Docosahexaenoic acid (DHA) supplementation during pregnancy has been recommended by several health organizations due to its role in neural, visual, and cognitive development. There are several fat sources available on the market for the manufacture of these dietary supplements with DHA. These fat sources differ in the lipid structure in which DHA is esterified, mainly phospholipids (PL) and triglycerides (TG) molecules. The supplementation of DHA in the form of PL or TG during pregnancy can lead to controversial results depending on the animal model, physiological status and the fat sources utilized. The intestinal digestion, placental uptake, and fetal accretion of DHA may vary depending on the lipid source of DHA ingested by the mother. The form of DHA used in maternal supplementation that would provide an optimal DHA accretion for fetal brain development, based on the available data obtained most of them from different animal models, indicates no consistent differences in fetal accretion when DHA is provided as TG or PL. Other related lipid species are under evaluation, e.g., lyso-phospholipids, with promising results to improve DHA bioavailability although more studies are needed. In this review, the evidence on DHA bioavailability and accumulation in both maternal and fetal tissues after the administration of DHA supplementation during pregnancy in the form of PL or TG in different models is summarized.
- DHA
- Pregnancy
- supplementation
- phospholipids
- triglycerides
1. Introduction
There is a growing interest in the effects of maternal diet consumed during pregnancy on both development and fetal programming of many physiological functions. During pregnancy and lactation there is an elevated docosahexaenoic acid (22:6 omega-3, DHA) requirement in the fetus and neonate as it is a critical building block of brain and retina [1,2,3][1][2][3]. In the last trimester of pregnancy, it is estimated a fetal accretion of 67 mg of omega-3 fatty acids (FA) per day, mainly DHA, and around 5% is delivered to the brain (3.1 mg/d) [4,5][4][5]. DHA conversion efficiency from α-linolenic acid (18:3 omega-3), its essential FA precursor, is very low (<1%) in fetus, placenta and newborns [6,7,8[6][7][8][9],9], being therefore insufficient to satisfy the high supply of DHA needed by the growing fetus [10,11][10][11]. Moreover, several studies have shown that supplementation with α-linolenic acid in human adults is not a good strategy to increase DHA levels, being necessary the direct supplementation with the preformed DHA molecule to observe: enhanced DHA status in blood and tissues [12], higher transfer of DHA to the fetus [13] or even to increase DHA secretion in human milk [14].
