Diseases of the kidney contribute a significant morbidity and mortality burden on society. Localized delivery of therapeutics directly into the kidney, via its arterial blood supply, has the potential to enhance their therapeutic efficacy while limiting side effects associated with conventional systemic delivery. Targeted delivery in humans is feasible given that we can access the renal arterial blood supply using minimally invasive endovascular techniques and imaging guidance. Techniques to reproduce or mimic this approach in a small animal model will allow for further pre-clinical translational studies investigating therapies for the treatment of renal pathologies.
- Locoregional delivery
- kidney disease
- microsurgery
1. Introduction
Kidney diseases contribute a significant morbidity and mortality burden on society: acute kidney injury (AKI) causes 9.5% of in-hospital mortality, and chronic kidney disease (CKD), most commonly due to hypertension and diabetes, has a staggering 14% prevalence in the United States[1]. Progression of both AKI and CKD can ultimately result in end stage renal disease, for which dialysis and kidney transplantation are the only treatment options. These treatment modalities are themselves demanding for patients and fraught with health complications, requiring multiple long visits to the hospital or the need to be on life-long immunosuppression. In addition to AKI and CKD, kidney cancer also contributes a significant burden on society with approximately 60,000 Americans diagnosed with this disease each year[2].
Although there have been significant advances for treating these pathologies, the majority of promising pharmacological and cellular therapies under investigation are administered by conventional intravenous (IV) injection[3][4]. Directly delivering therapeutics to organs, via their arterial blood supply, has many advantages over IV injection, including minimizing systemic side effects, maximizing the therapeutic efficacy at the target site, and avoidance of first pass metabolism in the liver or sequestration by the reticuloendothelial system and lungs. While intra-arterial (IA) delivery of therapeutics is already widely clinically utilized for the treatment of stroke[5], heart attacks[6], and liver cancer[7], there are comparatively few indications for IA delivery of therapeutics for pathologies of the kidney. In part, this can be attributed to limited pre-clinical studies given that performing non-terminal IA injection into the kidneys in small animal models is technically challenging.
The development of IA therapeutics for kidney disease relies on the availability of animal models and feasibility of surgical techniques. Previous studies that injected MSCs directly into the renal artery have used larger animal models, including cats[8], pigs[9], rats[10], and sheep[11]. Indeed, a meta-analysis of 21 studies applying MSC therapy to animal models of renal failure has suggested that IA delivery of MSCs has greater therapeutic effect compared to intravenous delivery[12]; it should be noted though that the studies used different animal and disease models, and none of them did a direct comparison between delivery routes. Regardless, these larger animals are often not readily accessible to researchers, or do not have as well-established models of kidney disease. Mice are by far the most comprehensive species in regards to models of renal pathology, with established models of acute kidney injury (AKI), chronic kidney disease (CKD), and kidney cancer, via nephrotoxic, genetic, autoimmune, metabolic, and ischemic etiologies[13][14].
