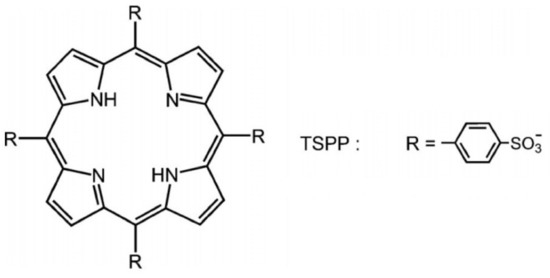
Photodynamic inactivation is known as a new antimicrobial photodynamic therapy (aPDT). It is based on the administration of a photosensitizer located in the bacterial/viral cell followed by exposure to light radiations (with a proper wavelength corresponding with the maximum value of absorption of the photosensitizer) that generate singlet oxygen or reactive oxygen species, which lead to the death of different microorganisms. This review will present an overview beyond the state-of-the-art of the photosensitizer types (based on tetra-p-sulphonated-phenyl porphyrin—TSPP, which is able to form cationic and J-aggregates forms at different pH values ((1–4) and concentrations around 10−5 M) and their applications of PDT for viruses, especially.
- aPDT
- photodynamic inactivation
- TSPP
1. Antimicrobial photodynamic therapy (aPDT)
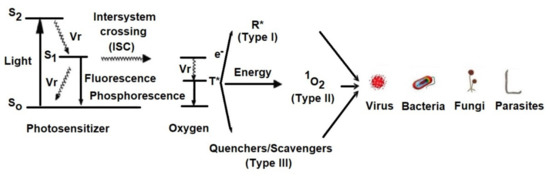
2. Mechanism of aPDT
-
Light excites the ground state photosensitizer to an excited singlet state.
-
The formation of triplets of excited sensitizer molecule (intersystem crossing).
-
However, this state is short lived and can decay to the ground state by radiative or non-radiative transition directly emitting light as fluorescence.
-
From this excited state, the photosensitive substance can then return to the ground state by phosphorescence.
-
The triplet excited state of the photosensitizer is able to react with oxygen in its triplet state, generating singlet oxygen (type II reaction) or initiating free radical chain reactions with superoxide and hydrogen peroxide ions as well as hydroxyl radicals (type I reaction).
2.1. Type I Mechanism
-
hydrogen transfer: 3S * + RH → SH + R˙
-
electron transfer: 3S * + RH → S− + RH+
-
formation of hydrogen dioxide
-
formation of superoxide anion
2.2. Type II Mechanism
-
intermolecular exchange
-
cellular oxidation
3. Light Sources
4. Photosensitizers Used for Photodynamic Inactivation of Microorganisms
| Photosensitizer | λabs (nm) | |
|---|---|---|
| Psoralen | 300–380 | |
| Acridine | 400–500 | |
| Cyanine | ||
| J-aggregate | 500–600 | |
| 422; 490; 707 | Porphyrin | 600–650 |
| H-aggregate | 401; 517; 552; 593; 650Perylenequinonoid | 600–650 |
| Phenothiazinium (methylene blue, toluidine blue O) |
620–660 | |
| Phthalocyanine | 660–700 | |
| Crystal violet | 550–610 | |
| Rose Bengal | 450–650 | |
| Neutral Red | 460–550 | |
| Congo Red | 400–560 | |
| Riboflavin | 300–600 | |
| Eosin B | 514–544 |
-
hydrophobic photosensitizers without peripheral substituents with electric charge and being slightly soluble in water or alcohol (phthalocyanines and naphthalocyanines, hematoporphyrin, hematoporphyrin derivative (HpD), porfimer sodium, and porphyrin precursors)
-
hydrophilic photosensitizers that contain three or more peripheral substituents with electric charge and have a high solubility in water at physiological pH.
-
amphiphilic photosensitizers that contain one or two peripheral substituents with electric charges, soluble in water or alcohol, at physiological pH. In their structure, there are always two regions, one hydrophobic (represented by porphyrin with electrically charged groups) and another hydrophilic [21].