2. DHA Recommendations and Health Outcomes
2.1. DHA Intake during the Perinatal Period
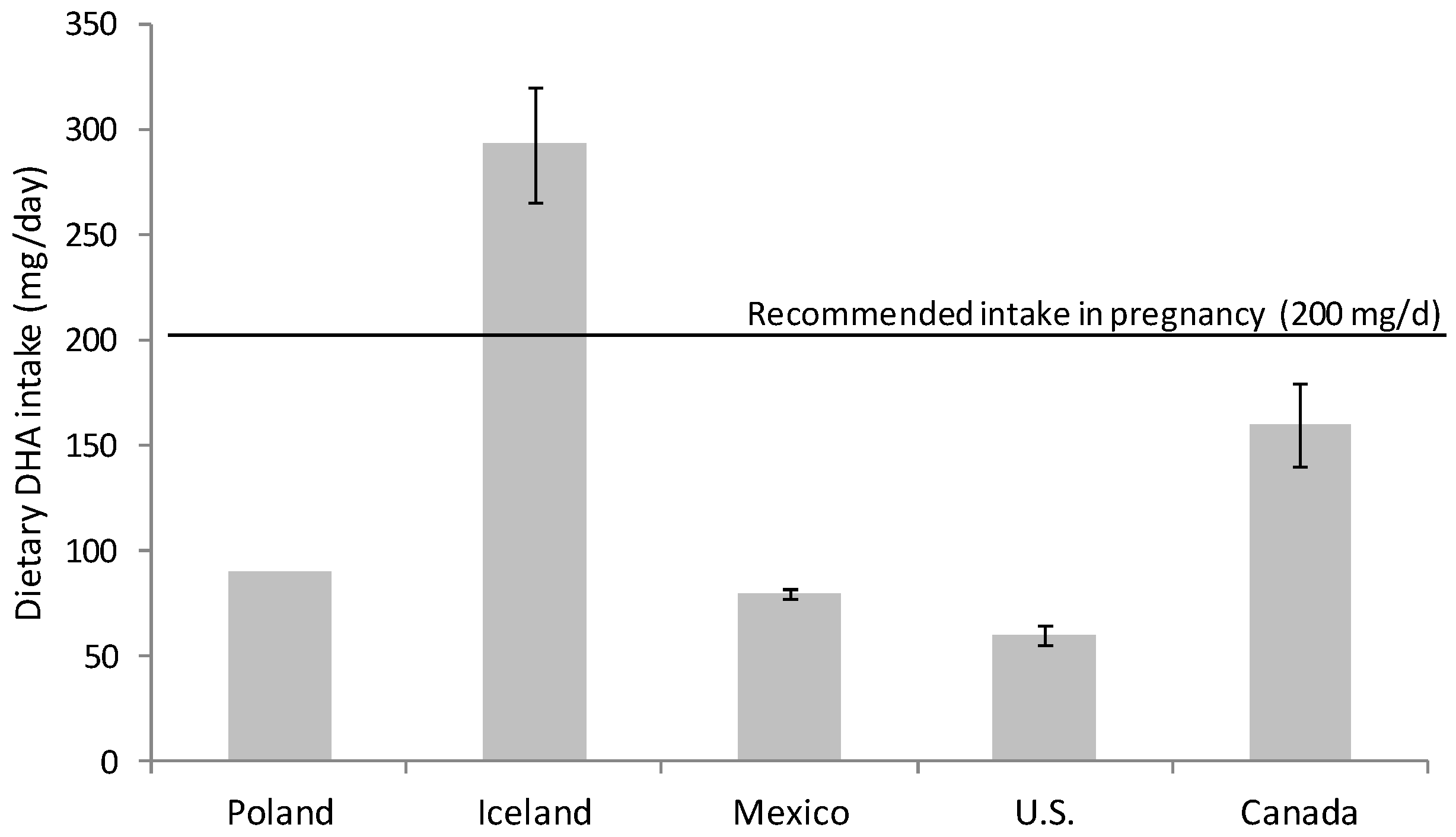
Omega-3 FA intake had fallen during the 20th century; the development of the modern vegetable oil industry, the use of cereal grains and the change in eating habits have produced a remarkable disparity in the ratio of consumption of omega-6 and omega-3 FA [15,16][15][16]. Omega-6 FA consumption, mainly in the form of linoleic acid (18:2 n-6), has increased at the expense of omega-3 FA (DHA and eicosapentaenoic acid (EPA, 20:5 n-3)) in the general population [15,16,17][15][16][17]. Nowadays, the intake of DHA in developed countries with free access to food of animal origin rich in micronutrients and omega-3 FA is highly variable. There are several studies evaluating DHA consumption that warn about inadequate dietary DHA intake for many women during pregnancy [18,19,20][18][19][20] (Figure 1). It is especially alarming the situation in western countries, for example in Canada and U.S. where DHA intakes are especially low, revealing that a majority of childbearing-age and pregnant women consume less than the recommended DHA dose [21,22][21][22] (Figure 1).
The preferential placental uptake and transfer of DHA to the fetus in relation to other FA (palmitic, oleic and linoleic acid) has been demonstrated by the administration of stable isotope-labelled FAs to pregnant women [25,26][25][26]. Moreover, the percentage of DHA and arachidonic acid (AA; 20:4 omega-6,) both in plasma and adipose tissue is higher in the neonate than in the mother, which reveals the important role of the placenta in the concentration of these FA in the fetal compartment [27,28][27][28]. This process is known as “biomagnification” and is defined as selective enrichment of these FA in fetal, with respect to maternal plasma [29]. AA and DHA concentration in non-esterified FA (NEFA) of the intervillous space of the placenta is 3–4 times higher than in maternal blood outside the placenta [30]. This fact implies that there is certain selectivity of placental tissue for the release of these long-chain polyunsaturated FA (LC-PUFA) from the circulating lipoproteins.
