1. Dietary NO3− and Endothelial Dysfunction Therapy
Until a decade ago, NO
3− was considered an unfavorable dietary-derived toxic compound, as it was wrongly associated with the development of some malignancies, such as gastric cancer. Strict standards regarding the levels of this anion were regulated in food [1]. The World Health Organization (WHO) defined an acceptable daily intake (ADI) of 3.7 mg of NO
was considered an unfavorable dietary-derived toxic compound, as it was wrongly associated with the development of some malignancies, such as gastric cancer. Strict standards regarding the levels of this anion were regulated in food [26]. The World Health Organization (WHO) defined an acceptable daily intake (ADI) of 3.7 mg of NO 3−
·kg
−1
body weight, the same content adopted by the European Food Safety Authority. For a normal adult weighing 70 kg, this content is equivalent to ~260 mg of NO
3−
·day
−1
. However, vegetarian diets commonly contain >300 mg of NO
3−
·day
−1 for 70 kg adults, higher than the ADI [2].
for 70 kg adults, higher than the ADI [27].
Recently, researchers have become interested in the biological NO
3−
role. Findings regarding the improvement of cardiovascular function have raised a biologically plausible and widely recognized hypothesis that the NO
3− present in vegetables may serve as a physiological substrate for NO generation which, in turn, promotes vasodilation and, consequently, improves cardiovascular function [3][4].
present in vegetables may serve as a physiological substrate for NO generation which, in turn, promotes vasodilation and, consequently, improves cardiovascular function [8,13].
NO
3−
is a nitric acid salt, while NO
2−
is a nitrous acid salt compound, formed by a single nitrogen bonded to three or two oxygen atoms, respectively. Both compounds can be obtained from endogenous and/or exogenous sources. The endogenous formation of NO
3−
and NO
2−
occurs through the NO metabolism via the L-arginine/NO pathway, as mentioned previously. On the other hand, the main potential exogenous source for the acquisition of NO
3−
and NO
2−
is through the dietary route. Through this pathway, NO is then generated by a non-enzymatic pathway from NO
2−
. Dietary NO
3−
is reduced to NO
2−
in the oral cavity by bacteria that produce the NO
3−-reductase enzyme [5]. The metabolic activities of commensal bacteria species, such as
-reductase enzyme [9]. The metabolic activities of commensal bacteria species, such as Granulicatella
,
Actinomyces
,
Veillonella
,
Prevotella
,
Neisseria
,
Haemophilus
, and
Rothia
genera that inhabit the oral cavity have a significant influence on NO
3−
to NO pathway. Previous studies have shown that individuals with a higher abundance of NO
3−
-reducing bacteria are able to generate more salivary NO
2−
and, consequently, NO, at a faster rate following dietary NO
3− ingestion [6]. However, enzymatic activity in the mouth and, consequently, the conversion of NO
ingestion [28]. However, enzymatic activity in the mouth and, consequently, the conversion of NO 3−
to NO
2− may be disrupted by antibiotic use or mouthwash rinsing, since both substances inactivate bacteria cells [7]. Subsequently, NO
may be disrupted by antibiotic use or mouthwash rinsing, since both substances inactivate bacteria cells [10]. Subsequently, NO 2−
reaches the stomach and, in this acidic environment, is protonated, forming nitrous acid (HNO
2
), which decomposes non-enzymatically to NO and other bioactive nitrogen oxides such as nitrogen dioxide (NO
2
), dinitrogen trioxide (N
2
O
3
) and the nitrosonium ion (NO
+) [5][4]. The remaining NO
) [9,13]. The remaining NO 3−
and NO
2−
in the jejunum are rapidly absorbed into the bloodstream or tissues, where their accumulation occurs in tandem with molecules endogenously synthesized by the L-arginine/NO pathway. Most NO
3− is excreted in urine, whereas a small portion is extracted by the salivary glands, concentrating this compound in the saliva, continuing the entero-salivary cycle [3][5]. A small part of plasma NO
is excreted in urine, whereas a small portion is extracted by the salivary glands, concentrating this compound in the saliva, continuing the entero-salivary cycle [8,9]. A small part of plasma NO 3−
and NO
2−
concentrations may suffer the action of xanthine oxidoreductase (XOR), which displays similar activity to NO
3−
-reductase. NO
2−
can also be reduced to bioactive NO by deoxyhemoglobin (deoxyHb) and deoxymyoglobin (deoxyMb), especially when O
2
levels are low. Other enzymes and compounds exhibiting redox potential, such as aldehyde oxidase (AO), aldehyde dehydrogenase (ALDH), carbonic anhydrase (CA), vitamin C (Vit C.) and polyphenols, display the ability to synthesize NO from NO
2− reduction [3].
