the CM of bacteria.
Alkyl hydrophobic norbornene-type polymers and the analogous oxanorbornene-based hydrophilic macromolecules, containing primary alkyl ammonium groups as cationic moieties were prepared by Ilker et al. (2004)
[70][70]. The first ones, although very active
versus representatives of Gram-negative bacteria, proved to be not selective for pathogens, thus resulting considerably toxic on mammalian cells, as established by the vesicle-dye leakage assays. On the contrary, the latter were less cytotoxic, but practically inactive
[70][70].
By random copolymerization of two types of alkyl hydrophobic norbornene monomers, it was possible to tune the overall hydrophobicity of the polymer achieving CAPs with high selectivity (>100) and considerable activity against
E. coli (MIC [µg/mL, (µΜ)] = 40, 2.6–3.3).
Similar results were obtained later by Gabriel et al. (2009), for slightly modified oxanorbornene-based hydrophilic polymers that proved to be endowed with low cytotoxicity and good selectivity, but were practically inactive
[71][71].
Later a good solution, in terms of preparing compounds with high antimicrobial activity on bacteria and low hemolytic toxicity on human cells, was to replace the primary ammonium group onto the side alkyl chain with the guanidinium one
[72][72].
In this regard, a polyguanidinium oxanorbornene (PGON) compound was synthesized from norbornene monomers via ring-opening metathesis polymerization (ROMP), which in time killing studies proved to be lethal for bacteria and not only bacteriostatic
[72][72].
A broad library of highly active antimicrobial FA oxanorbornene monomers were prepared and after ROMP and deprotection, provided several series of polynorbornene-derived polymers with tunable activity and selectivity
[73][73]. Polyamine oxanorbornene-based antimicrobial random copolymers, with high hydrophobicity were prepared by performing two different approaches. One strategy involved the copolymerization of two hydrophobic FA monomers with cationic primary ammonium groups on side alkyl chains, while the other consisted in copolymerizing one cationic primary ammonium oxanorbornene monomer and a hydrophobic alkyl amine oxanorbornene comonomer.
By following the second strategy, a series of copolymers endowed with significant antibacterial activity and tunable selectivity were prepared
[71][71].
Amphiphilic polyoxanorbornene-based polymers having different quaternary alkyl pyridinium side chains were synthesized by Eren et al. (2008), but with questionable success
[74][74].
Compounds with a C
4 side chain or shorter proved low antimicrobial activity and low hemolytic toxicity on human red blood cells, while compounds with a side chain longer than C
6 proved high antimicrobial effect, but low selectivity for bacterial over mammalian cells
[74][74].
Polymers Containing not Quaternized Amine Groups
For years, it was thought that fixing permanent cationic charges on polymers by quaternization of amine or phosphorus groups could be the best way to achieve polymers with enhanced antimicrobial effects. To disprove this belief, polymer systems encompassing not quaternary protonated amine groups were synthetized and their antimicrobial activity was evaluated and compared to that of
N-quaternized analogous derivatives.
In this regard, polystyrene-based polymers, containing tertiary amine groups susceptible of reversible protonation, exerted bactericidal activity similar to that of the peptide toxin melittin and somewhat lower activity than that of a potent derivative of the host defense peptide known as magainin II
[75][75].
For clarity, host defense peptide is another broader term to call CAMPs, which takes into account, that small cationic amphipathic peptides have strong potential not only as antimicrobials, but also as antibiofilm agents, immune modulators and anti-inflammatories
[76][76].
The not quaternary compounds, compared to the permanently cationic corresponding
N-quaternized macromolecules, showed far higher antimicrobial activity, suggesting that reversible
N-protonation leads to greater biocidal activity than irreversible
N-quaternization
[75][75].
Unfortunately, protonable amine polymers, not exerting their antibacterial activity by a detergent like membrane disruption mechanism, lacked the selectivity of magainin II and showed high hemolytic toxicity, mimicking the not selective melittin behavior
[75][75].
Amphiphilic methacrylamide random copolymers, bearing reversibly protonated primary or tertiary amine groups and encompassing a hydrocarbon hydrophobic side chains, were prepared and their antimicrobial and hemolytic properties were compared with those of similar macromolecules, containing quaternary ammonium groups
[77][77].
The not quaternized copolymers owing the primary amine groups proved considerable antimicrobial activity on
E. coli by a membrane-disrupting action
[77] [