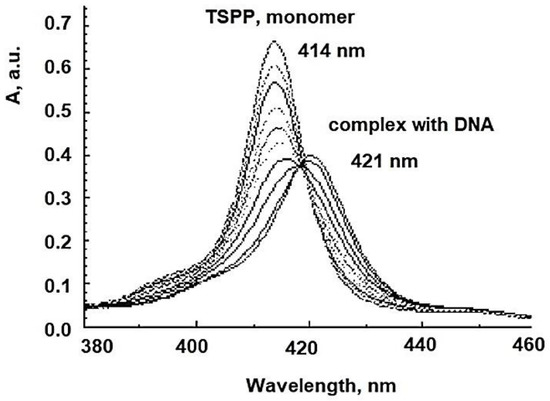
5. Anionic Photosensitizers as Anti-Viral Agent for aPDT
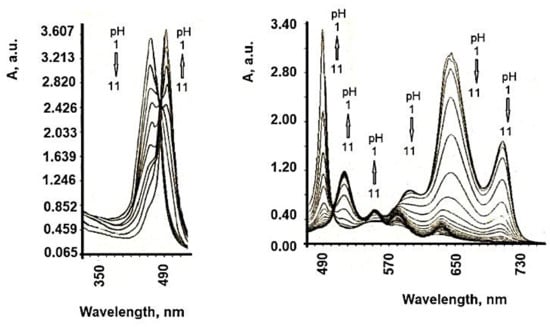
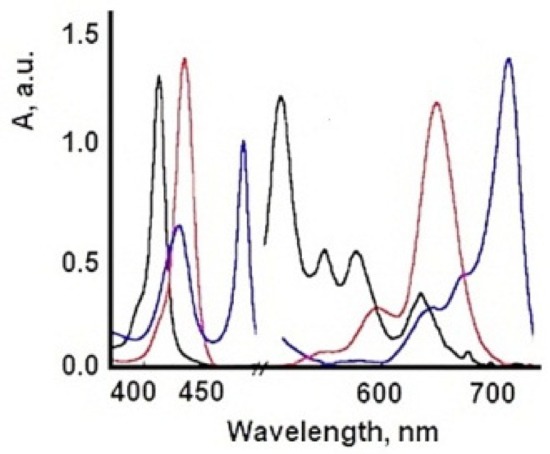
| TSPP Form | Absorption Bands (nm) |
|---|---|
| neutral | 412; 515; 551; 579; 633 |
| dication | 433; 550; 594; 644 |
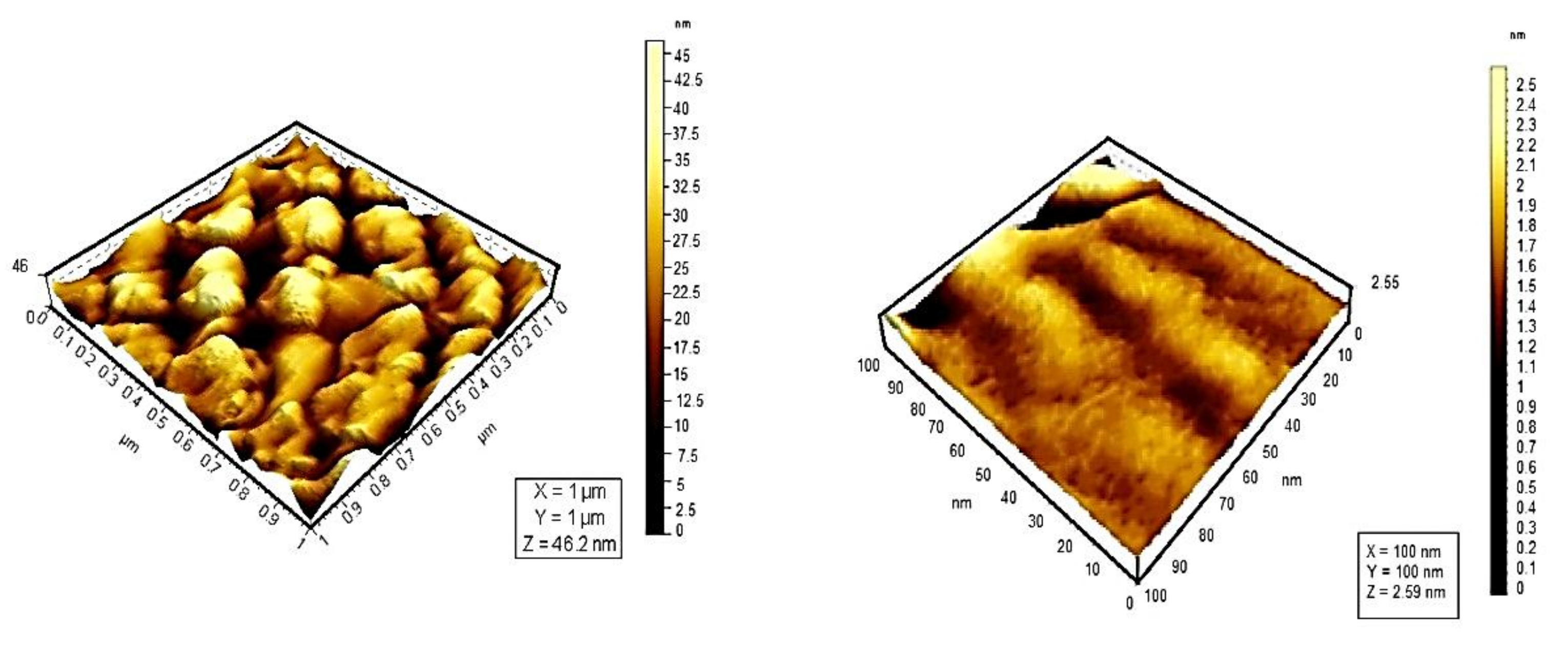
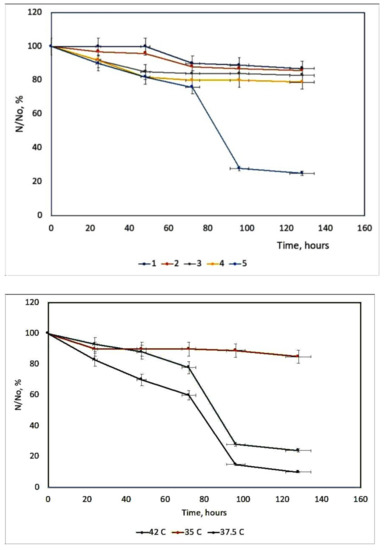
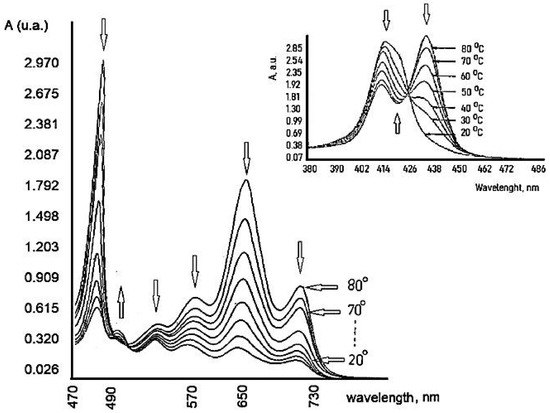
6. Influence of Dicationic (J-Aggregates) TSPP form on aPDT
References
- Nicholas, J.L. Photo(chemo)therapy: General principles. Clin. Dermatol. 1997, 15, 745–752.
- Ion, R.M. The photodynamic therapy of cancer-a photosensitisation or a photocatalytic process? Progr. Catal. 1997, 1, 55–62.
- Evensen, J.F.; Moan, J.; Winkelman, J.W. Toxic and phototoxic effects of tetraphenylporphine sulphonate and haematoporphyrin derivative in vitro. Int. J. Radiat. Biol. Relat. Stud. Phys. Chem. Med. 1987, 51, 477–491.
- Qian, P.; Evensen, J.F.; Rimington, C.; Moan, J. A comparison of different photosensitizing dyes with respect to uptake C3H-tumors and tissues of mice. Cancer Lett. 1987, 36, 1–10.
