Critical limb ischemia (CLI) constitutes the most severe form of peripheral arterial disease (PAD), a prevalent manifestation of atherosclerosis which involves the blockade of major systemic arteries other than those of the cerebral and coronary circulation, more common in legs than in arms. Overall, CLI patients suffer from chronic ischemic rest pain, ulcers, or gangrene, as well as an increased risk of cardiovascular events. CLI has a huge impact on the patients’ quality of life, being associated with an increased risk of amputations (fingers, toes, or extremities) and, moreover, an increase in mortality rates.
Currently, revascularization strategies (bypass grafting, angioplasty) remain the first option for CLI patients, although less than 45% of them are eligible for surgical intervention mainly due to associated comorbidities. Moreover, patients usually require amputation in the short-term. As an alternative to conventional treatments, therapeutic angiogenesis has arisen as a promising treatment for CLI patients, mainly those considered as “no-option”, due to the potential of this strategy to promote revascularization of ischemic tissues. Different approaches including angiogenic gene or cell-based therapies are currently under investigation.
- critical limb ischemia
- neovascularization
- angiogenesis
- arteriogenesis
- cell therapy
- secretomes
1. Critical Limb Ischemia
Critical Limb Ischemia (CLI) constitutes the most severe form of Peripheral Arterial Disease (PAD), a prevalent manifestation of atherosclerosis which involves the blockade of major systemic arteries other than those of the cerebral and coronary circulation [1], more common in legs than in arms [2]. PAD affects around 10–15% of adults, being an underestimated and underdiagnosed cardiovascular disease (CVD) due to its asymptomatic initial stages [3]. PAD is associated with risk factors such as older age, hypertension, dyslipidemia, or smoking [4], and it is more prevalent in diabetic people due to metabolic alterations such as angiogenesis impairment, inflammatory progression, or endothelial dysfunction [5][6][7][8][5,6,7,8]. CLI itself has an annual incidence of 0.35% and an average prevalence of 1.33%, affecting to 500–1000 people per 1 million population in Europe and the United States [9]. CLI patients are classified based on clinical criteria and hemodynamic parameters (i.e., pulse volume recordings, ankle and toe pressure values, rest pain, and tissue loss) [10][11][12][10,11,12] currently accepted in international consensus guidelines on PAD and CLI [12][13][14][15][16][12,13,14,15,16]. Overall, CLI patients suffer from chronic ischemic rest pain, ulcers, or gangrene, as well as an increased risk of cardiovascular events. CLI has a huge impact on the patients’ quality of life, being associated with an increased risk of amputations (fingers, toes, or extremities) and, moreover, an increase in mortality rates [15][17][18][19][20][15,17,18,19,20]. This debilitating disease causes high dependency on caregivers, requiring permanent local wound treatment, and the chronic use of pain-relieving medications, considerably diminishing patient’s quality of life [21].
Nowadays, the treatment of CLI remains highly variable and, in many situations, suboptimal [22]. Initial recommendations for CLI patients to prevent further cardiovascular events include smoking cessation, lipid lowering (statins mainly), antiplatelet therapies, or ACE inhibitors [16]. Alternatively, other medical strategies or pharmaceutical agents have been applied for the specific treatment of CLI patients (sympathectomy or spinal cord stimulation, iloprost) [23]. Unfortunately, these strategies do not seem to be totally effective in reducing limb-specific events [16], although larger studies/clinical trials are required in order to reach definitive conclusions.
The majority of CLI patients require revascularization interventions like bypass or angioplasty, having observed a significant improvement in the techniques and devices applied (cryoplasty, stent-grafts, drug-eluting balloons or stents, etc.) in the past decades. Nevertheless, the percentage of patients eligible for these strategies is not higher than 45% due to high comorbidity or surgical related issues such as difficult access due to narrow vessels, etc. Furthermore, patients that undergo surgery will usually require amputation at the short term [24]. Amputation rates are unacceptably high, typically exceeding 15–20% at 1 year and can vary by the presence of comorbid conditions [25] such as diabetes mellitus (DM), which elevates this rate up to 50% in CLI diabetic patients [26]. Diabetic patients have higher risk of suffering PAD/CLI and a negative outcome partly related to the abrogation of new vessel formation and remodeling of the pre-existing vasculature under hyperglycemic conditions [27]. Unfortunately, the increasing prevalence of PAD together with higher presence of other CLI risk factors (i.e., diabetes) and the rising number of people in advanced age provide little reason to believe that the number of patients suffering this disease will decrease in the near future [25]. The poor prognosis of CLI patients as well as their impaired quality of life makes compulsory to find effective and less invasive treatments. Moreover, the desirable treatment should be applicable to all CLI patients, because the actual percentage of ineligible patients is unacceptably high.
