Plasmonic biosensing has enabled the design of novel detection strategies capable of targeting individual molecules while evaluating their binding affinity and biological interactions.
- single-molecule analysis
- biosensors
- plasmonics
- nanoparticle
- nanostructure
- nucleic acids
- virus
- living-cells
1. Introduction
The detection of clinical biomarkers with relevant sensitivity is becoming a great concern for biomedical analysis [1,2,3][1][2][3]. The objective quantification of biological targets (i.e., nucleic acids, proteins, cells and microorganisms) enables both the identification of normal or pathological diagnostic outcomes and the monitoring of the biological response to therapeutic drugs or environmental agents. Typically, chemical and biological species are present in clinical samples at trace concentrations as low as attomolar levels [1,2,4][1][2][4]. Measuring such small amounts of biomolecules is particularly relevant not only for the early identification of cancer biomarkers but also for the rapid recognition of pathogenic agents and the prevention of potential outbreaks (Figure 1) [2,5][2][5]. For instance, the precise diagnosis of bacterial and virus infections such as tuberculosis, hepatitis, Acquired Immune Deficiency Syndrome (AIDS) and more recently coronavirus disease 2019 (COVID-19) may require the transduction of independent events occurring at single-molecule or cellular levels [2,4][2][4]. Single-molecule analysis is also needed when only a minimum volume of sample is available or multiplexed analysis is demanded to target various analytes simultaneously in a short time [3,6][3][6].
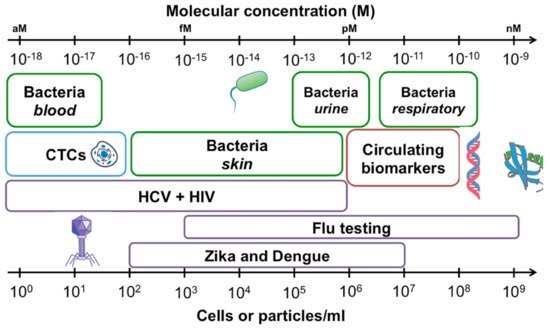
Figure 1. Concentrations of common analytes in biological samples expressed as cells or particles per milliliter of sample (bottom axis), and molarity (top axis). Bacterial concentrations are outlined in green, viral targets are outlined in purple, circulating tumor cells (CTCs) are shown in blue and circulating biomarkers including proteins and nucleic acids in red. Reprinted with permission from Kelley et al. [2] Copyright © 2017 American Chemical Society.
Meeting these challenges involves the monitoring of single-molecule interactions between reacting species via sensing devices that can distinguish binding kinetics in heterogeneous samples with sufficient specificity [1,4][1][4]. The observation of a signal from an individual molecule within the ensemble also relies on the transport of the analytes to the sensing region. Therefore, strategies to improve single molecule detection should take into consideration both the sensing mechanism and the diffusion rate, analyte concentration and sample volume [6].
Current research in single-molecule bioanalytical technologies has been primarily focused on configurations based on total internal reflection fluorescence (TIRF) and fluorescent-related methods [1,7,8][1][7][8]. Other optical detection techniques such as surface-enhanced Raman scattering (SERS), total internal reflection scattering (TIRS), dark-field microscopy (DFM), or near-field scanning optical microscopy (NSOM) have also demonstrated their value for the determination of single-molecule interactions [9,10][9][10].
From this perspective, plasmonic-based techniques take advantage of the unique optical and electronic properties of plasmonic nanostructures to assess the signals and kinetic distributions of single-molecule binding events occurring in real-time [11,12][11][12]. Nanoplasmonic sensing principles rely on the collective oscillation of free electrons known as localized surface plasmons at metallic interfaces or within nanostructures under light stimulation [13,14,15][13][14][15]. The possibility of providing sensitive and selective analysis using single-molecule sensing mechanisms depend on the signal amplification resulting from the enhancement of the intensity of the optical field [8]. Therefore, the manipulation of the physicochemical environment and composition of plasmonic nanostructures allow the precise confinement of the electromagnetic field into nanoscale volumes of only a few nanometers, which are named “hot spots”. The generation of extreme electric field gradients facilitates optical trapping of single molecules while providing longer dwelling times [16,17][16][17]. Particularly, the interaction between a biological receptor and its target analyte near the optical-field can be detected through the shift of the plasmon polariton resonance wavelength induced by a local refractive index change [13].