Several studies report beneficial effects of dietary NO
3− sources as a new physiological, therapeutic and nutritional approach to attain the intended cardioprotective effects by NO production stimulation [3][8][9]. However, dosage, supplementation regimen and individual health status must be considered to obtain the maximum cardioprotective effect following NO
sources as a new physiological, therapeutic and nutritional approach to attain the intended cardioprotective effects by NO production stimulation [8,29,30]. However, dosage, supplementation regimen and individual health status must be considered to obtain the maximum cardioprotective effect following NO 3−
intake. Furthermore, environmental factors such as temperature, exposure to sunlight, atmospheric humidity, water content and irradiation, as well as agricultural factors like plant genotype, fertilization, herbicide use, amount of available nitrogen, type of crop, soil conditions, nutrient availability and transport and, finally, storage conditions also influence NO
3−
contents in plants, and, consequently dietary NO
3− supplementation [10].
2. Dietary NO3− Vegetable Sources
Vegetables are the main source of dietary NO
3−
, corresponding to 85% of the daily intake, although NO
3− content can vary widely within members of distinct botanical families [11]. The NO
content can vary widely within members of distinct botanical families [32]. The NO 3− contents in plant organs can be classified from highest to lowest, as petiole ˃ leaf ˃ stem ˃ root ˃ tuber ˃ bulb ˃ fruit ˃ seed [12].
contents in plant organs can be classified from highest to lowest, as petiole ˃ leaf ˃ stem ˃ root ˃ tuber ˃ bulb ˃ fruit ˃ seed [33]. presents a list of vegetables commonly included in Western diets considered NO
3−
sources, classified according to NO
3−
contents, from the highest to the lowest.
Table 1.
Dietary NO
3−
sources classified from the highest to the lowest according to mean [and range] NO
3−
content.
The NO
3−
-rich vegetables within the
Amaranthaceae
family comprise beetroot (1300 mg·kg
−1
), beet greens (1852 mg·kg
−1
), Swiss chard (1690 mg·kg
−1
), and green spinach (≈2500 mg·kg
−1
), while a
Lamiaceae
family representative consists of basil (2292 mg·kg
−1
). Concerning the
Brassicaceae
family, the most representative members are bok choy (1933 mg·kg
−1
), black radish (1271 mg·kg
−1
), turnip (1018 mg·kg
−1
), mustard greens (1160 mg·kg
−1
), rocket or arugula (4677 mg·kg
−1
), kohlrabi (1769 mg·kg
−1
), and radish (≈2000 mg·kg
−1
).
Apiaceae
family members include coriander (2445 mg·kg
−1
), celery (1100 mg·kg
−1
) and parsley (2134 mg·kg
−1
), whereas
Asteraceae
family members include lettuce (≈1800 mg·kg
−1
), leaf chicory (1452 mg·kg
−1
), and butter leaf lettuce (2000 mg·kg
−1
). All these vegetables are included in the high NO
3−
-containing vegetable category of > 1000 mg·kg
−1
. Vegetables such as cabbage (513 mg·kg
−1
), curly kale (987 mg·kg
−1
), broccoli (≈300 mg·kg
−1
), broccoli raab (905 mg·kg
−1
), cauliflower (202 mg·kg
−1
) and Savoy cabbage (324 mg·kg
−1
), which belong to the
Brassicaceae
family; carrot (≈300 mg·kg
−1
) and fennel (363 mg·kg
−1
), both members of the
Apiaceae
family; artichokes (174 mg·kg
−1
), asparagus chicory (355 mg·kg
−1
), and endive (663 mg·kg
−1
), belonging to the
Asteraceae
family, garlic (183 mg·kg
−1
) and green onion (≈450 mg·kg
−1
) from the
Liliaceae
family; aubergine (314 mg·kg
−1
), capsicum (108 mg·kg
−1
) and potato (220 mg·kg
−1
), belonging
Solanaceae
family; courgette (416 mg·kg
−1
) and cucumber (240 mg·kg
−1
), pumpkin (894 mg·kg
−1
), from the
Cucurbitaceae
family member all contain intermediate NO
3−
concentrations ranging from 100 to 1000 mg·kg
−1 [12][13][14][15].
