The vaccination for the novel Coronavirus (COVID-19) is undergoing its final stages of analysis and testing. It is an impressive feat under the circumstances that we are on the verge of a potential breakthrough vaccination. This will help reduce the stress for millions of people around the globe, helping to restore worldwide normalcy. In this review, the analysis looks into how the new branch of Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) came into the forefront of the world like a pandemic. This review will break down the details of what COVID-19 is, the viral family it belongs to and its background of how this family of viruses alters bodily functions by attacking vital human respiratory organs, the circulatory system, the central nervous system and the gastrointestinal tract.
- COVID-19 Vaccination
1. Introduction
Covid-19 Breakdown and Background
By the end of January 2020, after succumbing to considerable pressure the World Health Organization (WHO) officially declared COVID-19 as a pandemic, a Public Health Emergency of International Concern (PHEIC). Due to action that was not taken sooner in many parts of the world, the virus spread to around 25 countries by early February 2020. With the numbers increasing, guidelines and criteria for diagnosis, treatment and preventative measures had to be established rapidly [1][2]. Viral detection using RT-PCR identified the SARS-CoV-2 virus to be the disease which caused this viral transmission worldwide. This virus bore significant similarity to that found present within bats and was of the same family as Severe Acute Respiratory Syndrome Coronavirus 1 (SARS-CoV-1) and Middle East Respiratory Syndrome Coronavirus (MERS-CoV), therefore significantly narrowing down the likelihood that this had been somehow transmitted from bats to humans (owing to bats being the main reservoir for this virus). COVID-19 has a high recombinant and mutation rate due to its unique replication capabilities, enabling it to adapt to new host cells and different target sites [3]. So far, Covid-19 has been defined by 17 known mutations (14 non-synonymous mutations and 3 deletions), eight of these mutations have been on the spike protein, the main target site for the vaccination, with at least three of these mutations having a significant biological effect. These mutations, in particular N501Y, can incur a substantial change in the binding domain, resulting in enhancing the binding affinity to the human ACE2 enzyme. Another mutation (P681H) that is located directly close to the spike protein has shown the potential to increase infection and transmission. In terms of deletions that have occurred to the viral genome, the deletion of two amino acids has shown a link indicating immune escapability in immunocompromised patients thus enhancing viral infectivity [4]. The transmission of COVID-19 is from human-to-human contact [5], the most common infections occur from sufferers who are asymptomatic, therefore transmitting the virus without being aware they are carriers [6].
Symptoms of COVID-19 as was mentioned consist of two states, the (i) symptomatic state and the (ii) asymptomatic state. The symptomatic state can be easily noticed through the patient showing multiple different symptoms, one of them being the Acute Respiratory Disease Syndrome (ARDS), which include fever, cough, tiredness, sore throat, headache, and myalgia. More severe symptoms include aches and pains, diarrhoea, conjunctivitis, loss of taste and smell, a rash on skin and discoloration of fingers or toes; the most severe cases include difficulty breathing, chest pains or pressure and can even lead to loss of speech and movement [7]—some of the symptoms can result in multiple organ failure and eventually death. ARDS patients who experience symptoms tend to carry underlying health conditions, a suppressed immune system or are of older age. According to the literature, asymptomatic COVID-19 sufferers are the main source of transmission; through their respiratory droplets being airborne, as well as transmitted through virus-contaminated containers and foods [8][9]. Asymptomatic carriers show no symptoms of the virus due to an immune system capable of combatting the virus. However, they are capable of infecting others, henceforth making the virus capable of spreading around and becoming sometimes untraceable. The only way to identify an asymptomatic patient is through the administration of an RT-PCR. This, therefore, makes it difficult for countries to conduct identification tests whilst attempting to control the spread of the virus [10].