It is well known that the maternal DHA intake, and hence maternal DHA levels, during pregnancy determines the DHA status of the newborn at birth and for several weeks following delivery [31,32,33,34][31][32][33][34]. Large observational studies have shown that women with low seafood intakes during pregnancy are prone to an increased risk of poor infant cognition and behavioral outcome [35,36][35][36]. Low levels of DHA and AA in maternal plasma and cord blood has been related to lower head circumference, lower birthweight, lower placental weight [32], and less cognitive and visual maturation during childhood [37,38][37][38]. Other studies found associations between omega-3 FA intake during pregnancy and lower risks of intrauterine growth restriction, preterm birth, allergies, and asthma in children [19,39,40][19][39][40]. However, some randomized controlled trials and meta-analysis reported inconsistent evidences and very few differences between child born from omega-3 supplemented vs. placebo mothers on long-term vision, growth and neurodevelopment outcomes [41,42,43,44,45][41][42][43][44][45]. Further follow-up studies are needed to assess the longer-term consequences and health outcomes for both mother and child of maternal omega-3 supplementation.
2.2. Dietary Recommendation during Pregnancy and Lactation
DHA dietary supplementation has been recommended by several health organizations [46,47][46][47]. European and global guidelines recommends the intake of at least 200 mg/d DHA during these periods, which can be met with two servings of fish per week [48,49,50][48][49][50]. The highest concentration of DHA is found in seafood, especially in oily fish (tuna, salmon, herring, mackerel, etc.) [51]. Smaller fishes are highly recommendable since they contain lower levels of methyl mercury and other contaminants than large-size predators [49]. Probably, the dose should be higher to detect significant effects on some outcomes but, due to the high variability in DHA intake from other sources, these recommendations are highly conservative.
DHA supplementation should be considered only if dietary consumption (natural sources) is not sufficient to meet the recommendations or when it is problematic due to food availability, socio-cultural dietary preferences, fish aversion, ethics issues (e.g., vegans), or other factors [52].
3. Conclusions
The animal model used to evaluate the materno-fetal transfer of DHA as PL or TG is a key issue since placental structure differs among the species used for such studies. Administration of DHA-rich PL produces a modest enrichment of DHA in PL plasma lipid fraction in piglets and pregnant sows compared to DHA-TG administration while similar or opposite results have been observed in other species. Intestinal digestion, re-esterification in both gut enterocytes and liver, as well as placental transfer processes reduce the impact of the dietary intervention with different lipid sources on fetal DHA levels. Dietary lipid utilization and bioavailability comprises several metabolic processes that are not completely understood and further research is needed. There are a limited number of studies evaluating placental and fetal accretion of DHA after the administration of different fat sources to pregnant animals (PL and TG). Despite some differences observed in placental DHA content between animal species, fetal DHA accretion and, especially, fetal brain DHA accumulation after PL or TG administration was similar. However, it is important to note that the use of animal models (rodents and pigs) in most studies might have some limitations in extrapolating results to humans. Lyso-PL have been proposed as a preferred physiological carrier of DHA to the brain, the available data on DHA Lyso-PL bioavailability with respect to other sources are promising and seem to indicate an increased DHA incorporation in some tissues but more studies are needed to evaluate their effects during pregnancy, fetal bioavailability, and long-term effects on neurodevelopment. In summary, although most of the results available were obtained in animal models, both PL and TG sources can be used for the manufacture of DHA supplements during pregnancy since they show a comparable bioavailability and promote similar DHA accretion in the fetus. The dose of DHA administered is perhaps more decisive than the fat source to increase fetal DHA status.
References
- Clandinin, M.T.; Chappell, J.E.; Leong, S.; Heim, T.; Swyer, P.R.; Chance, G.W. Intrauterine fatty acid accretion rates in human brain: Implications for fatty acid requirements. Early Hum. Dev. 1980, 4, 121–129.
- Martinez, M. Tissue levels of polyunsaturated fatty acids during early human development. J. Pediatr. 1992, 120, S129–S138.
- Coletta, J.M.; Bell, S.J.; Roman, A.S. Omega-3 Fatty acids and pregnancy. Rev. Obstet. Gynecol. 2010, 3, 163–171.