2. Currant status
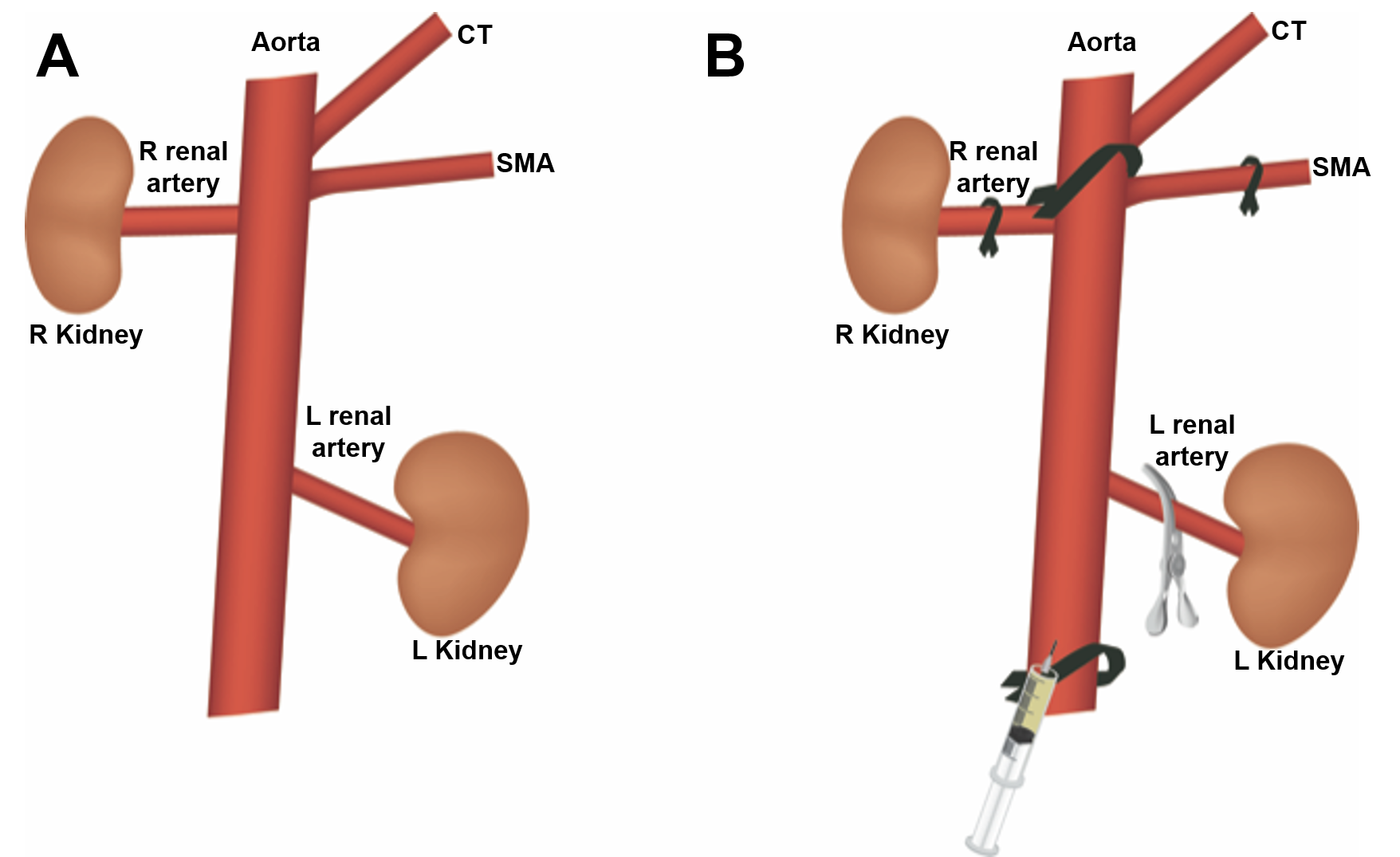
Recently, Ullah et al. reported the first microsurgical technique to deliver therapeutics into both kidneys in mice, via their arterial supply[15]. Their technique involves selective ligation of (1) the distal aorta 1 cm distal to the left renal artery, (2) the proximal aorta between the superior mesenteric artery (SMA) and celiac trunk (CT), and (3) the SMA origin. A 34-gauge needle was then inserted into the aorta with gentle back-tension applied on the distal suture to help keep it in a perfect line to facilitate safe cannulation. A metal clamp was applied temporarily on the left renal artery for the first part of the injection to direct any injected solution preferentially into the right renal artery. After 50% of the solution was injected, the metal clamp was then removed and the reminder of the solution was injected which now preferentially flowed into the left kidney.
Figure 1. (A) Normal anatomy of the mouse abdominal aorta, showing origin of the celiac trunk (CT), superior mesenteric artery (SMA), renal arteries, and kidneys. (B) Suture ligation sites, including the proximal aorta between the origins of the CT and SMA, the SMA, and the distal aorta. A metal clip is placed temporarily on the left renal artery to allow delivery of therapeutics first to the right renal artery.