- Boda, D.; Neagu, M.; Constantin, C.; Diaconeasa, A.; Ianosi, S.; Ion, R.M.; Amalinei, C.; Stanoiu, B.; Crauciuc, E.; Toma, O. New photosensitizers versus aminolevulinic acid (ala) in experimental photodynamic therapy of actinic keratosis—A case report. Ann. Alexandru Ioan Cuza Univ. Sect. Genet. Mol. Biol. 2009, 3, 62–69.
- Winkelman, J.W.; Collins, G.H. Neurotoxicity of tetraphenylporphinesulfonate TPPS4 and its relation to photodynamic therapy. Photochem. Photobiol. 1987, 46, 801–807.
- Streleckova, E.; Kodetova, D.; Pouckova, P.; Zadinova, M.; Lukas, E.; Rokyta, R.; Jirsa, M. Meso-tetra-(4-sulfonatophenyl)-porphine of low neurotoxicity. SB Lek. 1995, 96, 7–13.
- Huang, L.; El-Hussein, A.; Xuan, W.; Hamblin, M.R. Potentiation by potassium iodide reveals that the anionic porphyrin TPPS4 is a surprisingly effective photosensitizer for antimicrobial photodynamic inactivation. J. Photochem. Photobiol. B Biol. 2018, 178, 277–286.