- Clandinin, M.T.; Chappell, J.E.; Heim, T.; Swyer, P.R.; Chance, G.W. Fatty acid utilization in perinatal de novo synthesis of tissues. Early Hum. Dev. 1981, 5, 355–366.
- Georgieff, M.K.; Innis, S.M. Controversial nutrients that potentially affect preterm neurodevelopment: Essential fatty acids and iron. Pediatr. Res. 2005, 57, 99R–103R.
- Innis, S.M. Perinatal biochemistry and physiology of long-chain polyunsaturated fatty acids. J. Pediatr. 2003, 143, S1–S8.
- Pawlosky, R.J.; Hibbeln, J.R.; Novotny, J.A.; Salem, N., Jr. Physiological compartmental analysis of alpha-linolenic acid metabolism in adult humans. J. Lipid Res. 2001, 42, 1257–1265.
- Goyens, P.L.; Spilker, M.E.; Zock, P.L.; Katan, M.B.; Mensink, R.P. Conversion of alpha-linolenic acid in humans is influenced by the absolute amounts of alpha-linolenic acid and linoleic acid in the diet and not by their ratio. Am. J. Clin. Nutr. 2006, 84, 44–53.
- Chambaz, J.; Ravel, D.; Manier, M.C.; Pepin, D.; Mulliez, N.; Bereziat, G. Essential fatty acids interconversion in the human fetal liver. Biol. Neonate 1985, 47, 136–140.
- Uauy, R.; Hoffman, D.R.; Peirano, P.; Birch, D.G.; Birch, E.E. Essential fatty acids in visual and brain development. Lipids 2001, 36, 885–895.
- Salem, N., Jr.; Wegher, B.; Mena, P.; Uauy, R. Arachidonic and docosahexaenoic acids are biosynthesized from their 18-carbon precursors in human infants. Proc. Natl. Acad. Sci. USA 1996, 93, 49–54.
- Burdge, G.C.; Calder, P.C. Conversion of alpha-linolenic acid to longer-chain polyunsaturated fatty acids in human adults. Reprod. Nutr. Dev. 2005, 45, 581–597.
- De Groot, R.H.; Hornstra, G.; van Houwelingen, A.C.; Roumen, F. Effect of alpha-linolenic acid supplementation during pregnancy on maternal and neonatal polyunsaturated fatty acid status and pregnancy outcome. Am. J. Clin. Nutr. 2004, 79, 251–260.
- Innis, S.M. Polyunsaturated fatty acids in human milk: An essential role in infant development. Adv. Exp. Med. Biol. 2004, 554, 27–43.
- Sanders, T.A. Polyunsaturated fatty acids in the food chain in Europe. Am. J. Clin. Nutr. 2000, 71, 176S–178S.
- Blasbalg, T.L.; Hibbeln, J.R.; Ramsden, C.E.; Majchrzak, S.F.; Rawlings, R.R. Changes in consumption of omega-3 and omega-6 fatty acids in the United States during the 20th century. Am. J. Clin. Nutr. 2011, 93, 950–962.
- Leaf, A.; Weber, P.C. Cardiovascular effects of n-3 fatty acids. N. Engl. J. Med. 1988, 318, 549–557.
- Parra-Cabrera, S.; Stein, A.D.; Wang, M.; Martorell, R.; Rivera, J.; Ramakrishnan, U. Dietary intakes of polyunsaturated fatty acids among pregnant Mexican women. Matern. Child. Nutr. 2011, 7, 140–147.
- Nordgren, T.M.; Lyden, E.; Anderson-Berry, A.; Hanson, C. Omega-3 Fatty Acid Intake of Pregnant Women and Women of Childbearing Age in the United States: Potential for Deficiency? Nutrients 2017, 9, 197.
- Loosemore, E.D.; Judge, M.P.; Lammi-Keefe, C.J. Dietary intake of essential and long-chain polyunsaturated fatty acids in pregnancy. Lipids 2004, 39, 421–424.
- Innis, S.M.; Elias, S.L. Intakes of essential n-6 and n-3 polyunsaturated fatty acids among pregnant Canadian women. Am. J. Clin. Nutr. 2003, 77, 473–478.