The authors validated their technique by applying it to a mouse model of cisplatin-induced AKI, treated with IA injection of mesenchymal stromal cell (MSC)-derived extracellular vesicles (EVs). Mesenchymal stromal cells (MSCs) have shown promising results for kidney regeneration in the context of AKI and CKD[3]. The therapeutic effect of MSC-based therapies is thought to come from their ability to home to damaged tissue and secrete soluble factors with regenerative properties. Due to the paracrine nature of this mechanism, the proximity of infused MSCs to the injured site is critical for therapeutic efficacy[16]. However, it is known that when MSCs are injected IV, the vast majority of cells become trapped in the pulmonary microvasculature, in what is known as the pulmonary first-past effect[17][18]. To avoid this intrinsic limitation, many groups have opted to instead study stem cell-derived extracellular vesicles (EVs), which are cell-free membrane-bound particles that carry a cargo of regenerative molecules[19]. EVs have been shown to avoid the pulmonary first-pass effect and have a therapeutic effect on par with the MSCs from which they are derived[20]. However, unlike MSCs there is little knowledge on how to further optimize delivery of EVs to target organs[21]. Locoregional delivery, via the arterial blood supply, is perhaps one of the most straightforward and clinically translatable strategies for doing so.
References
- Kidney Disease Statistics for the United States . NIDDK. Retrieved 2020-5-19
- Rebecca L Siegel; Kimberly D. Miller; Ahmedin Jemal; Cancer statistics, 2020.. CA: A Cancer Journal for Clinicians 2020, 70, 7-30, 10.3322/caac.21590.
- Marina Morigi; Cinzia Rota; Giuseppe Remuzzi; Marina Morigi Ph.D.; Mesenchymal Stem Cells in Kidney Repair. Advanced Structural Safety Studies 2016, 1416, 89-107, 10.1007/978-1-4939-3584-0_5.
- Abigail L. Thomson; Allyson C. Berent; Chick Weisse; Catherine E. Langston; Intra-arterial renal infusion of autologous mesenchymal stem cells for treatment of chronic kidney disease in cats: Phase I clinical trial.. Journal of Veterinary Internal Medicine 2019, 33, 1353-1361, 10.1111/jvim.15486.
- Leonardo Rangel-Castilla; Gary B Rajah; Hakeem J Shakir; Jason M Davies; Kenneth V. Snyder; Adnan H. Siddiqui; Elad I. Levy; L. Nelson Hopkins; Acute stroke endovascular treatment: tips and tricks.. J Cardiovasc Surg (Torino) 2016, 57, 758-768.
- Ashleigh Dind; Usaid Allahwala; Kaleab N. Asrress; Sanjit S. Jolly; Ravinay Bhindi; Fracp; Contemporary Management of ST-Elevation Myocardial Infarction. Heart, Lung and Circulation 2017, 26, 114-121, 10.1016/j.hlc.2016.07.009.
- Ali Habib; Kush Desai; Ryan Hickey; Bartley Thornburg; Robert Lewandowski; Riad Salem; Locoregional Therapy of Hepatocellular Carcinoma. Clinics in Liver Disease 2015, 19, 401-420, 10.1016/j.cld.2015.01.008.
- Lei Zhang; Xiang-Yang Zhu; Yu Zhao; Alfonso Eirin; Lei Liu; Christopher M. Ferguson; Hui Tang; Amir Lerman; Lilach O. Lerman; Selective intrarenal delivery of mesenchymal stem cell-derived extracellular vesicles attenuates myocardial injury in experimental metabolic renovascular disease. Basic Research in Cardiology 2020, 115, 16, 10.1007/s00395-019-0772-8.
- Lei Zhang; Xiang-Yang Zhu; Yu Zhao; Alfonso Eirin; Lei Liu; Christopher M. Ferguson; Hui Tang; Amir Lerman; Lilach O. Lerman; Selective intrarenal delivery of mesenchymal stem cell-derived extracellular vesicles attenuates myocardial injury in experimental metabolic renovascular disease. Basic Research in Cardiology 2020, 115, 16, 10.1007/s00395-019-0772-8.