The design of high-spatial-resolution surface structures is very valuable for the surface-enhanced sensing of individual events (Figure 2) [18]. In this sense, plasmonic single-molecule approaches mainly rely upon the extinction spectra of metallic nanoparticles (NPs) and the capacity of optical nanoapertures (i.e., arrays of nanoholes, nanopores, nanowells) to confine the electromagnetic field into ultrasmall subwavelength dimensions [1,8,9,19,20,21,22,23][1][8][9][19][20][21][22][23]. Both types of plasmonic nanostructures have been successfully applied to the label-free quantification of single-proteins in the diagnosis of acute and chronic diseases in early stages, the recognition of single-nucleotide specificity and the tracking of independent biological processes in living cells inserted in nanopores [24,25,26][24][25][26].
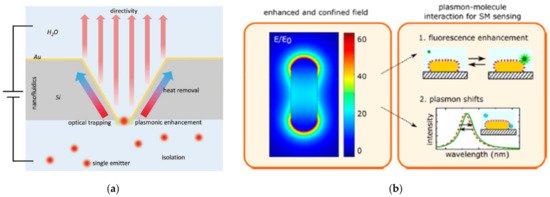
Figure 2. Schematic representation of plasmon-enhanced single-molecule sensing using nanoapertures and nanoparticles: (a) Nanoapertures (rectangle in an etched silicon film) in metal films (gold) for enhanced interaction with single emitters (shown as red glowing circles). The anisotropic etch produces a horn antenna for directive coupling. The aperture provides plasmonic enhancement for optical trapping. The aperture also serves as a nanopore for nanofluidic functionality, like monitoring ionic current from flow through the aperture Adapted with permission from Gordon et al. [27] Copyright © 2020 Wiley; (b) Nanoparticles: The plasmon resonance induces a strongly enhanced and tightly confined local field around the particles. The field shown here is for a gold nanorod that is excited on resonance. The local field mediates plasmon−molecule interactions, enabling enhanced single-molecule detection by monitoring plasmon-induced changes of the molecule (resulting in, e.g., fluorescence enhancement) or by monitoring molecule-induced changes of the plasmon (resulting in frequency-shifts of the plasmon). Adapted with permission from Taylor et al. [4] Copyright © 2017 American Chemical Society.
Although the fundamentals and functionalities of single-molecule biosensing have been extensively addressed in previous reviews [1,8,22][1][8][22], the specific role of plasmonic biosensors in the detection of single-molecule biological targets has still not been satisfactorily resolved. Therefore, the aim of this work is to summarize recent progress in plasmonic single-molecule biosensing schemes for biomedical applications. This review particularly concentrates on the quantification of clinically relevant biomarkers using plasmonic nanostructures such as metallic nanoparticles and optical nanoapertures. Recent research in the design of the new generation of plasmonic platforms for practical sensing of independent molecular interactions from the perspective of point of care testing is also reviewed.
2. Plasmonic Nanoapertures for Single-Molecule Analysis
The exploitation of plasmonic nanoapertures as sensing probes for single-molecule analysis has attracted great interest over the last decade. The formation of nanoapertures in an opaque metal film permits the confinement of light in the subwavelength regime, thereby limiting the interaction volume to the nanometer scale [28,29][28][29].
