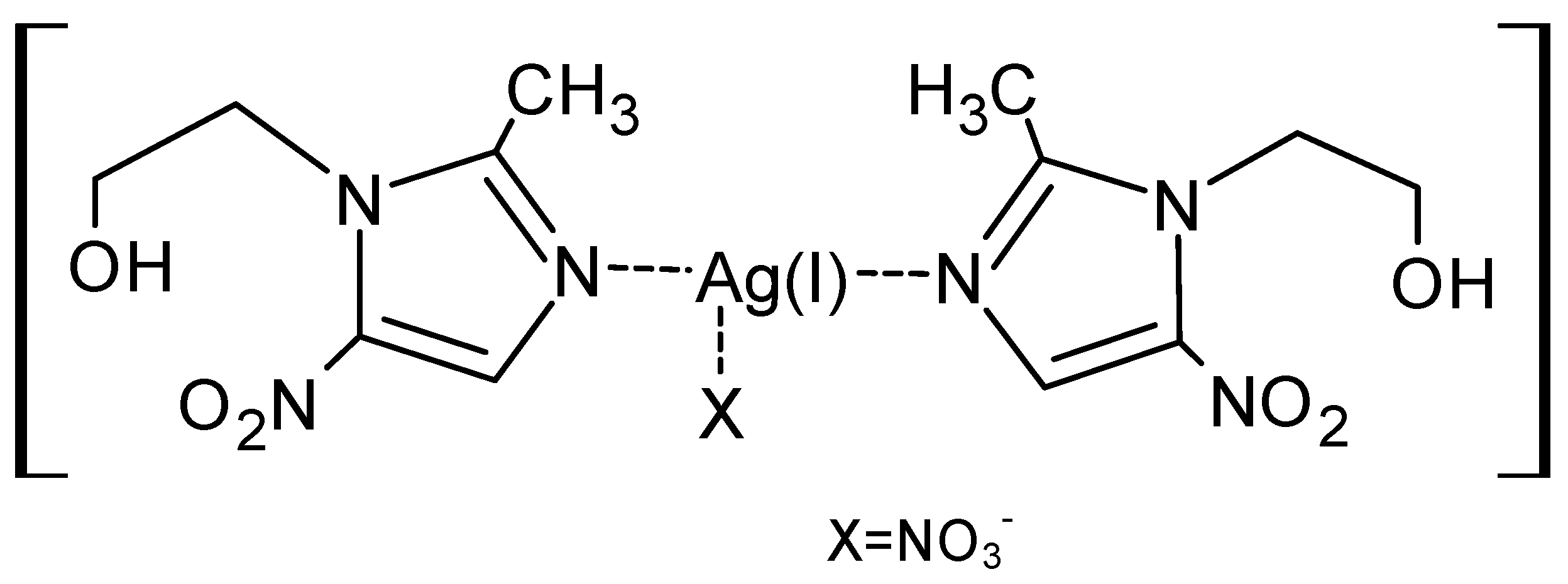The use of silver preparations in medicine is becoming increasingly popular.
- silver (I) complex
- ophthalmic diseases
- eye drops
- eye ointment
- Ocular Rosacea
- drops stabilization
1. Silver in Ophthalmology
1.1. History of Silver as an Antibacterial Agent
[1]
[2]
[3]
[6]
1.2. Toxicology
[7]
[8]
[9]
[10]
[13]
3
3
[16]
[16]
3
[17].
2. Application of Silver Compounds in Ophthalmology
[18]
Pseudomonas aeruginosa
Staphylococcus aureus
[19]
Staphylococcus aureus
[20]
[21]
[22]
[26].