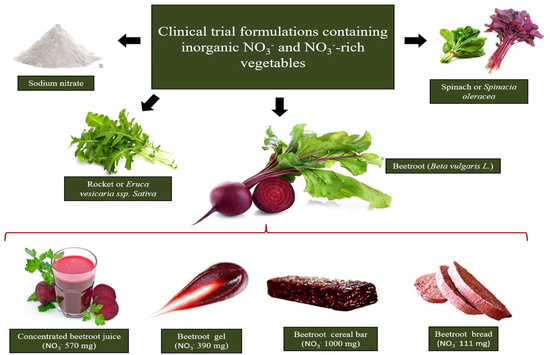
Among the vegetables considered the richest dietary NO
3−
sources, as listed in
, beetroot, rocket and spinach have been the most tested concerning dietary interventions, and all resulted in effective improvements in cardiovascular performance estimated through blood pressure reduction and vascular function amelioration (
).
Figure 1.
The richest sources of dietary NO
3−
tested in clinical interventions are beetroot, rocket and spinach. Beetroot formulation choice to supplement dietary NO
3−
relies on the design of beetroot-derived formulations containing pharmacological NO
3−
doses in a small serving portion.
NO
3−
vascular-effects depend on digestibility and bioavailability (bioacessibility), and better performances are obtained when NO
3−
intake originates from food matrices compared to NaNO
3− salt administration [16]. The beneficial effects of different NO
salt administration [37]. The beneficial effects of different NO 3−
-rich vegetables and NO
3−
doses in NO stimulation production and biochemical, hemodynamic, and vascular parameters in healthy or cardiovascular-compromised patients are summarized in
. It is important to note that, to the best of our knowledge, NO
3−
supplementation from green leaves has only been performed in healthy individuals, and it is unknown whether their effects can be extended to individuals presenting cardiovascular risk factors. In addition, although the cardiovascular protective effects of NO
3−
-enriched vegetables have been clearly demonstrated in clinical trials with healthy individuals, the large volume of juice vegetables used to achieve effective dietary NO
3−
concentrations can be a limiting factor in ensuring adherence to long-term nutritional interventions. However, this NO
3−
limitation does not impact supplementation by beetroot juice. Beet juice and other beetroot formulations can be ingested in comfortable serving portions to achieve threshold NO
3− concentrations in order to promote beneficial cardiovascular function effects.
concentrations in order to promote beneficial cardiovascular function effects.
Table 2.
Selected clinical trials from 2012 to 2020 compared considering administered NO
3−
content, intervention duration, level of systemic increase in NO evaluated by plasma NO
3−
and NO
2−
levels and improvements in primary and advanced hemodynamic parameters in healthy individuals and in patients presenting impaired vascular function.
A large volume of spinach comprising a serving portion of 250 g leaves containing 220 mg of NO
3−
were administrated to twenty-six healthy individuals, resulting in an increase in NO synthesis evidenced by an eight-fold increase in salivary NO
2−
and a seven-fold increase in salivary NO
3− at 120 min post-meal. Large artery elasticity indices were increased alongside lower pulse pressure and reduced systolic blood pressure (SBP) [25].
at 120 min post-meal. Large artery elasticity indices were increased alongside lower pulse pressure and reduced systolic blood pressure (SBP) [46].