Among nanoscale apertures, solid-state nanopores provide the basis for the development of stable and durable sensing platforms capable of achieving extremely low single-molecule detection limits with high specificity [1,30][1][30]. Nanopore sensors consist of an electrically thin insulating membrane traversed by 1–100 nm pores through which ions can cross [22,31][22][31]. When charged molecules pass through the pore, the application of a voltage between two electrodes produces the interaction with the membrane and the displacement of ions, causing the change of the ionic current. This displacement can be determined via optical or electrical sensing [1,22][1][22]. The optical sensing mainly depends on the change of the refractive index after the arrival of the target molecule to the hotspot of the plasmonic nanopore, wherein the electromagnetic field is strongly localized (Figure 3) [19]. Specifically, the optical transmission through the nanoaperture conducts the plasmon resonance to the far field in which the individual molecule can be detected. Monitoring of the plasmon resonance redshift can either be performed by registering the plasmon resonance peak or by tracking the scattered light intensity at a fixed excitation wavelength. In this way, plasmonic nanoapertures enable the ultrasensitive quantification of molecular interactions by combining the acquisition of bimodal optical and electrical data [32].
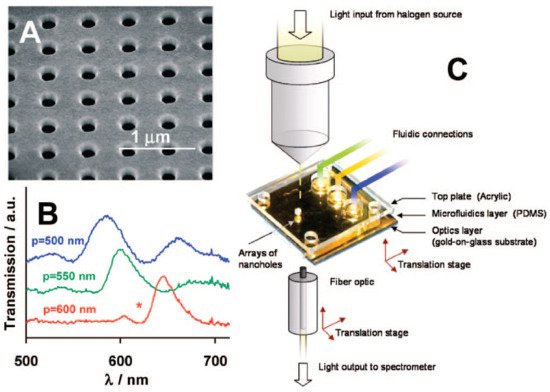
Figure 3. (A) Scanning electron micrograph (SEM) of an array of nanoholes in a gold film; (B) Extraordinary Optical Transmission (EOT) spectra for three arrays with different periodicities; (C) experimental setup to measure the EOT effect. The metal film is deposited on a glass slide, and the gold side of the array is exposed to solvents and aqueous solutions delivered by microfluidics. Adapted with permission from Gordon et al. [19] Copyright © 2008 American Chemical Society.
Since nanopore sensors facilitate the active transport of charged molecules at very low concentrations towards the nanopore aperture from long distances, the combination with plasmonic sensing can contribute to improve the diffusion of single-molecules at a plasmonic hotspot. In addition, the nanoscale diameter of the nanopore is comparable to the cross-section of the molecule, thus reducing the molecule motion to only one dimension. This feature allows both the increment of the dwelling time and the enhancement of the signal-to-background ratio [22]. Therefore, by controlling the nanopore diameter, analytes can be confined to a restricted space and the capture time increases significantly. Accordingly, the reduction in the nanopore size contributes to enhancing the signal-to-noise ratio results enabling highly selective single-molecule detection.
These interesting properties have paved the way for a variety of biological applications aimed at manipulating nanoscale molecules such as single-proteins, virus like-particles or nucleic acids. A detailed description of the fabrication of nanopores has been comprehensively reviewed in previous works and it is beyond the scope of this review [22,33][22][33]. Novel developments in plasmonic nanopore sensing schemes for evaluating biological processes including protein-protein interactions, translocation of single proteins, assessment of kinetic reactions, analysis of RNA structure, single nucleotide polymorphisms and DNA sequencing approaches are shown in this section (see Table 1).
Table 1. Key analytical features of plasmonic applications based on nanopores and nanohole arrays sensors classified according to the characteristics of target analyte, instrument configuration (namely sensing scheme or biological receptor) and detection format.