3. Silver (I) Complex with Metronidazole in Ophthalmology
Research on the use of new silver preparations combined with other active substances in ophthalmology is developing much more vigorously [27]. Numerous studies showed greater stability of metal ion complexes compared to metal ion salts [28]and lower toxicity due to the possibility of using lower concentrations of silver ions in complexes [27].The widest and best-known use of silver in medicine has been in combination with sulfadiazine (AgSD), where it becomes a topical antibacterial agent for the treatment of burns [29] and fungal keratitis [30][31]. The action of AgSD also demonstrates strong antibacterial potential against
E. coli
Staph. aureus
Klebsiella
Pseudomonas sp. [32].In terms of biological performance, studies in vitro, as well as in vivo, on retinal pigment epithelium with endophthalmitis in mouse and rabbit models confirm the cellular biocompatibility and antibacterial function of silver complex nanomaterials. The addition of photodynamic therapy with Ag-NPs as well as AuAgCu
2
Escherichia coli
Staphylococcus aureus
S. aureus for synergistic treatment of post-cataract surgery endophthalmitis [33][34].Ag-NPs conjugated with oleic acid or vildagliptin exhibit antiacanthamoebic activity that can be therapeutically applied against
Acanthamoeba castellanii, an opportunistic pathogen that is associated with blinding eye keratitis and a rare but fatal central nervous system infection [35][36].In our previous paper, we described, for the first time in the literature, the action of a metronidazole complex with well-soluble silver (I) salts in the form of drops and ointment in the treatment of ocular rosacea [37]. The use of a well-soluble silver (I) complex with metronidazole (
Figure 1.
Silver (I) nitrate complex with metronidazole [Ag(MTZ)
2
]NO
3
.
The antimicrobial effectiveness of silver preparations in ophthalmic diseases has been documented by many researchers. Complex silver (I) compounds seem to be a promising alternative to standard therapy and are, therefore, also considered as new generation antibiotics. Most silver (I) preparations are clinically used for topical applications. Few experimental results indicate the usefulness of intraocular or systemic administration of silver (I) preparations as an alternative or additional therapy in infectious and angiogenic eye diseases. New forms of silver (I) products will certainly find application in the treatment of many ophthalmic diseases. One of the most important features of the silver (I) complex is its capacity to break down bacterial resistance. It is very helpful to maintain the appropriate characteristics of the dosage form, e.g., pH and chemical, physical and pharmaceutical stabilities.
The antimicrobial effectiveness of silver preparations in ophthalmic diseases has been documented by many researchers. Complex silver (I) compounds seem to be a promising alternative to standard therapy and are, therefore, also considered as new generation antibiotics. Most silver (I) preparations are clinically used for topical applications. Few experimental results indicate the usefulness of intraocular or systemic administration of silver (I) preparations as an alternative or additional therapy in infectious and angiogenic eye diseases. New forms of silver (I) products will certainly find application in the treatment of many ophthalmic diseases. One of the most important features of the silver (I) complex is its capacity to break down bacterial resistance. It is very helpful to maintain the appropriate characteristics of the dosage form, e.g., pH and chemical, physical and pharmaceutical stabilities.References
- Hill, W.R.; Pillsbury, D.M. Argyria—The Pharmacology of Silver; Williams & Wilkins: Baltimore, MD, USA, 1939.
- Alexander, J.W. History of the medical use of silver. Surg. Infect. 2009, 10, 289–292.
- Rollings, N.B. Georgia Arbuckle Fix: Silver dollar surgeon. Am. Hist. Illus. 1985, 20, 20–21.
- Moore, D.L.; MacDonald, N.E. Canadian Paediatric Society, Infectious Diseases and Immunization Committee. Preventing ophthalmia neonatorum. Paediatr. Child Health 2015, 20, 93–96.
- Silva, L.R.; Gurgel, R.Q.; Lima, D.R.; Cuevas, L.E. Current usefulness of Credé’s method of preventing neonatal ophthalmia. Ann. Trop. Paediatr. 2008, 28, 45–48.
- Roe, A.L. Collosol argentum and its ophthalmic uses. Br. Med. J. 1915, 16, 104.
- Lansdown, A.B. A pharmacological and toxicological profile of silver as an antimicrobial agent in medical devices. Adv. Pharmacol. Sci. 2010, 2010, 910686.
- Jongerius, O.; Jongeneelen, F.I. Occupational Exposure Limits (Criteria Document for Metallic Silver); Commission of the European Communities: Brussels, Belgium, 1992.
- Venugopal, B.; Lucky, T.D. Metal. Toxicity in Mammals; Plenum Press: New York, NY, USA, 1978; Volume 2, pp. 86–99.
- Fung, M.C.; Bowen, D.L. Silver products for medical indications: Risk-benefit assessment. J. Toxicol. Clin. Toxicol. 1996, 34, 119–126.
