The high prevalence of type 2 diabetes mellitus (T2DM), together with the fact that current treatments are only palliative and do not avoid major secondary complications, reveals the need for novel approaches to treat the cause of this disease. Efforts are currently underway to identify therapeutic targets implicated in either the regeneration or re-differentiation of a functional pancreatic islet β-cell mass to restore insulin levels and normoglycemia. However, T2DM is not only caused by failures in β-cells but also by dysfunctions in the central nervous system (CNS), especially in the hypothalamus and brainstem.
- metabesity
- T2DM
- obesity
- inflammation
- pancreatic islet
- astrocytes
1. Introduction
Diabetes mellitus (DM) is currently a major social and economic burden worldwide. The global epidemic proportion of DM is one of the most important health problems of the 21st century, being the cause of four million deaths in 2017 [1]. According to the International Diabetes Federation, 425 million people were affected by DM in 2017 (8.8% worldwide prevalence), and this number is expected to rise to 629 million people by 2045 [1]. This alarming increase in the incidence of DM is mainly due to the increase in the number of type 2 DM (T2DM) sufferers, which accounts for 90–95% of all DM. T2DM is associated with environmental and nutritional factors, as well as lifestyle, operating on a genetic susceptibility background. Approximately 80% of T2DM patients are obese, and the increase in the global epidemic of obesity explains the dramatic explosion of T2DM incidence over the past two decades [1]. Over nutrition-derived metabolic dysfunctions, henceforth denoted as metabesity, contributes to the apparition of a chronic low-grade inflammation [2,3], which is recognized as the pathogenic link with T2DM. This inflammation initially targets peripheral tissues specialized in metabolism such as liver, adipose tissue, and pancreatic islets, and it plays a key role in insulin resistance and β-cell dysfunction [4,5,6]. Inflammation also affects the central nervous system (CNS), likely being one of the causes underlying the increased incidence of neurodegenerative diseases among T2DM patients. Of note, hypothalamic inflammation is arising as a key pathophysiological process in metabesity, linked to hypothalamic leptin and insulin resistance [7,8,9].
Diabetes mellitus (DM) is currently a major social and economic burden worldwide. The global epidemic proportion of DM is one of the most important health problems of the 21st century, being the cause of four million deaths in 2017 [1]. According to the International Diabetes Federation, 425 million people were affected by DM in 2017 (8.8% worldwide prevalence), and this number is expected to rise to 629 million people by 2045 [1]. This alarming increase in the incidence of DM is mainly due to the increase in the number of type 2 DM (T2DM) sufferers, which accounts for 90–95% of all DM. T2DM is associated with environmental and nutritional factors, as well as lifestyle, operating on a genetic susceptibility background. Approximately 80% of T2DM patients are obese, and the increase in the global epidemic of obesity explains the dramatic explosion of T2DM incidence over the past two decades [1]. Over nutrition-derived metabolic dysfunctions, henceforth denoted as metabesity, contributes to the apparition of a chronic low-grade inflammation [2][3], which is recognized as the pathogenic link with T2DM. This inflammation initially targets peripheral tissues specialized in metabolism such as liver, adipose tissue, and pancreatic islets, and it plays a key role in insulin resistance and β-cell dysfunction [4][5][6]. Inflammation also affects the central nervous system (CNS), likely being one of the causes underlying the increased incidence of neurodegenerative diseases among T2DM patients. Of note, hypothalamic inflammation is arising as a key pathophysiological process in metabesity, linked to hypothalamic leptin and insulin resistance [7][8][9].
T2DM is a progressive metabolic disorder classically defined by chronic hyperglycemia due to the combination of an abnormal insulin secretion by pancreatic islet β-cells and increased insulin resistance of insulin-target tissues (adipose tissue, skeletal muscle, liver, and brain) [10]. Guidelines for T2DM management include, as first-line therapy, serious lifestyle interventions such as physical exercise, while long-term add-on therapies include medication in order to increase insulin secretion and sensitivity [11]. Although these treatments are adequate to improve hyperglycemia, they alleviate symptoms rather than target the root-cause of the disease and lead to the development of secondary complications [12]. For example, sulfonylureas that impel insulin secretion were shown to cause β-cell death and patients are thenceforth confined to daily insulin injections to control glucose homeostasis [13]. However, new classes of pharmacological agents such as sodium glucose transporter 2 SGLT2 inhibitors and glucagon-like peptide-1 (GLP-1) receptor agonists significantly improve the prognosis of T2DM patients [14]. Nonetheless, a better understanding of T2DM etiology is mandatory to develop more effective therapies to avoid long-term complications and early death associated with this disease, and to restrain the apparition of neurodegenerative disorders such as Alzheimer’s disease, dubbed T3DM [15]. In recent years, the CNS gained much interest as a key regulator of glucose/energy homeostasis [16]. Thus, T2DM is not only caused by failures in pancreatic β-cells but also dysfunctions in the CNS that could lead to the development of this disease. Moreover, metabesity-associated chronic inflammation also affects the CNS-mediated control of blood glucose levels, exacerbating the disease. Herein, we review the evidence that supports a main role for the CNS in glucose homeostasis, with a special focus on astrocytes, and how interventions that simultaneously target both the CNS and pancreatic islets may synergistically improve the regulation of blood glucose levels and metabolism in T2DM patients.