| Target Analyte | Instrument Configuration | Detection Strategy | Reference |
|---|---|---|---|
| Nucleic acids | Nanopore-nanowells with fluorescence enhancement | Double-stranded single DNA molecules adsorption | [34] |
| Graphene quantum dots with a nanopore (density functional theory calculations.) | DNA-graphene interactions and nucleobase (adenine, thymine, cytosine, and guanine) rotation | [35] | |
| Nanopore, bowtie, and bowtie-nanopore structures and SPR materials (chromium, aluminum, rhodium and graphene) | DNA nucleotides shifts in the SPR spectra through Bowtie-nanopore structures | [36] | |
| Two elongated nanodiscs in a gold bowtie nanoantenna | Single-DNA molecules in the nanopore gap monitoring (l intensity of light backscattered from the antenna) | [37] | |
| Inverted-bowtie gold plasmonic nanopore | DNA-protein interactions and DNA translocation (optical signals under different illumination conditions) | [38] | |
| Nanohole arrays | Single-DNA origami triangles were captured on SiO2 bottom surface of gold nanoholes | [39] | |
| Proteins | Plasmonic nanopore tri-color fluorescence simulation and pattern-recognition algorithms | Individual protein translocations, human plasma proteome and cytokine recognition | [40] |
| Plasmonic nanopore acting as nano tweezer | Individual beta-amylase proteins optical trapping | [41] | |
| Infectious agents | Optical nanoantenna with direct physical fluorescence amplification | Single nucleotide variations in Zika artificial DNA origami buffer and human serum | [42] |
| Nanohole array | Single dengue virus-like particles and virucidal drug candidates (spectral shifts of transmission peaks) | [43] | |
| Organic compounds | Silver nanohole arrays and microscopy imaging | Individual rhodamine 6G (R6G) three-dimensional orientation | [44] |
(References would be added automatically after the entry is online)
References
- Akkilic, N.; Geschwindner, S.; Höök, F. Single-Molecule Biosensors: Recent Advances and Applications. Biosens. Bioelectron. 2020, 151, 111944.
- Kelley, S.O. What Are Clinically Relevant Levels of Cellular and Biomolecular Analytes? ACS Sens. 2017, 2, 193–197.
- Macchia, E.; Manoli, K.; Di Franco, C.; Scamarcio, G.; Torsi, L. New Trends in Single-Molecule Bioanalytical Detection. Anal. Bioanal. Chem. 2020, 412, 5005–5014.
- Taylor, A.B.; Zijlstra, P. Single-Molecule Plasmon Sensing: Current Status and Future Prospects. ACS Sens. 2017, 2, 1103–1122.
- Chen, H.; Liu, K.; Li, Z.; Wang, P. Point of Care Testing for Infectious Diseases. Clin. Chim. Acta 2019, 493, 138–147.
- Špačková, B.; Šípová-Jungová, H.; Käll, M.; Fritzsche, J.; Langhammer, C. Nanoplasmonic–Nanofluidic Single-Molecule Biosensors for Ultrasmall Sample Volumes. ACS Sens. 2021, 6, 73–82.
- Fabrizio, E.D.; Schlücker, S.; Wenger, J.; Regmi, R.; Rigneault, H.; Calafiore, G.; West, M.; Cabrini, S.; Fleischer, M.; van Hulst, N.F.; et al. Roadmap on Biosensing and Photonics with Advanced Nano-Optical Methods. J. Opt. 2016, 18, 063003.
- Ma, X.; Sim, S.J. Single Plasmonic Nanostructures for Biomedical Diagnosis. J. Mater. Chem. B 2020, 8, 6197–6216.
- Lee, S.; Sun, Y.; Cao, Y.; Kang, S.H. Plasmonic Nanostructure-Based Bioimaging and Detection Techniques at the Single-Cell Level. TrAC Trends Anal. Chem. 2019, 117, 58–68.
- Xiao, L.; Qiao, Y.; He, Y.; Yeung, E.S. Three Dimensional Orientational Imaging of Nanoparticles with Darkfield Microscopy. Anal. Chem. 2010, 82, 5268–5274.
- Armstrong, R.E.; Horáček, M.; Zijlstra, P. Plasmonic Assemblies for Real-Time Single-Molecule Biosensing. Small 2020, 16, 2003934.
- Halas, N.J.; Lal, S.; Chang, W.-S.; Link, S.; Nordlander, P. Plasmons in Strongly Coupled Metallic Nanostructures. Chem. Rev. 2011, 111, 3913–3961.
- Homola, J. Surface Plasmon Resonance Sensors for Detection of Chemical and Biological Species. Chem. Rev. 2008, 108, 462–493.
