For nearly a decade, researchers in the field of pediatric oncology have been using zebrafish as a model for understanding the contributions of genetic alternations to the pathogenesis of neuroblastoma (NB), and exploring the molecular and cellular mechanisms that underlie neuroblastoma initiation and metastasis.
- neuroblastoma
- zebrafish
- animal model
1. Introduction
Over the past ten years, zebrafish have become an increasingly popular tool for scientists conducting biomedical studies and other research. The species’ high fecundity rate, low cost of maintenance, and the ease of observation and genetic manipulation all contribute to its increasing use as an alternative and valuable vertebrate model system to study human disease. The expanding community of researchers using zebrafish has brought advanced technologies to the model, as well as a rapidly expanding inventory of transgenic and mutant lines that can be applied to different research niches. Cancer research using the zebrafish model can be traced back to 1965, when Dr. Mearle Stantion performed pioneered work to induce hepatic neoplasia in zebrafish with Diethylnitrosamine
[1]
. In 2003, the first zebrafish genetic cancer model was reported by Drs. David Langenau and Thomas Look, in which the MYC oncogene was overexpressed under control of the
rag2
promoter, resulting in the development of T cell leukemia in the transgenic animal
[2]
. Since then many more zebrafish cancer models have been developed to understand the pathogenesis of leukemia, melanoma, rhabdomyosarcoma, hepatocellular carcinoma and many other tumor types
. In particular, the zebrafish model has also shown exceptional promise in dissecting the contributions of genetic alterations that were identified from integrative genomic analyses of neuroblastoma (NB) to the pathogenesis of this devastating pediatric cancer.
NB is the most common extracranial solid tumor in children and accounts for ~10% of all childhood cancer-related deaths
[7]
. It is derived from transformed neural crest progenitor cells in the developing peripheral sympathetic nervous system (PSNS)
. High-risk patients with amplified
MYCN
and over 18 months of age are often presented with widespread metastasis at diagnosis. Over the past few years, the five-year event-free survival rate for children with high-risk disease remains lower than 50%
. Very recently, a Phase III trial of immunotherapy, consisting of Dinutuximab, granulocyte macrophage-colony stimulating factor (GM-CSF) and interleukin-2 (IL2), showed significantly increased five-year overall survival rate of patients with high-risk NB to ~70%
. This immunotherapy has been approved by FDA for the treatment of patients with high-risk NB who achieve at least a partial response to prior first-line multiagent, multimodality therapy
[12]
. Although the improved outcomes are observed with the inclusion of Dinutuximab as part of treatment regimens for newly diagnosed NB, the prognosis for the relapsed disease remains poor (<10% progression-free survival)
. Therefore, better understanding of the pathogenesis of this disease and developing novel and more effective therapies are needed.
As an important member of the
MYC
proto-oncogene family identified from NB patients
[16]
,
MYCN
amplification accounts for ~25% of NB cases and is associated with poor disease outcome
.
MYCN
is a bHLH transcription factor and is homologous to c-MYC structurally and functionally. It can promote neoplastic transformation of cultured mammalian cells and rat embryo fibroblasts
. In 1997, Dr. William Weiss developed the first animal model of NB by overexpressing
MYCN
under control of Tyrosine Hydroxylase (
TH
) in transgenic mice, which is by far still the most popular model for NB research
[22]
. Following Dr. Weiss’s effort, several genetically modified mouse (GEMM) lines with direct, conditional, inducible overexpression, knock-in or knockout of NB-relevant genes, including mutationally activated
ALK
(Anaplastic Lymphoma Receptor Tyrosine Kinase)
,
LIN28B
(Lin-28 Homolog B)
,
SV40
large T antigen (Simian Vacuolating Virus 40 TAg)
and others
were subsequently developed. These models demonstrated a sufficient induction of NB in mice, which resemble the features of human NBs
[24]
.
Although the mouse model provides valuable molecular insights on NB pathogenesis and opened the door for NB research, it has some disadvantages when compared to the zebrafish model. Neuroblastomas are different from adult tumors, in that they arise early in development; identifying the early onset of tumorigenesis in mice without euthanizing the animals is difficult and creates challenges in dissecting the molecular and cellular mechanisms underlying early onset tumor initiation. Zebrafish, by contrast, are translucent and develop from externally fertilized eggs, which allows for early detection of tumor onset in live animals. The zebrafish model is also more practical than GEMM, less expensive, and does not require sacrificing these animals to track tumor initiation and visualization of tumor growth. Therefore, the zebrafish model can serve as an alternative for the commonly used mouse model to conduct genetic research.
In 2012, the first zebrafish model of NB was generated and published by Zhu et al.
[33]
. Two oncogenes,
MYCN
and mutationally activated
ALK
(the most commonly mutated genes in primary neuroblastoma
and an attractive candidate for targeted therapy
), were expressed under control of the dopamine-beta-hydroxylase (
dβh
) promoter
[33]
. Following this initial effort on modeling NB in zebrafish, many new transgenic fish lines were developed, uncovering additional novel genetic alterations that cooperate with MYCN or c-MYC during NB pathogenesis. The evolution of zebrafish NB models has revealed the complexity of this disease at the molecular level and demonstrates the robustness of the model system in deepening our understanding of the molecular and cellular basis underlying NB pathogenesis. An overview of the NB zebrafish disease model workflow is illustrated in
.
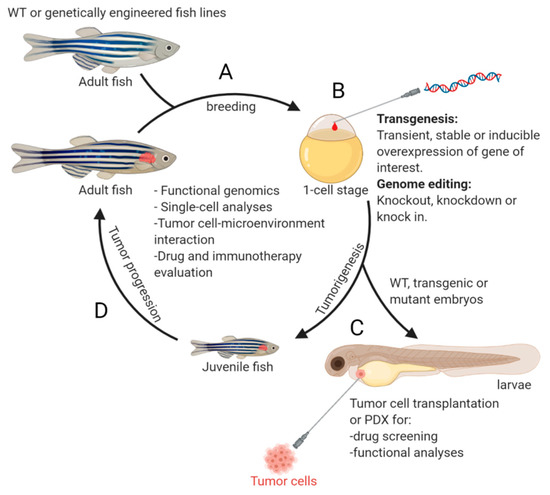
Of course, the advances in understanding NB have not been achieved without obstacles and challenges, some of which appear daunting. In this paper, we aim to:
-
Compare and contrast the zebrafish model with other popular lab animals used as disease models in order to help cancer and other biomedical researchers determine appropriate models for experimental applications (Table 1); and
| Zebrafish | Mouse | Fly | Worm | ||||||
|---|---|---|---|---|---|---|---|---|---|
| New Models Developed | Drugs Tested in the Zebrafish Models | Drugs applied in NB Treatment, Clinical Trials or other Animal Models | |||||||
| Transparency | Fully transparent at embryonic stage and remain translucency through adulthood. PTU can be used to inhibit pigmentation during early embryonic development. Mutant fish line without pigments are available. |
Not transparent | Transparent in larva stage and some parts of the adults | Transparent No pigmentation |
|||||
| Amsterdam, A. et al., 2009 [40] | Retroviral-mediated mutagenesis | Hagoromo Mutants | N/A | N/A | |||||
| Offspring size per mating | Up to 100 | ~3–12 | Up to 500 | Hermaphrodites, varies | |||||
| Zhu, S. et al., 2012 [33] | I-SceI meganuclease mediated transgenesis | Tg(dβh:EGFP-MYCN) and Tg(dβh:EGFP; dβh:ALKF1174L) transgenic fish lines | N/A | N/A | Genetic similarity (humans genome as reference) |
71% | 85% | 50% | 52% |
| Pei, D. et al., 2013 [41] | Morpholino-mediated gene knockdown & transient overexpression of structure variants | Embryos with gain or loss of function of phox2b/PHOX2B | 13–cis retinoic acid (at 1~100 nM) treatment of embryos | Applied to patients with high-risk NB as maintenance therapy after consolidation therapy [42][43] | Immune System | Underdeveloped adaptive immune system in larvae | |||
| He, S. et al., 2016 [44] | I-SceI meganuclease mediated transgenesis | Intact | Does not possess acquired/adaptive immunity | Does not possess acquired/adaptive immunity | |||||
