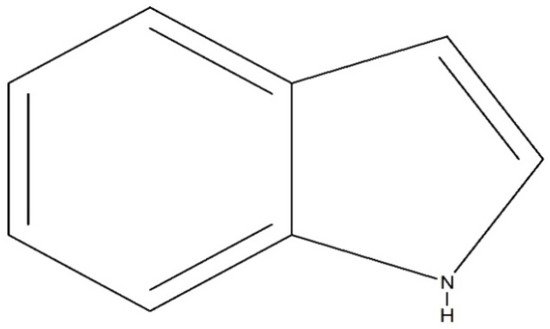
Indole (C8H7N) is a weakly basic molecule consisting of a pyrrole ring fused to a benzene nucleus, and ten π electrons move throughout the structure. The basic environment of indole alkaloids is thought to be caused by the delocalization of the lone pair of nitrogen electrons into the free circulation of the π electronic system. This results in indole becoming protonated at the C-3 position, which is thermodynamically more stable.Indole alkaloids have gained popularity due to their diverse pharmacological activities. Indole alkaloids have been identified in several prominent plant families, including Apocynaceae, Rubiaceae, Nyssaceae, and Loganiaceae, among others. Some of the identified indole alkaloid compounds have been highly effective in pre-clinical and clinical studies. Thousands of compounds containing the indole nucleus have been isolated from plant sources. Their pharmacological activities were assessed, with some now being examined in clinical trials and some already approved for therapeutic use in humans. Indole alkaloids are often characterized by their potent biological activities, which are relevant to the field of medicine, including anticancer, antibacterial, antiviral, antimalarial, antifungal, anti-inflammatory, antidepressant, analgesic, hypotensive, anticholinesterase, antiplatelet, antidiarrheal, spasmolytic, antileishmanial, lipid-lowering, antimycobacterial, and antidiabetic activities.
1. Indole Alkaloids
Nature has always been a blessing for the field of medicine, and peoples throughout history have used natural substances for the treatment of various diseases. The sources of natural substances can be both plants and animals, and an enormous number of pharmacologically active compounds have been derived from natural sources. Many compounds isolated from natural sources have been used as drugs for treatment purposes, either with or without modifications. Through the work of ongoing research, thousands of active compounds have been isolated from natural sources, which can be classified into multiple compound classes. Alkaloids refer to a broad class of compounds, and many of the isolated bioactive compounds have been further classified as indole alkaloids. Many of the therapeutically active indole alkaloids (
) are isolated from plants, and these compounds have had a noticeable impact on the practice of medicine. Adolf von Baeyer was the first to synthesize indole from oxindole using zinc dust in 1866 [1]. Due to the occurrence of adverse effects following treatment with existing drug molecules, the search for new compounds associated with fewer adverse effects has gained immense attention from medicinal chemists and other scientists worldwide. Some of the indole compounds that have since been developed, including vincristine and vinblastine (anticancer agents), reserpine (an antihypertensive agent), physostigmine (a cholinesterase inhibitor), and ajmaline (an anti-arrhythmic agent), are now used as therapeutic drugs.
Figure 1.
Chemical structure of indole.
2. Pharmacological Activities
2.1. Antimicrobial Activity
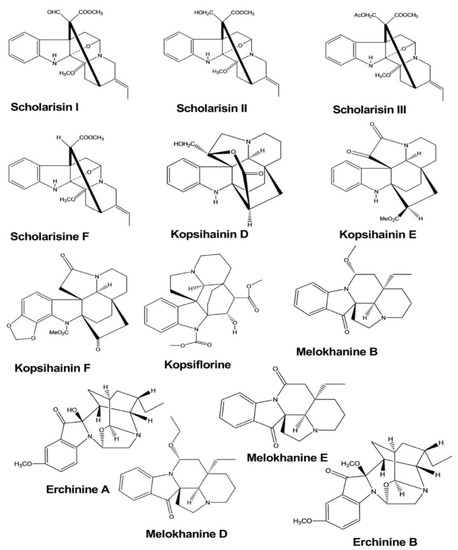
Scholarisins I, II, III, and scholarisine F were isolated from the leaves of Alstonia rupestris
, and the antifungal activities of these compounds were tested by the disk diffusion method. The zones of inhibition and the minimum inhibitory concentrations (MICs) were determined against five species of fungi, and these compounds displayed inhibitory activities against two species of fungi (Gibberella pulicaris
and Cercospora nicotianae), with MIC values of 0.64–0.69 mM, 1.37–1.44 µM, 1.80–1.91 µM, and 1.55–1.71 µM, respectively [2].