As an alternative to conventional treatments, therapeutic angiogenesis has arisen as a promising treatment for CLI patients, mainly those considered as “no-option”, due to the potential of this strategy to promote revascularization of ischemic tissues [28][29][30][31][32][33][28,29,30,31,32,33]. To date, different approaches including angiogenic gene or cell-based therapies are currently under investigation.
We have mainly focused on the use of angiogenic cell therapy for CLI (Figure 1), from animal/pre-clinical models designed to study CLI and the tools applied to test for revascularization in response to cell therapy, to the angiogenic therapies currently under evaluation in clinical trials. Moreover, recent alternatives derived from stem cell therapies, such as the use of secretomes, exosomes, or even microRNAs, will be described.
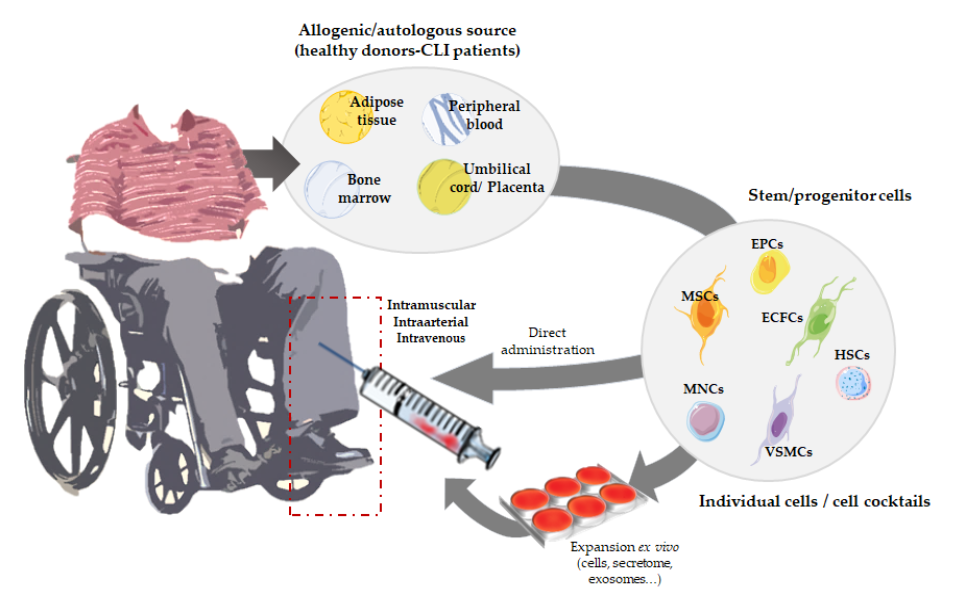
Figure 1.
Overview of angiogenic cell therapy for Critical Limb Ischemia (CLI).
2. Angiogenic Cell Therapy
Angiogenic therapy involves the use of angiogenic growth factors (VEGF, HIF-1a, FGF1, HGF, etc.) [33][34][33,64], gene transfer techniques using viral or non-viral vectors to transport a gene codifying for a therapeutic protein to the target tissues [35][65] or, alternatively, the use of angiogenic stem cells. All these strategies aim to improve revascularization by increasing the number/size of blood vessels, promoting blood flow recovery and therefore increasing tissue perfusion in the ischemic extremities [35][65]. Among them, cell-based therapies seem more efficient compared to protein- or gene-based approaches, not only because of their direct vasculogenic properties, but also due to their paracrine effect. Angiogenic cells can directly participate in the formation of new vessels, while in parallel they also provide endogenous growth factors, promoting vascular growth by paracrine fashion [36][37][66,67].
Thus, neovascularization can also be promoted by vasculogenesis, the novo formation of vessels mediated by circulating progenitors or stem cells [38][59]. Vasculogenesis was initially considered as an embryogenic process. However, post-natal vasculogenesis can also take place by incorporation of vascular stem or progenitor cells into vessel structures, allowing the formation of adult blood vessels [39][68]. To date, several strategies based on the use of stem and progenitor cells are being tested (Table 1), to promote vasculogenesis but also angiogenesis and arteriogenesis. The safety and efficacy of cell implantation therapies make of this less invasive treatment a feasible option for CLI patients.