- Zhang, Z.; Fulgoni, V.L.; Kris-Etherton, P.M.; Mitmesser, S.H. Dietary Intakes of EPA and DHA Omega-3 Fatty Acids among US Childbearing-Age and Pregnant Women: An Analysis of NHANES 2001–2014. Nutrients 2018, 10, 416.
- Gunnarsdottir, I.; Tryggvadottir, E.A.; Birgisdottir, B.E.; Halldorsson, T.I.; Medek, H.; Geirsson, R.T. Diet and nutrient intake of pregnant women in the capital area in Iceland. Laeknabladid 2016, 102, 378–384.
- Wierzejska, R.; Jarosz, M.; Wojda, B.; Siuba-Strzelinska, M. Dietary intake of DHA during pregnancy: A significant gap between the actual intake and current nutritional recommendations. Rocz. Panstw. Zakl. Hig. 2018, 69, 381–386.
- Gil-Sanchez, A.; Larque, E.; Demmelmair, H.; Acien, M.I.; Faber, F.L.; Parrilla, J.J.; Koletzko, B. Maternal-fetal in vivo transfer of [13C]docosahexaenoic and other fatty acids across the human placenta 12 h after maternal oral intake. Am. J. Clin. Nutr. 2010, 92, 115–122.
- Larque, E.; Demmelmair, H.; Berger, B.; Hasbargen, U.; Koletzko, B. In vivo investigation of the placental transfer of (13)C-labeled fatty acids in humans. J. Lipid Res. 2003, 44, 49–55.
- Crawford, M.A.; Costeloe, K.; Ghebremeskel, K.; Phylactos, A.; Skirvin, L.; Stacey, F. Are deficits of arachidonic and docosahexaenoic acids responsible for the neural and vascular complications of preterm babies? Am. J. Clin. Nutr. 1997, 66, 1032S–1041S.
- Bitsanis, D.; Crawford, M.A.; Moodley, T.; Holmsen, H.; Ghebremeskel, K.; Djahanbakhch, O. Arachidonic acid predominates in the membrane phosphoglycerides of the early and term human placenta. J. Nutr. 2005, 135, 2566–2571.
- Crawford, M.A.; Hassam, A.G.; Williams, G. Essential fatty acids and fetal brain growth. Lancet 1976, 1, 452–453.
- Benassayag, C.; Mignot, T.M.; Haourigui, M.; Civel, C.; Hassid, J.; Carbonne, B.; Nunez, E.A.; Ferre, F. High polyunsaturated fatty acid, thromboxane A2, and alpha-fetoprotein concentrations at the human feto-maternal interface. J. Lipid Res. 1997, 38, 276–286.
- Lauritzen, L.; Carlson, S.E. Maternal fatty acid status during pregnancy and lactation and relation to newborn and infant status. Matern. Child. Nutr. 2011, 7, 41–58.
- Guesnet, P.; Pugo-Gunsam, P.; Maurage, C.; Pinault, M.; Giraudeau, B.; Alessandri, J.M.; Durand, G.; Antoine, J.M.; Couet, C. Blood lipid concentrations of docosahexaenoic and arachidonic acids at birth determine their relative postnatal changes in term infants fed breast milk or formula. Am. J. Clin. Nutr. 1999, 70, 292–298.
- Elias, S.L.; Innis, S.M. Infant plasma trans, n-6, and n-3 fatty acids and conjugated linoleic acids are related to maternal plasma fatty acids, length of gestation, and birth weight and length. Am. J. Clin. Nutr. 2001, 73, 807–814.
- Helland, I.B.; Saugstad, O.D.; Smith, L.; Saarem, K.; Solvoll, K.; Ganes, T.; Drevon, C.A. Similar effects on infants of n-3 and n-6 fatty acids supplementation to pregnant and lactating women. Pediatrics 2001, 108, E82.
- Hibbeln, J.R.; Davis, J.M.; Steer, C.; Emmett, P.; Rogers, I.; Williams, C.; Golding, J. Maternal seafood consumption in pregnancy and neurodevelopmental outcomes in childhood (ALSPAC study): An observational cohort study. Lancet 2007, 369, 578–585.