- Strobbia, P.; Languirand, E.; Cullum, B.M. Recent Advances in Plasmonic Nanostructures for Sensing: A Review. Opt. Eng. 2015, 54, 100902.
- Masson, J.-F. Surface Plasmon Resonance Clinical Biosensors for Medical Diagnostics. ACS Sens. 2017, 2, 16–30.
- Mestres, P.; Berthelot, J.; Aćimović, S.S.; Quidant, R. Unraveling the Optomechanical Nature of Plasmonic Trapping. Light Sci Appl. 2016, 5, e16092.
- Kim, J.-D.; Lee, Y.-G. Trapping of a Single DNA Molecule Using Nanoplasmonic Structures for Biosensor Applications. Biomed. Opt. Express 2014, 5, 2471.
- Koya, A.N.; Cunha, J.; Guo, T.; Toma, A.; Garoli, D.; Wang, T.; Juodkazis, S.; Cojoc, D.; Proietti Zaccaria, R. Novel Plasmonic Nanocavities for Optical Trapping-Assisted Biosensing Applications. Adv. Opt. Mater. 2020, 8, 1901481.
- Gordon, R.; Sinton, D.; Kavanagh, K.L.; Brolo, A.G. A New Generation of Sensors Based on Extraordinary Optical Transmission. Acc. Chem. Res. 2008, 41, 1049–1057.
- Dahlin, A.B. Sensing Applications Based on Plasmonic Nanopores: The Hole Story. Analyst 2015, 140, 4748–4759.
- Khandelwal, G.; Umrao, S.; Nirala, N.R.; Sagar, S.S.; Kumar, V. Plasmonic Nanopores: A New Approach Toward Single Molecule Detection. In Nanomaterials: Biomedical, Environmental, and Engineering Applications; Kanchi, S., Ahmed, S., Sabela, M.I., Hussain, C.M., Eds.; John Wiley & Sons Inc.: Hoboken, NJ, USA, 2018; pp. 257–284. ISBN 978-1-119-37038-3.
- Spitzberg, J.D.; Zrehen, A.; van Kooten, X.F.; Meller, A. Plasmonic-Nanopore Biosensors for Superior Single-Molecule Detection. Adv. Mater. 2019, 31, 1900422.
- Celiksoy, S.; Ye, W.; Wandner, K.; Kaefer, K.; Sönnichsen, C. Intensity-Based Single Particle Plasmon Sensing. Nano Lett. 2021.
- Zhang, P.; Ma, G.; Dong, W.; Wan, Z.; Wang, S.; Tao, N. Plasmonic Scattering Imaging of Single Proteins and Binding Kinetics. Nat. Methods 2020, 17, 1010–1017.
- Zhang, Q.; Wu, Y.; Xu, Q.; Ma, F.; Zhang, C. Recent Advances in Biosensors for in Vitro Detection and in Vivo Imaging of DNA Methylation. Biosens. Bioelectron. 2021, 171, 112712.
- Gilboa, T.; Garden, P.M.; Cohen, L. Single-Molecule Analysis of Nucleic Acid Biomarkers–A Review. Anal. Chim. Acta 2020, 1115, 61–85.
- Gordon, R. Metal Nanoapertures and Single Emitters. Adv. Opt. Mater. 2020, 8, 2001110.
- Eftekhari, F.; Escobedo, C.; Ferreira, J.; Duan, X.; Girotto, E.M.; Brolo, A.G.; Gordon, R.; Sinton, D. Nanoholes as nanochannels: Flow-through plasmonic sensing. Anal. Chem. 2009, 81, 4308.
- Chen, C.; Juan, M.L.; Li, Y.; Maes, G.; Borghs, G.; Van Dorpe, P.; Quidant, R. Enhanced Optical Trapping and Arrangement of Nano-Objects in a Plasmonic Nanocavity. Nano Lett. 2012, 12, 125–132.