| Tg(dβh: GRD; dβh:mCherry | ) transgenic fish line | Isotretinoin (13-cis retinoic acid, at 1~2 µM) and Trametinib (MEK inhibitor, at 10~40 nM) treatment of juvenile fish | Trametinib is in clinical trials for the treatments of various types of cancers, including high-risk NB (see NCI clinical trial information). | Tumor visualization | Directly visualized in vivo by microscopy | Cannot be easily visualized inside the body | Directly visualized in vivo by microscopy | Directly visualized in vivo by microscopy | |
| Zhang, X. et al., 2017 [45] | I-SceI meganuclease mediated transgenesis | Tg(dβh:Gab2wt;dβh:EGFP) and Tg(dβh:ptpn11E69K-EGFP) transgenic fish lines | CBL0137 (FACT inhibitor, at 4 mM) and Trametinib (MEK inhibitor, at 2 μM) treatment of tumor-bearing fish | CBL0137 is in a clinical trial for the treatment of patients with advanced extremity melanoma or sarcoma with metastasis (see NCI clinical trial information). In TH-MYCN tumor-bearing mice, CBL0137 combined with panobinostat can ablate tumor completely (Oncology Times: December 20, 2018) |
Gene editing tools | ||||
| Morpholino | |||||||||
| Zhu, S. et al., 2017 [46] | I-SceI meganuclease mediated transgenesis | Tg(dβh:LMO1;dβh:mCherry) transgenic fish line | N/A | N/A | Established | Feasible but very limited | |||
| Radic-Sarikas, B. et al., 2017 [47 | Possible but not done yet | ] | Drug treatment | Possible but not done yet | |||||
| N/A | Lapatinib (EGFR inhibitor, at 2 µM) and YM155 (ABCB1 blocker, at 6.5 nM) treatment of tumor-bearing adult fish | Lapatinib is in clinical trials for the treatments of various types of cancers (see NCI clinical trial information). | Retroviral insertion mutagenesis screen | Feasible | Established | dβh:MYCNFeasible | ) transgenic fish linesFeasible | ||
| N/A | N/A | DNA co-injection (I-SceI) Transgenesis | Established, high efficiency | Hypothetical and not efficient | Hypothetical | ||||
| Zimmerman, M. W. et al., 2018 | Possible | ||||||||
| [49 | CRISPR/TALENs | Established | Established | Established | Established | ||||
| Tao, T. et al., 2017 [48] | I-SceI meganuclease mediated transgenesis | Tg(dβh:mCherry;dβh:DEF) and Tg(dβh:EGFP;] | I-SceI meganuclease mediated transgenesis | Tg(dβh:c-MYC; dβh:mCherry) transgenic fish line | N/A | N/A | |||
| Shen, J. et al., 2018 [50] | Injection of tumor cells into the yolk sac of zebrafish embryos | Zebrafish embryos xenografted with SK-N-BE(2)-C human NB cell line | Crizotinib (ALK/MET inhibitor, at 8 μM) and 20a (histone deacetylase inhibitor, at 100 μM) treatment of embryos transplanted with SK-N-BE(2)-C human NB cells. | Crizotinib is in clinical trials for the treatments of various types of cancers, including high-risk NB (see NCI clinical trial information). | Tumor transplantation/Xenograft application | Efficient | Moderate to difficult | N/A | N/A |
| Aveic, S. et al., 2018 [51] | Injection of tumor cells into the duct of Cuvier of zebrafish embryos | Tg(fli1:GFP) zebrafish embryos transplanted with NB3 and SH-SY5Y NB cell lines | TP-0903 (multi-kinase inhibitor) treatment of embryos transplanted with NB3 and SH-SY5Y NB cell lines | TP-0903 is in a clinical trial for the treatment of FLT3 mutated acute myeloid leukemia (see NCI clinical trial information). | Chimeric animal development | Mouse-zebrafish Chimeric | Human-mouse Chimeric | N/A | N/A |
| Seda, M. et al., 2019 [52] | Compound screen using Tg(sox10:gfp) transgenic larvae | N/A | Leflunomide was one of the top hits identified from a library of 640 compounds to regulate cartilage remodelling and NB cell viability. | Leflunomide is approved by FDA for the treatment of active rheumatoid arthritis. It is also in clinical trials for the treatments of various types of cancers (see NCI clinical trial information). | Syngeneic model | Yes | Yes | Yes | N/A |
| Drug screening | Established, high-throughput | Established, low-throughput | Established, high-throughput | Established, high-throughput |
| Publications | Approaches | |||
|---|---|---|---|---|
| Koach, J. et al., 2019 | ||||
| [ | ||||
| 53 | ||||
| ] | ||||
| Tol2 transposon- mediated transgenesis | ||||
| Tg(dβh:PA2G4) | ||||
| transgenic fish line | ||||
| WS6 (175.4 mg/kg, 5 μL) treatment of tumor-bearing fish | ||||
| WS6 can also suppress tumor growth in the | TH | - | MYCN | mouse model and mice xenografted with human NB cell lines [53]. |
| Tao, T. et al., 2020 [54] | I-SceI meganuclease mediated transgenesis | Tg(dβh:EGFP;dβh:LIN28B_WT) and Tg(dβh:EGFP;dβh:LIN28B_MU) transgenic fish lines | N/A | N/A |
| Shi, H. et al., 2020 [55] | CRISPR/Cas9-mediate gene knockout | arid1aa and arid1ab knockout fish lines | N/A | N/A |
| Dong, Z. et al., 2021 [56] | TALEN-mediate gene knockout | gas7 knockout fish line | N/A | N/A |
Since topics related to PSNS development in zebrafish and mammals and NB genetics have been previously covered in detail by ourselves and others [57][58], we will not be addressing these subjects in this review.
2. Advantages of Using Zebrafish as a Model for NB Research
2.1. Translucency of Juvenile and Adult Fish
2.1.1. Early Detection of Tumor Onset
ForSince the TH-MYCN mouse model, palpation is still the most common method used by researchers to examine tumor formationics related to PSNS [22][53]. Howdever, because tumlors predominantly originate from the abdominal ganglion structures [22], it can be tecpment in zebrafishnically challenging for researchers to detect the early onset of tumorigenesis and track the rate of tumor progression by touch. Recently, PET or MRI scanand mammals and NB genetics have been used to evaluate the growth of TH-MYCN tprevioumors in the presence or absence of different compounds, such as the Aurora A kinase or mTOR inhibitor [59][60]. Thely covered in detail by ourse tlvechniques seem promising and sensitive for monitoring tumor progression. However, they require expensives and others equipment[57][58], trainwed professionals, and lengthy procedures. In contrast, the onset of NB can be easily detected by fluorescent microscopy in the zebrafish model and large cohorts of animals (over 400 fish) can be monitored efficiently and regularly without harming the fish as early as 5 weeks post fertilization (wpf) will not be addressing these subjects in this review.
2. Advantages of Using Zebrafish as a Model for NB Research
2.1. Translucency of Juvenile and Adult Fish
2.1.1. Early Detection of Tumor Onset
2.1.2. Real-Time Monitoring of Tumor Progression and Metastasis
In additi
2.1.3. Efficient Evaluation of the Efficacy of Drug Treatment
2.2. Robustness in Genome Editing and Manipulation of Gene Expression
2.2.1. Retroviral-Mediated Mutagenesis
2.2.2. I-SceI Meganuclease-Mediated Transgenesis
2.2.3. Genome Editing with Clustered Regularly Interspaced Short Palindromic Repeats (CRISPR) and Transcription Activator-Like Effector Nucleases (TALENs)
2.2.4. Other Potentially Useful Methods
2.3. High Throughput Transplantation, Patient-Derived Xenograft (PDX) and In Vivo Drug Screening Using Zebrafish Larvae
Ton to being useful in the early detection of tumor onset, fluorescent-tagged tumor cells can be monitored for the growth of tumor, especially tumor metastasis in real-time. In transgenic fish with overexpression ofunderstand disease pathogenesis and screen or validate drug efficacy in vivo, scientists have successfully MYCN trand LMO1 oncogenes, fpluorescent-positiveanted tumor masses were observed using fluorescent microscopy in the distant regions from the primary tumor site, the interrenal gland region (IRG), as early as five weeks of age [46]. Usicells with different genetic alteration(s) or manipulated gene expression, as well as patieng pathological and immunohistochemical analyses with antibodies against neuroblastoma markers, such as Tyrosine Hydroxylase (TH) [61] and HuC [62], w-derived tumor cells into zebrafish at embryonidespreadc metastasis were detected in the orbit, gill, spleen, distal portion of kidneystage or adulthood and heart [4689]. AdditionSeveral sites of metastases, such as the bone, the liver and the pancreas, were also observed in another newly developed zebrafish model with loss of function of gas7 features of zebrafish larvae make them uniquely suited for these studies, includingene: [56].