2. Cell Types of the Hypothalamus Implicated in Energy Homeostasis
2.1. Hypothalamic Neurons
Several hypothalamic areas are involved in the control of energy homeostasis and glucose metabolism, including the arcuate (ARC), the ventromedial (VMH), the dorsomedial (DMH), the paraventricular (PVN) nuclei, and the lateral hypothalamic area (LHA) (
Figure 1A) [68]. These nuclei possess glucose sensing neurons that adapt their electrical activity and excitability in response to alterations in extracellular glucose concentrations [48,69,70], triggering response signals to normalize the glycemia. These populations are divided into glucose-excited neurons (GE), which increase their electrical activity when extracellular glucose concentration increased, and glucose-inhibited neurons (GI) which are activated by a decrease in extracellular glucose levels (A) [17]. These nuclei possess glucose sensing neurons that adapt their electrical activity and excitability in response to alterations in extracellular glucose concentrations [18][19][20], triggering response signals to normalize the glycemia. These populations are divided into glucose-excited neurons (GE), which increase their electrical activity when extracellular glucose concentration increased, and glucose-inhibited neurons (GI) which are activated by a decrease in extracellular glucose levels (
Figure 1B) [48,70,71]. GE neurons, located in the VMH, ARC, and PVN [71], use a similar glucose sensing mechanism as pancreatic β-cells [48,72], expressing GLUT-2 [73], GK [50], and KB) [18][20][21]. GE neurons, located in the VMH, ARC, and PVN [21], use a similar glucose sensing mechanism as pancreatic β-cells [18][22], expressing GLUT-2 [23], GK [24], and K
ATP channels that, in response to glucose, lead to membrane depolarization and neurotransmitter exocytosis [74]. In addition, GE neurons can also use alternative glucose sensing mechanisms [73,75,76], such as the sodium-dependent glucose transporters SGLT-1 or-3, which are involved in glucose-induced neuronal activity in GE neurons [72,77,78]. Alternatively, the heterodimeric G-protein-coupled receptors of the T1R family, expressed in several hypothalamic areas [79], are also activated by glucose [72,77]. Moreover, recent studies suggest that GE neurons in the VMN nucleus respond to glucose load with mitochondrial responses that activate a signal transduction cascade that ultimately promotes lowering of systemic glucose [80]. The second neuronal population, GI neurons, which are mainly situated in the LHA, ARC, and PVN nuclei, increase their firing activity under hypoglycemic conditions [48,71]. The mechanism used by GI to sense glucose levels is less clear. One of the proposed models suggests that a reduction in glucose uptake decreases ATP production and Nachannels that, in response to glucose, lead to membrane depolarization and neurotransmitter exocytosis [25]. In addition, GE neurons can also use alternative glucose sensing mechanisms [23][26][27], such as the sodium-dependent glucose transporters SGLT-1 or-3, which are involved in glucose-induced neuronal activity in GE neurons [22][28][29]. Alternatively, the heterodimeric G-protein-coupled receptors of the T1R family, expressed in several hypothalamic areas [30], are also activated by glucose [22][28]. Moreover, recent studies suggest that GE neurons in the VMN nucleus respond to glucose load with mitochondrial responses that activate a signal transduction cascade that ultimately promotes lowering of systemic glucose [31]. The second neuronal population, GI neurons, which are mainly situated in the LHA, ARC, and PVN nuclei, increase their firing activity under hypoglycemic conditions [18][21]. The mechanism used by GI to sense glucose levels is less clear. One of the proposed models suggests that a reduction in glucose uptake decreases ATP production and Na
+/K
+activity, resulting in the increase in cytoplasmic Na
+, which leads to membrane depolarization via activation of a chloride conductance [81]. Alternative models include the involvement of other glucose transporters such as GLUT-1, GLUT-2, or GLUT-3 and AMP, whose concentrations are increased by low levels of glucose, leading to AMPK activation and neuronal firing [82,83]. Noteworthy, both GI and GE neurons co-exist in the same hypothalamic nuclei [84], and are also involved in the control of energy homeostasis and food intake. In this context, GI and GE hypothalamic neurons have a functional endocannabinoid system, which modulates their electrical activity in response to variation in energy and food intake [85,86]., which leads to membrane depolarization via activation of a chloride conductance [32]. Alternative models include the involvement of other glucose transporters such as GLUT-1, GLUT-2, or GLUT-3 and AMP, whose concentrations are increased by low levels of glucose, leading to AMPK activation and neuronal firing [33][34]. Noteworthy, both GI and GE neurons co-exist in the same hypothalamic nuclei [35], and are also involved in the control of energy homeostasis and food intake. In this context, GI and GE hypothalamic neurons have a functional endocannabinoid system, which modulates their electrical activity in response to variation in energy and food intake [36][37].
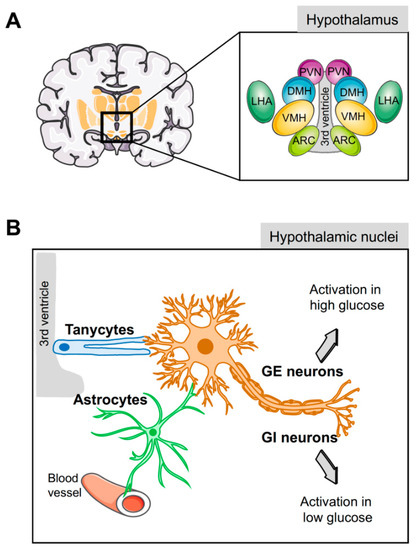
Glucose sensing by the hypothalamus. (
) Schematic representation of the different hypothalamic nuclei and their location surrounding the third ventricle. (
) Glucose-inhibited (GI) and glucose-excited (GE) neurons play a key role in glucose homeostasis; nevertheless, since these neurons are not located in direct contact with blood vessels or cerebrospinal fluid, sensing of circulating glucose levels is relayed through the hypothalamic glial cells (tanycytes and astrocytes). ARC, arcuate nucleus; VMH, ventromedial hypothalamic nucleus; DMH, dorsomedial hypothalamic nucleus; PVN, paraventricular nucleus; LHA, lateral hypothalamic area.