- Kato, H.; Komagoe, K.; Inoue, T.; Masuda, K.; Katsu, T. Structure–activity relationship of porphyrin-induced photoinactivation with membrane function in bacteria and erythrocytes. Photochem. Photobiol. Sci. 2018, 17, 954–963.
- Wainwright, M. Photodynamic antimicrobial chemotherapy (PACT). J. Antimicrob. Chemother. 1998, 42, 13–28.
- Bertoloni, G.; Lauro, F.M.; Cortella, G.; Merchat, M. Photosensitizing activity of hematoporphyrin on Staphylococcus aureus cells. Biochim. Biophys. Acta Gen. Subj. 2000, 1475, 169–174.
- Ion, R.M.; Planner, A.; Wiktorowicz, K.; Frackowiak, D. Incorporation of various porphyrins into human blood cells measured using the flow-cytometry, the absorption and emission spectroscopy. Acta Biochim. Pol. 1998, 45, 833–842.
- Salva, K.A. Photodynamic therapy: Unapproved uses, dosages, or indications. Clin. Derm. 2002, 20, 571–581.
- Juzeniene, A.; Juzenas, P.; Ma, L.-W.; Iani, V.; Moan, J. Effectiveness of different light sources for 5-aminolevulinic acid photodynamic therapy. Lasers Med. Sci. 2004, 19, 139–149.
- Koshi, E.; Mohan, A.; Rajesh, S.; Philip, K. Antimicrobial photodynamic therapy: An overview. J. Indian Soc. Periodontol. 2011, 15, 323–327.
- Kashef, N.; Huang, Y.-Y.; Hamblin, M.R. Advances in antimicrobial photodynamic inactivation at the nanoscale. Nanophotonics 2017, 6, 853–879.
- Hu, X.; Huang, Y.-Y.; Wang, Y.; Wang, X.; Hamblin, M.R. Antimicrobial photodynamic therapy to control clinically relevant biofilm infections. Front. Microbiol. 2018, 9, 1299.
- Kuo, W.-S.; Chang, C.-Y.; Chen, H.-H.; Hsu, C.-L.L.; Wang, J.-Y.; Kao, H.-F.; Chou, L.C.-S.; Chen, Y.-C.; Chen, S.-J.; Chang, W.-T.; et al. Two-photon photoexcited photodynamic therapy and contrast agent with antimicrobial graphene quantum dots. ACS Appl. Mater. Interfaces 2016, 8, 30467–30474.
- Wozniak, A.; Grinholc, M. Combined antimicrobial activity of photodynamic inactivation and antimicrobials–state of the art. Front. Microbiol. 2018, 9, 930.
- Bartolomeu, M.; Coimbra, S.; Cunha, A.; Neves, M.G.P.M.S.; Cavaleiro, J.A.S.; Faustino, M.A.F.; Almeida, A. Indirect and direct damage to genomic DNA induced by 5,10,15-tris(1-methylpyridinium-4-yl)-20-(pentafluorophenyl)porphyrin upon photody-namic action. J. Porph. Phthal. 2016, 20, 331–336.
- Wainwright, M.; McLean, A. Rational design of phenothiazinium derivatives and photoantimicrobial drug discovery. Dyes Pigments 2017, 136, 590–600.
- Yao, T.-T.; Wang, J.; Xue, Y.-F.; Yu, W.-J.; Gao, Q.; Ferreira, L.; Ren, K.-F.; Ji, J. A photodynamic antibacterial spray-coating based on the host–guest immobilization of the photosensitizer methylene blue. J. Mater. Chem. B 2019, 7, 5089–5095.
- Friedman, L.I.; Skripchenko, A.; Wagner, S.J. Photodynamic Inactivation of Pathogens in Blood by Phenothiazines and Oxygen. Patent WO/2001/049328, 28 December 2000.
- Ion, R.-M.; Boda, D. Porphyrin—Based supramolecular nanotubes generated by aggregation processes. Rev. Chim. 2008, 59, 205–207.
- Chatterjee, N.; Walker, G.C. Mechanisms of DNA damage, repair, and mutagenesis. Environ. Mol. Mutagen. 2017, 58, 235–263.
- Snipes, W.; Keller, G.; Woog, J.; Vickroy, T.; Deering, R.; Keith, A. Inactivation of lipid-containing viruses by hydrophobic photosensitizers and near-UV radiation. Photochem. Photobiol. 1979, 29, 780–785.