Although(i) there are anatomical differences between zebrafish and mammals, most of the organs in mammals have their functional equivalent counterparts in zebrafish. For example, the zebrafish IRG is equivalent to the human adrenal gland, which contains the chromaffin cells (an important cell lineage of origin for NB [63]) intansparent bodies that allow for easy tumor cell injection; (ii) ability to use trackable fluorescent-tagged cells following transplantation; (iii) an immature immune system during early embrposed with interrenal epithelial cells in the head kidneyyonic development, which reduces the chance [64]. Similarly, althoughf the lymph nodes are absent in zebrafish, the spleen and kidney (the sites where Timmune rejection of transplanted tumor cells, B cells, and dendritic cells reside) serve as secondary lymphoid organs in zebrafish [65][66]. The k; and (iv) availabilidnety marrows also function similarly to mammalian bone marrow [65]. Tof large clutche gills of zebrafish have a similar structure to the mammalian airways and fulfill the same gas-exchange function, making it analogous to the mammalian lung [67][68]. With embryos for transplantation. Multiple injection sites, such functional, physiological and anatomical homology of zebrafish to mammals, the metastases detected in the fish model match quite well to those metastatic sites commonly seen in human NB patients, including the bone marrow (70%), bone (55%), lymph nodes (30%), liver (30%), and brain (18%) [69].s the perivitelline space, pericardial space, yolk, retro-optical region, and brain, have been explored in In addition, increased numbers and networks of picrosirius red-stained collagen fibers, indicating the enhanced stiffness of extracellular matrix, were observed in the fish tumors with overexpression of both MYCN variety of studies to understand the mechanisms of tumor metastasis, and LMO1 [46], whgich is consistent with the concept that increased ECM stiffness contributes to enhanced metastasisgenesis, cellular intravasation/extravasation [90][7091][7192].
Drug Hscreence, thening on zebrafish model offers a unique advantage in dissecting the molecular and cellular basis and the contribution of microenvironment to NB metastasis.
2.1.3. Efficient Evaluation of the Efficacy of Drug Treatment
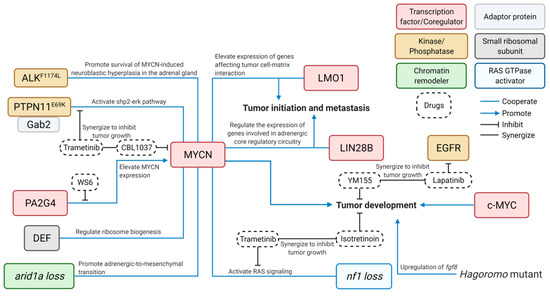
Ittransplants or xenografts is another exemplary usage isof challenging to develop effective and safe targeted therapies for cancer patients, especially for children. To do so requires using an entire organism to test the efficacy of new drugs or novel combinations thereof and evaluate the drug toxicity in order to develop new targeted therapies. Several NB-bearing zebrafish modelthis model. Both embryos and adults can be used in high-throughput drug-screening assays. Embryos are relatively easy to work with due to their permeability of small molecules have[93]. bReen used to demonstrate the effectiveness of novel inhibitors and combinations of new compounds in the treatment of NB, along with illuminating of the underlying mechanisms that may contribute to drug resistance in NB therapy (Figure 2).
For searchers have already performed small-molecule drug screeninstance,g using the dβh-MYCN zebrafish NB emodel (designated the MYCN line), Radic-Sabryos trikas et al. demonstrated that the epidermal growth factor receptor (EGFRnsplanted with neural crest stem cells (NCSCs) kinase[52]. Sinhibitor, lapatinib, can prolong and enhance the cytotoxicity of YM155, an anti-cancer drug, by inhibiting the multidrug-resistance efflux transporter ABCB1 [47]. This led ce NB is derived from to the synergistic inhibition of the growth of MYCN-overmpathoadrexpressing NB in vivo. Similarly, using the transgenic fish with overexpression of both MYCN al lineage of neurandl proliferation associated 2G4 (PA2G4), Koacrest ch et al. demonstrated that a lls, small molecule, WS6, can competitively bind to PA2G4 to prevent its interas that inhibit NCSC induction with MYCN, leading to destabilization and reduced expression of MYCN and in turn suppressed growth of NB [53]. Usimight be potentially useful for the NB treatment. Among the s640 FDA-ame zebrafishpproved drugs MYCN modeappl togiether with the transgenic fish overexpressing bothd in this screen, one MYCN and GAB2rug, Zhaleflung et al. demonstrated that the MEKomide, was identified to inhibitor trametinib can enhance the sensitivity of MYCN a NCSC inductiond. GAB2-ovLerexpressiflung NB to the treatment of CBL0137, a histone chaperone FACT omide, as an inhibitor [45]. In addition,f He et al. applied the MEK inhibitor (trametinib) and retinoid (isotretinoin) on the juvenile compound fish with loss of nf1 in thdihydroorotate dehydrogenase context(DHODH) of MYCN overexpression and demonstrated synergistic killing of tumor cells by this combination treatment [44]. Taken together, then immunose zebupprafish NB models showed considerable translational potential for investigating new strategies to improve tessive agent for the treatment of this devastating childhood tumor.
2.2. Robustness in Genome Editing and Manipulation of Gene Expression
Since fish deposit oocytes outside of the body for external fertilization, researchers can easily collect fertilized embryos in large batches to perform genetic editing at the single-cell stage effectively and efficiently. The whole process—from breeding the fish to injections of transgene constructs or genome editing reagents into a few hundred embryos—takes only two days. Zebrafish are relatively simple to use when compared to rodent models that require multiple time-consuming steps including a period of superovulation by pregnant mare serum (PMS) in the early afternoon followed by human chorionic gonadotropin treatment [72], labor-intensive oocyte harvesting, in vitro fertilization, and embryo implantation.
The relative ease-of-use of zebrafish embryos compared to mice allows researchers to rapidly and economically develop a large number of genetically engineered fish lines in a short period of time. Various genetic editing methods have proven effective in zebrafish and this article will focus on approaches used in NB research and how they may be applicable and transferable to other research settings.
2.2.1. Retroviral-Mediated Mutagenesis
Rpatients with rhetrovirus-mediated insertional mutagenesis in zebrafish was established in 1996 [73]. It imatoid arthritis, has already been s a phowerful forward-genetic approach for identifying genes that are critical for embryonic development [74].
In 2009, len to reduce proliferation and by Drs. Nancy Hopkins and Jacqueline Lees, a team of researchers performed a comprehensive insertional mutagenesis screen in zebrafish with a goal to identify genes susceptible to cancer. Four mutant fish lines with viral insertionsduce apoptosis in NB cells both in vitro and in vivo in[94]. thHe fbxw4 gene were identified. Interestingly, the mis-regulated fibroblast growth factor 8 (fgf8) in thnce, this result further dese mutants was found to contribute to neuroblastomagenesisonstrates [40].
Although the forward-genetic approach was successful, it was quite challenging to screen for fish with spontaneous development of NB and to effectively identify disease-driving genes. Therefore, the reverse-genetic strategy became a more popular approach to model NB using the zebrafish in the past decade.
