Pancreatic ductal adenocarcinoma (PDAC) is one of the most common and aggressive cancer types, with a five-year survival rate of only 2–9%.
- pancreatic cancer
- monoclonal antibodies
1. Introduction
In the absence of reliable biomarkers for use in the screening and early detection of pancreatic cancer, and more effective and less toxic therapeutic agents, it has been projected that pancreatic cancer will not only rise in incidence but also will take over breast, prostate and colorectal cancers and become the second leading cause of cancer death in the Western world by 2030 [1][2][3].
Treatment of patients with pancreatic cancer involves surgery, chemotherapy and radiotherapy. Although surgery is the only curative treatment, around 80% of patients are diagnosed at late stages of the disease and are not eligible for surgical resection. Adjuvant treatment with chemotherapy is beneficial given the high rate of locoregional relapse after surgery alone. Gemcitabine-based therapy was traditionally the mainstay for treatment of pancreatic cancer [4]. However, the results of the recently published ESPAC-4 trial showed that the combination of gemcitabine plus capecitabine increased median overall survival compared to gemcitabine alone (28.0 vs 25.5 months) with an acceptable toxicity profile, and an estimated 5-year survival of 28.8% for the combination group compared to 16.3% with gemcitabine monotherapy, making this combination the new standard of care in the adjuvant setting [5]. Treatment for borderline resectable or locally advanced unresectable tumours seems to yield better results with chemotherapy (e.g., FOLFIRINOX) rather than chemoradiotherapy, although more robust evidence from trials is needed [6].
Patients with metastatic disease are treated with either FOLFIRINOX or gemcitabine plus nab-paclitaxel as first-line in patients with good performance status [7][8]. Erlotinib was approved by the FDA for use in metastatic pancreatic cancer patients based on a study that showed a modest improvement in median survival in patients who received erlotinib plus gemcitabine compared to gemcitabine alone (6.4 vs 5.9 months) but the clinical relevance is controversial [9]. The combination of liposomal irinotecan, fluorouracil and folinic acid (NAPOLI-1 regimen) is the only currently approved second-line chemotherapy for patients with metastatic pancreatic cancer based on a phase 3 trial that showed median overall survival of 6.1 months for the triple combination compared to 4.2 months in patients receiving fluorouracil and folinic acid [10]. Therefore, it is essential to discover novel targets and to develop more effective, less toxic and pancreatic cancer specific therapeutic agents for the long-term benefit of patients with pancreatic cancer.
The advent of hybridoma technology by Köhler and Milstein in 1975, which allows the production of unlimited quantity of an antibody against any target antigen, has revolutionised many areas of biomedical research and medicine [11]. Further technological advances in genetic engineering allowed the production of less immunogenic and more effective types of mAbs (e.g., chimeric, humanised, fully human mAbs, antibody fragments and bispecific antibodies) for use in the treatment of patients with a range of diseases including cancer [12][13][14][15][16]. Indeed, mAb-based therapy is currently one of the two major types of targeted therapy and an attractive therapeutic alternative for the treatment of patients with a wide range of cancers. In this article, we provide a comprehensive review of monoclonal antibody-based agents that have been approved for the treatment of human cancers and the current state of preclinical and clinical studies with monoclonal antibody-based agents in pancreatic cancer. We shall also highlight some of the contributing factors for the poor response to therapy with mAbs, and emerging opportunities for more effective treatment of pancreatic cancer with antibody-based agents in combination with other treatments.
2. Therapeutic Antibodies Approved in Cancer
Over the past few decades, monoclonal antibody-based agents have been approved and used routinely in the treatment of a wide range of human diseases including cancer, infectious, autoimmune and metabolic diseases. Monoclonal antibody-based drugs can be developed by a variety of approaches such as hybridoma technology, phage display technology, the use of transgenic mouse or the single B-cell technique [15][17][18].
Depending on the target antigen and the antibody format, monoclonal antibody-based drugs can produce their anti-tumour activity by several mechanisms (Figure 1). Some mAbs are directed against growth factor receptors with high levels of expression in tumours cells and inhibit tumour growth by blocking the binding of growth factor to its receptor (e.g., anti-EGFR mAbs cetuximab and panitumumab), or by inhibiting receptor dimerization (e.g., anti-HER-2 mAb pertuzumab), consequently inhibiting the downstream cell signalling pathways. In contrast, other antibodies halt tumour growth by inhibiting angiogenesis (e.g., anti-VEGF blocking mAb bevacizumab), stimulating apoptosis (e.g., anti-CD20 mAb rituximab) or delivering lethal doses of radioisotopes (e.g., ibritumomab tiuxetan), or toxins to tumour sites (e.g., brentuximab vedotin, an anti-CD30 mAb conjugated to anti-microtubule agent monomethyl auristatin E). Other mAbs induce tumour killing by immune-mediated antibody-dependent cellular cytotoxicity (ADCC)/complement-dependent cytotoxicity (CDC, e.g., rituximab, trastuzumab) and immune checkpoint inhibition through targeting of PD-1/PD-L1 and CTLA-4 (e.g., nivolumab, pembrolizumab, atezolizumab, ipilimumab). Finally, other therapeutic mAbs are used as component of CAR-T cells, or as bispecific antibodies that induce tumour killing by simultaneous targeting of two different antigens on tumour cells, or bispecific immune cell engager by targeting one antigen on tumour cells and another antigen on T cells (e.g., catumaxomab, blinatumomab, Figure 1).