2.1. Cell Therapies Based on Single or Combined Isolated Cells
Mesenchymal stem cells (MSCs) are the most used cells in advanced therapies for CVDs [40]. MSCs can be isolated from bone marrow, peripheral blood, or adipose tissues, and from them we can obtain osteoblasts, chondrocytes, adipocytes, neurons, endothelial cells (ECs), skeletal muscle cells, and vascular smooth muscle cells (VSMCs) [41]. MSCs are reported to promote angiogenesis because of their capacity to induce ECs proliferation, migration, and tube formation, while decreasing apoptosis and fibrosis [40][42][43]. Furthermore, MSCs support neoangiogenesis, releasing soluble factors that contribute to stimulate angiogenesis [44]. These cells are thought to improve hind limb ischemia by secreting cytokines that regulate macrophage differentiation to M2, an anti-inflammatory phenotype [45]. Likewise, apart from MSCs, endothelial progenitor cells (EPCs) also represent an important group of cells used in vascular regeneration. In 1997, Asahara et al. demonstrated that CD34+ cells can be isolated from peripheral blood mononuclear cells (PB-MNCs) and differentiated in vitro into ECs, showing the potential use for collateral vessel growth augmentation in ischemic tissues [46]. Although CD34 is not a specific marker of a single cell type, it is mostly associated to EPCs. Many researchers have explored the potential of using EPCs in tissue engineering as an angiogenic source for vascular repairing [47][48]. In the past years, several isolation and culturing techniques for EPCs have been described. Besides, the controversy regarding the definition of EPC phenotypes remains, with different studies still presenting a variety of results in terms of surface-based EPC markers [49][47][50][51]. At least, two different sub-populations have been accepted and clearly defined, based on their differentiation status and the capability to form colonies: early EPCs (eEPCs) also named circulating angiogenic cells (CACs) or myeloid angiogenic cells (MACs), with hematopoietic phenotype, and late EPCs or endothelial colony forming cells (ECFCs), with endothelial phenotype [51]. EPCs have been thought to derive from hematopoietic stem cells (HSCs), some EPCs could be derived from a niche close to the vasa vasorum in the macro-vascular wall [52]. Despite the controversy regarding the nature of these cells, no one denies the potential of EPCs to promote therapeutic angiogenesis and neovascularization of ischemic tissues [53][54][52]. Overall, in response to injury, cytokines and growth factors mobilize EPCs from the bone marrow into the peripheral blood, which will then participate in neovascularization [53]. Very recently, we have shown how, first days after administration of CACs to ischemic CLI mice, these cells migrate into the ischemic tissues, modulating immune cells recruitment and promoting an increase of angiogenesis and arteriogenesis [49]. However, the administered cells do not remain in the ischemic tissues over time suggesting that they may promote vasculogenesis in a paracrine form [49][55]. Moreover, early EPCs do not seem to differentiate to ECs, with this role being assigned to ECFCs [51][56]. Indeed, different studies support that the regenerative properties of eEPCs are mainly due to paracrine effects, while ECFCs present vessel-forming activity in vivo [49][56]. Thus, a cell therapy mediated by both cell types, early, and late EPCs, could be a good strategy for CVDs. Yoon et al. evaluated this combined cell therapy, demonstrating a synergistic neovascularization involving several cytokines and matrix metalloproteinases (MMPs) [57]. Very recently, our group has also corroborated the potential of CACs to promote angiogenesis of ECFCs in vitro, and such effect was impaired under an atherosclerotic environment [58]. In the same way, different cell combinations have been tested. Rossi et al. demonstrated that co-injection of MSCs with ECFCs in a murine model of CLI increased vessel density and foot perfusion in greater ratio than cells individually administrated; corroborating the theory that MSCs support ECFC-mediated angiogenic processes [59]. Furthermore, their results indicated that MSCs accelerated muscle recovery via endoglin dependent mechanism. Similarly, the combination of EPCs and smooth muscle progenitor cells (SMPCs) has also been evaluated to treat CLI. This cell mixture improved vascular network formation, with both ECs and smooth muscle cells (SMCs) participating in vessel maturation and stability. Likewise, Foubert et al. demonstrated that co-administration of EPCs and SMPCs activates neovascularization resulting in a more effective therapy than these cells administrated separately [60]. Some studies suggest that SMCs may also originate from bone marrow-derived cells as SMPCs have been identified in peripheral blood [61].