- Zheng, J.; Zhang, K.; Liu, Y.; Wang, Y. Fatal acute arsenic poisoning by external use of realgar: Case report and 30 years literature retrospective study in China. Forensic Sci. Int. 2019, 300, e24–e30.
- Gulbranson, S.H.; Hud, J.A.; Hansen, R.C. Argyria following the use of dietary supplements containing colloidal silver protein. Cutis 2000, 66, 373–374.
- Claessens, D.; Zeitz, P.F.; Beckers, H. Blaugraue Verfärbung von Haut und Bindehaut [Bluish-gray discoloration of skin and conjunctiva]. Ophthalmologe 2020, 117, 66–68.
- Sarnat-Kucharczyk, M.; Pojda-Wilczek, D.; Mrukwa-Kominek, E. Diagnostic methods in ocular argyrosis: Case report. Doc. Ophthalmol. 2016, 133, 129–138.
- Pala, G.; Fronterré, A.; Scafa, F.; Scelsi, M.; Ceccuzzi, R.; Gentile, E.; Candura, S.M. Ocular argyrosis in a silver craftsman. J. Occup. Health 2008, 50, 521–524.
- Qin, G.; Tang, S.; Li, S.; Lu, H.; Wang, Y.; Zhao, P.; Li, B.; Zhang, J.; Peng, L. Toxicological evaluation of silver nanoparticles and silver nitrate in rats following 28 days of repeated oral exposure. Environ. Toxicol. 2017, 32, 609–618.
- Cvjetko, P.; Milošić, A.; Domijan, A.M.; Vinković Vrček, I.; Tolić, S.; Peharec Štefanić, P.; Letofsky-Papst, I.; Tkalec, M.; Balen, B. Toxicity of silver ions and differently coated silver nanoparticles in Allium cepa roots. Ecotoxicol. Environ. Saf. 2017, 137, 18–28.
- Leitão, J.H.; Sousa, S.A.; Leite, S.A.; Carvalho, M.F.N.N. Silver Camphor Imine Complexes: Novel Antibacterial Compounds from Old Medicines. Antibiotics 2018, 7, 65.
- Riau, A.K.; Aung, T.T.; Setiawan, M.; Yang, L.; Yam, G.H.F.; Beuerman, R.W.; Venkatraman, S.S.; Mehta, J.S. Surface Immobilization of Nano-Silver on Polymeric Medical Devices to Prevent Bacterial Biofilm Formation. Pathogens 2019, 8, 93.
- Luo, L.J.; Lin, T.Y.; Yao, C.H.; Kuo, P.Y.; Matsusaki, M.; Harroun, S.G.; Huang, C.C.; Lai, J.Y. Dual-functional gelatin-capped silver nanoparticles for antibacterial and antiangiogenic treatment of bacterial keratitis. J. Colloid Interface Sci. 2019, 536, 112–126.
- Fujimoto, T.; Hanaoka, N.; Konagaya, M.; Kobayashi, M.; Nakagawa, H.; Hatano, H.; Tsukahara-Kawamura, T.; Uchio, E.; Kaneko, H. Evaluation of a silver-amplified immunochromatography kit for adenoviral conjunctivitis. J. Med. Virol. 2019, 91, 1030–1035.
- Butler, M.R.; Prospero Ponce, C.M.; Weinstock, Y.E.; Orengo-Nania, S.; Chevez-Barrios, P.; Frankfort, B.J. Topical silver nanoparticles result in improved bleb function by increasing filtration and reducing fibrosis in a rabbit model of filtration surgery. Investig. Ophthalmol. Vis. Sci. 2013, 54, 4982–4990.
- Kalishwaralal, K.; BarathManiKanth, S.; Pandian, S.R.K.; Deepak, V.; Gurunathan, S. Silver nano—A trove for retinal therapies. J. Control. Release 2010, 145, 76–90.
- Pershadsingh, H.A.; Moore, D.M. PPARγ agonists: Potential as therapeutics for neovascular retinopathies. PPAR Res. 2008, 164273.
- Gurunathan, S.; Lee, K.J.; Kalishwaralal, K.; Sheikpranbabu, S.; Vaidyanathan, R.; Eom, S.H. Antiangiogenic properties of silver nanoparticles. Biomaterials 2009, 30, 6341–6350.