- Hu, R.; Tong, X.; Zhao, Q. Four Aspects about Solid-State Nanopores for Protein Sensing: Fabrication, Sensitivity, Selectivity, and Durability. Adv. Healthc. Mater. 2020, 9, 2000933.
- Gilboa, T.; Meller, A. Optical Sensing and Analyte Manipulation in Solid-State Nanopores. Analyst 2015, 140, 4733–4747.
- Peri, S.S.S.; Sabnani, M.K.; Raza, M.U.; Ghaffari, S.; Gimlin, S.; Wawro, D.D.; Lee, J.S.; Kim, M.J.; Weidanz, J.; Alexandrakis, G. Detection of Specific Antibody-Ligand Interactions with a Self-Induced Back-Action Actuated Nanopore Electrophoresis Sensor. Nanotechnology 2020, 31, 085502.
- Escobedo, C. On-Chip Nanohole Array Based Sensing: A Review. Lab Chip 2013, 13, 2445.
- Assad, O.N.; Gilboa, T.; Spitzberg, J.; Juhasz, M.; Weinhold, E.; Meller, A. Light-Enhancing Plasmonic-Nanopore Biosensor for Superior Single-Molecule Detection. Adv. Mater. 2017, 29, 1605442.
- Abasifard, M.; Ahmadi, V.; Fotouhi, B.; Roohi, R. DNA Nucleobases Sensing by Localized Plasmon Resonances in Graphene Quantum Dots with Nanopore: A First Principle Approach. J. Phys. Chem. C 2019, 123, 25309–25319.
- Fotouhi, B.; Ahmadi, V.; Faramarzi, V. Nano-Plasmonic-Based Structures for DNA Sequencing. Opt. Lett. 2016, 41, 4229.
- Shi, X.; Verschueren, D.V.; Dekker, C. Active Delivery of Single DNA Molecules into a Plasmonic Nanopore for Label-Free Optical Sensing. Nano Lett. 2018, 18, 8003–8010.
- Verschueren, D.V.; Pud, S.; Shi, X.; De Angelis, L.; Kuipers, L.; Dekker, C. Label-Free Optical Detection of DNA Translocations through Plasmonic Nanopores. ACS Nano 2019, 13, 61–70.
- Brassat, K.; Ramakrishnan, S.; Bürger, J.; Hanke, M.; Doostdar, M.; Lindner, J.K.N.; Grundmeier, G.; Keller, A. On the Adsorption of DNA Origami Nanostructures in Nanohole Arrays. Langmuir 2018, 34, 14757–14765.
- Ohayon, S.; Girsault, A.; Nasser, M.; Shen-Orr, S.; Meller, A. Simulation of Single-Protein Nanopore Sensing Shows Feasibility for Whole-Proteome Identification. PLoS Comput. Biol. 2019, 15, e1007067.
- Verschueren, D.; Shi, X.; Dekker, C. Nano-Optical Tweezing of Single Proteins in Plasmonic Nanopores. Small Methods 2019, 3, 1800465.
- Ochmann, S.E.; Vietz, C.; Trofymchuk, K.; Acuna, G.P.; Lalkens, B.; Tinnefeld, P. Optical Nanoantenna for Single Molecule-Based Detection of Zika Virus Nucleic Acids without Molecular Multiplication. Anal. Chem. 2017, 89, 13000–13007.
- Jackman, J.A.; Linardy, E.; Yoo, D.; Seo, J.; Ng, W.B.; Klemme, D.J.; Wittenberg, N.J.; Oh, S.-H.; Cho, N.-J. Plasmonic Nanohole Sensor for Capturing Single Virus-Like Particles toward Virucidal Drug Evaluation. Small 2016, 12, 1159–1166.
- Sahu, S.P.; Mahigir, A.; Chidester, B.; Veronis, G.; Gartia, M.R. Ultrasensitive Three-Dimensional Orientation Imaging of Single Molecules on Plasmonic Nanohole Arrays Using Second Harmonic Generation. Nano Lett. 2019, 19, 6192–6202.