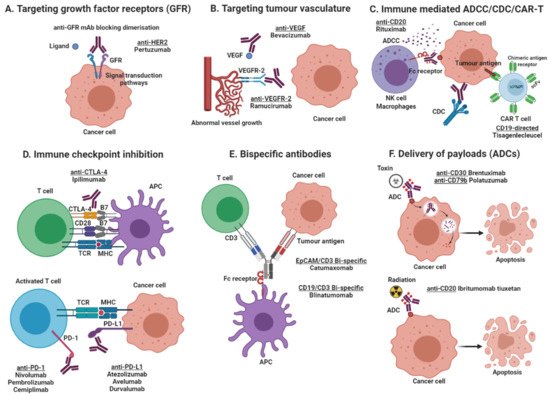
Figure 1. Mechanisms of action of monoclonal antibody-based products. (A) Targeting growth factor receptors, blocking the binding of an activating ligand and inhibiting receptor homo- and heterodimerization; (B) Targeting of tumour vasculature receptor or its ligands inhibiting angiogenesis; (C) Induction of apoptosis by recruitment of immune effector cells (ADCC) or activation of the complement cascade (CDC), and the use of antibody-based molecules to engineer T lymphocytes (CAR T cells); (D) Immune checkpoint inhibition by blockade of the PD-1/PD-L1 axis or CTLA-4 inhibitory receptors, increasing cytotoxic T cell activity; (E) Simultaneous targeting of two antigens, one on tumour cells and one on effector T cells, by using bispecific antibodies (BITE, bispecific T-cell enhancing); and (F) Delivery of payloads such as toxins and radioisotopes to tumour cells. Created with BioRender.com (accessed on 17 March 2021).
To date, 45 mAbs have been approved in the USA and/or the European Union (EU) for the treatment of patients with a wide range of cancers (Table 1). In particular, there has been a great deal of research interest in this area in recent years and a growing number of mAb approvals for different indications. Indeed, with the exception of checkpoint inhibitors, nearly half of the approved therapeutic antibodies are directed against one of the following six target antigens: CD19, CD20, the two members of the human epidermal growth factor receptor (HER) family namely EGFR and HER-2, VEGF and VEGFR (Table 1). Interestingly, several immune checkpoint inhibitors such as anti-CTLA-4 mAb ipilumumab, anti-PD-1 mAbs pembrolizumab and nivolumab, and anti-PD-L1 mAbs avelumab and durvalumab have been approved for a wide range of cancer types. Moreover, additional mAbs have been approved outside the USA and EU for treatment of various cancer types including nimotuzumab (in head and neck cancer, nasopharyngeal cancer and glioma) and vivatuxin (in lung cancer) [14].
Table 1. Monoclonal antibodies approved in the U.S. and/or European Union for cancer treatment.
| MAb Generic Name (Trade Name) | Target Antigen/Isotype | Cancer Type Indication | Date of Approval |
|---|
| Rituximab (Rituxan®) | CD20/Chimeric IgG1 | B-cell lymphoma, NHL | 1997 |
| Diffuse large B-cell, CD20+, NHL | 2006 | ||
| CLL | 2010 | ||
| Previously untreated follicular, CD20+, B-cell NHL | 2011 | ||
| Trastuzumab (Herceptin®) | HER-2/Humanised IgG1 | Metastatic breast cancer | 1998 |
| Early-stage breast cancer | 2006 | ||
| HER2 overexpressing metastatic gastric or GEJ adenoca | 2010 | ||
| Gentuzumab ozogamicin (Mylotarg®) | CD33/Humanised IgG4 | AML | 2000 * |
| Newly diagnosed, relapsed or refractory CD33+ AML | 2017 | ||
| Alemtuzumab (Campath®) | CD52/Humanised IgG1 | B-CLL | 2001 |
| Ibritumomab tiuxetan (Zevalin®) | CD20/Murine IgG1; conjugated to 90Y | NHL | 2002 |
| Tositumomab-I131 (Bexxar®) | CD20/Murine IgG2a; conjugated to 131I | NHL | 2003 * |
| Cetuximab (Erbitux®) | EGFR/Chimeric IgG1 | Metastatic CRC | 2004 |
| Locally or regionally advanced HNSCC or recurrent or metastatic HNSCC | 2006 | ||
| Recurrent locoregional disease and/or metastatic HNSCC (first-line) | 2011 | ||
| K-ras wild-type, EGFR-expressing metastatic CRC (first-line) | 2012 | ||
| Bevacizumab (Avastin®) | VEGF/Humanised IgG1 | Metastatic CRC | 2004 |
| Locally advanced, metastatic or recurrent NSCLC | 2006 | ||
| Metastatic HER-2 negative breast cancer | 2008 | ||
| Metastatic RCC | 2009 | ||
| GBM | 2009 | ||
| Metastatic CRC | 2013 | ||
| Persistent, recurrent or metastatic cervical cancer | 2014 | ||
| Platinum-resistant, recurrent epithelial ovarian, fallopian tube, or primary peritoneal cancer | 2014 | ||