, with MIC values of 0.64–0.69 mM, 1.37–1.44 µM, 1.80–1.91 µM, and 1.55–1.71 µM, respectively [5].
Kopsihainins D, E, F, and kopsiflorine were isolated from the twigs of Kopsia hainanensis.
The antibacterial activities of these four compounds were examined against Staphylococcus aureus using the disk diffusion method. These compounds exhibited inhibitory activity, forming antibacterial regions with diameters of 11.2, 9.1, 10.3, and 9.7 mm, respectively [3].
using the disk diffusion method. These compounds exhibited inhibitory activity, forming antibacterial regions with diameters of 11.2, 9.1, 10.3, and 9.7 mm, respectively [6].
Erchinines A and B have shown significant antibacterial and antifungal activities against Trichophyton rubrum
and Bacillus subtilis.
Both compounds were isolated from the roots of Ervatamia chinensis
, and they exhibited potent activity, with the MIC values of 0.78 and 0.78 μg/mL against Bacillus subtilis
and 12.5 and 6.25 μg/mL against Trichophyton rubrum, respectively [4].
, respectively [7].
Melokhanines B, D, E, and F exhibited excellent antibacterial activities against Pseudomonas aeruginosa
. The MIC values for these compounds were 5, 4, 2, and 2 μM, respectively. Moreover, melokhanine B and E also showed antibacterial activity against Enterococcus faecalis
, with a MIC value of 5 μM for both compounds () [5].
) [8].
Figure 2.
Antimicrobial activity of indole alkaloids.
2.2. Antiviral Activity
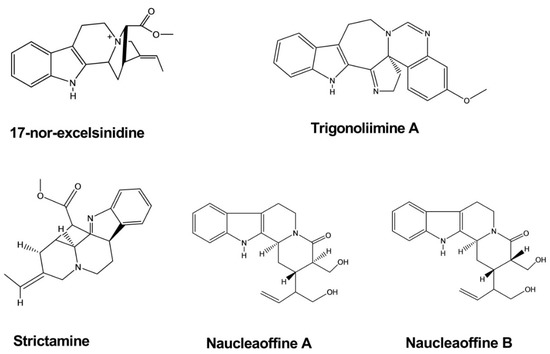
The compounds 17-nor-excelsinidine and strictamine were extracted from the twigs and leaves of Alstonia scholaris
. These compounds were shown to significantly inhibit herpes simplex virus (HSV) and adenovirus (ADV), with half-maximal effective concentrations (EC50) of 1.09 and 0.36 μg/mL against HSV and 0.94 and 0.28 against ADV, respectively [6].
) of 1.09 and 0.36 μg/mL against HSV and 0.94 and 0.28 against ADV, respectively [9].
Trigonoliimine A was extracted from the leaves of Trigonostemon lii
. This compound was evaluated for anti-HIV-1 activity using a microtiter syncytium formation infectivity assay and was found to exhibit a moderate level of inhibitory activity, with an EC50 value of 0.95 µg/mL [7].
value of 0.95 µg/mL [10].
Naucleaoffines A and B were isolated from the leaves and stems of Nauclea officinalis
. Both of these compounds have shown significant effects against HIV-1, with EC50
values of 0.06 and 0.23 µM, whereas the positive control demonstrated an EC50
value of 0.018 µM () [8].