2.2. Cell Therapies Based on Cellular Cocktails
As an alternative to the injection of a single cell type or the combination of two previously isolated cells, the administration of cellular cocktails derived from different niches, such as bone marrow, peripheral blood, or adipose tissue, is also a frequent approach to treat CLI. Indeed, the regenerative properties of mononuclear cells (MNCs) derived from either bone marrow or peripheral blood have been largely studied in the last years. Therapies employing bone marrow mononuclear cells (BM-MNCs) constitute a promising alternative for CLI patients to avoid or delay the onset of amputation [62][112]. BM-MNCs consist of a heterogeneous mix of multipotent stem cells working cooperatively as MSCs, HSCs, EPCs, monocytes, lymphocytes, and pluripotent stem cells [63][64][41,113]. We and other researchers have reported the beneficial effects of different combinations of BM-MNCs, representing an effective approach in promoting new vessel formation, perfusion recovery, and CLI reversal [63][44][65][66][67][68][69][70][71][72][73][41,100,114,115,116,117,118,119,120,121,122]. In the ischemic tissue, BM-MNCs produce and secrete different cytokines and growth factors [74][123] and increase neovascularization and collateral vessel formation in limb ischemia [75][79]. Moreover, Kikuchi-Taura et al. have recently described that transplantation of BM-MNCs into a murine stroke model promoted ECs angiogenesis by gap junction mediated cell–cell interactions, elucidating a new theory of how cell-based therapies work, and suggesting that stem cells supply energy to injured cells [76][124]. This study suggested that, under hypoxic conditions, transplanted BM-MNCs are capable to transfer small molecules to ECs via gap junction interactions, leading to HIF-1α activation, which induced upregulation of VEGF uptake into ECs and ECs autophagy suppression [76][124].
Alternatively to BM-MNCs, PB-MNCs are formed by circulating cells with angiogenic potential, thereby several studies involving the administration of these cells to treat CLI have also shown promising results [77][78][125,126]. Li et al. made a comparison between CD34+ and CD34- cells in PB-MNCs, concluding that both induce neovascularization, but only CD34+ incorporate into new capillaries [79][89]. PB-MNCs promote revascularization in ischemic limbs, even more when they are combined with platelet-rich plasma (PRP) [80][90]. PRP, a source of platelets, cytokines, and growth factors, participates in ECs proliferation and differentiation, interacting with important cell receptors related with angiogenesis [80][90]. Furthermore, in order to achieve high stem cell concentrations, hematopoietic growth factors are frequently used to induce cell mobilization. For example, prior PB-MNCs harvesting, progenitor cells are usually mobilized injecting granulocyte colony-stimulating factor (G-CSF) [77][78][81][82][125,126,127,128]. BM-MNCs and PB-MNCs treatments have been compared, and no significant differences have been observed between them [83][84][129,130]. Remarkably, without previous mobilization, PB-MNCs show higher concentration of mature cells as red blood cells, platelets, lymphocytes, and monocytes, while BM-MNCs show higher levels of EPCs [85][131].
The use of adipose tissue-derived stem cells (ASCs) has increased in the last years, due to the easier accessibility, abundance, and less painful collection compared to other sources such as bone marrow [86][132]. The stromal vascular fraction (SVF) derived from adipose tissue contains heterogeneous cell populations such as mesenchymal progenitor/stem cells, pre-adipocytes, endothelial cells, pericytes, T cells, and M2 macrophages. SVF-derived mesenchymal progenitor/stem cells, usually referred as ASCs themselves, can be easily expanded in vitro and have the potential to differentiate into multiple lineages, including myogenic, osteogenic, neurogenic, and hematopoietic pathways [87][88][89][90][91][133,134,135,136,137]. The angiogenic properties of these cells have been correlated with a strong paracrine activity, secreting an important number of angiogenesis-related cytokines [90][136]. Moreover, the administration of ASCs to CLI mice promotes a significant recovery of blood flow in ASCs treated mice compared to ischemic, non-treated ones [87][133]. Very recently, Liu J et al. have shown that the regenerative properties of transplanted ASCs might correlate with an immunomodulatory effect promoted by these cells. In presence of ASCs, a higher number of macrophages can be found in the muscle, with increased presence of M2 macrophages [92][91], and its administration in a murine model of CLI induces an angiogenic process in the ischemic tissue [87][133]. The clear advantages of using these cells are easy access and isolation. ASCs are highly abundant in adipose tissue, making almost unnecessary culture expansion of these cells. Moreover, adipose tissue harvesting requires a minimally invasive intervention [93][138]. A pilot study using adipose-derived regenerative cells (ADRCs) in CLI patients has been recently published [94][139].
Finally, other cells with multi-differentiation potential such as amniotic fluid derived stem cells (AFSCs) or umbilical cord blood and placenta tissue derived stem/progenitor cells have also been considered. Placenta-derived MSCs stromal-like cells (PLX-PAD) in CLI mice are currently being tested in a Phase III trial (PACE Trial) with atherosclerotic CLI patients (NCT03006770) after promising results in animal assays [95]. Unfortunately, the low availability of these cells together with ethics concerns related to their use, has limited their translation as cell therapies.