| Stage III or IV epithelial ovarian, fallopian tube, or primary peritoneal cancer after initial surgical resection | 2018 | ||
| Panitumumab (Vectibix®) | EGFR/Human IgG2 | Metastatic CRC | 2006 |
| nitumumab (Vectibix®) | EGFR/Human IgG2 | Metastatic CRC | 2006 |
| Ofatumumab (Arzerra®) | CD20/Human IgG1 | CLL | 2009 |
| CLL (previously untreated) | 2014 | ||
| Recurrent or progressive CLL | 2016 | ||
| Catumaxomab (Removab®) | EpCAM/ CD3/Bi-specific Rat IgG2b/mouse IgG2a |
Malignant ascites (in Europe) | 2009 |
| Ipilimumab (Yervoy®) | CTLA-4/Human IgG1 | Unresectable or metastatic melanoma | 2011 |
| Cutaneous melanoma with pathological involvement of regional lymph nodes | 2015 | ||
| Intermediate or poor risk, previously untreated advanced RCC (in combination with nivolumab) | 2018 | ||
| MSI-H or dMMR metastatic CRC (in combination with nivolumab) | 2018 | ||
| Brentuximab vedotin (Adcentris®) | CD30/Chimeric IgG1; conjugated to monomethyl auristatin E | ALCL and HL | 2011 |
| cHL (as consolidation post-auto-HSCT) | 2015 | ||
| pcALCL or CD30-expressing MF | 2017 | ||
| Previously untreated stage III or IV cHL | 2018 | ||
| Previously untreated systemic ALCL or other CD30-expressing peripheral T-cell lymphomas | 2018 | ||
| Pertuzumab (Perjecta®) | HER-2/Humanised IgG1 | HER-2 positive metastatic breast cancer | 2012 |
| HER-2 positive, locally advanced, inflammatory, or early-stage breast cancer (in combination with trastuzumab as neoadjuvant therapy) | 2013 | ||
| HER-2 positive early breast cancer at high risk or recurrence | 2017 | ||
| Denosumab (Xgeva®) | RANKL/Human IgG2 | Unresectable giant cell tumour of bone | 2013 |
| Ado-trastuzumab emtansine (Kadcyla®) | HER-2/Humanised IgG1; conjugated to DM1 | HER-2 positive, metastatic breast cancer | 2013 |
| HER-2 positive breast cancer with residual invasive disease | 2019 | ||
| Obinutuzumab (Gazyva®) | CD20/Humanised IgG1 | CLL (previously untreated) | 2013 |
| FL | 2016 | ||
| Previously untreated stage II bulky, III or IV FL | 2017 | ||
| Ramucirumab (Cyramza®) | VEGFR-2/Recombinant IgG1 | Advanced or metastatic, gastric or GEJ adenocarcinoma | 2014 |
| Metastatic NSCLC | 2014 | ||
| Metastatic CRC | 2015 | ||
| HCC | 2019 | ||
| First-line treatment of metastatic NSCLC (in combination with erlotinib) | 2020 | ||
| Pembrolizumab (Keytruda®) |
PD-1 receptor/Humanised IgG4 | Unresectable or metastatic melanoma and disease progression following ipilimumab | 2014 |
| Unresectable and metastatic melanoma (initial treatment) | 2015 | ||
| Metastatic NSCLC | 2016 | ||
| Recurrent or metastatic HNSCC | 2016 | ||
| Refractory cHL | 2017 | ||
| Previously untreated metastatic non-squamous NSCLC | 2017 | ||
| Locally advanced or metastatic urothelial carcinoma | 2017 | ||
| Unresectable or metastatic MSI-H or dMMR solid tumours | 2017 | ||
| Recurrent locally advanced or metastatic gastric or GEJ adenocarcinoma | 2017 | ||
| Recurrent or metastatic cervical cancer | 2018 | ||
| Refractory PMBCL | 2018 | ||
| First-line treatment metastatic non-squamous NSCLC | 2018 | ||
| First-line treatment metastatic squamous NSCLC | 2018 | ||
| HCC | 2018 | ||
| Recurrent locally advanced or metastatic Merkel cell carcinoma | 2018 | ||
| Melanoma with involvement of lymph nodes following complete resection | 2019 | ||
| First-line treatment stage III or metastatic NSCLC | 2019 | ||
| First-line treatment advanced RCC | 2019 | ||
| Metastatic or unresectable recurrent HNSCC | 2019 | ||
| Metastatic or unresectable recurrent | 2019 | ||
| Metastatic SCLC | 2019 | ||
| HNSCC | 2019 | ||
| Advanced oesophageal squamous cell cancer | 2019 | ||
| Metastatic SCLC | 2019 | ||
| Advanced endometrial carcinoma that is not MSI-H or dMMR | 2019 | ||