) [11].
Figure 3.
Antiviral activity of indole alkaloids.
2.3. Antidepressant Activity
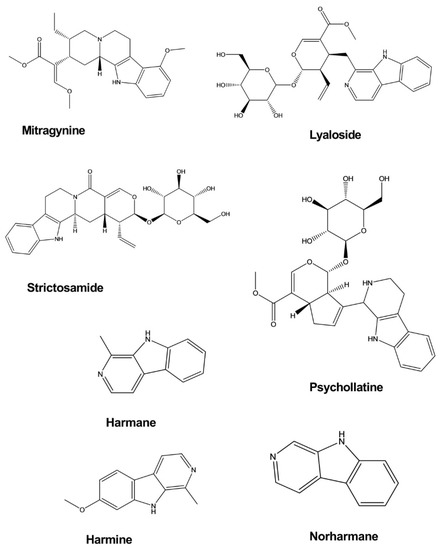
Mitragynine is an indole alkaloid isolated from Mitragyna speciosa Korth. The antidepressant activity of mitragynine was examined using the forced swim test (FST) and tail suspension test (TST) in a mouse model of depression. Mitragynine significantly decreased the immobility periods of mice in both the FST and TST without noticeable effects on locomotor activity in the open-field test when administered at doses of 10 and 30 mg/kg. The release of corticosterone in mice exposed to the FST and TST was found to be considerably diminished following treatment with mitragynine at doses that provided effective antidepressant effects [9].
. The antidepressant activity of mitragynine was examined using the forced swim test (FST) and tail suspension test (TST) in a mouse model of depression. Mitragynine significantly decreased the immobility periods of mice in both the FST and TST without noticeable effects on locomotor activity in the open-field test when administered at doses of 10 and 30 mg/kg. The release of corticosterone in mice exposed to the FST and TST was found to be considerably diminished following treatment with mitragynine at doses that provided effective antidepressant effects [12].
Lyaloside and strictosamide exhibited an inhibitory effect against monoamine oxidase (MAO), although this effect was not significant. However, these compounds may represent new leads for the development of analogs with potential antidepressant effects. Lyaloside and stratosamide inhibited MAO-A with half-maximal inhibitory concentrations (IC50
) values of 50.04 ± 1.09 and 132.5 ± 1.33 μg/mL, respectively, and MAO-B inhibition occurred at IC50
values of 306.6 ± 1.40 and 162.8 ± 1.26 μg/mL. Lyaloside and strictosamide were isolated from Psychotria suterella
and Psychotria laciniata, respectively [10].
, respectively [13].
Harmane, norharmane, and harmine exhibited antidepressant-like activity when administered to mice subjected to the FST. In a dose-dependent manner, these compounds decreased the immobility duration with a 50% effective dose (ED50) of 11.5 mg/kg by intraperitoneal (i.p.) administration for harmane, 8.5 mg/kg i.p. for norharmane, and 8 mg/kg i.p. for harmine. These effects do not appear to be mediated by presynaptic monoaminergic mechanisms but are likely caused by an inverse-agonistic mechanism that involves the benzodiazepine receptors [11].
) of 11.5 mg/kg by intraperitoneal (i.p.) administration for harmane, 8.5 mg/kg i.p. for norharmane, and 8 mg/kg i.p. for harmine. These effects do not appear to be mediated by presynaptic monoaminergic mechanisms but are likely caused by an inverse-agonistic mechanism that involves the benzodiazepine receptors [14].
Psychollatine was isolated from the plant Psychotria umbellate
. Psychollatine increased the number of crossings, rearings, and head-dips of treated mice during the hole-board test at the doses of 7.5 and 15 mg/kg. In the light/dark test, psychollatine increased the time spent in the light area and the latency to the first entry into the dark compartment when administered at a dose of 7.5 mg/kg. In the FST, psychollatine significantly diminished the duration of immobility in mice at doses of 3 and 7.5 mg/kg () [12].