2.2.2. I-SceI Meganuclease-Mediated Transgenesis
Usimportant application of zebrafish as an unbiased in vivo system for effective drug screening. a reverse-genetic approach, the first transgenic Recently, zebrafish model of neuroblastoma was developed in Dr. Thomas Look’s laboratory in 2012. Two stable transgenic lines with overexpression of MYCN or ALKF1174L wtransplanted with human NB cells have bere generated using meganuclease (I-SceI)-mediated transgenic strategy [33]. It is in used to demportant to note that human genes, rather than zebrafish genes, are commonly chosen for developing a transgenic zebrafish NB model. Doing so allows researchers to avoid the potential issues of targeting transgenes or validating the efficacy of drugs designed to interact with human proteins.
Thenstrate the effect of a new multi-kinase drug, TP-0903, on reducing extravasation and inducing tumor I-SceI-mediated transgenesis is highly efficient in zebrafish and frogs, although the transgene integration rate seems low in other species [75][76]. The strll death, suggesting the therapeutic potentiategyl to build transgene constructs for a NB zebrafish modelof this compound for the NB treatment is simi[51].
Alar to that of thehough TH-MYCNPDX mouse model.s However, thare TH prcomotnsider did not work well in zebrafish. Therefore,ed the gold standard for the dβhin prvivomoter was used instead to drive expressi validation of genes of interest in the PSNS [33]. The dβh-EGFP-MYCN condrug efficacy, the struct was used to generate the first zebrafish NB model (designated as MYCN line). Due to the instability of MYCN protein, EGFP-MYCN expression was visible under fluorescence microscopy but was not strongdies led by Drs. Ferreira and Fior, have demonstrated that the patient-derived zebrafish xenografts [33]. Hence(zPDX, researchers applied a strategy of co-injection of dβh-EGFP olso called cancer dβh-mCherry DNA with dβh promoter-dri“aven atransgene constructs containing genes of interest for the subsequent development of multiple transgenic lines.
Thars”) can be used to sense co-ianjected DNA has been demonstrated to be co-integrated into the fish genome, leading to their co-expression in the PSNS cells [33]cer behavior and screen for potential novel therapies. The EGFP expreUssion is detectable in the embryos as early as one-day post fertilization [33]. Hence, ing a panel of zebrafish xenografts withe EGFP-positive embryos can be easily identified and sorted using fluorescent microscopy, which eliminates additional genotyping procedures after the transgenic line is confirmed. This approach is cost-efficient for conducting large-scale experiments (which require screening hundreds of transgenic fish) at a time when lab space is at a premium.
Uspatient-derived colorectal cancers, Ferreira and Fior rapidly screened the available therapeutingc a similar transgenesis approach, researchers have developed a panel of transgenic lines with overexpression of control reporters or genes of interest. These include transgenic fish lines overexpressingoptions for the colorectal cancers and predicted the treatment outcomes EGFP [3392] or mCherry [4695], alone as a control group, or NB-relevant genes includinwhich set the groundwork for using ALKwtzPDX [33],as PTPN11a [45], GAB2 [45], LIN28B WT [54], LIN28B_MU [54], LMO1 [46], ranpid DEF [48]. Interestingly, most of the aforementioned genes have been shown to cooperate with MYCN to contribute to the pathogenesis of NB through different cellular mechanisms (Figure 2). Hvivo screening platform for future persowever, none of these genes can sufficiently drive NB tumorigenesis when they were overexpressed alone, suggestalized cancer treatments. Following that MYCN is a potent oncogene and a key driver for the NB initiation.
Recentlyese efforts, a newer shigh-table transgenichroughput zebrafish line with overexpression of MYCNxenograft assay of under control of the dβh preuroblastomotera was developed in Dr. Thomas Look’s lab [48]. Instperformead of using the DNA construct carrying a dβh-EGFP-MYCN fusto confirm cannabion gene, two transgenic constructs,oid receptor dβh-MYCN2 (cDCNA+3′UTRR2) and dβh-EGFP, were coMinjected into the one-cell stage of wild-type embryos in developing this line (designated asgen-activated protein kinase 8 TgMYCN_TT(MAPK8). The advantage of the coinjection approach has been illustrated above. The major difference of the TgMYCN_TT ls promising candidates for the treatment of hine to tghe MYCN l-rine is the inclusion of the 3′ UTR ofk NB and to MYCN gene, containing microRNA rdecognition sites [77], in tntify the tdransgene construct. Thus, the MYCN expression iugs GW405833 an the TgMYCN_TT line could beAS601245 regulated at the post-transcriptional level, which might link to the increased penetrance of NB to ~70% by 29 wpfas the most effective and well-tolerated CNR2 and [48]MAPK8 in the TgMYCN_TT linargeted as compared to ~30% in the MYCN lcompounds to inhine by 25 wpf [46]. Morit theover, overexpression of c-MYC—agrowth of highly expressed MYC-family gene in the MYCN nenon-gramplified high-risk NBs—in the PSNS of transgenic fts in zebrafish c[96].
Tould also induce NB tumorigenesisbetter [49], thus mimplicating the key oncogenic role of MYC family genes in NB pathogenesis.
2.2.3. Genome Editing with Clustered Regularly Interspaced Short Palindromic Repeats (CRISPR) and Transcription Activator-Like Effector Nucleases (TALENs)
CRISPR the cytokine-enriched microenvironment found in human patients can effectively target genes of interest and achieve up to 75–99% modification rate in zebrafishor xenotransplantation, Dr. Berman’s [78][79]. This high-throughput targeting can generate close to 30% germline mutations [80] wip generated the firsth as low as 1% of off-target effects [81]. Shi et umal. generatnized arid1aa and arid1ab knockout zebrafish using this robust technology in the NB model with MYCN oy overexpression to examine the relevance of ARID1A ag trans a NB suppressor gene in vivo and found that both arid1aa and arid1ab defs encoding human hematopoiciency increases penetration of MYCN-drtic-specifivenc NB in zebrafish [55]. Alternativelycytokines, targeted deletion in tsuche zebrafish genome can be achieved efficiently using the TALENs system [82]. With this method, as GM-CSF, stem cell factor (SCF), or stroma gas7l knockout fish line was generated and bred with theell-derived factor 1α (SDF1α). MYCN tTransgenic line,s which led to development of widespread metastasis in the compound fishith overexpression of each of the individual [56].
2.2.4. Other Potentially Useful Methods
Givgen that zebrafish modeling for NB research has only become common in the last decade, there are still a plethora of research methods in the zebrafish that have not been applied to the NB study, but that have potential to be used in the near future. For example, the Cre-Lox recombination, a site-specific recombinase technology, has been widely used in different model systems. Briefly, a ubiquitously or tissue-specifically expressed tyrosine recombinase enzyme (Cre) can be used to recombine a pair of short target sequences, called the Lox sequences, leading to manipulation of DNA sequence at specific mentioned above were developed first using Tol2 transposon-mediated transgenic approach and then incrossed to generate a compound transgenic fish line with overexpression of all of the aforementioned cytokines (GM-CSF, SCF, and SDF1α) (designated GSS fish) sites[97]. Such Cre-LoxP conditional expression approach has been used to target the oncogenic Kras/Ras pathway under control of atient-derived leukemias transplanted into the nestinGSS promotzer [83]. Gbrafiven tshat many lineage-specific promoters are available in zebrafish, this system is highly applicable when studies require specific activation of gene of interest in a given subset of cell popul exhibit improved survival, self-renewal ability and broader clonal representation. For example, Langenau et al. was able to express mouse c-Myc in lymphoblasts ofTherefore, the GSS fish establish a new standard for zebrafish using this experimental approach [84]. Hexenotrat shock inducible promoters also appear to be applicable which has been used to track individual retina-neuron axon pathways [85]. Addisplantation thationally, tamoxifen-inducible Cre recombinase can conditionally activate gene expression when needed, such as CreERT2 in zebrafish [86]. The whmore accurately recapitulates the human colnte system can be easily integrated using CRISPR/Cas9 in zebrafish [87]; xt for evaluating persona detaliled review on the zebrafish Cre-LoxP system has previously beenzed treatment published [88].