The ARC nucleus, located in the floor of the third ventricle, plays an important role sensing peripheral signals that relay the systemic energy status [17]. Expression of receptors for metabolic hormones, such as leptin, ghrelin, and insulin, [17][38][39][40], and the capacity to sense several nutrients, including glucose and free fatty acids [19][21], allow this hypothalamic nucleus to respond to hormonal and nutritional inputs regulating energy homeostasis and metabolism to compensate for whole-body demands. A network of GE and GI antagonistic neuronal populations that together control the feeding behavior and energy homeostasis composes the ARC nucleus. GI neurons [41] express the orexigenic neuropeptide Y (NPY) and the agouti-related peptide (AgRP), which are stimulated under caloric restriction conditions [42][43] to induce feeding, inhibit energy expenditure, and regulate glucose metabolism [44][45][46]. In contrast, GE neurons [47] are activated in response to caloric excess to inhibit feeding and increase energy expenditure and weight loss [46][48]. This second population expresses anorexigenic proopiomelanocortin (POMC)-derived peptides such as α-melanocyte stimulating hormone (α-MSH) and cocaine-and-amphetamine-regulated transcript (CART). Animal model studies showed that alterations in these neuronal populations affect glucose metabolism in peripheral tissues [49][50]. Acute activation of AgRP neurons impairs both insulin sensitivity and glucose tolerance [51], and reduces energy expenditure [52], while the lack of AgRP neurons increases energy expenditure [53]. Additionally, intracerebroventricular (icv) administration of NPY, mainly produced by ARC neurons, induces insulin secretion [54]. POMC and AgRP neurons from the ARC project into the VMH, which senses hypoglycemia and induces a counter-regulatory response to restore normal glucose levels. This counter-regulatory response is mediated by the steroidogenic-factor 1 (SF1)-expressing neurons [55]. Ablation of these VMH SF1 neurons impairs the recovery from insulin-induced hypoglycemia, while their activation, under normoglycemic conditions, raises blood glucose levels by increasing glucagon secretion and inhibiting glucose-stimulated insulin secretion [56][55].
Although it is clear that GI and GE neurons play a key role in glucose sensing, these glucosensing neurons are not in direct contact with blood or cerebrospinal fluid, suggesting that other actors may be involved in this function [104,105]. In this regard, hypothalamic glial cells arise as central players for glucose sensing due to their strategic anatomical position connecting neurons with blood vessel and cerebrospinal fluid.Although it is clear that GI and GE neurons play a key role in glucose sensing, these glucosensing neurons are not in direct contact with blood or cerebrospinal fluid, suggesting that other actors may be involved in this function [57][58]. In this regard, hypothalamic glial cells arise as central players for glucose sensing due to their strategic anatomical position connecting neurons with blood vessel and cerebrospinal fluid.
2.2. Hypothalamic Glial Cells
Virchow in 1846 initially coined the term neuroglia to describe a connective substance embedding neurons in the brain. Subsequently, the Spanish neuroscientist Ramón y Cajal reported on glial cells and their role in brain physiology [106]. The direct contact of glial cells to blood vessels and glucose sensing neurons raises the possibility that these cells mediate and bridge glucose signaling [105,107,108]. Indeed, the hypothalamic astrocytes and tanycytes, two types of glial cells, participate in the regulation of glucose and energy homeostasis [17,104,107,109] by modulating both peripheral and central glucose levels [60] and providing energy substrates to neighbouring neurons.
Virchow in 1846 initially coined the term neuroglia to describe a connective substance embedding neurons in the brain. Subsequently, the Spanish neuroscientist Ramón y Cajal reported on glial cells and their role in brain physiology [59]. The direct contact of glial cells to blood vessels and glucose sensing neurons raises the possibility that these cells mediate and bridge glucose signaling [58][60][61]. Indeed, the hypothalamic astrocytes and tanycytes, two types of glial cells, participate in the regulation of glucose and energy homeostasis [62][57][60][63] by modulating both peripheral and central glucose levels [64] and providing energy substrates to neighbouring neurons.
Tanycytes are specialised ependymoglial cells surrounding the lateral walls and floor of the third ventricle [110]. Cell bodies of tanycytes are in direct contact with the cerebral spinal fluid (CSF) and have a long process projected into the hypothalamus, a privileged position to sense hormones and nutritional signals from either the periphery or the CSF and relay them to the brain (
Tanycytes are specialised ependymoglial cells surrounding the lateral walls and floor of the third ventricle [65]. Cell bodies of tanycytes are in direct contact with the cerebral spinal fluid (CSF) and have a long process projected into the hypothalamus, a privileged position to sense hormones and nutritional signals from either the periphery or the CSF and relay them to the brain (
Figure 1B) [110]. Astrocytes also have a strategic physiological location in close proximity to the blood–brain barrier (BBB) and, with their end-feet covering the surface of capillaries, support the hypothesis that astrocytes supply neuron energetic demands in a process known as the astrocyte–neuron lactate shuttle (
B) [65]. Astrocytes also have a strategic physiological location in close proximity to the blood–brain barrier (BBB) and, with their end-feet covering the surface of capillaries, support the hypothesis that astrocytes supply neuron energetic demands in a process known as the astrocyte–neuron lactate shuttle (
Figure 1B) [107,111]. In the absence of glucose, the lactate derived from astrocytic glycogen metabolism maintains neuronal function [112].
B) [60][66]. In the absence of glucose, the lactate derived from astrocytic glycogen metabolism maintains neuronal function [67].
Recent evidence highlights the important role of these two hypothalamic glial cells in glucose sensing and glucose homeostasis. Central icv injection of fibroblast growth factor 1 (FGF1) induces diabetes remission in animal models of T2DM in a process mediated by tanycyte activation that enhances insulin-independent glucose clearance [56,113]. Noteworthy, this effect of FGF1 administration is accompanied by a preservation of β-cell function [113]. Additionally, image analyses of rat brain slices showed that, in response to glucose, tanycytes display an increase in intracellular Ca
Recent evidence highlights the important role of these two hypothalamic glial cells in glucose sensing and glucose homeostasis. Central icv injection of fibroblast growth factor 1 (FGF1) induces diabetes remission in animal models of T2DM in a process mediated by tanycyte activation that enhances insulin-independent glucose clearance [68][69]. Noteworthy, this effect of FGF1 administration is accompanied by a preservation of β-cell function [69]. Additionally, image analyses of rat brain slices showed that, in response to glucose, tanycytes display an increase in intracellular Ca
2+ stimulating glycolysis and lactate release [110,114,115,116,117]. This lactate is then relayed to POMC neurons in the ARC triggering exocytosis of αMSH to produce satiety [118]. This mechanism is dependent on the GLUT-2, as inhibition of this transporter specifically in tanycytes disrupts the hypothalamic glucosensing mechanism, reducing glucose uptake and lactate production. Consequently, POMC neuronal activity is interrupted and the control of feeding behavior is altered [119]. Noteworthy, the astrocytic glucose transporters have also a critical role in glucosensing. As an example of this, the re-expression of Glut2 in astrocytes of Glut2 null mice restores glucagon secretion in response to hypoglycemia [120,121], indicating that hypothalamic astrocytes via GLUT-2 are a key part of the central glucose sensing machinery and play a key role in the regulation of glycemia. Likewise, repression of hypothalamic GLUT-1 levels due to sustained hyperglycemia blunted the decrease in systemic glucose production, whereas GLUT-1 over-expression in hypothalamic astrocytes restored glucose-sensing capacity of these cells [122]. Nevertheless, glucose transporters are not the only mechanism used by these glial cells to sense glucose. The fact that tanycytes also respond to non-metabolizable glucose analogs, 2-deoxyglucose and methyl glucopyranoside [115,117], suggests that additional glucosensing mechanisms are implicated. In this regard, G-protein-coupled receptors (GPCRs) were proposed to be implicated in glucose sensing and regulation [123]. Interestingly, the GPCR cannabinoid CB1 receptor, a well-known player in energy homeostasis, is expressed in astrocytes and facilitates neuron–astrocyte communication [124]. Although the role of the CB1 receptor in glucose sensing and homeostasis is still unknown, some evidence points to the regulation of the leptin signaling pathway and glycogen storage in astrocytes [125].