) [15].
Figure 4.
Antidepressant activity of indole alkaloids.
2.4. Anticancer Activity
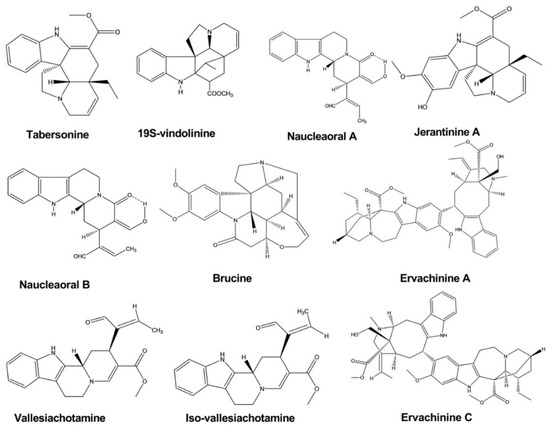
Tabersonine was isolated from the leaves and twigs of Melodinus fusiformis
. This compound exhibited significant anticancer activity against five human tumor cell lines, with IC50 values of 4.6, 5.6, 14.8, 9.9, and 12.1 against SW480, SMMC-7721, HL-60, MCF-7, and A-549 cells, respectively [13].
values of 4.6, 5.6, 14.8, 9.9, and 12.1 against SW480, SMMC-7721, HL-60, MCF-7, and A-549 cells, respectively [16].
Brucine displayed significant cytotoxic activity against the human hepatoma cell line HepG2, with IC50
values of 0.65, 0.32, and 0.10 mM at a different time interval after treatment. Brucine was isolated from the seeds of Strychnos nux-vomica L. Strychnine and isostrychnine, which were extracted from the same plant, also showed cytotoxic activity [14].
L. Strychnine and isostrychnine, which were extracted from the same plant, also showed cytotoxic activity [17].
Naucleaorals A and B were isolated from the roots of Nauclea orientalis
. Both compounds showed cytotoxic activity against the KB (human epidermoid carcinoma) and HeLa (human cervical carcinoma) cell lines. However, Naucleaoral A showed substantial cytotoxicity, with an IC50
value of 4.0 μg/mL against HeLa cells, whereas Naucleaoral B showed only very moderate cytotoxicity, with IC50 values of 7.8 and 9.5 μg/mL against the two cell lines [15].
values of 7.8 and 9.5 μg/mL against the two cell lines [18].
Vallesiachotamine and iso-vallesiachotamine were isolated from the fruits of Anthocephalus cadamba (Roxb) Miq
. Both compounds showed potent anticancer activity, with IC50 values of 4.24 and 3.79 μM, respectively, against the human lung cancer cell line H1299 after 72 h of incubation [16].
values of 4.24 and 3.79 μM, respectively, against the human lung cancer cell line H1299 after 72 h of incubation [19].
Ervachinines A, C, and D exhibited significant inhibitory effects against five cancer cell lines, including HL-60 human myeloid leukemia cells, SMMC-7721 hepatocellular carcinoma cells, A-549 lung cancer cells, MCF-7 breast cancer cells, and SW480 colon cancer cells. Except for ervachinine D against MCF-7 cells, all three compounds displayed IC50
values in the range of 0.84–4.63 μM against all five cancer cell lines. These compounds were extracted from the Ervatamia chinensis whole plant [17].
whole plant [20].
Jerantinines A and B were extracted from the plant Tabernaemontana corymbosa
. These compounds exhibited inhibitory effects against three cancer cell lines, as determined by the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay. The half-maximal growth inhibitory concentration (GI50) was calculated, which showed that jerantinine A displayed an inhibitory effect against breast, colon, and lung carcinoma cell lines, with the GI50 values less than 4.00 μM. Jerantinine B showed significant activity against all cell lines except MCF-7 cells, with GI50 values less than 1.00 μM. Jerantinine A also blocked the ability of cancer cells to form colonies () [18].