| Advanced oesophageal squamous cell cancer | 2019 | ||
| BCG-unresponsive, high-risk, non-muscle invasive bladder cancer with carcinoma in situ with or without papillary tumours | 2020 | ||
| Advanced endometrial carcinoma that is not MSI-H or dMMR | 2019 | ||
| Unresectable or metastatic tumour mutational burden-high (TMB H) solid tumours | 2020 | ||
| BCG-unresponsive, high-risk, non-muscle invasive bladder cancer with carcinoma in situ with or without papillary tumours | 2020 | ||
| Recurrent or metastatic CSCC not curable by surgery or radiation | 2020 | ||
| Unresectable or metastatic tumour mutational burden-high (TMB H) solid tumours | 2020 | ||
| First-line treatment unresectable or metastatic MSI-H or dMMR CRC | 2020 | ||
| Recurrent or metastatic CSCC not curable by surgery or radiation | 2020 | ||
| R/R cHL | 2020 | ||
| First-line treatment unresectable or metastatic MSI-H or dMMR CRC | 2020 | ||
| Locally recurrent unresectable or metastatic TNBC whose tumours express PD-L1 | 2020 | ||
| R/R cHL | 2020 | ||
| Blinatumomab (Blincyto®) | CD19/Bispecific CD19-directed CD3 T-cell engager | Philadelphia chromosome-negative R/R B-cell precursor ALL | 2014 |
| Locally recurrent unresectable or metastatic TNBC whose tumours express PD-L1 | 2020 | ||
| R/R B-cell precursor ALL | 2017 | ||
| Blinatumomab (Blincyto®) | CD19/Bispecific CD19-directed CD3 T-cell engager | Philadelphia chromosome-negative R/R B-cell precursor ALL | 2014 |
| B-cell precursor ALL in first or second complete remission with MRD >/= 0.1% | 2018 | ||
| R/R B-cell precursor ALL | 2017 | ||
| Nivolumab (Opdivo®) | PD-1 receptor/Human IgG4 | Unresectable or metastatic melanoma | 2014 |
| B-cell precursor ALL in first or second complete remission with MRD >/= 0.1% | 2018 | ||
| BRAF V600 wild-type, unresectable or metastatic melanoma (in combination with ipilimumab) | 2015 | ||
| Nivolumab (Opdivo®) | PD-1 receptor/Human IgG4 | Unresectable or metastatic melanoma | 2014 |
| Metastatic NSCLC | 2015 | ||
| BRAF V600 wild-type, unresectable or metastatic melanoma (in combination with ipilimumab) | 2015 | ||
| Advanced RCC | 2015 | ||
| Metastatic NSCLC | 2015 | ||
| cHL | 2016 | ||
| Advanced RCC | 2015 | ||
| Recurrent or metastatic HNSCC | 2016 | ||
| cHL | 2016 | ||
| Locally advanced or metastatic urothelial carcinoma | 2017 | ||
| Recurrent or metastatic HNSCC | 2016 | ||
| MSI-H or dMMR metastatic CRC | 2017 | ||
| Locally advanced or metastatic urothelial carcinoma | 2017 | ||
| HCC | 2017 | ||
| MSI-H or dMMR metastatic CRC | 2017 | ||
| Melanoma with involvement of lymph nodes or metastatic disease following complete resection | |||
| HCC | |||
| 2017 | |||
| 2017 | |||
| Intermediate or poor risk, previously untreated advanced RCC (in combination with ipilimumab) | 2018 | ||
| Melanoma with involvement of lymph nodes or metastatic disease following complete resection | 2017 | ||
| Metastatic SCLC | 2018 | ||
| Intermediate or poor risk, previously untreated advanced RCC (in combination with ipilimumab) | 2017 | ||
| HCC (in combination with ipilimumab) | 2018 | ||
| Metastatic SCLC | 2018 | ||
| First-line treatment metastatic NSCLC whose tumours express PD-L1 (in combination with ipilimumab) | 2020 | ||
| HCC (in combination with ipilimumab) | 2018 | ||
| Unresectable advanced, recurrent or metastatic oesophageal squamous cell carcinoma (ESCC) | 2020 | ||
| First-line treatment metastatic NSCLC whose tumours express PD-L1 (in combination with ipilimumab) | 2018 | ||
| Unresectable malignant pleural mesothelioma (first-line; in combination with ipilimumab) | 2020 | ||
| Unresectable advanced, recurrent or metastatic oesophageal squamous cell | 2020 | ||
| First-line treatment advanced renal cell carcinoma (in combination with cabozantinib) | 2021 | ||