An amount of 800 mg NO
3−
intake was supplied through four different vegetable drinks, namely beetroot juice (116 g), rocket salad (196 g), spinach (365 g) or NaNO
3−
(1.1 g) prepared in water, which triggered an increase in NO
3−
and NO
2− plasma concentrations. SBP declined after 150 min of beetroot juice ingestion (from 118 ± 2 to 113 ± 2 mm Hg) and a rocket salad beverage (from 122 ± 3 to 116 ± 2 mm Hg), which was sustained for at least 300 min after ingestion of the spinach beverage (from 118 ± 2 to 111 ± 3 mm Hg). Diastolic blood pressure (DBP) also declined after 150 min ingestion of all beverages and was sustained at lower levels for 300 min after rocket salad or spinach ingestion [27].
plasma concentrations. SBP declined after 150 min of beetroot juice ingestion (from 118 ± 2 to 113 ± 2 mm Hg) and a rocket salad beverage (from 122 ± 3 to 116 ± 2 mm Hg), which was sustained for at least 300 min after ingestion of the spinach beverage (from 118 ± 2 to 111 ± 3 mm Hg). Diastolic blood pressure (DBP) also declined after 150 min ingestion of all beverages and was sustained at lower levels for 300 min after rocket salad or spinach ingestion [48].
All NO
3−
rich-vegetable drinks were more efficient than NaNO
3−
in reducing both SBP and DBP, and beetroot was the most effective considering the food weight/NO
3− content ratio [27]. However, to the best of our knowledge, the lowest effective volume of beetroot able to promote beneficial vascular effects was 70 mL of beetroot juice containing 6.45 mmol NO
content ratio [48]. However, to the best of our knowledge, the lowest effective volume of beetroot able to promote beneficial vascular effects was 70 mL of beetroot juice containing 6.45 mmol NO 3−
(403 mg), which was administered to 24 older and overweight volunteers for three weeks. This supplementation regimen and the offered dose promoted 2.3-fold and 7.3-fold increases in urinary and salivary NO
3−, respectively, and resulted in a 7.3 mm Hg decrease in SBP [29].
, respectively, and resulted in a 7.3 mm Hg decrease in SBP [50].
Beetroot consumption is noteworthy as a convenient and attractive alternative to obtain cardioprotective NO
3−
effects in both healthy individuals and those presenting risk factors for CVD diseases, due to the distinct but smart formulations (traditional or novel) that can be prepared to fulfill effective pharmacological dietary NO
3−
concentrations. An attractive and compact NO
3−
-enriched-beetroot gel has been formulated in an attempt to provide an enriched NO
3− food product able to promote the claimed cardioprotective effects while still being easy to administer and facilitate adherence to nutritional therapy [23]. Acute supplementation with 100 g of beetroot gel containing 390 mg of NO
food product able to promote the claimed cardioprotective effects while still being easy to administer and facilitate adherence to nutritional therapy [44]. Acute supplementation with 100 g of beetroot gel containing 390 mg of NO 3−
promoted a decrease in SBP (−6.2 mm Hg), DBP (−5.2 mm Hg), and heart rate (−7 bpm) in a pilot study conducted with healthy individuals. However, NO
3−
supplementation had to be adjusted to treat hypertensive individuals, since similar doses in compromised vascular individuals do not alter hemodynamic parameters. The non-susceptibility of 27 treated hypertensive patients was clearly demonstrated by the intake of 7.0 mmol (434 mg) of NO
3−
in 140 mL of beetroot juice for 7 days, resulting in increased NO synthesis, assessed by plasmatic, urinary and salivary NO
3−
and NO
2−, but with no differences in home and 24 h ambulatory, SBP and DBP [26]. These results indicate that, in order to ameliorate primary hemodynamic parameters, high doses of dietary NO
, but with no differences in home and 24 h ambulatory, SBP and DBP [47]. These results indicate that, in order to ameliorate primary hemodynamic parameters, high doses of dietary NO 3−
combined with a long-term intervention can be applied to treat individuals presenting impaired endothelial function. Furthermore, in an unprecedented clinical trial, patients displaying at least three risk factors for the development of CVD, including hypertension, were chronically supplemented for three weeks with an enriched NO
3−
beetroot-cereal bar providing 589 mg of NO
3−
in 60 g of the intervention product, resulting in 14.0 mm Hg and 6.5 mm Hg decreases in SBP and DBP, respectively, in response to ~15-fold or ~7-fold increased plasma NO
3−
and NO
2− concentrations, respectively. Endothelial function in the treated volunteers was improved and arterial stiffness was reduced by 14% [24][30].
concentrations, respectively. Endothelial function in the treated volunteers was improved and arterial stiffness was reduced by 14% [45,51].