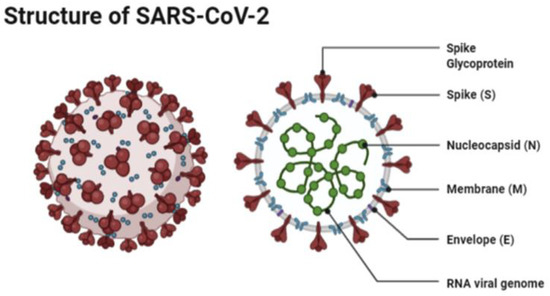
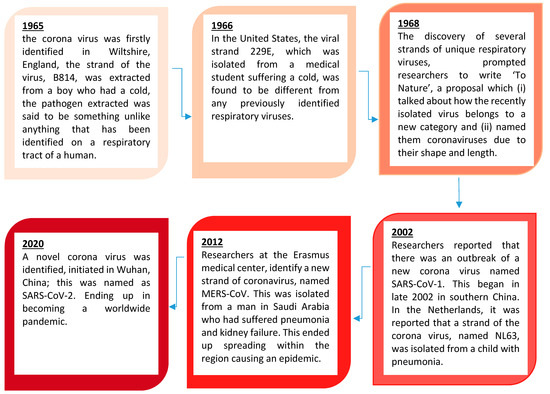
The structure of SARS-CoV-2 has shown to have a single strand enveloped RNA (sRNA). The size of the virus is between 50 and 150 nm in diameter, its linearity and positive-sense RNA genome is large. It belongs to the CoV-2 family, which was firstly found in the mid 1960s [11][12]. This family of viruses has a spherical shape, with envelopes containing helical nucleo-capsids and nucleoproteins; these are associated with the genomic structure of RNAs. The virus is capable of attaching itself to the host cells of its target due to a trimer of spike glycol-proteins, which include hemagglutinin esterase; there are also integral membrane and envelope proteins [13]. The virus can infect humans. Although animals will have varying immune responses to the virus, their immune system is more equipped to combat it and therefore it does not spread between animals as quickly as it does between humans. The virus targets the respiratory, hepatic, gastro-intestinal and neurological systems [12]. The name Coronavirus is due to the presence of the crown-like structures identified when scanned under the electron microscope. The viral structure consists of envelopes that contain helical nucleo-capsids and nucleoproteins (N), these are associated with the RNA genome. Embedded in the envelope is a 2 nm trimer of spike glycoproteins (S), this is the main source of the virus’s attachment to the receptor of the host cells. Within the virus, it also consists of integral membranes (M) and envelope proteins (E). Beta-Coronaviruses have an additional membrane glycoprotein named hemagglutinin esterase, which contains 5–7 nm long spikes (See
Figure 1). There are different families of the Coronavirus; the International Committee on Taxonomy of Viruses (ICTV) has separated them into different genres depending on their activity and structure. The genres are named; Alpha, Beta, Gamma and Delta Coronaviruses [14]. Several human Coronaviruses (alpha-CoVs, HCoVs-NL63, beta-CoVs, HCoVs-OC43, HCoVs-229E, HCoVs-HKU1, MERS-CoV, SARS-CoV and ARDS have been identified [15]. New versions of the Coronavirus will appear due to the large genomic potential, rapid mutation capabilities, high prevalence and wide distribution within the bird and animal kingdom. The emergence of CoVs is due to birds being able to carry this viral form and transfer it from area to area through flying and being capable of inhabiting in between groups [16][17].
The most common test to detect the virus is through RT-PCR. This test requires both a nasal and throat swab. The test detects the RNA of the virus, which may be present within a patient prior to the formation of antibodies or symptoms. With this test, early-stage detection can be achieved. The RT-PCR targets two parts to the virus, the Open Reading Frame Gene (ORFG) and the viral nucleo-capsid regions. The test works through the reverse transcription of the RNA of the virus into a complementary DNA (cDNA) [21]. This is then amplified in the Real-Time Polymerase Chain Reaction thermal cycle. The dye used produces fluorescent signals, whereby the RT-PCR is then capable of automatically forming a curve, thus giving a quantitative analysis for the presence of the SARS-CoV-2 virus at the nucleic acid level. RT-PCR can detect the virus in asymptomatic persons, however, the test is capable of giving false-negatives hence patients may be tested twice before being confirmed as positive or negative [21][23]. The RT-PCR kit remains an effective kit to use in the identification of SAR-CoV-2, however, one of the main worries for challenges arising from this analysis is that cases may have gone undetected; several studies have shown that the clinical sensitivity of the analysis of respiratory swabs was at around 70% effective. This was due to the timing of these swabs, the type of specimen obtained and the quality of the sample taken. The viruses present in the upper respiratory tract for the first several days following the onset of symptoms, hence after 5 days of symptoms, it becomes increasingly difficult to identify the virus via RT-PCR. In the latter stages, for correct and accurate reading, swabs from the lower respiratory tract will yield a higher rate of detection. Due to these nuances, it has been challenging laboratory professionals to truly define the clinical sensitivity of SARS-CoV-2 real-time PCR and has required that negative results be interpreted in the context of the timing of the sample [24]. An example of problems faced for SAR-CoV-2 RT-PCR testing is shown in
Figure 2.
2. The Research and History Ortho-Coronavirinae
Ortho-Coronavirinae.
3. Vaccinations Proposed for SARS-CoV-1
-
Inactivated SARS-CoV based vaccine: this vaccine expressed several structural proteins such as nucleo-capsid, membranes and spike proteins [27]. These are thought to induce an immune reaction that is capable of stimulating an immune response. The inactivated virus was intended for use as a first-generation vaccine, this is due to the ease of generation of these inactivated viral particles. The next step was the replacement of the inactivated viral vaccine by a second vaccine based around fragments containing neutralizing epitopes that are safer and more efficacious to use. Several reports have shown that SARS-CoV-1 was inactivated with formaldehyde, UV light, and β-propiolactone which can induce virus-neutralizing antibodies in immunized animals [28,29,30].
