Teneurins have been identified in vertebrates as four different genes (TENM1-4), coding for membrane proteins that are mainly involved in embryonic and neuronal development.
- teneurins
- cancer
- tumor progression
- metastasis
1. Introduction
Teneurins belong to a conserved family of transmembrane proteins that are involved in cell–cell and cell-extracellular matrix interactions, with a pivotal role in embryonic development and nervous system function. Teneurin homologs have been functionally implicated in axon guidance [1], neurite outgrowth [1][2][1,2], and transcriptional regulation [3][4][5][3,4,5], as well as in cell proliferation and adhesion [1]. Teneurin genes were first described in vertebrates in 1998 [6] as orthologs of the Ten-m/odd Oz (Odz) genes that had previously been independently identified in Drosophila by two groups using two different strategies [7][8][7,8]. Baumgartner and colleagues discovered the Ten-m gene by screening the Drosophila genome with a Tenascin-a probe that covers its epidermal growth factor (EGF)-like domain [7], while Levine and colleagues described the Odz gene via a tyrosine phosphorylation-based screening [8]. Initially, Ten-m/Odz proteins were defined as pair-rule genes involved in Drosophila segmentation and were mainly expressed in the central nervous system and heart [7][8][7,8]. Later, as Ten-m mutants failed to display a pair-rule phenotype, a different role was proposed and Ten-m/Odz genes were demonstrated to code for transmembrane proteins with a possible oscillator function [9][10][11][9,10,11]. In mice and humans, four different Ten-m paralogous genes (Ten-m1–4) code for large type II transmembrane proteins, called teneurin (TENM)1–4.
2. Common Structural and Functional Features of Mammalian Teneurins
All four of the identified mammalian teneurins are type II transmembrane proteins and possess strong sequence homology, consist of about 2800 amino acids, and entail a complex set of functional domains. Although their involvement in cell adhesion has been well documented, teneurins are unusual in that they lack the classical domains that are generally observed in cell adhesion molecules, such as immunoglobulin, cadherin, laminin-a, neurexin and sex hormone-binding globulin, and integrin domains. All teneurins have a C-terminal extracellular domain, including a globular part, followed by an Ig-like domain (composed of a carboxypeptidase-like domain and of a cysteine-rich domain), which is involved in teneurins dimerization, and by eight EGF modules (Figure 1).
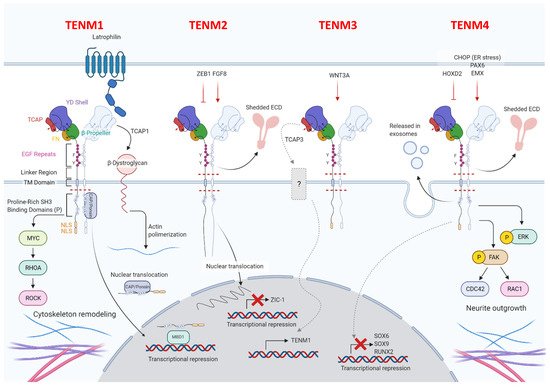
Figure 1.
The globular part of all teneurins is composed of a fibronectin plug, a protein-protein interaction domain, called 6-bladed β-propeller, a tyrosine-aspartic acid (YD)-repeat barrel, an RHS core protein domain, and a teneurin C-terminal associated peptide (TCAP), which is involved in the interaction between teneurins and latrophilins [12][17]. TCAP has recently been demonstrated to also possess nuclease activity and an apoptogenic role [13][18]. Teneurin proteins cross the membrane via hydrophobic residues that are linked to an N-terminal intracellular domain, which is characterized by one or more proline-rich SRC Homology 3 (SH3)-binding domains in the part closest to the transmembrane region, conserved tyrosines, which have been predicted to be phosphorylated, nuclear localization sequences, and a basic sequence motive RKRK, which may be a proteolytic cleavage site [14][19]. Despite the great homology, some structural differences between teneurins exist, and these are mainly related to the number of proline-rich SH3-binding domains, and also to the presence or absence of predicted nuclear localization sequences and proteolytic cleavage sites [14][19]. Details on the distinguishing features of each teneurin will be described in the following dedicated paragraphs.
Teneurins, following dimerization, act as transcriptional regulators after the release of the intracellular domain. They can form both homo- and heterodimers. Electrophoretic and electron-microscopic analyses of TENM1 have shown that homodimerization most likely takes place between the EGF-like modules and the hydrophobic regions of two teneurins that are arrayed side-by-side on the same cell [15][20]. Recent evidence has revealed that homodimerization can also occur between two identical teneurins expressed on different cells in close proximity [16][21]. This trans-homodimerization of teneurins has been reported to allow the correct matching between axons and their targets to occur, thus contributing to correct circuit-wiring in the nervous system [17][18][19][22,23,24]. Upon homodimerization, teneurins’ intracellular domains are cleaved close to the plasma membrane and translocate into the nucleus, where they regulate gene expression via direct and indirect interactions with transcription factors [20][25].
In addition, all teneurins can mediate cell–cell interaction by forming heterodimers [21][26] with different proteins, among which are the adhesion G-protein-coupled receptors latrophilins and the actin-crosslinking protein filamin. While the trans-heterodimerization between teneurins and latrophilins is involved in synapse formation and organization [22][23][27,28] and in cellular communication [24][29], teneurins and filamin interactions are involved in cell motility [9]. Recent X-ray crystallographic studies have revealed that the teneurin–latrophilin binding involves the presence of another protein, the fibronectin leucine-rich repeat transmembrane protein (FLRT) [25][30].
Teneurins exist as splice variants which differ for the presence or absence of nine- and seven-amino acid insertions at the EGF repeats and the β-propeller regions, respectively [16][26][27][21,31,32]. The different teneurins’ alternative variants interact with different ligands, mediating either teneurin homodimerization or heterodimerization with latrophilins, leading to the activation of different biological functions [16][21]. This, together with the possibility that they can be cleaved and released, is likely to contribute to the high level of complexity in these molecules and their functions. Indeed, amino acid sequence analyses have shown that teneurin proteins possess cleavage sites in the extracellular domain, leading to potential for the extracellular release of teneurins. One cleavage site can be located between the transmembrane and EGF-like domains, and is susceptible to proteolytic cleavage in vitro [15][20], while the other is situated near the C-terminus of the protein [28][33] and is responsible for the secretion of the TCAP domain, which is a bioactive neuropeptide involved in several functions.
Besides their wide expression in developing embryos, all teneurins can be found in several adult tissues, suggesting that they have a potential role in normal physiology. However, both their function and whether their expression is essential in normal adult tissues are currently unknown.
An ever-growing amount of evidence has demonstrated the aberrant expression [29][34] as well as somatic alterations and chromosomal rearrangements [30][35] of teneurins in human tumors of different histotypes. Their involvement in tumor cell signaling, tumorigenic regulatory pathways and drug resistance mechanisms have also been described [29][34]. Moreover, thanks to an analysis of open-access molecular and clinical datasets of tumor patients, the possible prognostic impact of teneurins has also been reported [30][35]. However, their functional role in tumor initiation and malignant progression has not yet been fully elucidated. Herein, we review available data about the general features of teneurins with a particular focus on their involvement in cancer, in order to highlight the potential role that teneurins may be able to play as novel tumor biomarkers and targets for therapeutic approaches.