3. Plasma NO3−/NO2− Increments on Cardiovascular Health and Impaired Cardiovascular Functions
It is known that plasma NO
3−
and NO
2−
concentrations are dependent on ingested NO
3− [31], but the minimum increase in NO
[52], but the minimum increase in NO 3−
/NO
2−
plasma levels necessary to promote hemodynamic responses may differ between healthy individuals and those with compromised cardiovascular function. In a clinical trial where healthy men received dietary supplementation, 3.5-fold and 1.6-fold increases of plasma NO
3−
and NO
2−
, respectively, resulted in significant DBP reductions and increases in endothelium-independent vasodilatation. This small but effective plasma increase was generated after the acute intake of beetroot bread (NO
3− 1.1 mmol) [18]. On the other hand, Haun et al. [28] reported plasma NO
1.1 mmol) [39]. On the other hand, Haun et al. [49] reported plasma NO x
(~3-fold) and NO
2−
(less than 1.5-fold) increases, albeit without any changes in hemodynamic parameters such as heart rate, DBP, SBP, FMD, radial artery pulse waves (PWA), central mean arterial pressure (CMAP) and central pulse pressure (CPP), after the acute intake of red spinach extract (NO
3− 1.45 mmol) by 15 healthy subjects. Although the dose used by Haun et al. [28] was slightly higher than by Hobbs et al. [18] trial, plasma NO
1.45 mmol) by 15 healthy subjects. Although the dose used by Haun et al. [49] was slightly higher than by Hobbs et al. [39] trial, plasma NO 3−
(>3.5-fold) and NO
2−
(>1.6-fold) increases should be a determinant factor in choosing the dose required to benefit healthy populations.
In individuals with impaired cardiovascular function, the administered NO
3−
dose should be able to meet two requirements: (i) promote an increase in systemic NO
3−
and NO
2−
higher than observed in healthy individuals; (ii) be administered in a chronic and uninterrupted manner.
Hypertensive pregnant women exhibited ~10- and ~1.5-fold increases in plasma NO
3−
and NO
2−
, respectively, after 7 days of daily supplementation with NO
3−
(6.45 mmol in beetroot juice). No significant differences were observed in plasma NO
3−
and NO
2− levels measured 24 h after the initial dose, and even in the following 7-days of daily supplementation [32]. Similarly, a 1-week intake of beetroot juice (NO
levels measured 24 h after the initial dose, and even in the following 7-days of daily supplementation [53]. Similarly, a 1-week intake of beetroot juice (NO 3−
~6.45 mmol) in 27 treated hypertensive individuals resulted in a three-fold increase in plasma NO
3−
and NO
2−, with no differences in home and 24-h ambulatory blood pressures [33]. Finally, 24 overweight older subjects supplemented for 3 weeks with concentrated beet juice (~4.8–6.45 mmol) exhibited urinary NO
, with no differences in home and 24-h ambulatory blood pressures [54]. Finally, 24 overweight older subjects supplemented for 3 weeks with concentrated beet juice (~4.8–6.45 mmol) exhibited urinary NO 3−
values ~3-fold higher greater than the baseline and beneficial SBP effects after juice intake. However, both urinary NO
3− and SBP returned to baseline levels 24 h after ingestion and in the first week following the end of supplementation [19]. These findings demonstrate that acute treatments able to promote systemic increases in NO
and SBP returned to baseline levels 24 h after ingestion and in the first week following the end of supplementation [40]. These findings demonstrate that acute treatments able to promote systemic increases in NO 3−
and NO
2−
at levels similar to those observed in healthy individuals do not benefit individuals presenting cardiovascular risks.