| carcinoma (ESCC) | 2020 | ||
| Dinutuximab (Unituxin®) | GD2/Chimeric IgG1 | High-risk neuroblastoma | 2015 |
| Unresectable malignant pleural mesothelioma (first-line; in combination with ipilimumab) | 2020 | ||
| Daratumumab (Darzalex®) | CD38/Human IgG1 | MM | 2015 |
| First-line treatment advanced renal cell carcinoma (in combination with cabozantinib) | 2021 | ||
| Newly diagnosed MM ineligible for autologous SCT | 2019 | ||
| Dinutuximab (Unituxin®) | GD2/Chimeric IgG1 | High-risk neuroblastoma | 2015 |
| Newly diagnosed MM eligible for autologous SCT | 2019 | ||
| Daratumumab (Darzalex®) | CD38/Human IgG1 | MM | 2015 |
| Necitumumab (Portrazza®) | EGFR/Human IgG1 | Metastatic squamous NSCLC (first-line) | 2015 |
| Newly diagnosed MM ineligible for autologous SCT | 2019 | ||
| Elotuzumab (Empliciti®) |
SLAMF7/Humanised IgG1 | MM | 2015 |
| Newly diagnosed MM eligible for autologous SCT | 2019 | ||
| Atezolizumab (Tecentriq®) | PD-L1/Humanised IgG1 | Locally advanced or metastatic urothelial carcinoma | 2016 |
| Necitumumab (Portrazza®) | EGFR/Human IgG1 | Metastatic squamous NSCLC (first-line) | 2015 |
| Metastatic NSCLC | 2016 | ||
| Elotuzumab (Empliciti®) |
SLAMF7/Humanised IgG1 | MM | 2015 |
| First-line treatment metastatic non-squamous NSCLC | 2018 | ||
| Atezolizumab (Tecentriq®) | PD-L1/Humanised IgG1 | Locally advanced or metastatic urothelial carcinoma | 2016 |
| Unresectable locally-advanced or metastatic TNBC | 2019 | ||
| Metastatic NSCLC | 2016 | ||
| Extensive-stage SCLC | 2019 | ||
| First-line treatment metastatic non-squamous NSCLC | 2018 | ||
| Unresectable or metastatic HCC (in combination with bevacizumab) | 2020 | ||
| Unresectable locally-advanced or metastatic TNBC | 2019 | ||
| BRAF V600 mutation-positive unresectable or metastatic melanoma | 2020 | ||
| Extensive-stage SCLC | 2019 | ||
| Olaratumab (Lartruvo®) |
PDGFRα/Human IgG1 | Metastatic soft-tissue sarcoma | 2016 |
| Unresectable or metastatic HCC (in combination with bevacizumab) | 2020 | ||
| Avelumab (Bavencio®) | PD-L1/Human IgG1 | Metastatic Merkel cell carcinoma | 2017 |
| BRAF V600 mutation-positive unresectable or metastatic melanoma | 2020 | ||
| Locally advanced or metastatic urothelial carcinoma | 2017 | ||
| Olaratumab (Lartruvo®) |
PDGFRα/Human IgG1 | Metastatic soft-tissue sarcoma | 2016 |
| Advanced RCC | 2019 | ||
| Avelumab (Bavencio®) | PD-L1/Human IgG1 | Metastatic Merkel cell carcinoma | 2017 |
| Maintenance treatment in locally advanced or metastatic urothelial carcinoma (UC) | 2020 | ||
| Locally advanced or metastatic urothelial carcinoma | 2017 | ||
| Durvalumab (Imfinzi®) | PD-L1/Human IgG1 | Locally advanced or metastatic urothelial carcinoma | 2017 |
| Advanced RCC | 2019 | ||
| Unresectable stage III NSCLC | 2018 | ||
| Maintenance treatment in locally advanced or metastatic urothelial carcinoma (UC) | 2020 | ||
| Extensive-stage SCLC | 2020 | ||
| Durvalumab (Imfinzi®) | PD-L1/Human IgG1 | Locally advanced or metastatic urothelial carcinoma | 2017 |
| Rituximab+hyaluronidase human (Rituxan hycela®) | CD20/Chimeric IgG1 | Follicular lymphoma, DLBCL and CLL | 2017 |
| Unresectable stage III NSCLC | 2018 | ||
| Inotuzumab ozogamicin (Besponsa®) | CD22/Humanised IgG4; conjugated with calicheamicin | R/R B-cell precursor ALL | 2017 |
| Extensive-stage SCLC | 2020 | ||
| Mogamulizumab (Poteligeo®) | CCR4/Humanised IgG1 | R/R mycosis fungoides or Sezary syndrome | 2018 |
| Rituximab+hyaluronidase human (Rituxan hycela®) | CD20/Chimeric IgG1 | Follicular lymphoma, DLBCL and CLL | 2017 |
| Moxetumomab pasudotox-tdfk (Lumoxiti®) | CD-22/Immunotoxin; IgG1 fragment fused to Pseudomonas exotoxin PE38 | R/R hairy cell leukaemia | 2018 |