stimulating glycolysis and lactate release [65][70][71][72][73]. This lactate is then relayed to POMC neurons in the ARC triggering exocytosis of αMSH to produce satiety [74]. This mechanism is dependent on the GLUT-2, as inhibition of this transporter specifically in tanycytes disrupts the hypothalamic glucosensing mechanism, reducing glucose uptake and lactate production. Consequently, POMC neuronal activity is interrupted and the control of feeding behavior is altered [75]. Noteworthy, the astrocytic glucose transporters have also a critical role in glucosensing. As an example of this, the re-expression of Glut2 in astrocytes of Glut2 null mice restores glucagon secretion in response to hypoglycemia [76][77], indicating that hypothalamic astrocytes via GLUT-2 are a key part of the central glucose sensing machinery and play a key role in the regulation of glycemia. Likewise, repression of hypothalamic GLUT-1 levels due to sustained hyperglycemia blunted the decrease in systemic glucose production, whereas GLUT-1 over-expression in hypothalamic astrocytes restored glucose-sensing capacity of these cells [78]. Nevertheless, glucose transporters are not the only mechanism used by these glial cells to sense glucose. The fact that tanycytes also respond to non-metabolizable glucose analogs, 2-deoxyglucose and methyl glucopyranoside [71][73], suggests that additional glucosensing mechanisms are implicated. In this regard, G-protein-coupled receptors (GPCRs) were proposed to be implicated in glucose sensing and regulation [79]. Interestingly, the GPCR cannabinoid CB1 receptor, a well-known player in energy homeostasis, is expressed in astrocytes and facilitates neuron–astrocyte communication [80]. Although the role of the CB1 receptor in glucose sensing and homeostasis is still unknown, some evidence points to the regulation of the leptin signaling pathway and glycogen storage in astrocytes [81].
3. Brain/Islet Glucose Homeostasis Axis Orchestrated by HMG20A?
Glucose homeostasis is maintained through a network of different organs and tissues that respond to alterations in glucose levels in an organized manner orchestrated by the brain. As described here, the hypothalamus senses glucose fluctuations and activates neuronal circuits that contribute to the regulation of insulin and glucagon secretion by pancreatic endocrine cells, as well as glucose metabolic pathways in liver, fat, and muscle, altogether resulting in the maintenance of glucose homeostasis. Nonetheless, a centralized model of glucosensing relies, as a last resort, on the adequate function of the pancreatic islets; thus, a brain–islet axis is indispensable for the fine-tuning of glucose homeostasis. Interestingly, both pancreatic β-cell and CNS cells possess similar developmental genetic programs that include common key transcription factors such as NEUROD and ISL1, as well as sharing identical regulated exocytosis machinery [23,182,183]. As such, potential epigenetic master regulators could foreseeably orchestrate both tissues/organs in cooperatively controlling glucose homeostasis. We and others showed that HMG20A is involved in neuronal and β-cell mature function [41,184]. As HMG20A regulates the expression of key genes such as NEUROD, GK, and GLUTs common to both astrocytes and β-cells, as well as genes such as insulin and GFAP restricted to each cell type, it is tempting to speculate that this chromatin remodeling factor is a common master regulator in β-cells and astrocytes, integrating inputs to altered glucose levels and stressful physiological conditions that can influence glucose metabolism (
Glucose homeostasis is maintained through a network of different organs and tissues that respond to alterations in glucose levels in an organized manner orchestrated by the brain. As described here, the hypothalamus senses glucose fluctuations and activates neuronal circuits that contribute to the regulation of insulin and glucagon secretion by pancreatic endocrine cells, as well as glucose metabolic pathways in liver, fat, and muscle, altogether resulting in the maintenance of glucose homeostasis. Nonetheless, a centralized model of glucosensing relies, as a last resort, on the adequate function of the pancreatic islets; thus, a brain–islet axis is indispensable for the fine-tuning of glucose homeostasis. Interestingly, both pancreatic β-cell and CNS cells possess similar developmental genetic programs that include common key transcription factors such as NEUROD and ISL1, as well as sharing identical regulated exocytosis machinery [82][83][84]. As such, potential epigenetic master regulators could foreseeably orchestrate both tissues/organs in cooperatively controlling glucose homeostasis. We and others showed that HMG20A is involved in neuronal and β-cell mature function [85][86]. As HMG20A regulates the expression of key genes such as NEUROD, GK, and GLUTs common to both astrocytes and β-cells, as well as genes such as insulin and GFAP restricted to each cell type, it is tempting to speculate that this chromatin remodeling factor is a common master regulator in β-cells and astrocytes, integrating inputs to altered glucose levels and stressful physiological conditions that can influence glucose metabolism (
). This premise is currently under investigation.