2.3. High Throughput Transplantation, Patient-Derived Xenograft (PDX) and In Vivo Drug Screening Using Zebrafish Larvae
To understand disease pathogenesis and screen or validate drug efficacy in vivo, scientists have successfully transplanted tumor cells with different genetic alteration(s) or manipulated gene expression, as well as patient-derived tumor cells into zebrafish at embryonic stage or adulthood [89]. Several features of zebrafish larvae make them uniquely suited for these studies, including: (i) transparent bodies that allow for easy tumor cell injection; (ii) ability to use trackable fluorescent-tagged cells following transplantation; (iii) an immature immune system during early embryonic development, which reduces the chance of the immune rejection of transplanted tumor cells; and (iv) availability of large clutches of embryos for transplantation. Multiple injection sites, such as the perivitelline space, pericardial space, yolk, retro-optical region, and brain, have been explored in a variety of studies to understand the mechanisms of tumor metastasis, angiogenesis, cellular intravasation/extravasation [90][91][92].
Drug screening on zebrafish transplants or xenografts is another exemplary usage of this model. Both embryos and adults can be used in high-throughput drug-screening assays. Embryos are relatively easy to work with due to their permeability of small molecules [93]. Researchers have already performed small-molecule drug screening using zebrafish embryos transplanted with neural crest stem cells (NCSCs) [52]. Since NB is derived from the sympathoadrenal lineage of neural crest cells, small molecules that inhibit NCSC induction might be potentially useful for the NB treatment. Among the 640 FDA-approved drugs applied in this screen, one drug, leflunomide, was identified to inhibit NCSC induction. Leflunomide, as an inhibitor of dihydroorotate dehydrogenase (DHODH) and an immunosuppressive agent for the treatment of patients with rheumatoid arthritis, has already been shown to reduce proliferation and induce apoptosis in NB cells both in vitro and in vivo [94]. Hence, this result further demonstrates the important application of zebrafish as an unbiased in vivo system for effective drug screening. Recently, zebrafish transplanted with human NB cells have been used to demonstrate the effect of a new multi-kinase drug, TP-0903, on reducing extravasation and inducing tumor cell death, suggesting the therapeutic potential of this compound for the NB treatment [51].
Although PDX mouse models are considered the gold standard for the in vivo validation of drug efficacy, the studies led by Drs. Ferreira and Fior, have demonstrated that the patient-derived zebrafish xenografts (zPDX, also called cancer “avatars”) can be used to sense cancer behavior and screen for potential novel therapies. Using a panel of zebrafish xenografts with patient-derived colorectal cancers, Ferreira and Fior rapidly screened the available therapeutic options for the colorectal cancers and predicted the treatment outcomes [92][95], which set the groundwork for using zPDX as a rapid in vivo screening platform for future personalized cancer treatments. Following these efforts, a high-throughput zebrafish xenograft assay of neuroblastoma was performed to confirm cannabinoid receptor 2 (CNR2) and Mitogen-activated protein kinase 8 (MAPK8) as promising candidates for the treatment of high-risk NB and to identify the drugs GW405833 and AS601245 as the most effective and well-tolerated CNR2 and MAPK8 targeted compounds to inhibit the growth of xenografts in zebrafish [96].
To better mimic the cytokine-enriched microenvironment found in human patients for xenotransplantation, Dr. Berman’s group generated the first humanized zebrafish by overexpressing transgenes encoding human hematopoietic-specific cytokines, such as GM-CSF, stem cell factor (SCF), or stromal cell-derived factor 1α (SDF1α). Transgenic lines with overexpression of each of the individual gene mentioned above were developed first using Tol2 transposon-mediated transgenic approach and then incrossed to generate a compound transgenic fish line with overexpression of all of the aforementioned cytokines (GM-CSF, SCF, and SDF1α) (designated GSS fish) [97]. Patient-derived leukemias transplanted into the GSS zebrafish exhibit improved survival, self-renewal ability and broader clonal representation. Therefore, the GSS fish establish a new standard for zebrafish xenotransplantation that more accurately recapitulates the human context for evaluating personalized treatment [97].
.
References
- Stanton, M.F. Diethylnitrosamine-Induced Hepatic Degeneration and Neoplasia in the Aquarium Fish, Brachydanio Rerio. J. Natl. Cancer Inst. 1965, 34, 117–130.
- Langenau, D.M.; Traver, D.; Ferrando, A.A.; Kutok, J.L.; Aster, J.C.; Kanki, J.P.; Lin, S.; Prochownik, E.; Trede, N.S.; Zon, L.I.; et al. Myc-induced T cell leukemia in transgenic zebrafish. Science 2003, 299, 887–890.
- Etchin, J.; Kanki, J.P.; Look, A.T. Zebrafish as a model for the study of human cancer. Methods Cell Biol. 2011, 105, 309–337.
- Feitsma, H.; Cuppen, E. Zebrafish as a cancer model. Mol. Cancer Res. 2008, 6, 685–694.
- Benjamin, D.C.; Hynes, R.O. Intravital imaging of metastasis in adult Zebrafish. BMC Cancer 2017, 17, 660.
- Kim, I.S.; Heilmann, S.; Kansler, E.R.; Zhang, Y.; Zimmer, M.; Ratnakumar, K.; Bowman, R.L.; Simon-Vermot, T.; Fennell, M.; Garippa, R.; et al. Microenvironment-derived factors driving metastatic plasticity in melanoma. Nat. Commun. 2017, 8, 14343.
- Colon, N.C.; Chung, D.H. Neuroblastoma. Adv. Pediatr. 2011, 58, 297–311.
- Maris, J.M. Recent advances in neuroblastoma. N. Engl. J. Med. 2010, 362, 2202–2211.
- Brodeur, G.M. Neuroblastoma: Biological insights into a clinical enigma. Nat. Rev. Cancer 2003, 3, 203–216.
- Pearson, A.D.; Pinkerton, C.R.; Lewis, I.J.; Imeson, J.; Ellershaw, C.; Machin, D. High-dose rapid and standard induction chemotherapy for patients aged over 1 year with stage 4 neuroblastoma: A randomised trial. Lancet Oncol. 2008, 9, 247–256.
- Proust-Houdemont, S.; Pasqualini, C.; Blanchard, P.; Dufour, C.; Benhamou, E.; Goma, G.; Semeraro, M.; Raquin, M.A.; Hartmann, O.; Valteau-Couanet, D. Busulfan-melphalan in high-risk neuroblastoma: The 30-year experience of a single institution. Bone Marrow Transplant. 2016, 51, 1076–1081.
- Yu, A.L.; Gilman, A.L.; Ozkaynak, M.F.; London, W.B.; Kreissman, S.G.; Chen, H.X.; Smith, M.; Anderson, B.; Villablanca, J.G.; Matthay, K.K.; et al. Anti-GD2 antibody with GM-CSF, interleukin-2, and isotretinoin for neuroblastoma. N. Engl. J. Med. 2010, 363, 1324–1334.
- Yu, A.L.; Gilman, A.L.; Ozkaynak, M.F.; Naranjo, A.; Diccianni, M.B.; Gan, J.; Hank, J.A.; Batova, A.; London, W.B.; Tenney, S.C.; et al. Long-term follow-up of a Phase III Study of ch14.18 (Dinutuximab) + Cytokine Immunotherapy in Children with High-risk Neuroblastoma: COG Study ANBL0032. Clin. Cancer Res. 2021.
- Basta, N.O.; Halliday, G.C.; Makin, G.; Birch, J.; Feltbower, R.; Bown, N.; Elliott, M.; Moreno, L.; Barone, G.; Pearson, A.D.; et al. Factors associated with recurrence and survival length following relapse in patients with neuroblastoma. Br. J. Cancer 2016, 115, 1048–1057.
- London, W.B.; Bagatell, R.; Weigel, B.J.; Fox, E.; Guo, D.; Van Ryn, C.; Naranjo, A.; Park, J.R. Historical time to disease progression and progression-free survival in patients with recurrent/refractory neuroblastoma treated in the modern era on Children’s Oncology Group early-phase trials. Cancer-Am. Cancer Soc. 2017, 123, 4914–4923.
- Schwab, M.; Alitalo, K.; Klempnauer, K.H.; Varmus, H.E.; Bishop, J.M.; Gilbert, F.; Brodeur, G.; Goldstein, M.; Trent, J. Amplified DNA with limited homology to myc cellular oncogene is shared by human neuroblastoma cell lines and a neuroblastoma tumour. Nature 1983, 305, 245–248.