- Le Gall, T.; Lemercier, G.; Chevreux, S.; Tucking, K.-S.; Ravel, J.; Thetiot, F.; Jonas, U.; Schönherr, H.; Montier, T. Ruthenium (II) polypyridyl complexes as photosensitizers for antibacterial photodynamic therapy: A structure-activity study on clinical bac-terial strains. ChemMedChem 2018, 13, 2229–2239.
- Minnock, A.; Vernon, D.I.; Schofield, J.; Griffiths, J.; Parish, J.H.; Brown, S.B. Mechanism of uptake of a cationic water-soluble pyridinium zinc phthalocyanine across the outer membrane of Escherichia coli. Antimicrob. Agents Chemo-ther. 2000, 44, 522–527.
- Hancock, R.E.W. Alterations in outer membrane permeability. Annu. Rev. Microbiol. 1984, 38, 237–264.
- Hancock, R.E. The bacterial outer membrane as a drug barrier. Trends Microbiol. 1997, 5, 37–42.
- Hancock, R.E.; Farmer, S.W. Mechanism of uptake of deglucoteicoplanin amide derivatives across outer membranes of Escherichia coli and Pseudomonas aeruginosa. Antimicrob. Agents Chemother. 1993, 37, 453–456.
- Vieira, C.; Gomes, A.T.; Mesquita, M.Q.; Moura, N.M.M.; Neves, M.G.P.M.S.; Faustino, M.A.F.; Almeida, A. An insight into the potentiation effect of potassium iodide on aPDT e_cacy. Front. Microbiol. 2018, 9, 2665–2670.
- Vieira, C.; Santos, A.; Mesquita, M.Q.; Gomes, A.T.P.C.; Neves, M.G.P.M.S.; Faustino, M.A.F.; Almeida, A. Advances in aPDT based on the combination of a porphyrinic formulation with potassium iodide: Effectiveness on bacteria and fungi plankton-ic/biofilm forms and viruses. J. Porph. Phthal. 2019, 23, 534–545.
- Costa, L.; Tomé, J.P.C.; Neves, M.D.G.P.M.S.; Tomé, A.C.; Cavaleiro, J.A.S.; Cunha, A.; Faustino, M.A.F.; Almeida, A. Sus-ceptibility of non-enveloped DNA- and RNA-type viruses to photodynamic inactivation. Photochem. Photobiol. Sci. 2012, 11, 1520–1530.
- Wu, J.J.; Li, N.; Li, K.A.; Liu, F. J-aggregates of diprotonated tetrakis(4- sulfonatophenyl)porphyrin induced by ionic liquid 1-butyl-3-methylimidazolium tetrafluoroborate. J. Phys. Chem. B 2008, 112, 8134–8138.
- Kemnitz, K.; Sakaguchi, T. Water-soluble porphyrin monomer-dimer systems: Fluorescence dynamics and thermodynamic properties. Chem. Phys. Lett. 1992, 196, 497–502.
- Ribo, J.M.; Crusats, J.; Farrera, J.-A.; Valero, M.L. Aggregation in water solutions of tetrasodium diprotonated meso-tetrakis (4-sulfonatophenyl) porphyrin. J. Chem. Soc. Chem. Comm. 1974, 6, 681–690.
- Hattori, S.; Ishii, K. Magneto-chiral dichroism of aromatic conjugated systems. Opt. Mater. Express 2014, 4, 2423–2432.
- Farjtabar, A.; Gharib, F.; Farajtabar, A. Solvent effect on protonation constants of 5, 10, 15, 20-tetrakis(4-sulfonatophenyl)porphyrin in different aqueous solutions of methanol and ethanol. J. Solut. Chem. 2010, 39, 231–244.
- Kobayashi, T. (Ed.) J-Aggregates; World Scientific Publishing: Singapore, 1996.
- Corsini, A.; Herrmann, O. Aggregation of meso-tetra-(p-sulphonatophenyl) porphine and its Cu(II) and Zn (II) complexes in aqeous solution. Talanta 1986, 33, 335–339.
- Cunderlikova, B.; Bjørklund, E.G.; Pettersen, E.O.; Moan, J. pH-dependent spectral properties of HpIX, TPPS2a, mTHPP and mTHPC. Photochem. Photobiol. 2001, 74, 246–252.