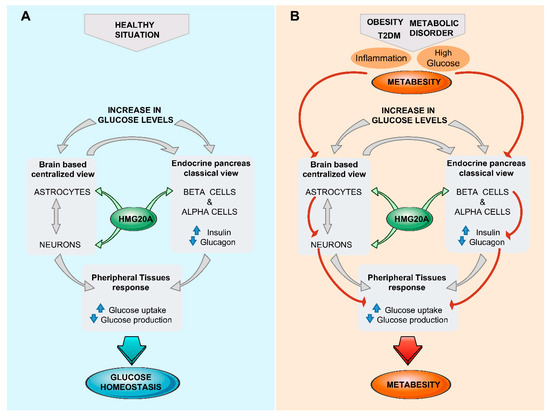
Figure 2.
The brain (astrocyte)/islet axis in metabesity. (
A
) In response to increased glucose levels, a dialogue between brain astrocytes/neurons and islets coordinates glucose homeostasis by peripheral tissue relaying. The chromatin-remodeling factor HMG20A is a common denominator expressed in both astrocytes and islets that facilitates integration of input signals. (
B
) In the advent of metabesity, characterized by obesity, insulin resistance, and inflammation, which alter the expression of HMG20A, the brain/islet axis is short-circuited, precipitating the disease state. T2DM, type 2 diabetes mellitius.
References
- IDF Diabetes Atlas. Available online: (accessed on 9 January 2019).
- Velloso, L.A.; Araujo, E.P.; de Souza, C.T. Diet-induced inflammation of the hypothalamus in obesity. Neuroimmunomodulation 2008, 15, 189–193.
- van Greevenbroek, M.M.; Schalkwijk, C.G.; Stehouwer, C.D. Obesity-associated low-grade inflammation in type 2 diabetes mellitus: Causes and consequences. Neth. J. Med. 2013, 71, 174–187.
- Lontchi-Yimagou, E.; Sobngwi, E.; Matsha, T.E.; Kengne, A.P. Diabetes mellitus and inflammation. Curr. Diab. Rep. 2013, 13, 435–444.
- Shoelson, S.E.; Lee, J.; Goldfine, A.B. Inflammation and insulin resistance. J. Clin. Investig. 2006, 116, 1793–1801.
- Eguchi, K.; Nagai, R. Islet inflammation in type 2 diabetes and physiology. J. Clin. Investig. 2017, 127, 14–23.
- Douglass, J.D.; Dorfman, M.D.; Fasnacht, R.; Shaffer, L.D.; Thaler, J.P. Astrocyte IKKβ/NF-κB signaling is required for diet-induced obesity and hypothalamic inflammation. Mol. Metab. 2017, 6, 366–373.
- Fruhwurth, S.; Vogel, H.; Schurmann, A.; Williams, K.J. Novel Insights into How overnutrition disrupts the hypothalamic actions of leptin. Front. Endocrinol. (Lausanne) 2018, 9, 89.
- Ono, H. Molecular mechanisms of hypothalamic insulin resistance. Int. J. Mol. Sci. 2019, 20, 1317.
- WHO. Global Report on Diabetes. World Health Organization. 2016. Available online: (accessed on 13 March 2019).
- Davies, M.J.; D’Alessio, D.A.; Fradkin, J.; Kernan, W.N.; Mathieu, C.; Mingrone, G.; Rossing, P.; Tsapas, A.; Wexler, D.J.; Buse, J.B. Management of hyperglycaemia in type 2 diabetes, 2018. A consensus report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetologia 2018, 61, 2461–2498.
- Roglic, G.; Unwin, N. Mortality attributable to diabetes: Estimates for the year 2010. Diabetes Res. Clin. Pract. 2010, 87, 15–19.
- Maedler, K.; Carr, R.D.; Bosco, D.; Zuellig, R.A.; Berney, T.; Donath, M.Y. Sulfonylurea induced β-cell apoptosis in cultured human islets. J. Clin. Endocrinol. Metab. 2005, 90, 501–506.
- Munir, K.M.; Davis, S.N. Are SGLT2 inhibitors or GLP-1 receptor agonists more appropriate as a second-line therapy in type 2 diabetes? Expert Opin. Pharmacother. 2018, 19, 773–777.
- Kandimalla, R.; Thirumala, V.; Reddy, P.H. Is Alzheimer’s disease a type 3 diabetes? A critical appraisal. Biochim. Biophys. Acta Mol. Basis. Dis. 2017, 1863, 1078–1089.
- Lopez-Gambero, A.J.; Martinez, F.; Salazar, K.; Cifuentes, M.; Nualart, F. Brain glucose-sensing mechanism and energy homeostasis. Mol. Neurobiol. 2018.
- Williams, K.W.; Elmquist, J.K. From neuroanatomy to behavior: Central integration of peripheral signals regulating feeding behavior. Nat. Neurosci. 2012, 15, 1350–1355.
- Thorens, B. Sensing of glucose in the brain. Handb. Exp. Pharmacol. 2012, 277–294.
- Jordan, S.D.; Konner, A.C.; Bruning, J.C. Sensing the fuels: Glucose and lipid signaling in the CNS controlling energy homeostasis. Cell. Mol. Life Sci. 2010, 67, 3255–3273.
- Oomura, Y.; Kimura, K.; Ooyama, H.; Maeno, T.; Iki, M.; Kuniyoshi, M. Reciprocal activities of the ventromedial and lateral hypothalamic areas of cats. Science 1964, 143, 484–485.
- Routh, V.H.; Hao, L.; Santiago, A.M.; Sheng, Z.; Zhou, C. Hypothalamic glucose sensing: Making ends meet. Front. Syst. Neurosci. 2014, 8, 236.
- Yang, X.J.; Kow, L.M.; Funabashi, T.; Mobbs, C.V. Hypothalamic glucose sensor: Similarities to and differences from pancreatic β-cell mechanisms. Diabetes 1999, 48, 1763–1772.
- Kang, L.; Routh, V.H.; Kuzhikandathil, E.V.; Gaspers, L.D.; Levin, B.E. Physiological and molecular characteristics of rat hypothalamic ventromedial nucleus glucosensing neurons. Diabetes 2004, 53, 549–559.