- Huang, M.; Weiss, W.A. Neuroblastoma and MYCN. Cold Spring Harb. Perspect. Med. 2013, 3, a014415.
- Brodeur, G.M.; Seeger, R.C.; Schwab, M.; Varmus, H.E.; Bishop, J.M. Amplification of N-myc in untreated human neuroblastomas correlates with advanced disease stage. Science 1984, 224, 1121–1124.
- Seeger, R.C.; Brodeur, G.M.; Sather, H.; Dalton, A.; Siegel, S.E.; Wong, K.Y.; Hammond, D. Association of multiple copies of the N-myc oncogene with rapid progression of neuroblastomas. N. Engl. J. Med. 1985, 313, 1111–1116.
- Schwab, M.; Varmus, H.E.; Bishop, J.M. Human N-myc gene contributes to neoplastic transformation of mammalian cells in culture. Nature 1985, 316, 160–162.
- Land, H.; Parada, L.F.; Weinberg, R.A. Tumorigenic conversion of primary embryo fibroblasts requires at least two cooperating oncogenes. Nature 1983, 304, 596–602.
- Weiss, W.A.; Aldape, K.; Mohapatra, G.; Feuerstein, B.G.; Bishop, J.M. Targeted expression of MYCN causes neuroblastoma in transgenic mice. EMBO J. 1997, 16, 2985–2995.
- Berry, T.; Luther, W.; Bhatnagar, N.; Jamin, Y.; Poon, E.; Sanda, T.; Pei, D.; Sharma, B.; Vetharoy, W.R.; Hallsworth, A.; et al. The ALK(F1174L) mutation potentiates the oncogenic activity of MYCN in neuroblastoma. Cancer Cell 2012, 22, 117–130.
- Heukamp, L.C.; Thor, T.; Schramm, A.; De Preter, K.; Kumps, C.; De Wilde, B.; Odersky, A.; Peifer, M.; Lindner, S.; Spruessel, A.; et al. Targeted expression of mutated ALK induces neuroblastoma in transgenic mice. Sci. Transl. Med. 2012, 4, 141ra191.
- Cazes, A.; Lopez-Delisle, L.; Tsarovina, K.; Pierre-Eugene, C.; De Preter, K.; Peuchmaur, M.; Nicolas, A.; Provost, C.; Louis-Brennetot, C.; Daveau, R.; et al. Activated Alk triggers prolonged neurogenesis and Ret upregulation providing a therapeutic target in ALK-mutated neuroblastoma. Oncotarget 2014, 5, 2688–2702.
- Hennchen, M.; Stubbusch, J.; Abarchan-El Makhfi, I.; Kramer, M.; Deller, T.; Pierre-Eugene, C.; Janoueix-Lerosey, I.; Delattre, O.; Ernsberger, U.; Schulte, J.B.; et al. Lin28B and Let-7 in the Control of Sympathetic Neurogenesis and Neuroblastoma Development. J. Neurosci. 2015, 35, 16531–16544.
- Molenaar, J.J.; Domingo-Fernandez, R.; Ebus, M.E.; Lindner, S.; Koster, J.; Drabek, K.; Mestdagh, P.; van Sluis, P.; Valentijn, L.J.; van Nes, J.; et al. LIN28B induces neuroblastoma and enhances MYCN levels via let-7 suppression. Nat. Genet. 2012, 44, 1199–1206.
- Servenius, B.; Vernachio, J.; Price, J.; Andersson, L.C.; Peterson, P.A. Metastasizing neuroblastomas in mice transgenic for simian virus 40 large T (SV40T) under the olfactory marker protein gene promoter. Cancer Res. 1994, 54, 5198–5205.
- Iwakura, H.; Ariyasu, H.; Kanamoto, N.; Hosoda, K.; Nakao, K.; Kangawa, K.; Akamizu, T. Establishment of a novel neuroblastoma mouse model. Int. J. Oncol. 2008, 33, 1195–1199.
- Hattori, Y.; Kanamoto, N.; Kawano, K.; Iwakura, H.; Sone, M.; Miura, M.; Yasoda, A.; Tamura, N.; Arai, H.; Akamizu, T.; et al. Molecular characterization of tumors from a transgenic mouse adrenal tumor model: Comparison with human pheochromocytoma. Int. J. Oncol. 2010, 37, 695–705.
- Kamili, A.; Atkinson, C.; Trahair, T.N.; Fletcher, J.I. Mouse models of high-risk neuroblastoma. Cancer Metastasis Rev. 2020, 39, 261–274.
- Ornell, K.J.; Coburn, J.M. Developing preclinical models of neuroblastoma: Driving therapeutic testing. BMC Biomed. Eng. 2019, 1.
- Zhu, S.; Lee, J.S.; Guo, F.; Shin, J.; Perez-Atayde, A.R.; Kutok, J.L.; Rodig, S.J.; Neuberg, D.S.; Helman, D.; Feng, H.; et al. Activated ALK collaborates with MYCN in neuroblastoma pathogenesis. Cancer Cell 2012, 21, 362–373.
- Mosse, Y.P.; Laudenslager, M.; Longo, L.; Cole, K.A.; Wood, A.; Attiyeh, E.F.; Laquaglia, M.J.; Sennett, R.; Lynch, J.E.; Perri, P.; et al. Identification of ALK as a major familial neuroblastoma predisposition gene. Nature 2008, 455, 930–935.
- Chen, Y.; Takita, J.; Choi, Y.L.; Kato, M.; Ohira, M.; Sanada, M.; Wang, L.; Soda, M.; Kikuchi, A.; Igarashi, T.; et al. Oncogenic mutations of ALK kinase in neuroblastoma. Nature 2008, 455, 971–974.
- Janoueix-Lerosey, I.; Lequin, D.; Brugieres, L.; Ribeiro, A.; de Pontual, L.; Combaret, V.; Raynal, V.; Puisieux, A.; Schleiermacher, G.; Pierron, G.; et al. Somatic and germline activating mutations of the ALK kinase receptor in neuroblastoma. Nature 2008, 455, 967–970.
- George, R.E.; Sanda, T.; Hanna, M.; Frohling, S.; Luther, W., 2nd; Zhang, J.; Ahn, Y.; Zhou, W.; London, W.B.; McGrady, P.; et al. Activating mutations in ALK provide a therapeutic target in neuroblastoma. Nature 2008, 455, 975–978.
- Carpenter, E.L.; Mosse, Y.P. Targeting ALK in neuroblastoma—Preclinical and clinical advancements. Nat. Rev. Clin. Oncol. 2012, 9, 391–399.
- Umapathy, G.; Mendoza-Garcia, P.; Hallberg, B.; Palmer, R.H. Targeting anaplastic lymphoma kinase in neuroblastoma. APMIS 2019, 127, 288–302.
- Amsterdam, A.; Lai, K.; Komisarczuk, A.Z.; Becker, T.S.; Bronson, R.T.; Hopkins, N.; Lees, J.A. Zebrafish Hagoromo mutants up-regulate fgf8 postembryonically and develop neuroblastoma. Mol. Cancer Res. 2009, 7, 841–850.
- Pei, D.; Luther, W.; Wang, W.; Paw, B.H.; Stewart, R.A.; George, R.E. Distinct neuroblastoma-associated alterations of PHOX2B impair sympathetic neuronal differentiation in zebrafish models. PLoS Genet. 2013, 9, e1003533.
- Brodeur, G.M.; Hogarty, M.; Bagatell, R.; Mosse, Y.P.; Maris, J.M. Neuroblastoma. In Principles and Practice of Pediatric Oncology, 7th ed.; Pizzo, P.A., Poplack, D., Eds.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2016; pp. 772–792.
- Pinto, N.R.; Applebaum, M.A.; Volchenboum, S.L.; Matthay, K.K.; London, W.B.; Ambros, P.F.; Nakagawara, A.; Berthold, F.; Schleiermacher, G.; Park, J.R.; et al. Advances in Risk Classification and Treatment Strategies for Neuroblastoma. J. Clin. Oncol. 2015, 33, 3008–3017.