) [21].
Figure 5.
Anticancer activity of indole alkaloids.
2.5. Anti-Inflammatory Activity
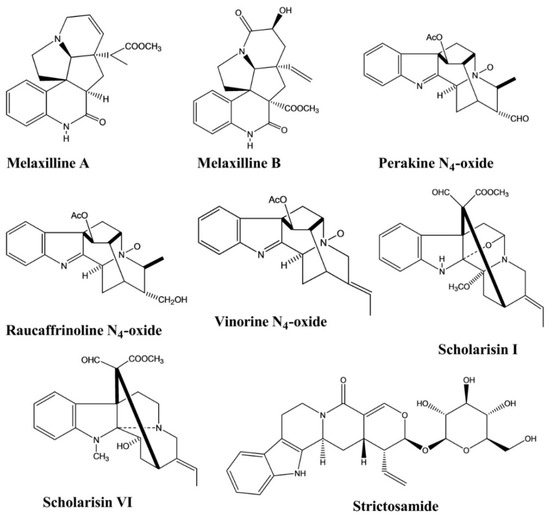
Melaxillines A and B, two alkaloids containing an indole nucleus, were isolated from the roots of Melodinus axillaris
. Both compounds showed potent anti-inflammatory activity, as assessed using an in vitro assay to measure the inhibition of β-glucuronidase secretion induced by platelet-activating factor (PAF) in rat polymorphonuclear leukocytes (PMNs). The IC50 values of these two compounds were 1.51 and 2.62 μM, respectively [19].
values of these two compounds were 1.51 and 2.62 μM, respectively [22].
Perakine N4
-oxide, raucaffrinoline N4
-oxide, and vinorine N4
-oxide were isolated from Alstonia yunnanensis. An in vitro anti-inflammatory activity assay revealed selective cyclooxidase 2 (COX-2) inhibition by these three compounds, with inhibitory values of 94.77%, 88.09%, and 94.05%, respectively; however, none of these compounds displayed any significant COX-1 inhibition (<45%) [20].
. An in vitro anti-inflammatory activity assay revealed selective cyclooxidase 2 (COX-2) inhibition by these three compounds, with inhibitory values of 94.77%, 88.09%, and 94.05%, respectively; however, none of these compounds displayed any significant COX-1 inhibition (<45%) [23].
Scholarisins I and VI, two monoterpenoid indole alkaloids, were isolated from Alstonia rupestris. Both compounds displayed the selective inhibition of COX-2, with inhibitory values of 96.4% and 95.5%, respectively, with no significant inhibitory effects against COX-1 [2].
. Both compounds displayed the selective inhibition of COX-2, with inhibitory values of 96.4% and 95.5%, respectively, with no significant inhibitory effects against COX-1 [5].
Strictosamide showed anti-inflammatory activity against a mouse model of ear edema induced by 12-O-tetradecanoylphorbol-13-acetate (TPA) at doses of 20 and 40 mg/kg. Strictosamide administration significantly diminished the ear swelling rates from 143.9 ± 8.8 to 108.4 ± 11.7 and 103.5 ± 16.0, representing 24.7% and 28.1% inhibition against inflammation, respectively. Strictosamide substantially blocked peritoneal capillary permeability induced by acetic acid in mice, with inhibitory rates of 23.3% and 33.4% at doses of 20 and 40 mg/kg, respectively. In another test, strictosamide significantly reduced leukocyte counts induced by carboxymethylcellulose sodium (CMC–Na) at doses of 10, 20, and 40 mg/kg, resulting in reductions of 46.0%, 49.1%, and 58.7%, respectively () [21].
) [24].
Figure 6.
Anti-inflammatory activity of indole alkaloids.