- Levin, B.E.; Routh, V.H.; Kang, L.; Sanders, N.M.; Dunn-Meynell, A.A. Neuronal glucosensing: What do we know after 50 years? Diabetes 2004, 53, 2521–2528.
- Moriyama, R.; Tsukamura, H.; Kinoshita, M.; Okazaki, H.; Kato, Y.; Maeda, K. In vitro increase in intracellular calcium concentrations induced by low or high extracellular glucose levels in ependymocytes and serotonergic neurons of the rat lower brainstem. Endocrinology 2004, 145, 2507–2515.
- Fioramonti, X.; Lorsignol, A.; Taupignon, A.; Penicaud, L. A new ATP-sensitive K+ channel-independent mechanism is involved in glucose-excited neurons of mouse arcuate nucleus. Diabetes 2004, 53, 2767–2775.
- Song, Z.; Routh, V.H. Differential effects of glucose and lactate on glucosensing neurons in the ventromedial hypothalamic nucleus. Diabetes 2005, 54, 15–22.
- O’Malley, D.; Reimann, F.; Simpson, A.K.; Gribble, F.M. Sodium-coupled glucose cotransporters contribute to hypothalamic glucose sensing. Diabetes 2006, 55, 3381–3386.
- Gonzalez, J.A.; Reimann, F.; Burdakov, D. Dissociation between sensing and metabolism of glucose in sugar sensing neurones. J. Physiol. 2009, 587, 41–48.
- Ren, X.; Zhou, L.; Terwilliger, R.; Newton, S.S.; de Araujo, I.E. Sweet taste signaling functions as a hypothalamic glucose sensor. Front. Integr. Neurosci. 2009, 3, 12.
- Toda, C.; Kim, J.D.; Impellizzeri, D.; Cuzzocrea, S.; Liu, Z.W.; Diano, S. UCP2 regulates mitochondrial fission and ventromedial nucleus control of glucose responsiveness. Cell 2016, 164, 872–883.
- Silver, I.A.; Erecinska, M. Glucose-induced intracellular ion changes in sugar-sensitive hypothalamic neurons. J. Neurophysiol. 1998, 79, 1733–1745.
- Fioramonti, X.; Marsollier, N.; Song, Z.; Fakira, K.A.; Patel, R.M.; Brown, S.; Duparc, T.; Pica-Mendez, A.; Sanders, N.M.; Knauf, C.; et al. Ventromedial hypothalamic nitric oxide production is necessary for hypoglycemia detection and counterregulation. Diabetes 2010, 59, 519–528.
- Murphy, B.A.; Fakira, K.A.; Song, Z.; Beuve, A.; Routh, V.H. AMP-activated protein kinase and nitric oxide regulate the glucose sensitivity of ventromedial hypothalamic glucose-inhibited neurons. Am. J. Physiol. Cell Physiol. 2009, 297, C750–C758.
- Wang, R.; Liu, X.; Hentges, S.T.; Dunn-Meynell, A.A.; Levin, B.E.; Wang, W.; Routh, V.H. The regulation of glucose-excited neurons in the hypothalamic arcuate nucleus by glucose and feeding-relevant peptides. Diabetes 2004, 53, 1959–1965.
- Romero-Zerbo, S.Y.; Bermudez-Silva, F.J. Cannabinoids, eating behaviour, and energy homeostasis. Drug Test. Anal. 2014, 6, 52–58.
- Bermudez-Silva, F.J.; Cardinal, P.; Cota, D. The role of the endocannabinoid system in the neuroendocrine regulation of energy balance. J. Psychopharmacol. 2012, 26, 114–124.
- Kohno, D.; Yada, T. Arcuate NPY neurons sense and integrate peripheral metabolic signals to control feeding. Neuropeptides 2012, 46, 315–319.
- Coppari, R.; Ichinose, M.; Lee, C.E.; Pullen, A.E.; Kenny, C.D.; McGovern, R.A.; Tang, V.; Liu, S.M.; Ludwig, T.; Chua, S.C., Jr.; et al. The hypothalamic arcuate nucleus: A key site for mediating leptin’s effects on glucose homeostasis and locomotor activity. Cell Metab. 2005, 1, 63–72.
- Wang, Q.; Liu, C.; Uchida, A.; Chuang, J.C.; Walker, A.; Liu, T.; Osborne-Lawrence, S.; Mason, B.L.; Mosher, C.; Berglund, E.D.; et al. Arcuate AgRP neurons mediate orexigenic and glucoregulatory actions of ghrelin. Mol. Metab. 2014, 3, 64–72.
- Muroya, S.; Yada, T.; Shioda, S.; Takigawa, M. Glucose-sensitive neurons in the rat arcuate nucleus contain neuropeptide Y. Neurosci. Lett. 1999, 264, 113–116.
- Liu, T.; Kong, D.; Shah, B.P.; Ye, C.; Koda, S.; Saunders, A.; Ding, J.B.; Yang, Z.; Sabatini, B.L.; Lowell, B.B. Fasting activation of AgRP neurons requires NMDA receptors and involves spinogenesis and increased excitatory tone. Neuron 2012, 73, 511–522.
- Yang, Y.; Atasoy, D.; Su, H.H.; Sternson, S.M. Hunger states switch a flip-flop memory circuit via a synaptic AMPK-dependent positive feedback loop. Cell 2011, 146, 992–1003.
- Luquet, S.; Perez, F.A.; Hnasko, T.S.; Palmiter, R.D. NPY/AgRP neurons are essential for feeding in adult mice but can be ablated in neonates. Science 2005, 310, 683–685.
- Gropp, E.; Shanabrough, M.; Borok, E.; Xu, A.W.; Janoschek, R.; Buch, T.; Plum, L.; Balthasar, N.; Hampel, B.; Waisman, A.; et al. Agouti-related peptide-expressing neurons are mandatory for feeding. Nat. Neurosci. 2005, 8, 1289–1291.
- Atasoy, D.; Betley, J.N.; Su, H.H.; Sternson, S.M. Deconstruction of a neural circuit for hunger. Nature 2012, 488, 172–177.