- He, S.; Mansour, M.R.; Zimmerman, M.W.; Ki, D.H.; Layden, H.M.; Akahane, K.; Gjini, E.; de Groh, E.D.; Perez-Atayde, A.R.; Zhu, S.; et al. Synergy between loss of NF1 and overexpression of MYCN in neuroblastoma is mediated by the GAP-related domain. Elife 2016, 5.
- Zhang, X.; Dong, Z.; Zhang, C.; Ung, C.Y.; He, S.; Tao, T.; Oliveira, A.M.; Meves, A.; Ji, B.; Look, A.T.; et al. Critical Role for GAB2 in Neuroblastoma Pathogenesis through the Promotion of SHP2/MYCN Cooperation. Cell Rep. 2017, 18, 2932–2942.
- Zhu, S.; Zhang, X.; Weichert-Leahey, N.; Dong, Z.; Zhang, C.; Lopez, G.; Tao, T.; He, S.; Wood, A.C.; Oldridge, D.; et al. LMO1 Synergizes with MYCN to Promote Neuroblastoma Initiation and Metastasis. Cancer Cell 2017, 32, 310–323.e315.
- Radic-Sarikas, B.; Halasz, M.; Huber, K.V.M.; Winter, G.E.; Tsafou, K.P.; Papamarkou, T.; Brunak, S.; Kolch, W.; Superti-Furga, G. Lapatinib potentiates cytotoxicity of YM155 in neuroblastoma via inhibition of the ABCB1 efflux transporter. Sci. Rep. 2017, 7, 3091.
- Tao, T.; Sondalle, S.B.; Shi, H.; Zhu, S.; Perez-Atayde, A.R.; Peng, J.; Baserga, S.J.; Look, A.T. The pre-rRNA processing factor DEF is rate limiting for the pathogenesis of MYCN-driven neuroblastoma. Oncogene 2017, 36, 3852–3867.
- Zimmerman, M.W.; Liu, Y.; He, S.; Durbin, A.D.; Abraham, B.J.; Easton, J.; Shao, Y.; Xu, B.; Zhu, S.; Zhang, X.; et al. MYC Drives a Subset of High-Risk Pediatric Neuroblastomas and Is Activated through Mechanisms Including Enhancer Hijacking and Focal Enhancer Amplification. Cancer Discov. 2018, 8, 320–335.
- Shen, J.; Najafi, S.; Stable, S.; Fabian, J.; Koeneke, E.; Kolbinger, F.R.; Wrobel, J.K.; Meder, B.; Distel, M.; Heimburg, T.; et al. A kinome-wide RNAi screen identifies ALK as a target to sensitize neuroblastoma cells for HDAC8-inhibitor treatment. Cell Death Differ. 2018, 25, 2053–2070.
- Aveic, S.; Corallo, D.; Porcu, E.; Pantile, M.; Boso, D.; Zanon, C.; Viola, G.; Sidarovich, V.; Mariotto, E.; Quattrone, A.; et al. TP-0903 inhibits neuroblastoma cell growth and enhances the sensitivity to conventional chemotherapy. Eur. J. Pharmacol. 2018, 818, 435–448.
- Seda, M.; Geerlings, M.; Lim, P.; Jeyabalan-Srikaran, J.; Cichon, A.C.; Scambler, P.J.; Beales, P.L.; Hernandez-Hernandez, V.; Stoker, A.W.; Jenkins, D. An FDA-Approved Drug Screen for Compounds Influencing Craniofacial Skeletal Development and Craniosynostosis. Mol. Syndromol. 2019, 10, 98–114.
- Koach, J.; Holien, J.K.; Massudi, H.; Carter, D.R.; Ciampa, O.C.; Herath, M.; Lim, T.; Seneviratne, J.A.; Milazzo, G.; Murray, J.E.; et al. Drugging MYCN Oncogenic Signaling through the MYCN-PA2G4 Binding Interface. Cancer Res. 2019, 79, 5652–5667.
- Tao, T.; Shi, H.; Mariani, L.; Abraham, B.J.; Durbin, A.D.; Zimmerman, M.W.; Powers, J.T.; Missios, P.; Ross, K.N.; Perez-Atayde, A.R.; et al. LIN28B regulates transcription and potentiates MYCN-induced neuroblastoma through binding to ZNF143 at target gene promotors. Proc. Natl. Acad. Sci. USA 2020, 117, 16516–16526.
- Shi, H.; Tao, T.; Abraham, B.J.; Durbin, A.D.; Zimmerman, M.W.; Kadoch, C.; Look, A.T. ARID1A loss in neuroblastoma promotes the adrenergic-to-mesenchymal transition by regulating enhancer-mediated gene expression. Sci. Adv. 2020, 6, eaaz3440.
- Dong, Z.; Yeo, K.S.; Lopez, G.; Zhang, C.; Dankert Eggum, E.N.; Rokita, J.L.; Ung, C.Y.; Levee, T.M.; Her, Z.P.; Howe, C.J.; et al. GAS7 Deficiency Promotes Metastasis in MYCN-driven Neuroblastoma. Cancer Res. 2021.
- Corallo, D.; Candiani, S.; Ori, M.; Aveic, S.; Tonini, G.P. The zebrafish as a model for studying neuroblastoma. Cancer Cell Int. 2016, 16, 82.
- Casey, M.J.; Stewart, R.A. Zebrafish as a model to study neuroblastoma development. Cell Tissue Res. 2018, 372, 223–232.
- Quarta, C.; Cantelli, E.; Nanni, C.; Ambrosini, V.; D’Ambrosio, D.; Di Leo, K.; Angelucci, S.; Zagni, F.; Lodi, F.; Marengo, M.; et al. Molecular imaging of neuroblastoma progression in TH-MYCN transgenic mice. Mol. Imaging Biol. 2013, 15, 194–202.
- Almeida, G.S.; Panek, R.; Hallsworth, A.; Webber, H.; Papaevangelou, E.; Boult, J.K.; Jamin, Y.; Chesler, L.; Robinson, S.P. Pre-clinical imaging of transgenic mouse models of neuroblastoma using a dedicated 3-element solenoid coil on a clinical 3T platform. Br. J. Cancer 2017, 117, 791–800.
- Teitelman, G.; Baker, H.; Joh, T.H.; Reis, D.J. Appearance of catecholamine-synthesizing enzymes during development of rat sympathetic nervous system: Possible role of tissue environment. Proc. Natl. Acad. Sci. USA 1979, 76, 509–513.
- Marusich, M.F.; Furneaux, H.M.; Henion, P.D.; Weston, J.A. Hu neuronal proteins are expressed in proliferating neurogenic cells. J. Neurobiol. 1994, 25, 143–155.
- Dong, R.; Yang, R.; Zhan, Y.; Lai, H.D.; Ye, C.J.; Yao, X.Y.; Luo, W.Q.; Cheng, X.M.; Miao, J.J.; Wang, J.F.; et al. Single-Cell Characterization of Malignant Phenotypes and Developmental Trajectories of Adrenal Neuroblastoma. Cancer Cell 2020, 38, 716–733.e716.
- Hsu, H.J.; Lin, G.; Chung, B.C. Parallel early development of zebrafish interrenal glands and pronephros: Differential control by wt1 and ff1b. Development 2003, 130, 2107–2116.
- Renshaw, S.A.; Trede, N.S. A model 450 million years in the making: Zebrafish and vertebrate immunity. Dis. Model. Mech. 2012, 5, 38–47.
- Lieschke, G.J.; Trede, N.S. Fish immunology. Curr. Biol. 2009, 19, R678–R682.
- Menke, A.L.; Spitsbergen, J.M.; Wolterbeek, A.P.; Woutersen, R.A. Normal anatomy and histology of the adult zebrafish. Toxicol. Pathol. 2011, 39, 759–775.
- Progatzky, F.; Cook, H.T.; Lamb, J.R.; Bugeon, L.; Dallman, M.J. Mucosal inflammation at the respiratory interface: A zebrafish model. Am. J. Physiol. Lung Cell. Mol. Physiol. 2016, 310, L551–L561.