- Kadish, K.M.; Maiya, G.B.; Araullo, C.; Guillard, R. Micellar effects on the aggregation of tetraanionic porphyrins. Spectroscopic characterization of free-base meso-tetrakis(4-sulfonatophenyl)porphyrin, (TPPS)H2, and (TPPS)M (M = Zn(II), Cu(II), V02+) in aqueous micellar media. Inorg. Chem. 1989, 28, 2125–2131.
- Kano, K.; Takei, M.; Hashimoto, S. Cationic porphyrins in water. 1H NMR and fluorescence studies on dimer and molecular complex formation. J. Phys. Chem. 1990, 94, 181–187.
- Kano, K.; Tanaka, N.; Minamizono, H.; Kawakita, Y. Tetraarylporphyrins as probes for studying mechanism of inclusion-complex formation of cyclodextrins. Effect of microscopic environment on inclusion of ionic guests. Chem. Lett. 1996, 25, 925–926.
- Valanciunaite, J.; Poderys, V.; Bagdonas, S.; Rotomskis, R.; Selskis, A. Protein induced formation of porphyrin (TPPS4) nanostructures. J. Phys. Conf. Ser. 2007, 61, 1207–1211.
- Aggarwal, L.P.F.; Borissevitch, I.E. On the dynamics of the TPPS4 aggregation in aqueous solutions: Successive formation of H and J aggregates. Spectrochim. Acta Part A Mol. Biomol. Spectr. 2006, 63, 227–233.
- Faraon, V.; Ion, R.-M.; Pop, S.-F.; Van-Staden, R.; Van-Staden, J.-F. Porphyrins as molecular nanomaterials. In Proceedings of the SPIE 7821, Advanced Topics in Optoelectronics, Microelectronics, and Nanotechnologies, V, 78212H, Constanta, Romania, 4 December 2010.
- Jori, G.; Coppellotti, O. Inactivation of pathogenic microorganisms by photodynamic techniques: Mechanistic aspects and perspective applications. Anti-Infect. Agents Med. Chem. 2007, 6, 913–931.
- Gottfried, V.; Kimmel, S. Temperature effects on photosensitized processes. J. Photochem. Photobiol. B Biol. 1991, 8, 419–430.
- Kochevar, I.E.; Bouvier, J.; Lynch, M.; Lin, C.W.; Chi-Wei, L. Influence of dye and protein location on photosensitization of the plasma membrane. Biochim. Biophys. Acta Biomembr. 1994, 1196, 172–180.
- Lytle, C.; Carney, P.; Felten, R.; Bushar, H.; Straight, R. Inactivation and mutagenesis of herpes virus by photodynamic treatment with therapeutic dyes. Photochem. Photobiol. 1989, 50, 367–371.
- Ion, R.M.; Safta, I.; Natile, G. Photodynamic Inactivation of Herpes Simplex Viruses with Porphyrin Derivatives. Available online: (accessed on 24 February 2021).
- Smetana, Z.; Ben-Hur, E.; Mendelson, E.; Salzberg, S.; Wagner, P.; Malik, Z. Herpes simplex virus proteins are damaged following photodynamic inactivation with phthalocyanines. J Photochem Photobiol B. 1998, 15, 77–83.
- Gradova, M.A.; Kuryakov, V.N.; Lobanov, A.V. The role of the counterions in self-assembly of j-aggregates from meso-aryl substituted porphyrin diacids in aqueous solutions. Macroheterocycles 2015, 8, 244–251.
- Conrado, P.C.V.; Sakita, K.M.; Arita, G.S.; Galinari, C.B.; Gonçalves, R.S.; Lopes, L.D.G.; Lonardoni, M.V.C.; Teixeira, J.J.V.; Bonfim-Mendonça, P.S.; Kioshima, E.S. A systematic review of photodynamic therapy as an antiviral treatment: Potential guidance for dealing with SARS-CoV-2. Photodiagnosis Photodyn Ther. 2021, 34, 102221.
- Frackowiak, D.; Planner, A.; Ion, R.M.; Wiktorowicz, K. Incorporation of dyes in resting and stimulated leukocytes. In Synthesis, Properties and Applications of Near-Infrared Dyes in High Technology Fields; NATO ASI Series; Daehne, S., Ed.; Springer: Dordrecht, The Netherlands, 1998.
- Afrasiabi, S.; Pourhajibagher, M.; Raoofian, R.; Tabarzad, M.; Bahador, A. Therapeutic applications of nucleic acid aptamers in microbial infections. J. Biomed. Sci. 2020, 27, 6–13.