- Ibrahim, N.; Bosch, M.A.; Smart, J.L.; Qiu, J.; Rubinstein, M.; Ronnekleiv, O.K.; Low, M.J.; Kelly, M.J. Hypothalamic proopiomelanocortin neurons are glucose responsive and express KATP channels. Endocrinology 2003, 144, 1331–1340.
- Zhan, C.; Zhou, J.; Feng, Q.; Zhang, J.E.; Lin, S.; Bao, J.; Wu, P.; Luo, M. Acute and long-term suppression of feeding behavior by POMC neurons in the brainstem and hypothalamus, respectively. J. Neurosci. 2013, 33, 3624–3632.
- Konner, A.C.; Janoschek, R.; Plum, L.; Jordan, S.D.; Rother, E.; Ma, X.; Xu, C.; Enriori, P.; Hampel, B.; Barsh, G.S.; et al. Insulin action in AgRP-expressing neurons is required for suppression of hepatic glucose production. Cell Metab. 2007, 5, 438–449.
- Hill, J.W.; Elias, C.F.; Fukuda, M.; Williams, K.W.; Berglund, E.D.; Holland, W.L.; Cho, Y.R.; Chuang, J.C.; Xu, Y.; Choi, M.; et al. Direct insulin and leptin action on pro-opiomelanocortin neurons is required for normal glucose homeostasis and fertility. Cell Metab. 2010, 11, 286–297.
- Steculorum, S.M.; Ruud, J.; Karakasilioti, I.; Backes, H.; Engstrom Ruud, L.; Timper, K.; Hess, M.E.; Tsaousidou, E.; Mauer, J.; Vogt, M.C.; et al. AgRP neurons control systemic insulin sensitivity via myostatin expression in brown adipose tissue. Cell 2016, 165, 125–138.
- Krashes, M.J.; Koda, S.; Ye, C.; Rogan, S.C.; Adams, A.C.; Cusher, D.S.; Maratos-Flier, E.; Roth, B.L.; Lowell, B.B. Rapid, reversible activation of AgRP neurons drives feeding behavior in mice. J. Clin. Investig. 2011, 121, 1424–1428.
- Joly-Amado, A.; Denis, R.G.; Castel, J.; Lacombe, A.; Cansell, C.; Rouch, C.; Kassis, N.; Dairou, J.; Cani, P.D.; Ventura-Clapier, R.; et al. Hypothalamic AgRP-neurons control peripheral substrate utilization and nutrient partitioning. EMBO J. 2012, 31, 4276–4288.
- Sainsbury, A.; Rohner-Jeanrenaud, F.; Cusin, I.; Zakrzewska, K.E.; Halban, P.A.; Gaillard, R.C.; Jeanrenaud, B. Chronic central neuropeptide Y infusion in normal rats: Status of the hypothalamo-pituitary-adrenal axis, and vagal mediation of hyperinsulinaemia. Diabetologia 1997, 40, 1269–1277.
- Meek, T.H.; Nelson, J.T.; Matsen, M.E.; Dorfman, M.D.; Guyenet, S.J.; Damian, V.; Allison, M.B.; Scarlett, J.M.; Nguyen, H.T.; Thaler, J.P.; et al. Functional identification of a neurocircuit regulating blood glucose. Proc. Natl. Acad. Sci. USA 2016, 113, E2073–E2082.
- Garfield, A.S.; Shah, B.P.; Madara, J.C.; Burke, L.K.; Patterson, C.M.; Flak, J.; Neve, R.L.; Evans, M.L.; Lowell, B.B.; Myers, M.G., Jr.; et al. A parabrachial-hypothalamic cholecystokinin neurocircuit controls counterregulatory responses to hypoglycemia. Cell Metab. 2014, 20, 1030–1037.
- Levin, B.E.; Magnan, C.; Dunn-Meynell, A.; Le Foll, C. Metabolic sensing and the brain: Who, what, where, and how? Endocrinology 2011, 152, 2552–2557.
- Cortes-Campos, C.; Elizondo, R.; Carril, C.; Martinez, F.; Boric, K.; Nualart, F.; Garcia-Robles, M.A. MCT2 expression and lactate influx in anorexigenic and orexigenic neurons of the arcuate nucleus. PLoS ONE 2013, 8, e62532.
- Garcia-Marin, V.; Garcia-Lopez, P.; Freire, M. Cajal’s contributions to glia research. Trends Neurosci. 2007, 30, 479–487.
- Pellerin, L. How astrocytes feed hungry neurons. Mol. Neurobiol. 2005, 32, 59–72.
- Yang, L.; Qi, Y.; Yang, Y. Astrocytes control food intake by inhibiting AGRP neuron activity via adenosine A1 receptors. Cell Rep. 2015, 11, 798–807.
- Marty, N.; Dallaporta, M.; Thorens, B. Brain glucose sensing, counterregulation, and energy homeostasis. Physiology 2007, 22, 241–251.
- Elizondo-Vega, R.; Cortes-Campos, C.; Barahona, M.J.; Oyarce, K.A.; Carril, C.A.; Garcia-Robles, M.A. The role of tanycytes in hypothalamic glucosensing. J. Cell Mol. Med. 2015, 19, 1471–1482.
- Lam, T.K.; Gutierrez-Juarez, R.; Pocai, A.; Rossetti, L. Regulation of blood glucose by hypothalamic pyruvate metabolism. Science 2005, 309, 943–947.
- Rodriguez, E.M.; Blazquez, J.L.; Pastor, F.E.; Pelaez, B.; Pena, P.; Peruzzo, B.; Amat, P. Hypothalamic tanycytes: A key component of brain-endocrine interaction. Int. Rev. Cytol. 2005, 247, 89–164.
- Machler, P.; Wyss, M.T.; Elsayed, M.; Stobart, J.; Gutierrez, R.; von Faber-Castell, A.; Kaelin, V.; Zuend, M.; San Martin, A.; Romero-Gomez, I.; et al. In vivo evidence for a lactate gradient from astrocytes to neurons. Cell Metab. 2016, 23, 94–102.