2.6. Analgesic Activity
Brucine and brucine N-oxide were extracted from the seeds of Strychnos nux-vomica
. Three different tests, including the hot plate test, writhing test, and formalin test, were conducted to determine whether these compounds exerted analgesic effects. In the formalin test, brucine showed potent inhibitory effects against both the early- and late-phase pain stimuli at doses ranging from 7.5 to 30 mg/kg. However, brucine N-oxide exhibited a significant inhibitory effect only against the late phase. In the writhing test, brucine (15 and 30 mg/kg) and brucine N-oxide (50 and 200 mg/kg) showed significant inhibition of the writhing response to the i.p. administration of acetic acid in mice. In the hot plate test, the ED50 value of brucine N-oxide was five and six times greater than that of brucine 30 and 60 min after drug administration, respectively, which indicated that brucine prolonged the pain threshold of mice in a dose-dependent manner [22].
value of brucine N-oxide was five and six times greater than that of brucine 30 and 60 min after drug administration, respectively, which indicated that brucine prolonged the pain threshold of mice in a dose-dependent manner [25].
Mitragynine and 7-hydroxymitragynine were isolated from the plant Mitragyna speciosa [23][24]. The antinociceptive effects of mitragynine were examined using the hot plate test, which revealed a dose-dependent response at doses ranging from 3–35 mg/kg. The latency period increased after the administration of the 15 mg/kg dose. The most significant antinociceptive effect was observed at the 35 mg/kg mitragynine dose, which corresponded with the longest latency time [23]. Similarly, 7-hydroxymitragynine demonstrated antinociceptive activity in a dose-dependent manner (2.5–10 mg/kg) in the tail-flick and hotplate tests. The maximum possible effect (MPE) value of 7-hydroxymitragynine (5 mg/kg, subcutaneous (s.c.)) reached 100% between 15 and 30 min after administration in the tail-flick test. In the hotplate test, the MPE value of 7-hydroxymitragynine (10 mg/kg, s.c.) reached 94% at 15 min after administration [24].
[26,27]. The antinociceptive effects of mitragynine were examined using the hot plate test, which revealed a dose-dependent response at doses ranging from 3–35 mg/kg. The latency period increased after the administration of the 15 mg/kg dose. The most significant antinociceptive effect was observed at the 35 mg/kg mitragynine dose, which corresponded with the longest latency time [26]. Similarly, 7-hydroxymitragynine demonstrated antinociceptive activity in a dose-dependent manner (2.5–10 mg/kg) in the tail-flick and hotplate tests. The maximum possible effect (MPE) value of 7-hydroxymitragynine (5 mg/kg, subcutaneous (s.c.)) reached 100% between 15 and 30 min after administration in the tail-flick test. In the hotplate test, the MPE value of 7-hydroxymitragynine (10 mg/kg, s.c.) reached 94% at 15 min after administration [27].
Strictosamide exhibited analgesic activity in the writhing test, with no such effect observed in the hot plate test. The i.p. injection of strictosamide reduced acetic acid-induced writhing in mice in a dose-dependent manner. Strictosamide remarkably lengthened the pain latency of mice at doses of 20 and 40 mg/kg, resulting in latency periods 336.5 s and 345.8 s, respectively. When the writhing activity was counted, a significant reduction in writhing activity was observed for the 40 mg/kg dose of strictosamide, which reduced the count to 9.7 compared with the positive drug. The inhibition observed at doses of 20 and 40 mg/kg were 37.0% and 49.7%, respectively. This compound was isolated from the Nauclea officinalis [21].
[24].
Umbellatine was isolated from the leaves of Psychotria umbellate
. Analgesic activity was investigated by conducting the tail-flick test, hot plate test, formalin test, and capsaicin-induced pain test. In the four test models, umbellatine exhibited good activity against tail-flick test and hot plate test and significant activity against formalin test and capsaicin-induced pain test at the doses of 100–300 mg/kg (