| Inotuzumab ozogamicin (Besponsa®) | CD22/Humanised IgG4; conjugated with calicheamicin | R/R B-cell precursor ALL | 2017 |
| Cemiplimab-rwlc (Libtayo®) | PD-1/Human IgG4 | Metastatic CSCC or locally advanced CSCC who are not candidates for curative surgery or curative radiation | 2018 |
| Mogamulizumab (Poteligeo®) | CCR4/Humanised IgG1 | R/R mycosis fungoides or Sezary syndrome | 2018 |
| Locally advanced and metastatic basal cell carcinoma | 2021 | ||
| Moxetumomab pasudotox-tdfk (Lumoxiti®) | CD-22/Immunotoxin; IgG1 fragment fused to Pseudomonas exotoxin PE38 | R/R hairy cell leukaemia | 2018 |
| First-line treatment of advanced NSCLC whose tumors have high PD-L1 expression | 2021 | ||
| Cemiplimab-rwlc (Libtayo®) | PD-1/Human IgG4 | Metastatic CSCC or locally advanced CSCC who are not candidates for curative surgery or curative radiation | 2018 |
| Trastuzumab + hyaluronidase oysk (Herceptin Hylecta®) | HER-2/Humanised IgG1 | HER-2 overexpressing breast cancer | 2019 |
| Locally advanced and metastatic basal cell carcinoma | 2021 | ||
| Polatuzumab vedotin-piiq (Polivy®) | CD79b-directed ADC; conjugated to MMAE | R/R DLBCL | 2019 |
| First-line treatment of advanced NSCLC whose tumors have high PD-L1 expression | 2021 | ||
| Enfortumab vedotin-ejfv (Padcev®) | Nectin-4-directed ADC; conjugated to MMAE | Locally advanced or metastatic urothelial cancer | 2019 |
| Trastuzumab + hyaluronidase oysk (Herceptin Hylecta®) | HER-2/Humanised IgG1 | HER-2 overexpressing breast cancer | 2019 |
| Fam-trastuzumab deruxtecan-nxki (Enhertu®) | HER-2 directed ADC; conjugated to topoisomerase inhibitor | Unresectable or metastatic HER2-positive breast cancer | 2019 |
| Polatuzumab vedotin-piiq (Polivy®) | CD79b-directed ADC; conjugated to MMAE | R/R DLBCL | 2019 |
| Locally advanced or metastatic HER2-positive gastric or GEJ adenocarcinoma | 2021 | ||
| Enfortumab vedotin-ejfv (Padcev®) | Nectin-4-directed ADC; conjugated to MMAE | Locally advanced or metastatic urothelial cancer | 2019 |
| Isatuximab-irfc (Sarclisa®) | CD38 | MM | 2020 |
| Fam-trastuzumab deruxtecan-nxki (Enhertu®) | HER-2 directed ADC; conjugated to topoisomerase inhibitor | Unresectable or metastatic HER2-positive breast cancer | 2019 |
| Sacituzumab govitecan-hziy (Trodelvy®) | Trop-2 directed ADC; conjugated to SN-38 | Metastatic TNBC | 2020 |
| Locally advanced or metastatic HER2-positive gastric or GEJ adenocarcinoma | 2021 | ||
| Daratumumab and hyaluronidase-fihj (Darzalex faspro®) | CD38/Human IgG1 | Newly diagnosed or R/R MM | 2020 |
| Isatuximab-irfc (Sarclisa®) | CD38 | MM | 2020 |
| Tafasitamab-cxix (Monjuvi®) | CD19/Humanised Fc-modified cytolytic | R/R DLBCL | 2020 |
| Sacituzumab govitecan-hziy (Trodelvy®) | Trop-2 directed ADC; conjugated to SN-38 | Metastatic TNBC | 2020 |
| Belantamab mafodotin-blmf (Blenrep®) | BCMA/ADC conjugated to microtubule inhibitor monomethyl auristatin F (MMAF) | R/R MM | 2020 |
| Daratumumab and hyaluronidase-fihj (Darzalex faspro®) | CD38/Human IgG1 | Newly diagnosed or R/R MM | 2020 |
| Naxitamab (Danyelza®) | GD2/Humanised IgG1 | R/R high-risk neuroblastoma in the bone or bone marrow (in combination with GM-CSF) | 2020 |
| Tafasitamab-cxix (Monjuvi®) | CD19/Humanised Fc-modified cytolytic | R/R DLBCL | 2020 |
| Margetuximab-cmkb (Margenza®) | HER2/Fc engineered chimeric IgG1 | Metastatic HER2-positive breast cancer | 2020 |
| Belantamab mafodotin-blmf (Blenrep®) | BCMA/ADC conjugated to microtubule inhibitor monomethyl auristatin F (MMAF) | R/R MM | 2020 |
| Naxitamab (Danyelza®) |
| GD2/Humanised IgG1 |
| R/R high-risk neuroblastoma in the bone or bone marrow (in combination with GM-CSF) |
| 2020 |
| Margetuximab-cmkb (Margenza®) |
| HER2/Fc engineered chimeric IgG1 |
| Metastatic HER2-positive breast cancer |
| 2020 |