- Oken, E.; Wright, R.O.; Kleinman, K.P.; Bellinger, D.; Amarasiriwardena, C.J.; Hu, H.; Rich-Edwards, J.W.; Gillman, M.W. Maternal fish consumption, hair mercury, and infant cognition in a U.S. Cohort. Environ. Health Perspect. 2005, 113, 1376–1380.
- Innis, S.M. Fatty acids and early human development. Early Hum. Dev. 2007, 83, 761–766.
- Lauritzen, L.; Brambilla, P.; Mazzocchi, A.; Harslof, L.B.; Ciappolino, V.; Agostoni, C. DHA Effects in Brain Development and Function. Nutrients 2016, 8, 6.
- Emmett, P.M.; Jones, L.R.; Golding, J. Pregnancy diet and associated outcomes in the Avon Longitudinal Study of Parents and Children. Nutr. Rev. 2015, 73, 154–174.
- De Giuseppe, R.; Roggi, C.; Cena, H. n-3 LC-PUFA supplementation: Effects on infant and maternal outcomes. Eur. J. Nutr. 2014, 53, 1147–1154.
- Smithers, L.G.; Gibson, R.A.; Makrides, M. Maternal supplementation with docosahexaenoic acid during pregnancy does not affect early visual development in the infant: A randomized controlled trial. Am. J. Clin. Nutr. 2011, 93, 1293–1299.
- Makrides, M.; Gibson, R.A.; McPhee, A.J.; Yelland, L.; Quinlivan, J.; Ryan, P. Effect of DHA supplementation during pregnancy on maternal depression and neurodevelopment of young children: A randomized controlled trial. JAMA 2010, 304, 1675–1683.
- Middleton, P.; Gomersall, J.C.; Gould, J.F.; Shepherd, E.; Olsen, S.F.; Makrides, M. Omega-3 fatty acid addition during pregnancy. Cochrane Database Syst. Rev. 2018, 11, CD003402.
- Larque, E.; Demmelmair, H.; Gil-Sanchez, A.; Prieto-Sanchez, M.T.; Blanco, J.E.; Pagan, A.; Faber, F.L.; Zamora, S.; Parrilla, J.J.; Koletzko, B. Placental transfer of fatty acids and fetal implications. Am. J. Clin. Nutr. 2011, 94, 1908S–1913S.
- Newberry, S.J.; Chung, M.; Booth, M.; Maglione, M.A.; Tang, A.M.; O’Hanlon, C.E.; Wang, D.D.; Okunogbe, A.; Huang, C.; Motala, A.; et al. Omega-3 Fatty Acids and Maternal and Child Health: An Updated Systematic Review. Evid. Rep. Technol. Assess 2016, 1–826.
- Innis, S.M. The role of dietary n-6 and n-3 fatty acids in the developing brain. Dev. Neurosci. 2000, 22, 474–480.
- Lauritzen, L.; Hansen, H.S.; Jorgensen, M.H.; Michaelsen, K.F. The essentiality of long chain n-3 fatty acids in relation to development and function of the brain and retina. Prog. Lipid Res. 2001, 40, 1–94.
- World Health Organization. Fats and fatty acids in human nutrition. In Proceedings of the Joint FAO/WHO Expert Consultation, Geneva, Switzerland, 10–14 November 2008; pp. 5–300.
- Koletzko, B.; Cetin, I.; Brenna, J.T. Dietary fat intakes for pregnant and lactating women. Br. J. Nutr. 2007, 98, 873–877.
- Panel on Dietetic Products, Nutrition and Allergies. Scientific opinion of the Panel on Dietary Reference Values for fats, including saturated fatty acids, polyunsaturated fatty acids, monounsaturated fatty acids, trans fatty acids, and cholesterol. EFSA J. 2010, 8, 1461.
- Lee, J.H.; O’Keefe, J.H.; Lavie, C.J.; Harris, W.S. Omega-3 fatty acids: Cardiovascular benefits, sources and sustainability. Nat. Rev. Cardiol. 2009, 6, 753–758.
- Kris-Etherton, P.M.; Hill, A.M. N-3 fatty acids: Food or supplements? J. Am. Diet. Assoc. 2008, 108, 1125–1130.