- Michela De Martino; Sandro Zonta; Teresa Rampino; Marilena Gregorini; F. Frassoni; G. Piotti; G. Bedino; Lorenzo Cobianchi; Antonio Dal Canton; Paolo Dionigi; et al.Mario Alessiani Mesenchymal Stem Cells Infusion Prevents Acute Cellular Rejection in Rat Kidney Transplantation. Transplantation Proceedings 2010, 42, 1331-1335, 10.1016/j.transproceed.2010.03.079.
- Luc Behr; Mehrak Hekmati; Gaëlle Fromont; Nicolas Borenstein; Laure-Hélène Noel; Martine Lelièvre-Pégorier; Kathleen Laborde; Intra Renal Arterial Injection of Autologous Mesenchymal Stem Cells in an Ovine Model in the Postischemic Kidney. Nephron Physiology 2006, 107, p65-p76, 10.1159/000109821.
- Yan Wang; Juan He; Xiaohua Pei; Weihong Zhao; Systematic review and meta-analysis of mesenchymal stem/stromal cells therapy for impaired renal function in small animal models. Nephrology 2013, 18, 201-208, 10.1111/nep.12018.
- Timothy Hewitson; Takahiko Ono; Gavin J. Becker; Small Animal Models of Kidney Disease: A Review. Advanced Structural Safety Studies 2008, 466, 41-57, 10.1007/978-1-59745-352-3_4.
- Yin-Wu Bao; Yuan Yuan; Jiang-Hua Chen; Weiqiang Lin; Kidney disease models: tools to identify mechanisms and potential therapeutic targets. Zool. Res. 2017, 39, 72-86, 10.24272/j.issn.2095-8137.2017.055.
- Mujib Ullah; Daniel D. Liu; Sravanthi Rai; Mehdi Razavi; Jeff Choi; Jing Wang; Waldo Concepcion; Avnesh S Thakor; A Novel Approach to Deliver Therapeutic Extracellular Vesicles Directly into the Mouse Kidney via Its Arterial Blood Supply. Cells 2020, 9, 937, 10.3390/cells9040937.
- Marina Morigi; Paolo De Coppi; Cell Therapy for Kidney Injury: Different Options and Mechanisms - Mesenchymal and Amniotic Fluid Stem Cells. Nephron Experimental Nephrology 2014, 126, 59-59, 10.1159/000360667.
- Sonja Schrepfer; T. Deuse; H. Reichenspurner; M.P. Fischbein; R.C. Robbins; M.P. Pelletier; Stem Cell Transplantation: The Lung Barrier. Transplantation Proceedings 2007, 39, 573-576, 10.1016/j.transproceed.2006.12.019.
- Elke Eggenhofer; Volker Benseler; Alexander Kroemer; F. C. Popp; E. K. Geissler; H. J. Schlitt; C. Baan; M. H. Dahlke; Martin J. Hoogduijn; Mesenchymal stem cells are short-lived and do not migrate beyond the lungs after intravenous infusion. Frontiers in Immunology 2012, 3, 2012, 10.3389/fimmu.2012.00297.
- Lin-Li Lv; Wei‐Jun Wu; Ye Feng; Zuo‐Lin Li; Tao‐Tao Tang; Bi-Cheng Liu; Therapeutic application of extracellular vesicles in kidney disease: promises and challenges. Journal of Cellular and Molecular Medicine 2017, 22, 728-737, 10.1111/jcmm.13407.
- Donald Phinney; Mark F. Pittenger; Concise Review: MSC-Derived Exosomes for Cell-Free Therapy. STEM CELLS 2017, 35, 851-858, 10.1002/stem.2575.
- Mujib Ullah; Daniel D. Liu; Avnesh S Thakor; Mesenchymal Stromal Cell Homing: Mechanisms and Strategies for Improvement.. iScience 2019, 15, 421-438, 10.1016/j.isci.2019.05.004.