ADC: antibody drug conjugate; ALCL: anaplastic large cell lymphoma; ALL: acute lymphoblastic leukaemia; AML: acute myeloid leukaemia; auto-HSCT: autologous hematopoietic stem cell transplantation; BCG: Bacillus Calmette-Guerin; B-CLL: B-cell chronic lymphocytic leukaemia; BCMA: B-cell maturation antigen; cHL: classical Hodgkin lymphoma; CLL: chronic lymphocytic leukaemia; CRC: colorectal cancer; CSCC: cutaneous squamous cell carcinoma; CTLA-4: cytotoxic T lymphocyte antigen-4; DLBCL: diffuse large B-cell lymphoma; dMMR: mismatch repair deficient; EGFR: epidermal growth factor receptor; EpCAM: epithelial cell adhesion molecule; FL: follicular lymphoma; GBM: glioblastoma multiforme; GD2: surface disialoganglioside GD2; GEJ: gastroesophageal junction; GM-CSF: granulocyte-macrophage colony-stimulating factor; HCC: hepatocellular carcinoma; HER-2: human epidermal growth factor receptor-2; HNSCC: head and neck squamous cell carcinoma; MM: multiple myeloma; MMAE: monomethyl auristatin E; MRD: minimal residual disease; MSI-H: microsatellite instability-high; NHL: non-Hodgkin lymphoma; NSCLC: non-small-cell lung cancer; pcALCL: primary cutaneous anaplastic large cell lymphoma; PDGFRα: platelet-derived growth factor receptor alpha; PD-1: programmed death-1 receptor; PD-L1: programmed death ligand-1; PMBCL: primary mediastinal large B-cell lymphoma; RANKL: receptor activator of nuclear factor-kappa B ligand; RCC: renal cell carcinoma; R/R: relapsed or refractory; SCLC: small cell lung cancer; SCT: stem cell transplant; SLAMF7: signalling lymphocytic activation molecule F7; TNBC: triple-negative breast cancer. VEGF: vascular endothelial growth factor; VEGFR-2: vascular endothelial growth factor receptor 2. * withdrawn; Taken from: https://www.fda.gov/drugs/resources-information-approved-drugs/hematologyoncology-cancer-approvals-safety-notifications. Updated as of 12 March 2021.
However, despite such advances, to date no antibody-based drugs have been approved for the treatment of patients with pancreatic cancer [19]. Some of the contributing factors are the harsh desmoplastic microenvironment of pancreatic cancer, the heterogeneous nature of tumours and the lack of reliable predictive biomarkers and companion diagnostic tests to select patients who are more likely to respond to such therapy [20][21][22][23]. In the following sections, we discuss the results of preclinical studies and clinical trials with antibody-based agents in pancreatic cancer. We will also highlight the importance of antibody-based technology and other approaches in the discovery of cell surface antigens with high levels of expression in pancreatic cancer (i.e., additional therapeutic targets) and in the development of mAb-based targeted therapy for patients with pancreatic cancer.
References
- McGuigan, A.; Kelly, P.; Turkington, R.C.; Jones, C.; Coleman, H.G.; McCain, R.S. Pancreatic cancer: A review of clinical diagnosis, epidemiology, treatment and outcomes. World J. Gastroenterol. 2018, 24, 4846–4861.
- Rahib, L.; Smith, B.D.; Aizenberg, R.; Rosenzweig, A.B.; Fleshman, J.M.; Matrisian, L.M. Projecting Cancer Incidence and Deaths to 2030: The Unexpected Burden of Thyroid, Liver, and Pancreas Cancers in the United States. Cancer Res. 2014, 74, 2913–2921.
- Rawla, P.; Sunkara, T.; Gaduputi, V. Epidemiology of Pancreatic Cancer: Global Trends, Etiology and Risk Factors. World J. Oncol. 2019, 10, 10–27.
- Burris, H.A.; Moore, M.J.; Andersen, J.; Green, M.R.; Rothenberg, M.L.; Modiano, M.R.; Cripps, M.C.; Portenoy, R.K.; Storniolo, A.M.; Tarassoff, P.; et al. Improvements in survival and clinical benefit with gemcitabine as first-line therapy for patients with advanced pancreas cancer: A randomized trial. J. Clin. Oncol. 1997, 15, 2403–2413.