- Mourad, R.; Helaly, F.; Darwesh, O.; Sawy, S.E. Antimicrobial and physicomechanical natures of silver nanoparticles incorporated into silicone-hydrogel films. Cont. Lens Anterior Eye 2019, 42, 325–333.
- Radko, L.; Stypuła-Trębas, S.; Posyniak, A.; Żyro, D.; Ochocki, J. Silver (I) Complexes of the Pharmaceutical Agents Metronidazole and 4-Hydroxymethylpyridine: Comparison of Cytotoxic Profile for Potential Clinical Application. Molecules 2019, 24, 1949. Kalinowska-Lis, U.; Felczak, A.; Chęcińska, L.; Zawadzka, K.; Patyna, E.; Lisowska, K.; Ochocki, J. Synthesis, characterization and antimicrobial activity of water-soluble silver(i) complexes of metronidazole drug and selected counter-ions. Dalton Trans. 2015, 44, 8178–8189.
- Żyro, D.; Śliwińska, A.; Szymczak-Pajor, I.; Stręk, M.; Ochocki, J. Light Stability, Pro-Apoptotic and Genotoxic Properties of Silver (I) Complexes of Metronidazole and 4-Hydroxymethylpyridine against Pancreatic Cancer Cells In Vitro. Cancers 2020, 12, 3848.
- Modak, S.M.; Sampath, L.; Fox, C.L.J. Combined topical use of silver sulfadiazine and antibiotics as a possible solution to bacterial resistance in burn wounds. J. Burn Care Rehabil. 1988, 9, 359–363.
- FlorCruz, N.V.; Evans, J.R. Medical interventions for fungal keratitis. Cochrane Database Syst. Rev. 2015, 9, CD004241.
- Mohan, M.; Gupta, S.K.; Kalra, V.K.; Vajpayee, R.B.; Sachdev, M.S. Topical silver sulphadiazine—A new drug for ocular keratomycosis. Br. J. Ophthalmol. 1988, 72, 192–195.
- Rai, M.K.; Deshmukh, S.D.; Ingle, A.P.; Gade, A.K. Silver nanoparticles: The powerful nanoweapon against multidrug-resistant bacteria. J. Appl. Microbiol. 2012, 112, 841–852.
- Chen, H.; Yang, J.; Sun, L.; Zhang, H.; Guo, Y.; Qu, J.; Jiang, W.; Chen, W.; Ji, J.; Yang, Y.W.; et al. Synergistic Chemotherapy and Photodynamic Therapy of Endophthalmitis Mediated by Zeolitic Imidazolate Framework-Based Drug Delivery Systems. Small 2019, 15, e1903880.
- Ye, Y.; He, J.; Qiao, Y.; Qi, Y.; Zhang, H.; Santos, H.A.; Zhong, D.; Li, W.; Hua, S.; Wang, W.; et al. Mild temperature photothermal assisted anti-bacterial and anti-inflammatory nanosystem for synergistic treatment of post-cataract surgery endophthalmitis. Theranostics 2020, 10, 8541–8557.
- Anwar, A.; Abdalla, S.A.O.; Aslam, Z.; Shah, M.R.; Siddiqui, R.; Khan, N.A. Oleic acid-conjugated silver nanoparticles as efficient antiamoebic agent against Acanthamoeba castellanii. Parasitol. Res. 2019, 118, 2295–2304.
- Anwar, A.; Siddiqui, R.; Raza Shah, M.; Khan, N.A. Antidiabetic Drugs and Their Nanoconjugates Repurposed as Novel Antimicrobial Agents against Acanthamoeba castellanii. J. Microbiol. Biotechnol. 2019, 29, 713–720.
- Waszczykowska, A.; Żyro, D.; Jurowski, P.; Ochocki, J. Effect of treatment with silver (I) complex of metronidazole on ocular rosacea: Design and formulation of new silver drug with potent antimicrobial activity. J. Trace Elem. Med. Biol. 2020, 61, 126531.
- Kalinowska-Lis, U.; Felczak, A.; Chęcińska, L.; Zawadzka, K.; Patyna, E.; Lisowska, K.; Ochocki, J. Synthesis, characterization and antimicrobial activity of water-soluble silver(i) complexes of metronidazole drug and selected counter-ions. Dalton Trans. 2015, 44, 8178–8189.