- DuBois, S.G.; Kalika, Y.; Lukens, J.N.; Brodeur, G.M.; Seeger, R.C.; Atkinson, J.B.; Haase, G.M.; Black, C.T.; Perez, C.; Shimada, H.; et al. Metastatic sites in stage IV and IVS neuroblastoma correlate with age, tumor biology, and survival. J. Pediatr. Hematol. Oncol. 1999, 21, 181–189.
- Levental, K.R.; Yu, H.; Kass, L.; Lakins, J.N.; Egeblad, M.; Erler, J.T.; Fong, S.F.; Csiszar, K.; Giaccia, A.; Weninger, W.; et al. Matrix crosslinking forces tumor progression by enhancing integrin signaling. Cell 2009, 139, 891–906.
- Paszek, M.J.; Zahir, N.; Johnson, K.R.; Lakins, J.N.; Rozenberg, G.I.; Gefen, A.; Reinhart-King, C.A.; Margulies, S.S.; Dembo, M.; Boettiger, D.; et al. Tensional homeostasis and the malignant phenotype. Cancer Cell 2005, 8, 241–254.
- Luo, C.; Zuniga, J.; Edison, E.; Palla, S.; Dong, W.; Parker-Thornburg, J. Superovulation strategies for 6 commonly used mouse strains. J. Am. Assoc. Lab. Anim. Sci. 2011, 50, 471–478.
- Gaiano, N.; Amsterdam, A.; Kawakami, K.; Allende, M.; Becker, T.; Hopkins, N. Insertional mutagenesis and rapid cloning of essential genes in zebrafish. Nature 1996, 383, 829–832.
- Allende, M.L.; Amsterdam, A.; Becker, T.; Kawakami, K.; Gaiano, N.; Hopkins, N. Insertional mutagenesis in zebrafish identifies two novel genes, pescadillo and dead eye, essential for embryonic development. Genes Dev. 1996, 10, 3141–3155.
- Thermes, V.; Grabher, C.; Ristoratore, F.; Bourrat, F.; Choulika, A.; Wittbrodt, J.; Joly, J.S. I-SceI meganuclease mediates highly efficient transgenesis in fish. Mech. Dev. 2002, 118, 91–98.
- Pan, F.C.; Chen, Y.; Loeber, J.; Henningfeld, K.; Pieler, T. I-SceI meganuclease-mediated transgenesis in Xenopus. Dev. Dyn. 2006, 235, 247–252.
- Powers, J.T.; Tsanov, K.M.; Pearson, D.S.; Roels, F.; Spina, C.S.; Ebright, R.; Seligson, M.; de Soysa, Y.; Cahan, P.; Theissen, J.; et al. Multiple mechanisms disrupt the let-7 microRNA family in neuroblastoma. Nature 2016, 535, 246–251.
- Hwang, W.Y.; Fu, Y.; Reyon, D.; Maeder, M.L.; Tsai, S.Q.; Sander, J.D.; Peterson, R.T.; Yeh, J.R.; Joung, J.K. Efficient genome editing in zebrafish using a CRISPR-Cas system. Nat. Biotechnol. 2013, 31, 227–229.
- Jao, L.E.; Wente, S.R.; Chen, W. Efficient multiplex biallelic zebrafish genome editing using a CRISPR nuclease system. Proc. Natl. Acad. Sci. USA 2013, 110, 13904–13909.
- Varshney, G.K.; Pei, W.; LaFave, M.C.; Idol, J.; Xu, L.; Gallardo, V.; Carrington, B.; Bishop, K.; Jones, M.; Li, M.; et al. High-throughput gene targeting and phenotyping in zebrafish using CRISPR/Cas9. Genome Res. 2015, 25, 1030–1042.
- Hruscha, A.; Krawitz, P.; Rechenberg, A.; Heinrich, V.; Hecht, J.; Haass, C.; Schmid, B. Efficient CRISPR/Cas9 genome editing with low off-target effects in zebrafish. Development 2013, 140, 4982–4987.
- Bedell, V.M.; Wang, Y.; Campbell, J.M.; Poshusta, T.L.; Starker, C.G.; Krug, R.G., 2nd; Tan, W.; Penheiter, S.G.; Ma, A.C.; Leung, A.Y.; et al. In vivo genome editing using a high-efficiency TALEN system. Nature 2012, 491, 114–118.
- Seok, S.H.; Na, Y.R.; Han, J.H.; Kim, T.H.; Jung, H.; Lee, B.H.; Emelyanov, A.; Parinov, S.; Park, J.H. Cre/loxP-regulated transgenic zebrafish model for neural progenitor-specific oncogenic Kras expression. Cancer Sci. 2010, 101, 149–154.
- Langenau, D.M.; Feng, H.; Berghmans, S.; Kanki, J.P.; Kutok, J.L.; Look, A.T. Cre/lox-regulated transgenic zebrafish model with conditional myc-induced T cell acute lymphoblastic leukemia. Proc. Natl. Acad. Sci. USA 2005, 102, 6068–6073.
- Robles, E.; Filosa, A.; Baier, H. Precise lamination of retinal axons generates multiple parallel input pathways in the tectum. J. Neurosci. 2013, 33, 5027–5039.
- Felker, A.; Mosimann, C. Contemporary zebrafish transgenesis with Tol2 and application for Cre/lox recombination experiments. Methods Cell Biol. 2016, 135, 219–244.
- Burg, L.; Palmer, N.; Kikhi, K.; Miroshnik, E.S.; Rueckert, H.; Gaddy, E.; MacPherson Cunningham, C.; Mattonet, K.; Lai, S.L.; Marin-Juez, R.; et al. Conditional mutagenesis by oligonucleotide-mediated integration of loxP sites in zebrafish. PLoS Genet. 2018, 14, e1007754.
- Carney, T.J.; Mosimann, C. Switch and Trace: Recombinase Genetics in Zebrafish. Trends Genet. 2018, 34, 362–378.
- Veinotte, C.J.; Dellaire, G.; Berman, J.N. Hooking the big one: The potential of zebrafish xenotransplantation to reform cancer drug screening in the genomic era. Dis. Model. Mech. 2014, 7, 745–754.
- Haney, M.G.; Moore, L.H.; Blackburn, J.S. Drug Screening of Primary Patient Derived Tumor Xenografts in Zebrafish. J. Vis. Exp. 2020, e60996.
- Cabezas-Sainz, P.; Pensado-Lopez, A.; Sainz, B., Jr.; Sanchez, L. Modeling Cancer Using Zebrafish Xenografts: Drawbacks for Mimicking the Human Microenvironment. Cells 2020, 9, 1978.
- Costa, B.; Estrada, M.F.; Mendes, R.V.; Fior, R. Zebrafish Avatars towards Personalized Medicine-A Comparative Review between Avatar Models. Cells 2020, 9, 293.
- Pichler, F.B.; Laurenson, S.; Williams, L.C.; Dodd, A.; Copp, B.R.; Love, D.R. Chemical discovery and global gene expression analysis in zebrafish. Nat. Biotechnol. 2003, 21, 879–883.
- Zhu, S.; Yan, X.; Xiang, Z.; Ding, H.F.; Cui, H. Leflunomide reduces proliferation and induces apoptosis in neuroblastoma cells in vitro and in vivo. PLoS ONE 2013, 8, e71555.
- Fior, R.; Povoa, V.; Mendes, R.V.; Carvalho, T.; Gomes, A.; Figueiredo, N.; Ferreira, M.G. Single-cell functional and chemosensitive profiling of combinatorial colorectal therapy in zebrafish xenografts. Proc. Natl. Acad. Sci. USA 2017, 114, E8234–E8243.
- Almstedt, E.; Elgendy, R.; Hekmati, N.; Rosen, E.; Warn, C.; Olsen, T.K.; Dyberg, C.; Doroszko, M.; Larsson, I.; Sundstrom, A.; et al. Integrative discovery of treatments for high-risk neuroblastoma. Nat. Commun. 2020, 11, 71.
- Rajan, V.; Melong, N.; Hing Wong, W.; King, B.; Tong, S.R.; Mahajan, N.; Gaston, D.; Lund, T.; Rittenberg, D.; Dellaire, G.; et al. Humanized zebrafish enhance human hematopoietic stem cell survival and promote acute myeloid leukemia clonal diversity. Haematologica 2020, 105, 2391–2399.