- Wender, R.; Brown, A.M.; Fern, R.; Swanson, R.A.; Farrell, K.; Ransom, B.R. Astrocytic glycogen influences axon function and survival during glucose deprivation in central white matter. J. Neurosci. 2000, 20, 6804–6810.
- Scarlett, J.M.; Rojas, J.M.; Matsen, M.E.; Kaiyala, K.J.; Stefanovski, D.; Bergman, R.N.; Nguyen, H.T.; Dorfman, M.D.; Lantier, L.; Wasserman, D.H.; et al. Central injection of fibroblast growth factor 1 induces sustained remission of diabetic hyperglycemia in rodents. Nat. Med. 2016, 22, 800–806.
- Scarlett, J.M.; Muta, K.; Brown, J.M.; Rojas, J.M.; Matsen, M.E.; Acharya, N.K.; Secher, A.; Ingvorsen, C.; Jorgensen, R.; Hoeg-Jensen, T.; et al. Peripheral mechanisms mediating the sustained antidiabetic action of FGF1 in the brain. Diabetes 2019, 68, 654–664.
- Garcia, M.; Millan, C.; Balmaceda-Aguilera, C.; Castro, T.; Pastor, P.; Montecinos, H.; Reinicke, K.; Zuniga, F.; Vera, J.C.; Onate, S.A.; et al. Hypothalamic ependymal-glial cells express the glucose transporter GLUT2, a protein involved in glucose sensing. J. Neurochem. 2003, 86, 709–724.
- Frayling, C.; Britton, R.; Dale, N. ATP-mediated glucosensing by hypothalamic tanycytes. J. Physiol. 2011, 589, 2275–2286.
- Orellana, J.A.; Saez, P.J.; Cortes-Campos, C.; Elizondo, R.J.; Shoji, K.F.; Contreras-Duarte, S.; Figueroa, V.; Velarde, V.; Jiang, J.X.; Nualart, F.; et al. Glucose increases intracellular free Ca2+ in tanycytes via ATP released through connexin 43 hemichannels. Glia 2012, 60, 53–68.
- Benford, H.; Bolborea, M.; Pollatzek, E.; Lossow, K.; Hermans-Borgmeyer, I.; Liu, B.; Meyerhof, W.; Kasparov, S.; Dale, N. A sweet taste receptor-dependent mechanism of glucosensing in hypothalamic tanycytes. Glia 2017, 65, 773–789.
- Meda, P.; Schuit, F. Glucose-stimulated insulin secretion: The hierarchy of its multiple cellular and subcellular mechanisms. Diabetologia 2013, 56, 2552–2555.
- Barahona, M.J.; Llanos, P.; Recabal, A.; Escobar-Acuna, K.; Elizondo-Vega, R.; Salgado, M.; Ordenes, P.; Uribe, E.; Sepulveda, F.J.; Araneda, R.C.; et al. Glial hypothalamic inhibition of GLUT2 expression alters satiety, impacting eating behavior. Glia 2018, 66, 592–605.
- Marty, N.; Dallaporta, M.; Foretz, M.; Emery, M.; Tarussio, D.; Bady, I.; Binnert, C.; Beermann, F.; Thorens, B. Regulation of glucagon secretion by glucose transporter type 2 (glut2) and astrocyte-dependent glucose sensors. J. Clin. Investig. 2005, 115, 3545–3553.
- Thorens, B.; Guillam, M.T.; Beermann, F.; Burcelin, R.; Jaquet, M. Transgenic reexpression of GLUT1 or GLUT2 in pancreatic β cells rescues GLUT2-null mice from early death and restores normal glucose-stimulated insulin secretion. J. Biol. Chem. 2000, 275, 23751–23758.
- Chari, M.; Yang, C.S.; Lam, C.K.; Lee, K.; Mighiu, P.; Kokorovic, A.; Cheung, G.W.; Lai, T.Y.; Wang, P.Y.; Lam, T.K. Glucose transporter-1 in the hypothalamic glial cells mediates glucose sensing to regulate glucose production in vivo. Diabetes 2011, 60, 1901–1906.
- Bolborea, M.; Dale, N. Hypothalamic tanycytes: Potential roles in the control of feeding and energy balance. Trends Neurosci. 2013, 36, 91–100.
- Navarrete, M.; Diez, A.; Araque, A. Astrocytes in endocannabinoid signalling. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2014, 369, 20130599.
- Bosier, B.; Bellocchio, L.; Metna-Laurent, M.; Soria-Gomez, E.; Matias, I.; Hebert-Chatelain, E.; Cannich, A.; Maitre, M.; Leste-Lasserre, T.; Cardinal, P.; et al. Astroglial CB1 cannabinoid receptors regulate leptin signaling in mouse brain astrocytes. Mol. Metab. 2013, 2, 393–404.
- Gauthier, B.R.; Wollheim, C.B. Synaptotagmins bind calcium to release insulin. Am. J. Physiol. Endocrinol. Metab. 2008, 295, E1279–E1286.
- Dolai, S.; Xie, L.; Zhu, D.; Liang, T.; Qin, T.; Xie, H.; Kang, Y.; Chapman, E.R.; Gaisano, H.Y. Synaptotagmin-7 functions to replenish insulin granules for exocytosis in human islet β-cells. Diabetes 2016, 65, 1962–1976.
- Turecek, J.; Regehr, W.G. Synaptotagmin 7 mediates both facilitation and asynchronous release at granule cell synapses. J. Neurosci. 2018, 38, 3240–3251.
- Mellado-Gil, J.M.; Fuente-Martin, E.; Lorenzo, P.I.; Cobo-Vuilleumier, N.; Lopez-Noriega, L.; Martin-Montalvo, A.; Gomez, I.G.H.; Ceballos-Chavez, M.; Gomez-Jaramillo, L.; Campos-Caro, A.; et al. The type 2 diabetes-associated HMG20A gene is mandatory for islet β cell functional maturity. Cell Death Dis. 2018, 9, 279.
- Wynder, C.; Hakimi, M.A.; Epstein, J.A.; Shilatifard, A.; Shiekhattar, R. Recruitment of MLL by HMG-domain protein iBRAF promotes neural differentiation. Nat. Cell Biol. 2005, 7, 1113–1117.