-
S-protein based vaccines: several recombinant based vaccines that have expressed the spike protein in SARS-CoV-1 were assessed in pre-clinical studies [31]. Reports have shown that candidate DNA vaccines encoding the spike protein stimulated an immune response. This led to the study showing that injected mice are protected for SARS-CoV-1. Wang et al. have produced higher titres of neutralizing antibodies and demonstrated that major and minor neutralizing epitopes are located in the S1 and S2 subunits, respectively [32]. Other groups also found neutralizing epitopes in the S2 subunit [33,34]. Bisht et al. [35] have shown that intranasal or intramuscular inoculations of mice with highly attenuated Modified Vacciniavirus Ankara (MVA) vaccines encoding full-length SARS-CoV-1 S protein. This also produced neutralizing antibodies. Bukreyev et al. [36] reported that mucosal immunization of African green monkeys with an attenuated parainfluenza virus expressing S protein resulted in the production of neutralizing antibodies and protected animals from infection by challenge with SARS-CoV-1. These data suggest that the S protein can induce neutralizing antibodies and protective responses in immunized animals [37].
4. Vaccinations Proposed for MERS-CoV
-
Recombinant MERS-CoV: unlike the SARS-CoV-1 vaccine, the MERS-CoV vaccine was constructed based on the recombinant viruses using reverse genetics. This resulted in expressed marker mutations, which allowed for replication-competent, propagation-defective MERS-CoV vaccines [45].
-
Viral-Vector-based MERS vaccine: this is similar to the vector-based SARS vaccine; MERS vaccines can also be constructed using viral vectors that express major MERS-CoV proteins, normally the S protein. Several such MERS vaccine candidates have been developed and/or tested for efficacy in mouse models or camels [46,47,48,49]. Viral vectors expressing full-length S protein of MERS-CoV induced S-specific antibody responses and/or T-cell responses in a mouse model via the intramuscular route, showed effective in vitro neutralization for MERS-CoV infection [47,50]. Additionally, vaccination of mice with an MVA-based full-length S vaccine-elicited MERS-CoV-specific CD8+ T cell response and neutralizing antibodies, protecting mice against MERS-CoV [48,49]. Intra-nasally or intra-muscularly administered MVA-S vaccine-induced mucosal immunity resulted in a significant reduction of excreted infectious viruses and viral RNA transcripts [45,46].Vaccines based on fragments containing neutralizing epitopes: fragments that were responsible for the virus binding to receptors within a host cell were targeted. Patients and animals that became infected with SAR-CoV-1 reacted strongly to this type of vaccine. They were immunized and inactivated with a receptor-binding domain (RBD) [29,38]. Absorption of antibodies by RBD showed the capability for removal of most of the neutralizing antibodies, RBD-specific antibodies isolated from these antisera have potent neutralizing activity [38,39]. The immunized mice were protected from SARS-CoV-1. The antibodies purified from the antisera against SARS-CoV-1 significantly inhibited RBD binding to ACE2 [29,38,40,41]. This suggested that RBD contains the major neutralizing epitopes in the S protein and is an ideal SARS vaccine candidate because RBD contains the receptor-binding site, which is critical for virus attachment to the target cell for infection [42,43,44]. Antibodies specific for RBD are expected to block the binding of the virus to the target cell. Therefore, RBD induces higher titers of neutralizing antibodies than those vaccines expressing the full-length S protein [31,32,35,37,43].
-
Nanoparticle-based MERS vaccine: in recent years nanoparticles have been at the forefront of many research projects, this has allowed them to have the potential to develop a MERS vaccine. Nanoparticles containing MERS-CoV full-length S proteins can be prepared and purified from pellets of infected baculovirus insect cells. In the absence of adjuvants, these nanoparticles induced a lower level of MERS-CoV producing antibodies in mice. While in the presence of adjuvants, such as aluminium hydroxide (Alum) or Matrix M1, such antibodies were significantly increased and maintained. Thus, adjuvants are required for MERS nanoparticle vaccines and different adjuvants function differently in promoting the immunogenicity of these vaccines [45].
-
DNA-based MERS vaccine: Like the full-length S gene of SARS-CoV-1, DNA encoding full-length S protein of MERS-CoV is utilized to develop MERS vaccines [31,51]. Indeed, intramuscular injections of mice with a synthetic DNA encoding full-length S proteins of MERS-CoV elicited potent virus-neutralizing antibodies and cellular immune responses, as represented by the secretion of INF-γ, TNF-α, and/or IL-2 cytokines in CD4+ and/or CD8+ T cells, as well as the production of antibodies in immunized camels.
-
Subunit MERS vaccines: Protein-based subunit vaccines against MERS-CoV have been developed [52,53,54]. While some are designed on the basis of the full-length S1 proteins [55], the majority of them are based on viral RBD [53,54,56,57]. These RBD-based vaccines are evaluated for immunogenicity and protective immunity in a number of MERS-CoV mice models. The antigenicity and functionality of these RBD proteins have also been extensively investigated. In general, subunit vaccines might not induce immune responses as strong as those induced by other vaccine types mentioned above. However, the immunogenicity of subunit vaccines could be significantly promoted in the presence of an ideal adjuvant via an appropriate route [52]. In addition, it is also essential to maintain a suitable conformation of the protein antigen in the vaccine, such as the MERS-CoV RBD proteins [53,56]. In terms of safety consideration, subunit vaccines should be accounted as the safest vaccine type. They do not contain viral genetic materials, but only include essential antigens for eliciting protective immune responses, thus excluding the possibility of recovering virulence or inducing adverse reactions [58,59,60].