On the other hand, clinical trials lasting more than 3 weeks or comprising higher NO
3−
doses than usually applied (6–7 mmol) resulted in better hemodynamic outcomes. Hypertensive subjects treated for 4 weeks with beetroot juice (NO
3−
6.4 mmol) exhibited substantial increases in NO
3−
and NO
2− plasma levels (~5.5 and ~2.7, respectively). In addition, this intervention provided sustained BP lowering of 7.7/5.2 mm Hg 24 h after the treatment, with clinical BP reduced by 7.7/2.4 mm Hg and home BP, by 8.1/3.8 mm Hg [20]. In this trial, SBP and DBP reduction peaks occurred only in the last week, highlighting the importance of a prolonged intervention.
plasma levels (~5.5 and ~2.7, respectively). In addition, this intervention provided sustained BP lowering of 7.7/5.2 mm Hg 24 h after the treatment, with clinical BP reduced by 7.7/2.4 mm Hg and home BP, by 8.1/3.8 mm Hg [41]. In this trial, SBP and DBP reduction peaks occurred only in the last week, highlighting the importance of a prolonged intervention.
In another trial, supplementation for 3 weeks with a high dose of dietary NO
3−
concentrate in a 60 g beetroot cereal bar (9.5 mmol) resulted in ~15- and ~7-fold increases in plasma NO
3−
and NO
2−, respectively. This was accompanied by a considerable reduction in BP (−14.0/−6.5 mm Hg) and improvement in central hemodynamic and endothelial function parameters such as arterial stiffness, augmentation and index pressures, aortic systolic and pulse pressures and cutaneous microvascular conductance [24].
, respectively. This was accompanied by a considerable reduction in BP (−14.0/−6.5 mm Hg) and improvement in central hemodynamic and endothelial function parameters such as arterial stiffness, augmentation and index pressures, aortic systolic and pulse pressures and cutaneous microvascular conductance [45].
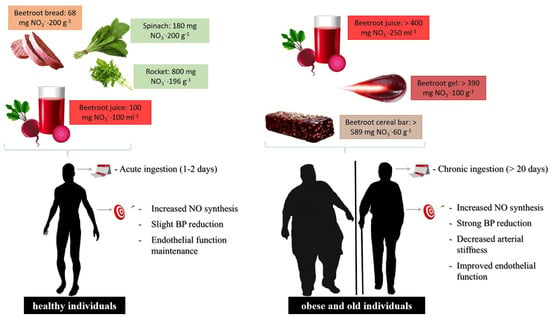
Based on these reports, individuals presenting physiopathological conditions that affect the cardiovascular system require a dietary therapy that associates high NO
3−
doses capable of promoting systemic increases in NO
3−
and NO
2−
to levels higher than found in healthy individuals and in addition, is administered continuously (
).
Figure 2.
Food formulations and supplementation regimen of dietary NO
3−
in healthy or cardiovascular-compromised patients. For individuals presenting risk factors for the development of cardiovascular disease, the dietary NO
3−
dose should be higher to promote the systemic elevation of plasma NO
3−
and NO
2−
levels compared to healthy individuals, increasing NO generation by the NO
3−
/NO
2−
pathway, where increased levels must be administered through chronic and uninterrupted supplementation.
In short, the aforementioned studies discussed herein suggest that frequent daily dietary NO
3−
doses for long periods of time would be necessary to promote beneficial effects on blood pressure and endothelial function in populations presenting compromised vascular responsiveness. A systematic review and meta-analysis study of randomized controlled trials demonstrated that supplementation of inorganic NO
3−
from beetroot juice over 14 days provoked decreases in SBP (−3.55 mm Hg; 95% CI: −4.55, −2.54 mm Hg) and DBP (−1.32 mm Hg; 95% CI: −1.97, −0.68 mm Hg). Furthermore, beneficial dietary NO
3−
effects on endothelial function were associated with dose, age, and body mass index (BMI), where chronic beetroot juice supplementation improved flow-mediated dilation (FMD) and endothelium functional effects according to the administered NO
3−
contents (β = 0.04, SE = 0.01,
p
< 0.001), age (β = −0.01, SE = 0.004,
p
= 0.02) and BMI (β = −0.04, SE = 0.02,
p = 0.05) [34]. A critical review of experimental data shows that chronic dietary NO
= 0.05) [55]. A critical review of experimental data shows that chronic dietary NO 3−
ingestion is a positive vascular endothelium effector promoting vasodilatation and reducing blood pressure in compromised vascular responsiveness individuals.