- Neoptolemos, J.P.; Palmer, D.H.; Ghaneh, P.; Psarelli, E.E.; Valle, J.W.; Halloran, C.M.; Faluyi, O.; O’Reilly, D.A.; Cunningham, D.; Wadsley, J.; et al. Comparison of adjuvant gemcitabine and capecitabine with gemcitabine monotherapy in patients with resected pancreatic cancer (ESPAC-4): A multicentre, open-label, randomised, phase 3 trial. Lancet 2017, 389, 1011–1024.
- Neoptolemos, J.P.; Kleeff, J.; Michl, P.; Costello, E.; Greenhalf, W.; Palmer, D.H. Therapeutic developments in pancreatic cancer: Current and future perspectives. Nat. Rev. Gastroenterol. Hepatol. 2018, 15, 333–348.
- Conroy, T.; Desseigne, F.; Ychou, M.; Bouché, O.; Guimbaud, R.; Bécouarn, Y.; Adenis, A.; Raoul, J.L.; Gourgou-Bourgade, S.; de la Fouchardière, C.; et al. FOLFIRINOX versus Gemcitabine for Metastatic Pancreatic Cancer. N. Engl. J. Med. 2011, 364, 1817–1825.
- Von Hoff, D.D.; Ervin, T.; Arena, F.P.; Chiorean, E.G.; Infante, J.; Moore, M.; Seay, T.; Tjulandin, S.A.; Ma, W.W.; Saleh, M.N.; et al. Increased Survival in Pancreatic Cancer with nab-Paclitaxel plus Gemcitabine. N. Engl. J. Med. 2013, 369, 1691–1703.
- Moore, M.J.; Goldstein, D.; Hamm, J.; Figer, A.; Hecht, J.R.; Gallinger, S.; Au, H.J.; Murawa, P.; Walde, D.; Wolff, R.A.; et al. Erlotinib plus gemcitabine compared with gemcitabine alone in patients with advanced pancreatic cancer: A phase III trial of the National Cancer Institute of Canada Clinical Trials Group. J. Clin. Oncol. 2007, 25, 1960–1966.
- Wang-Gillam, A.; Li, C.-P.; Bodoky, G.; Dean, A.; Shan, Y.-S.; Jameson, G.; Macarulla, T.; Lee, K.H.; Cunningham, D.; Blanc, J.F.; et al. Nanoliposomal irinotecan with fluorouracil and folinic acid in metastatic pancreatic cancer after previous gemcitabine-based therapy (NAPOLI-1): A global, randomised, open-label, phase 3 trial. Lancet 2016, 387, 545–557.
- Köhler, G.; Milstein, C. Continuous cultures of fused cells secreting antibody of predefined specificity. Nature 1975, 256, 495–497.
- Modjtahedi, H.; Ali, S.; Essapen, S. Therapeutic application of monoclonal antibodies in cancer: Advances and challenges. Br. Med. Bull. 2012, 104, 41–59.
- Pillay, V.; Gan, H.K.; Scott, A.M. Antibodies in oncology. N. Biotechnol. 2011, 28, 518–529.
- Scott, A.M.; Allison, J.P.; Wolchok, J.D. Monoclonal antibodies in cancer therapy. Cancer Immun. 2012, 12, 14.
- Lu, R.M.; Hwang, Y.C.; Liu, I.J.; Lee, C.C.; Tsai, H.Z.; Li, H.J.; Wu, H.C. Development of therapeutic antibodies for the treatment of diseases. J. Biomed. Sci. 2020, 27, 1.
- Huang, S.; van Duijnhoven, S.M.J.; Sijts, A.; van Elsas, A. Bispecific antibodies targeting dual tumor-associated antigens in cancer therapy. J. Cancer. Res. Clin. Oncol. 2020, 146, 3111–3122.
- Buss, N.A.P.S.; Henderson, S.J.; McFarlane, M.; Shenton, J.M.; de Haan, L. Monoclonal antibody therapeutics: History and future. Curr. Opin. Pharmacol. 2012, 12, 615–622.
- Modjtahedi, H. Monoclonal Antibodies as Therapeutic Agents: Advances and Challenges. Iran. J. Immunol. 2005, 2, 3–20.
- Kaplon, H.; Reichert, J.M. Antibodies to watch in 2021. mAbs 2021, 13, 1860476.
- Neesse, A.; Bauer, C.A.; Öhlund, D.; Lauth, M.; Buchholz, M.; Michl., P.; Tuveson, D.A.; Gress, T.M. Stromal biology and therapy in pancreatic cancer: Ready for clinical translation? Gut 2019, 68, 159–171.
- Cros, J.; Raffenne, J.; Couvelard, A.; Poté, N. Tumor Heterogeneity in Pancreatic Adenocarcinoma. Pathobiology 2018, 85, 64–71.
- Duffy, M.J.; Crown, J. Companion biomarkers: Paving the pathway to personalized treatment for cancer. Clin. Chem. 2013, 59, 1447–1456.
- Agarwal, A.; Ressler, D.; Snyder, G. The current and future state of companion diagnostics. Pharmgenom. Pers. Med. 2015, 8, 99–110.