However, only beetroot supplementation has been tested in acute and chronic assays in individuals with impaired cardiovascular function. Although the NO
3−
content of green leaves is able to fulfill the effective NO
3−
concentration in such patients, beetroot formulations may be the best non-drug strategy, since beetroot-derived formulations can concentrate the pharmacological NO
3−
dosage in a small serving portion of an attractive food product, favoring continuous intake and better adherence to this nutritional intervention. This may explain the well-documented and consistent cardioprotective effects of beet products in both healthy individuals and those presenting risk factors for the development of CVD when compared with other rich-NO
3−
vegetables, such as green leaves, assayed in clinical trials.
Furthermore, it is important to note that, in addition to NO
3, vegetables are also a source of numerous phytochemicals able to increase eNOS activity in endothelial cells and contribute to NO synthesis [35][36]. Due to the great variety of polyphenols and other bioactive compounds in vegetables, it is difficult to point out individual or synergistic effects on NO generation. However, only NO
, vegetables are also a source of numerous phytochemicals able to increase eNOS activity in endothelial cells and contribute to NO synthesis [56,57]. Due to the great variety of polyphenols and other bioactive compounds in vegetables, it is difficult to point out individual or synergistic effects on NO generation. However, only NO 3−
has been directly associated to the cardioprotective effect, since it provides the physiological substrate for NO generation via the NO
3−
-NO
2−/NO enterosalivary pathway [3]. The administration of the same food matrix, depleted in NO
/NO enterosalivary pathway [8]. The administration of the same food matrix, depleted in NO 3−
, used as a placebo in the clinical trials had no effect on NO synthesis and hemodynamic parameters, proving that NO
3−
is probably the active principle. The remaining phytochemicals after NO
3−
removal, including polyphenols, which are preserved in the placebo, may promote a discrete increase in NO production but it seems they are not effective in promoting hemodynamic improvements, similar to the effect observed when NO
3− at concentrations under the pharmacological threshold is administered.
at concentrations under the pharmacological threshold is administered.
4. Conclusions
Vegetables are important health-promoting foods in a balanced diet, due to the presence of bioactive compounds, including dietary NO
3−
. Vegetables that belong to the green leaf group, such as rocket, green spinach, basil, radish, Swiss chard and bok choy, in addition to red beetroot, are considered the richest sources of dietary NO
3−
. Increasing dietary NO
3−
ingestion results in beneficial effects in many physiological and clinical settings. Several clinical interventions with different NO
3−
-rich vegetables have been reported as affecting metabolic and cardiovascular functions by increasing NO concentrations and improving endothelial function by reducing BP and arterial stiffness. However, minimal or no hemodynamic and vascular beneficial effects in healthy individuals have been observed following acute NO
3−
ingestion. To obtain the maximum cardioprotective effects of NO
3−
intake, patient health status, as well as NO
3−
dosage and supplementation regimen, must be considered.
The aforementioned studies suggest that frequent daily doses up to 6.0 mmol of dietary NO
3−
for long periods of time (≥3 weeks) are required to promote beneficial blood pressure and endothelial function effects, mainly in populations with compromised vascular responsiveness such as hypertensive, metabolic syndrome, obese and older individuals.
Only beetroot supplementation has been tested in acute and chronic assays in individuals with impaired cardiovascular function. Although the NO
3−
content of green leaves or other vegetables could fulfill the effective NO
3−
concentration in healthy individuals, patients with impaired vascular function require a higher dose able to provide systemic increases in NO
3−
and NO
2−
to levels higher than those achieved in healthy individuals. Beet formulations are easier, attractive, accessible and were the only vegetable shown to be effective in promoting increased systemic NO production at the magnitude necessary to achieve the expected pharmacological effects in individuals presenting cardiovascular disease risk factors.